Abstract
Growth inhibitory factor (GIF) is a small protein belonging to the metallothionein family that has the capacity to inhibit neuronal survival and neurite formation in vitro. This study was conducted to investigate the role of GIF in the brain afflicted with ischemic injury. We used the in situ hybridization technique and Northern blot analysis to study the changes in GIF messenger RNA (mRNA) expression in a rat focal ischemia model. On the first day, the expression tended to decrease in the hemisphere ipsilateral to the injury. It returned to normal levels on the second day except for the central area of the middle cerebral artery (MCA) territory. On the third and fourth day, the expression increased diffusely in the hemisphere of the affected side, including the subcortical area. Two weeks after ischemia, the GIF mRNA expression increased again but only in the pert-infarcted area. Down-regulation of GIF on the first day in the cortex ipsilateral to the infarction might promote neurite sprouting. The subsequent increase in GIF mRNA expression on the third and fourth day might be a symptom of neurons attempting to inhibit excessive neurite outgrowth, or to protect themselves against toxicity caused by oxygen radicals. The later increase in the limited area around the infarction may be related to astroglial reaction. Growth inhibitory factor may play an important role in regulating the central nervous system after ischemic insults.
Several neurotrophic factors have been found to increase in the brain after various injuries including ischemic insults (Finklestein et al., 1988; Frautschy et al., 1991; Garcia et al., 1992; Gomez et al., 1992; Lindholm et al., 1992). These factors increase around the wound and are thought to take part in rescuing neurons and repairing the damaged network. However, some regulatory mechanism must exist to maintain the neuronal network in order. Certain inhibitory factors, which suppress excessive neurite elongation, can be assumed to take part in such a mechanism. We focused on growth inhibitory factor (GIF) as one of the inhibitory factors because it has been shown to inhibit survival and neurite formation of cortical neurons in vitro (Tsuji et al., 1992).
Growth inhibitory factor was originally discovered by Uchida et al. (Uchida et al., 1989) in relation to Alzheimer's disease. Although it is abundantly present in the normal brain, the level of GIF is greatly reduced in the brain of the patient with Alzheimer's disease (Uchida et al., 1989; Uchida et al., 1991). Growth inhibitory factor is found to be a member of the metallothionein (MT) family and named also as MT-III (Palmiter et al., 1992). Purified GIF is a small 68-amino acid protein with a molecular weight of 6.9 kd. Human GIF cDNA was cloned by Tsuji et al. (1992), and rat GIF cDNA was cloned by Kobayashi et al (1993). Growth inhibitory factor messenger RNA (mRNA) expression changes during brain development. The rat GIF mRNA showed monophasic increase at days 10 to 17, suggesting its role in regulation of neurite elongation (Kobayashi et al., 1993). Although both MT-I and GIF have high sequence homology, only GIF has the capacity to inhibit neuronal survival and neurite formation in vitro (Tsuji et al., 1992). Numerous studies have examined MT gene expression in the liver (e.g., Ebadi et al., 1995). GIF exists only in the brain (Kobayashi et al., 1993), although other members of the MT family, MT-I and MT-II, are found in all organs of the body (Masters et al., 1994). Metallothionein-III is expressed predominantly in neurons that sequester zinc in synaptic vesicles (Masters et al., 1994; Yuguchi et al., 1995). However, less is known about GIF gene expression after injury in the mature brain.
In situ hybridization technique and Northern blot analysis were used to investigate changes in GIF mRNA expression after focal cerebral ischemia in rats. Possible roles of GIF in the brain after ischemic injury are discussed.
MATERIALS AND METHODS
Experimental model
Techniques for generating middle cerebral artery (MCA) occlusion have been used previously in our laboratory (Kataoka et al., 1991; Tamura et al., 1981). Forty-three male Sprague Dawley rats (body weight, 250 g) were used in this study. Using general anesthesia with an intraperitoneal injection of chloral hydrate (300 mg/kg), the left MCA was exposed via a small opening in the skull and dura. The MCA was cauterized at the level of the olfactory tract. The sham operation was performed in the same manner but without coagulation of the MCA (n = 6). A rectal thermometer and heating unit were used to maintain the rectal temperature constantly at 37°C throughout the experiment.
The animals were deeply anesthetized with an intraperitoneal injection of pentobarbital (60 mg/kg) and decapitated one hour (n = 4), 12 hours (n = 2), one day (n = 7), two days (n = 1), three days (n = 6), four days (n = 6), seven days (n = 6), and 14 days (n = 5) after surgery.
All animal experiments have been conducted according to the Osaka University Medical School Guideline for the Care and Use of Laboratory Animals. Animal surgeries and experimental procedure were approved by the Osaka University Medical School Animal Care and Use Committee.
Tissue preparation and immunohistochemistry
The brains were quickly removed and frozen at −80°C. Serial coronal sections (10-μm thick) were obtained from the frozen brains with a cryostat and mounted on slides coated with 3-aminopropyltriethoxysilane and stored in a tightly closed case at −80°C.
Besides being used for the in situ hybridization study, adjacent sections were stained with hematoxylin and eosin to determine the size of the infarction and immunohistochemically for glial fibrillary acidic protein (GFAP). The primary antibody for GFAP was supplied by Boehringer Mannheim Biochemica GmbH (Mannheim, Germany). Immunohistochemical staining was performed by using the avidin-biotin complex method. The GFAP antibody (20 μg/mL) was used at a dilution of 1:40. The staining procedure consisted of overnight incubation in the primary antibody with 5% normal horse serum at 4°C after quenching the endogenous peroxidase activity with 0.3% H2O2, followed by brief washes, and incubation in a cocktail of biotinylated anti-mouse immunoglobulin Ig G/Ig M for 30 minutes at room temperature. The sections were then washed, incubated in avidin-biotin complex with peroxidase for 30 minutes, washed again, and finally reacted with diaminobenzidine and H2O2 to visualize the end product. The sections were lightly counter-stained with hematoxylin.
In situ hybridization
The unique 3′-non-coding sequence of rat GIF cDNA (pcD2RDIF2) was used to avoid cross hybridization to MT-I and MT-II mRNA. Production of RNA probes and in situ hybridization for GIF were performed according to the previously reported procedure (Yuguchi et al., 1995). Hybridization was performed overnight at 55°C and followed by a subsequent high-stringency washing at 65°C.
For semiquantitative analysis of GIF mRNA expression, the optical density of the cortex, marginal zone, and caudate putamen on the macroautoradiogram was measured with a densitometer (Sakura PDA-15, Konishiroku, Tokyo, Japan). Regions of interest were selected in these three areas as well as in the symmetrically corresponding areas on the contralateral hemisphere. The optical density of the region of interest was then measured three times and the mean values of the counts were used for further analysis. The optical density ratio of the lesion side was calculated by dividing the optical density of the lesion side by that of the contralateral side. Statistical analysis was performed with a one-way analysis of variance for each area followed by multiple t-test with Bonferroni correction for comparison to a control group.
To clarify the cellular localization of GIF mRNA expression, we also synthesized digoxigenin-labeled single strand RNA probes with changing the [35S]-UTP to digoxigenin-labeled-UTP. In situ hybridization for GIF were performed with the same conditions (Yuguchi et al., 1995). Hybridized digoxigenin-labeled probes were detected by Nucleic Acid Detection Kit (Boehringer Mannheim Biochemica GmbH, Mannheim, Germany) according to the manufacturer's instructions. After color reaction, the slides were rinsed with 10 mmol/L Tris-HCl pH 8.0 with 1 mmol/L EDTA. Controls included hybridization with the sense (mRNA) probe; RNase A treatment (20 μg/mL) before hybridization; and use of neither antisense RNA probe nor anti-digoxigenin antibody. All three control experiments showed no positive signals.
Northern blot analysis
Total cytoplasmic RNA was isolated from the bilateral rat cortex with the acid of guanidinium-phenol-chloroform method (Chomczynski et al., 1987) and 15 μg of samples were fractionated by electrophoresis through 1% agarose gels containing formaldehyde. Then, the RNA was transferred onto nylon membranes (Hybond-H+, Amersham, Buckinghamshir, U.K.). After prehybridization at 65°C, hybridization was performed overnight using random-primed, 32P-labeled probes with a specific activity of 2 ×: 109 counts per minute per mg at 65°C. The membranes were washed in 2 × SSC/1% SDS for 1 hour at 50°C and subsequently, in 0.1 × SSC for 30 minutes at room temperature. Then, the membranes were exposed to Kodak XAR 5 film (Eastman Kodak Company, Rochester, NY, U.S.A.) with intensifying screens at −70°C for 5 days. After quantification of the hybridized probe, it was removed with a boiling solution of 0.1% SDS poured on the membrane and allowed to return to room temperature. Hybridization and washing for the GAPDH probe was carried out in the same manner. Finally, X-ray film was placed on the membrane at −80°C for 1 day.
RESULTS
Consecutive sections were hybridized with both the sense and antisense probes throughout this study. Significant signals were observed only in the sections hybridized with the antisense probes (Fig. 1A, B), thus confirming the specificity of the hybridization signals. In sham-operated animals, GIF mRNA was expressed consistently in the hippocampus, the cerebral cortex, and the thalamus. On the first day after MCA occlusion, reduced staining with hematoxylin and eosin of the central zone of the MCA territory was noted suggesting infarction (Fig. 2B), while the expression of GIF mRNA disappeared in this area (Fig. 2A). Interestingly, the expression of GIF mRNA was reduced only slightly in the affected hemisphere except for this central area of the MCA territory in comparison with that on the contralateral side (Fig. 2A, C, D). After 2 days, the expression pattern, except in the infarcted area had not remarkably changed in comparison with that of sham-operated animals.
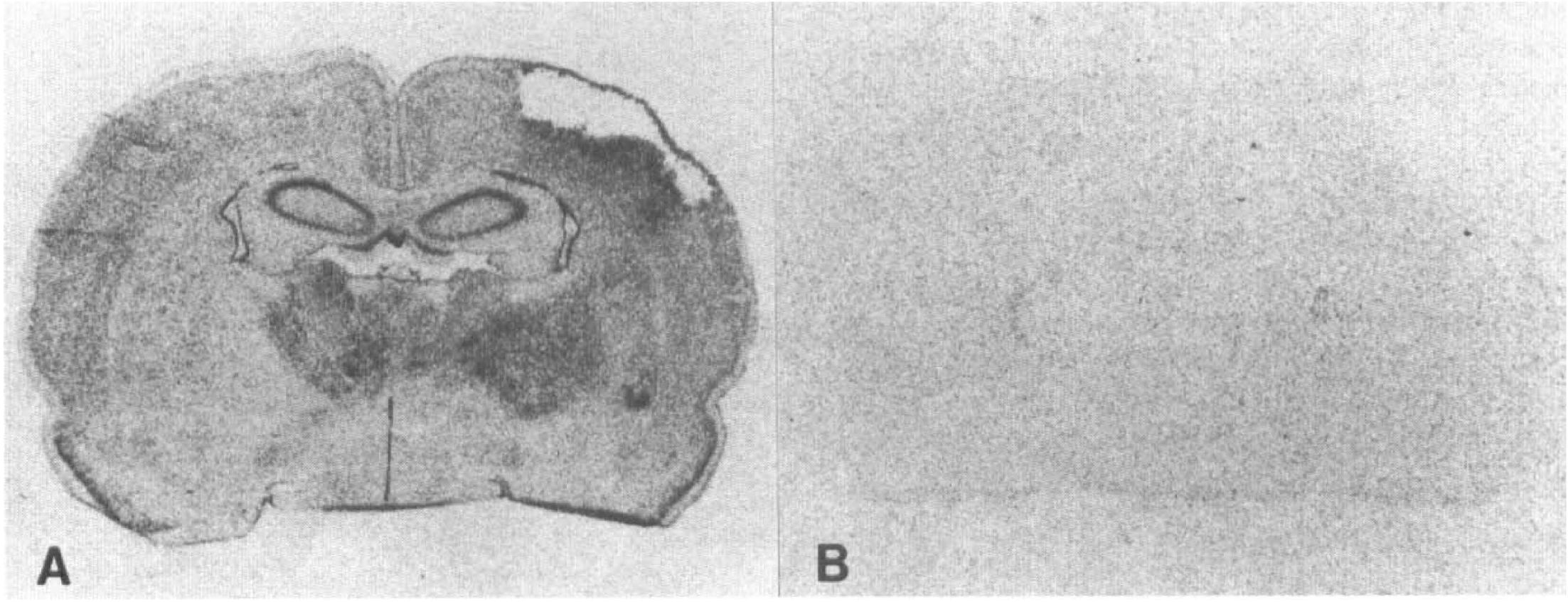
Macroautoradiograms of in situ hybridization for growth inhibitory factor (GIF) messenger RNA (mRNA) 2 weeks after middle cerebral artery (MCA) occlusion. An anti-sense probe (
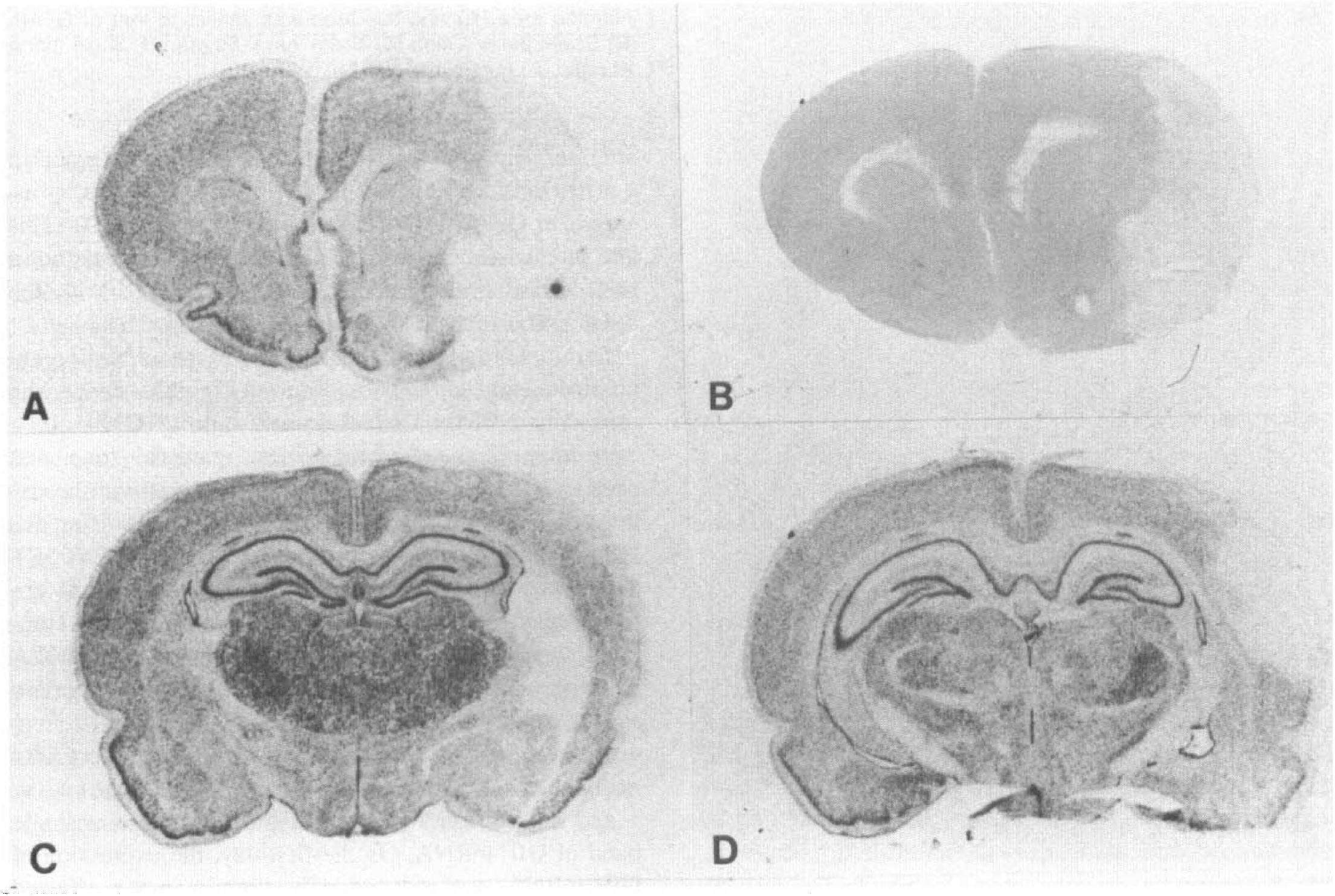
Macroautoradiograms of in situ hybridization for GIF mRNA at different levels of the brain sections (
However, in animals observed 3 and 4 days after surgery, the expression increased remarkably on the affected side cortex. This increase in GIF mRNA expression extended over the entire ipsilateral cortex and subcortical area except for the infarcted area (Fig. 3A, B, C). However, this increase was only transient and had already returned to normal levels by day 7. Two weeks after surgery, GIF mRNA expression increased again in a restricted area adjacent to the infarction, where an increase in GFAP was detected (Fig. 4A, B, C, D). This late intensification of GIF mRNA expression was not a false signal because the sections hybridized with the sense probe revealed no such signals (Fig. 1B).
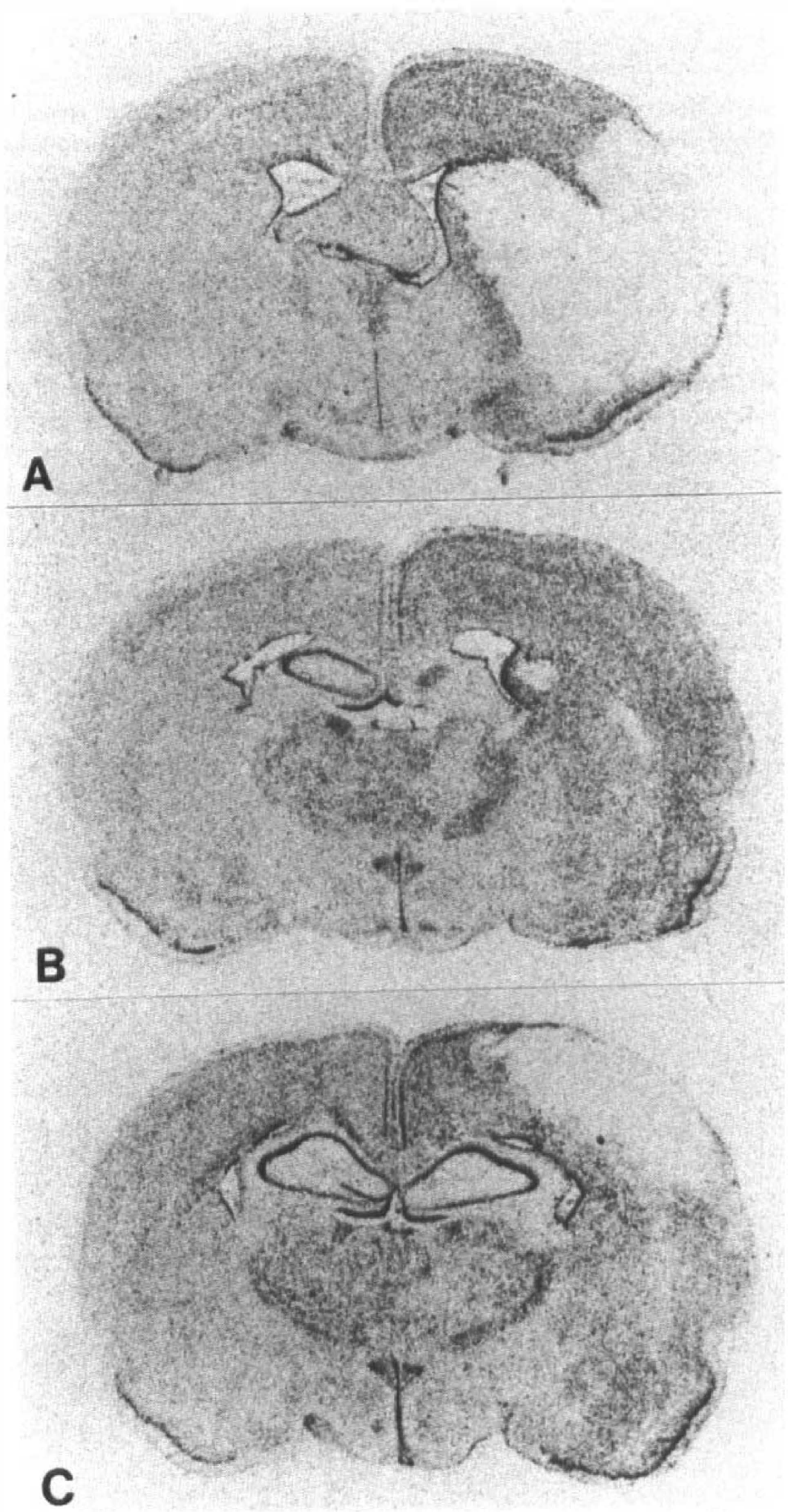
Macroautoradiograms of in situ hybridization for GIF mRNA at different levels of the brain sections 4 days after MCA occlusion. The gene expression increased diffusely in the affected side cortex, caudate putamen, and subcortical white matter except for the central zone of the MCA territory.
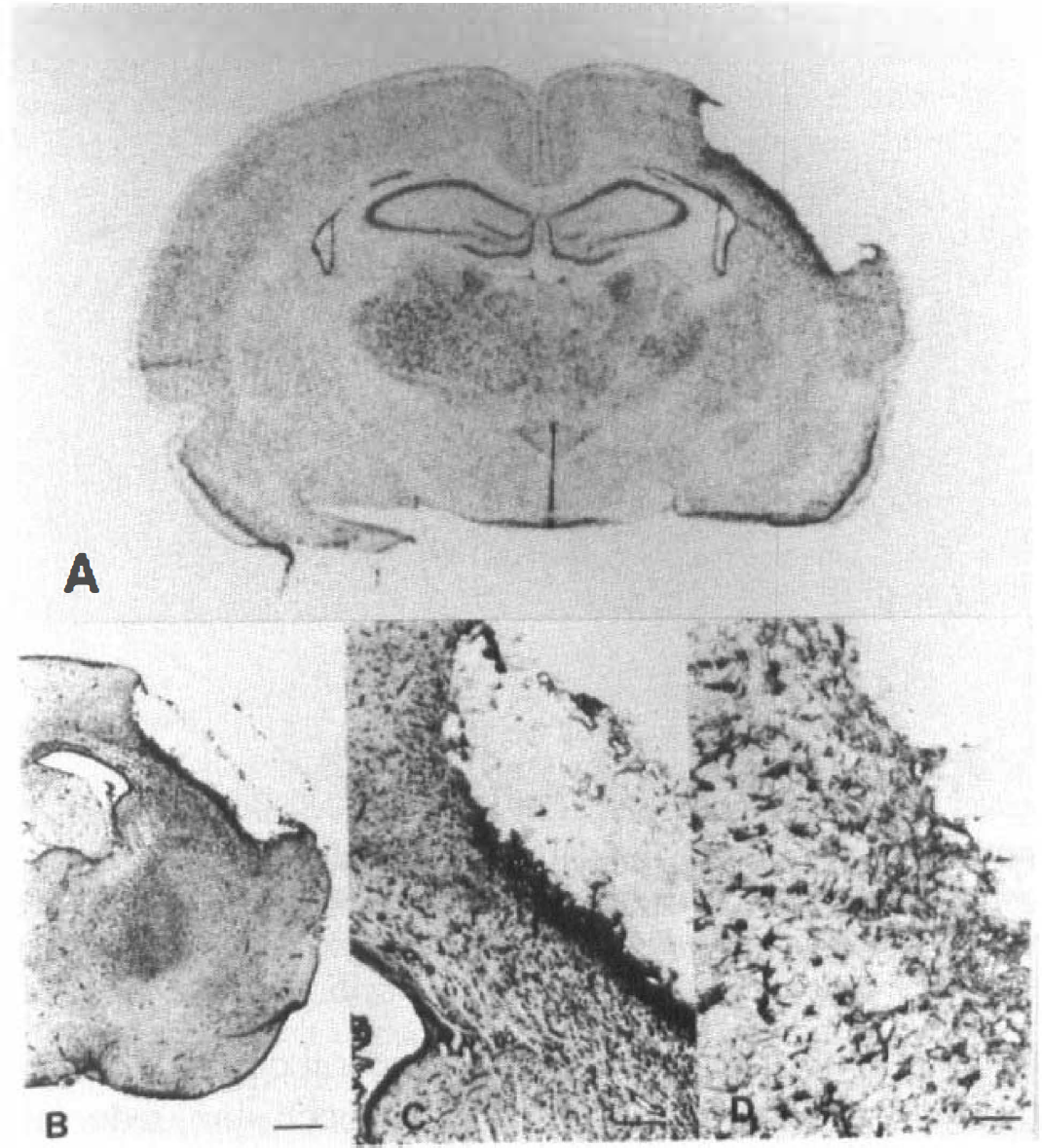
Macroautoradiogram of in situ hybridization for GIF mRNA (
Semiquantitative evaluation of macroautoradiogram confirmed the above observation. Fig. 5 shows the time course of the optical density ratio of GIF mRNA expression in the cerebral cortex, marginal zone, and caudate putamen (Fig. 5A, B, C). Analysis with a one-way analysis of variance revealed significant difference in each area (P < 0.001 for A and B; P = 0.003 for C). There was a significant (P < 0.05) difference in the optical density ratio on days 1, 3, and 4 in all areas compared with that of sham controls. Two weeks after MCA occlusion, a significant difference in optical density ratio was seen only in the marginal zone (Fig. 5B).
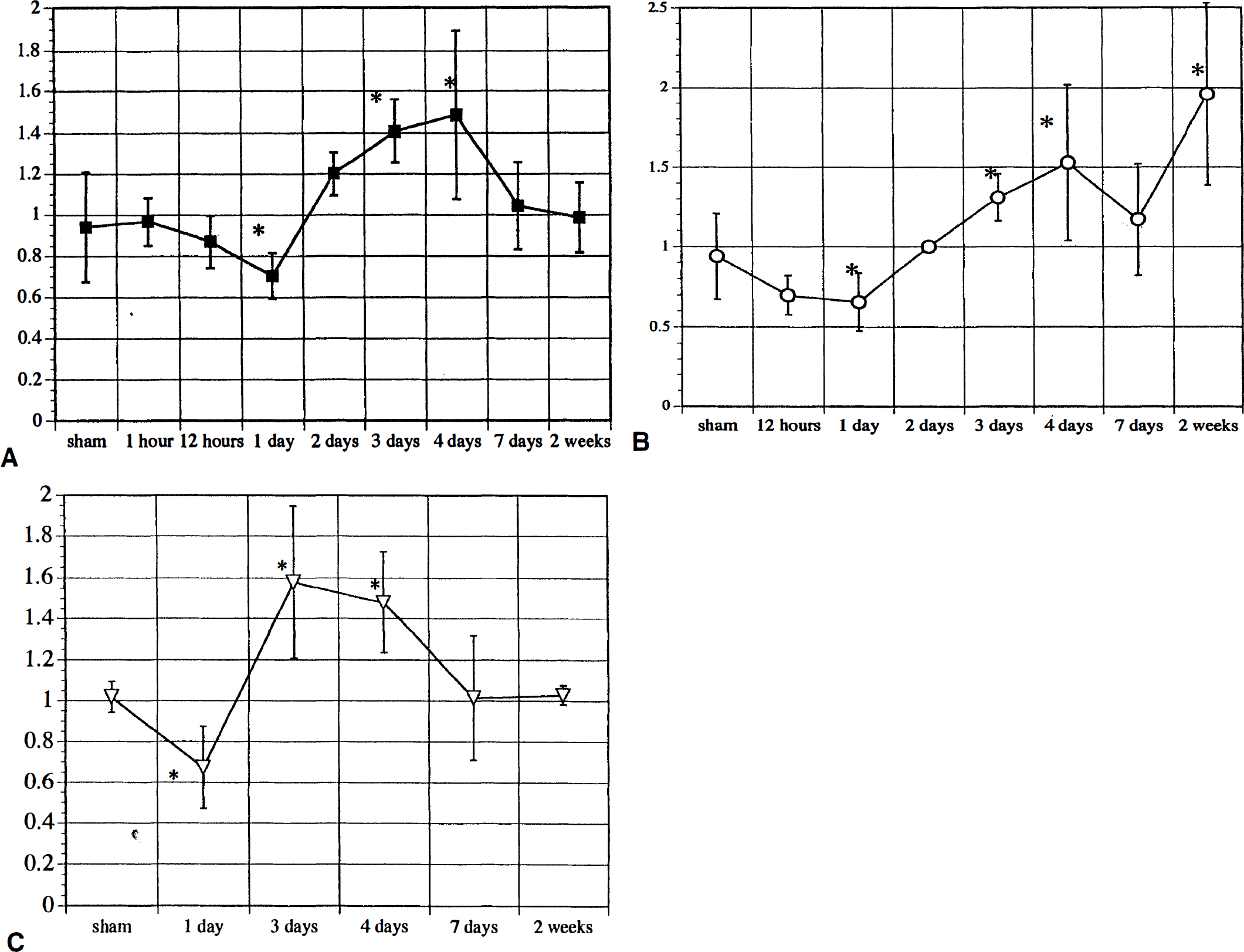
The time course of the optical density ratio on the macroautoradiogram for GIF mRNA expression in the cerebral cortex (
Besides in situ hybridization, we did Northern blot analysis of rat GIF mRNA in the cerebral cortex on day 1 and day 4 after MCA occlusion. It showed a single band of GIF mRNA. On the first day, the expression of GIF mRNA was reduced only slightly in the affected side cortex. Four days after MCA occlusion, the expression increased remarkably on the affected side cortex (Fig. 6). Densitometry revealed that the ratio of ipsilateral to contralateral GIF mRNA corrected with GAPDH mRNA was 0.72 for 1 day and 3.94 for 4 days after MCA occlusion respectively. Northern blot analysis thus confirmed the results of in situ hybridization.
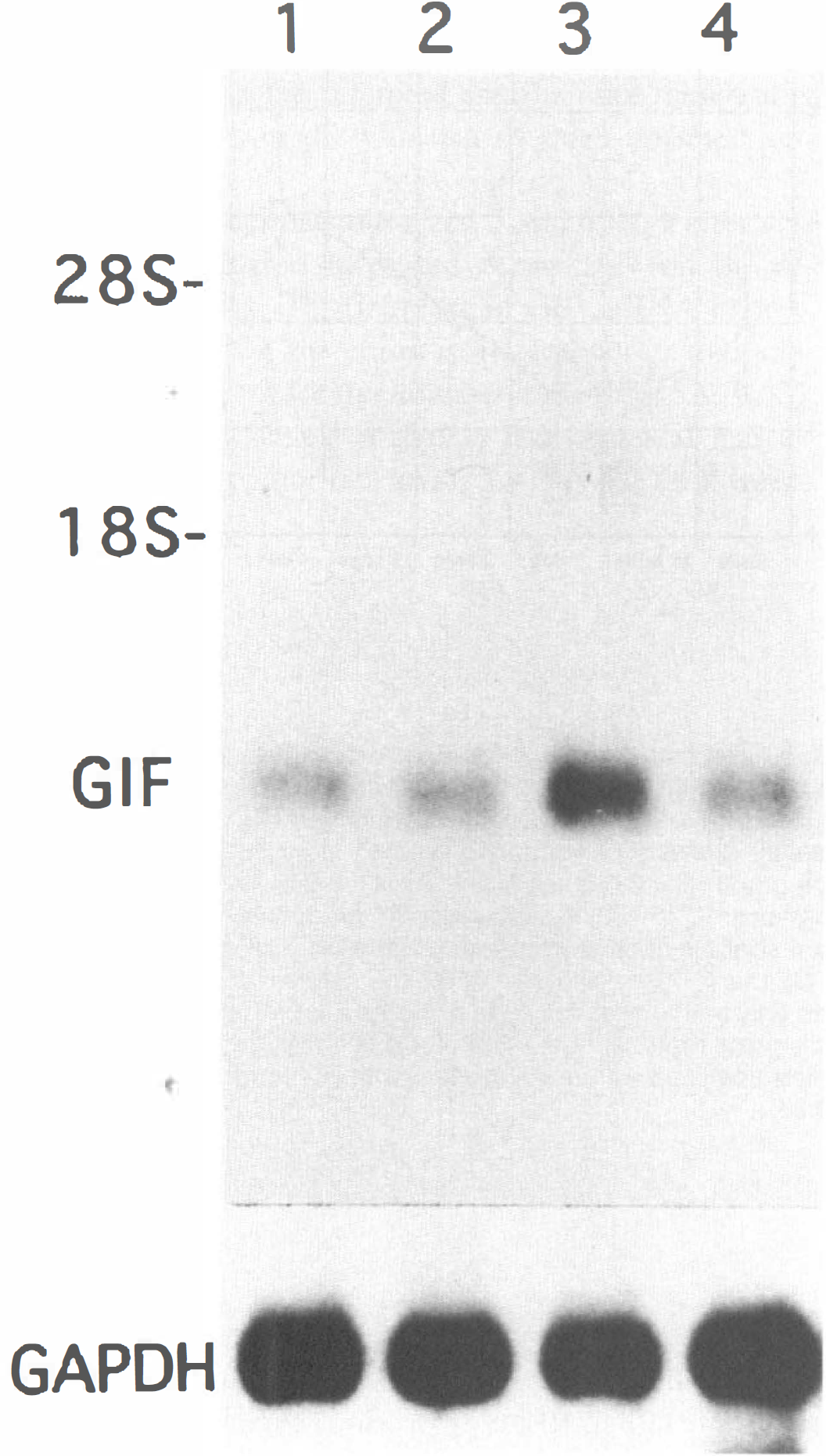
Northern blot of total RNA (15 μg per lane) from the cortices of rats with MCA occlusion using a specific probe for rat GIF. Samples were obtained and pooled from three animals. Lane 1: ipsilateral cortex one day after MCA occlusion; lane 2: contralateral cortex one day after MCA occlusion; lane 3: ipsilateral cortex four days after MCA occlusion; lane 4; contralateral cortex four days after MCA occlusion. Single band was obtained with the GIF probe. Densitometry revealed that the ratio of ipsilateral to contralateral GIF mRNA corrected with GAPDH mRNA was 0.72 for 1 day and 3.94 for 4 days after MCA occlusion respectively.
To define the cellular origin of GIF, we used digoxigenin-labeled probes, as the in situ hybridization with these can provide better microscopical orientation than that with radioactive probes. In the contralateral cortex, GIF mRNA signals were observed mainly on neurons (Fig. 7A). Four days after surgery, the density of the GIF mRNA-positive cells had increased in the ipsilateral cortex (Fig. 7B). Growth inhibitory factor mRNA is localized on the large cells corresponding neurons in higher magnification (Fig. 7C). Two weeks after surgery, the signals had increased in the small cells adjacent to the infarction (Fig. 7D). These cells are presumably astrocytes when we compare the hybridized sections with the adjacent ones stained for GFAP.
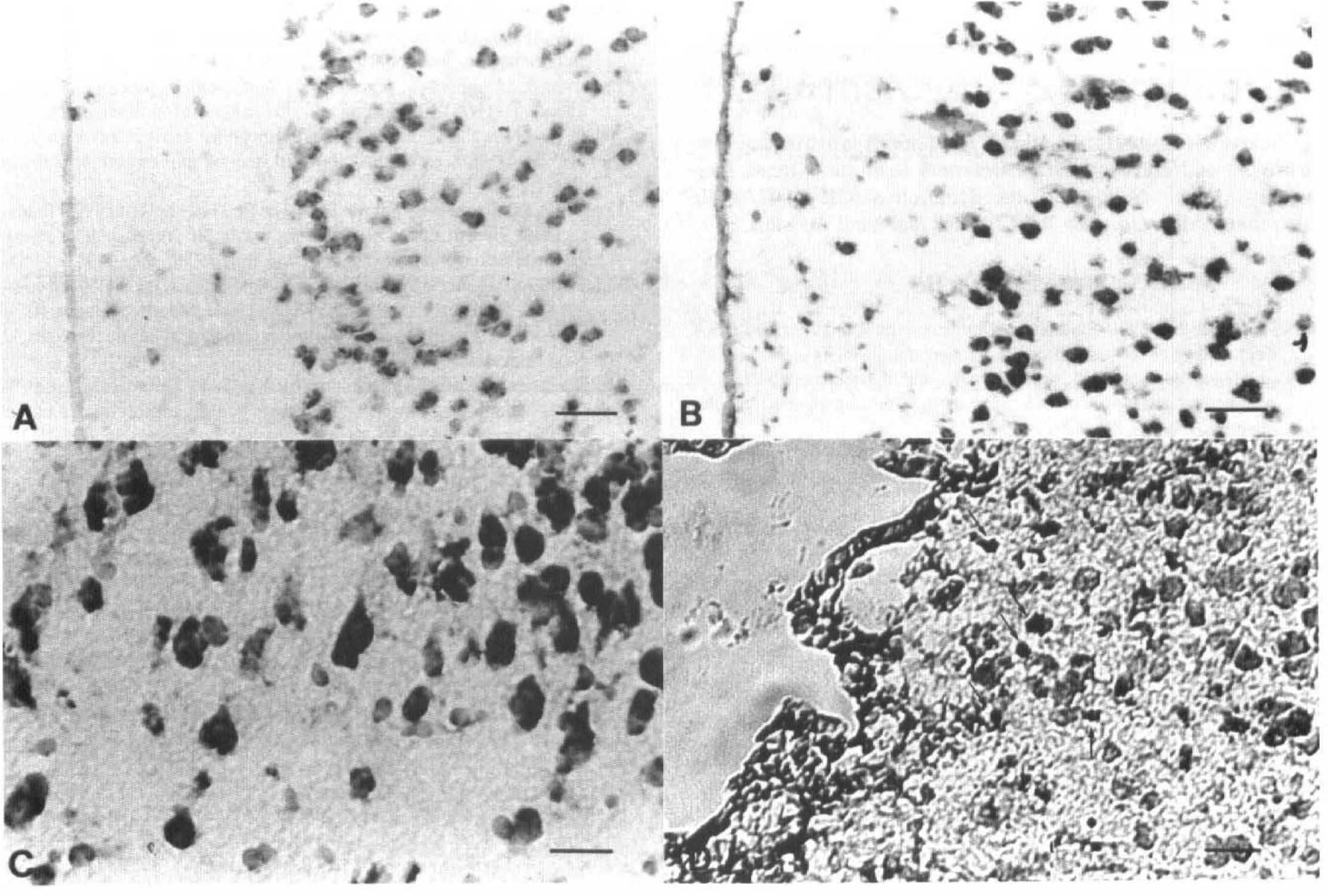
Light-field microphotograms of in situ hybridization for GIF mRNA with digoxigenin-labeled probe (
DISCUSSION
We identified the time course and regional expression of GIF mRNA in the rat brain after MCA occlusion. Three distinct patterns in space distribution were observed in GIF mRNA expression. First, one day after MCA occlusion, the expression was suppressed slightly in the cerebral hemisphere of the occlusion side. Second, the expression increased 3 and 4 days after occlusion throughout the ipsilateral cerebral cortex, white matter, and caudate putamen except for the central area of the MCA territory. Third, 2 weeks after MCA occlusion, the expression increased only in the region adjacent to the infarction.
The mechanism by which focal ischemia triggers an extensive increase in GIF mRNA expression is not yet clarified. Regional expression of c-fos mRNA, an immediate early gene, after focal ischemia has been reported. Expression of c-fos mRNA was increased by 15 minutes after MCA occlusion and continued to increase at 1 and 3 hours throughout the ipsilateral cerebral cortex (Welsh et al., 1992). We identified the same pattern of c-fos expression after unilateral cortical ablation (Kohmura et al., 1995). Spreading depression has been suggested to be one of the key mechanisms for this induction of c-fos mRNA (Dragunow et al., 1990; Kohmura et al., 1995). The induction of immediate early genes and their gene products should be followed by further responses of late gene. Since the pattern for the increase in GIF mRNA expression in the cerebral cortex 4 days after MCA occlusion resembled somewhat that for c-fos mRNA, it is possible that the same signal transduction mechanism may be involved in GIF mRNA expression after focal ischemia.
Growth inhibitory factor inhibits neuronal survival and neurite formation (Tsuji et al., 1992; Uchida et al., 1991). Neurons have the essential capacity to elongate neurites. However, if the neurite extension is uncontrolled, the nervous system may become chaotic. Therefore, continuous inhibition of neurons is indispensable. Growth inhibitory factor may act as an inhibitory factor in the brain. The present study showed that occlusion of the MCA suppressed expression of GIF mRNA on day 1. This result can be interpreted as follows: the original inhibition of neurite elongation is eliminated in the ipsilateral hemisphere after MCA occlusion so that neurons begin to sprout. However, this suppression of GIF mRNA expression was restricted to only one day after ischemia and was followed by a transient increase. This may reflect the general concept that neurons in the central nervous system do not have any regenerative capacity. In contrast, changes in GIF mRNA expression are quite different in the peripheral nervous system after injury, where regenerative capacity is well known. We previously studied the expression of GIF mRNA in the facial nucleus after facial nerve transection and found that this expression was dramatically suppressed after nerve transection. The suppression continued for up to 5 weeks (Yuguchi et al., 1995). Thus, in the peripheral nervous system, neurite extension can be facilitated continuously for purposes of regeneration.
As mentioned earlier, GIF is a member of the metallothionein family and identical to MT-III (Palmiter et al., 1992). Growth inhibitory factor is abundantly present in the normal brain (Uchida et al., 1991), so that GIF may play an important role as a methallothionein in the brain. Numerous studies have examined MT gene expression in the liver, in which MT plays important roles in the homeostasis of essential metal ions, such as copper and zinc (Cousins et al., 1985), detoxication of heavy metals, such as cadmium and mercury, and the scavenging of free radicals to protect against oxidative stress (Abel et al., 1989; Thornalley et al., 1985). In the present study, we found a transient increase in GIF mRNA expression on the affected side cerebral cortex on days 3 and 4. Besides inhibiting effect on the excessive sprouting of neurites, GIF might be involved in protecting neurons against ischemic insults by scavenging free radicals (Abel, 1989; Thornalley et al., 1985).
Another consideration about the meaning of changes in GIF is its possible function in relation to zinc. Zinc is an abundant cation in the brain with a specific distribution pattern (Frederickson et al., 1989), and is supposed to affect synaptic transmission in certain areas of the central nervous system. In the areas containing high contents of zinc, such as the retina, the pineal gland, and the hippocampus, unique isoforms of MT are synthesized on a continuous basis (Ebadi et al., 1995). Zinc ion is contained in synaptic vesicles and released into the synaptic cleft during excitation (Frederickson et al., 1989). Growth inhibitory factor (MT-III) seems to coordinate zinc, copper, and cadmium with the same stoichiometry and affinity as other MT (Masters et al., 1994). Growth inhibitory factor may therefore play an important role in maintaining the homeostasis of zinc in the brain and thereby regulate synaptic transmission. After MCA occlusion, synaptic transmission in the cerebral cortex should be altered. An alteration in the expression of GIF mRNA after MCA occlusion may therefore reflect a change in synaptic transmission. More recently, it is reported that excessive exposure to extracellular zinc can damage central neurons (Koh et al, 1996). Growth inhibitory factor might also be involved in protecting neurons against ischemic insults by reducing extracellular zinc.
Two weeks after surgery, GIF mRNA increased again, but the region was limited to the peri-infarcted area. Many GFAP-positive cells can be observed in this area with GFAP immunohistochemistry. Therefore, this late response may be related to reactive gliosis, and the expression of GIF in the nonneuronal cells may contribute to the regulation of DNA transcription, because MT adjusts the amount of zinc available for zinc-dependent transcription factors.
On the basis of these results, it is suggested that GIF can be assumed to play important roles in maintaining the central nervous system and to respond to ischemic insult in order to protect the central nervous system. Other factors having inhibitory activities should be also considered. It has been known that central nervous system myelin has contact-dependent growth inhibitory activity and the inhibitory protein is characterized by McKerracher et al. (McKerracher et al., 1994). The identified myelin-associated glycoprotein strongly inhibits neurite outgrowth from both developing cerebellar and adult dorsal root ganglion neurons in vitro. This inhibition is reversed by an anti-myelin-associated glycoprotein antibody (Mukhopadhyay et al., 1994). In contrast, myelin-associated glycoprotein promotes neurite outgrowth from newborn DRG neurons. These results suggest that myelin-associated glycoprotein may be responsible, in part, for the lack of central nervous system nerve regeneration in vivo and may influence, both temporally and spatially, regeneration in the peripheral nervous system. Myelin-associated glycoprotein can be, of course, an additional major inhibitory factor within the central nervous system. The role of myelin-associated glycoprotein after cerebral ischemia should be also studied in future.
Footnotes
Abbreviations used
Acknowledgements
We thank Dr. Shoji Tsuji from the Department of Neurology, Brain Research Institute, Niigata University Medical School, for the donation of GIF cDNA. We also thank Miss Chiyako Imai for her technical assistance.
