Abstract
Neuronal thread proteins (NTP) are a family of phosphoproteins expressed during neuritic sprouting. The 15 to 18 kD NTP cluster is associated with development and neuronal differentiation, whereas the 21 kD and 39 to 42 kD species are overexpressed in Alzheimer's disease, correlating with neurodegenerative sprouting and synaptic disconnection. Empirical observations suggested that NTP might also be modulated with central nervous system injury and stroke. In this study of both human and experimental (rat) focal cerebral infarcts, in situ hybridization and immunocytochemical staining revealed NTP gene expression up-regulated in perifocal neurons. These findings were confirmed by quantitative Northern and Western blot analyses. Moreover, Western blot analysis demonstrated selectively increased expression of the 15 to 18 kD NTP species during the acute, subacute, and healing phases of cerebral infarction in both humans and experimental animals, corresponding with the expected period of neuronal repair. These results suggest an additional role for the 15 to 18 kD NTP species in neuritic sprouting required for neuronal regeneration after injury in the mature central nervous system.
Keywords
Neuronal thread proteins (NTP) are a family of molecules expressed in brain (Ozturk et al., 1989; de la Monte, et al., 1992; de la Monte and Wands, 1992; de la Monte, et al., 1992) and neuroectodermal tumor cell lines (Xu et al., 1993; Xu et al., 1995). Neuronal thread protein expression is increased in neuronal cells during proliferation (Xu et al., 1993), differentiation (Xu et al., 1995; de la Monte et al., 1996), brain development (de la Monte et al., 1996a), and Alzheimer's disease (AD) neurodegeneration (Ozturk et al., 1989; de la Monte, et al., 1992; de la Monte and Wands, 1992; de la Monte, et al., 1992). In vitro studies showed NTP up-regulation, phosphorylation, and translocation from the perikarya to cell processes and growth cones with growth factor-induced neuritic sprouting and neuronal differentiation (Xu et al., 1995; de la Monte et al., 1996a; de la Monte et al., 1996b). In contrast, low-level NTP expression is a feature of normal mature brains, and confluent or serum-starved cultures with absent DNA synthesis (de la Monte et al., 1992; Xu et al., 1993; de la Monte et al., 1996b). Therefore, NTP expression is modulated with cell proliferation and neuritic growth. In the adult brain, NTP may be functionally important for synaptic plasticity and neuronal repair.
In neuroectodermal cells, six distinct NTP-immunoreactive molecules have been identified (42 kD, 39 kD, 26 kD, 21 kD, 17 to 18 kD, and 15 kD, respectively) in immunoprecipitation studies, quantitative immunoradiometric assays, and Western blot analyses (Xu et al., 1993; Xu et al 1995; de la Monte et al., 1996b). However, metabolic labeling studies showed that the 42 kD, 26 kD, and 17 to 18 kD NTP molecules represent phosphorylated forms of the 39 kD, 21 kD, and 15 kD NTP species, respectively, and that the 21 kD NTP species may be derived from a precursor molecule, possibly the 39 kD species (Xu et al., 1995; de la Monte et al., 1996b). The 39 kD and its phosphorylated 42 kD form are detectable in all neuronal cells. The 21 kD and its phosphorylated 26 kD form are expressed in the mature brain (de la Monte et al., 1996a). The 15 kD and its 17 to 18 kD phosphorylated form are abundantly expressed in the developing brain (de la Monte et al., 1996a) and in undifferentiated primitive neuroectodermal tumor cells (Xu et al., 1993; Xu et al., 1995). The 21 kD and 39 to 42 kD NTP species are overexpressed and accumulated in brains with AD (de la Monte and Wands 1992; de la Monte et al., 1992; de la Monte, et al., 1996c). Alzheimer's disease dementia is correlated with widespread cortical neuritic sprouting (Ihara 1988; Kowall and Kosik 1987; Tabaron et al., 1989; Scheibel and Tomiyasu 1978; Masliah et al., 1991; de la Monte and Bloch 1997), and progressive accumulation of NTP (de la Monte et al., 1992), suggesting that increased NTP expression in AD reflects neuronal loss and synaptic disconnection.
Our working hypothesis is that both the 15 to 18 kD and 21 to 29 kD NTP clusters are up-regulated during neuritic sprouting, with the former linked to growth of immature neuronal cells, and the latter to synaptic plasticity in the mature brain. Previous studies showed that immature and undifferentiated neuronal cells express predominantly 15 to 18 kD NTP, but with growth factor-induced differentiation and brain maturation, the most abundant forms of NTP detected are 21 to 29 kD (Xu et al., 1995; de la Monte, et al., 1996a; de la Monte et al., 1996b). In the adult brain and differentiated quiescent neurons, NTP expression is low-level, correlating with markedly reduced synaptic turnover and requirements for growth. However, pilot studies showed striking modulation of NTP after acute injury in the mature central nervous system (de la Monte et al., 1990). In addition, in brains with AD plus cerebral infarcts, we detected high levels of both the 21 kD and 17 to 18 kD NTP species by Western blot analysis (de la Monte SM and Wands JR, unpublished). To examine its potential role in central nervous system injury, we characterized NTP gene expression during different phases of spontaneous (human) and experimental (rat) focal cerebral infarction using brain-specific nucleic acid and antibody probes generated with the recently isolated AD7c-NTP cDNA (de la Monte and Wands 1993; de la Monte et al., 1995a) and the corresponding recombinant fusion protein (de la Monte, et al., 1996c). This study shows increased NTP expression in histologically intact neurons distributed in peri-infarction zones, and selective up-regulation of the 18 kD NTP species with a time course corresponding to the expected period of regenerative sprouting.
METHODS
Source of tissue
Nondemented control brains with incidental cerebral infarcts were identified in the database of the Alzheimer's Disease Research Center Brain Bank at the Massachusetts General Hospital. As dictated by the protocol at the Alzheimer's Disease Research Center Brain Bank at the Massachusetts General Hospital, 42 standardized blocks (2 × 2 × 0.5 cm) were dissected and then bisected with adjacent structures snap frozen in isopentane cooled with dry ice, or fixed in 10% neutral buffered formalin (de la Monte, 1989). The specimens used in this study included paired fixed and frozen blocks of cerebral cortex and underlying white matter with acute (<5 days), subacute (7 to 14 days), healing (2 to 4 weeks), or remote (>6 months) infarcts, together with the adjacent uninvolved tissue, and samples of intact cerebral tissue distant from the infarcts. Previous studies revealed similar levels of NTP expression in different regions of control neocortex (de la Monte, et al, 1992). Formalin-fixed paraffin-embedded sections were used for in situ hybridization and immunocytochemical staining studies. Frozen tissue blocks (stored at −80°C) were used to extract protein for Western blot analysis. All brains were harvested within 18 hours of death.
Experimental stroke model
An experimental middle cerebral artery (MCA) focal stroke model (Garcia et al., 1993; Fisher et al., 1995) was purchased from Zivic-Miller Laboratories, Inc (Zelienople, PA, U.S.A.). Unilateral permanent occlusion of an MCA was accomplished by the intraluminal thread technique (Fisher et al., 1995), in anesthetized adult male Sprague-Dawley rats (250 to 300 g). Briefly, after ligating the external carotid and occipital arteries, the internal carotid artery was isolated, and a 4-0 nylon monofilament with a rounded tip was introduced into the external carotid artery, and advanced into the internal carotid artery lumen to the bifurcation and origin of the middle cerebral artery (Kawamura et al., 1991). The catheter tip was advanced to the ostium of the MCA, occluding blood flow from the anterior cerebral artery and internal carotid artery. The uninfarcted hemispheres and sham-operated brains (N = 3) served as controls. Brains were harvested 1, 2, 3, and 7 days after surgery. Fresh frozen cerebral hemispheres were processed in pairs for Northern and Western blot analyses. Immersion fixed (Histochoice, Armresco, Solon, OH, U.S.A.) whole brains sectioned in the coronal plane along standardized landmarks were embedded in paraffin and used for immunocytochemical staining and in situ hybridization studies. In pilot studies, we observed no difference in the quality of the in situ hybridization signals or immunocytochemical staining results using immersion fixed compared with perfusion fixed cerebral tissue.
Nucleic acid AD7c-neuronal thread protein probes
In situ hybridization studies were performed with antisense and sense (negative control) cRNA probes labeled with [11-digoxigenin]UTP (Boehringer-Mannheim, Indianapolis, IN, U.S.A.) using SP6 or T7 DNA-dependent RNA polymerase (Melton et al., 1984). Probes were generated using the AD7c-NTP cDNA (de la Monte and Wands 1993; de la Monte et al, 1995a) subcloned into a pGEM7 vector (Promega, Madison, WI, U.S.A.) and linearized in both orientations using either Xho1 or Kpn1 restriction endonuclease. Northern blot analysis was performed using [α-32P]dCTP-labeled AD7c-NTP cDNA probes (specific activity of 108 cpm/µg DNA) generated with random hexamer primers (Feinberg and Vogelstein 1983; Ausubel et al., 1994) and Klenow large fragment DNA polymerase (Amersham, Arlington Heights, IL).
Northern blot analysis
RNA was extracted from snap-frozen samples of brain by the Chomczynski-Sacchi single-step RNA isolation method (Chomczynski and Sacci, 1987) using a commercially prepared acid guanidinium thiocyanate-phenol solution (Trizol reagent; Gibco-BRL, Grand Island, NY, U.S.A.). Samples containing 15 µg of total RNA were fractionated in 1.2% agarose/3% formaldehyde gels (Ausubel et al., 1994), and transferred to Hybond-N Plus membrane (Amersham, Arlington Heights, IL, U.S.A.). The membranes were hybridized for 8 hours at 42°C with 106 dpm/mL of [32P]dCTP-labeled AD7c-NTP cDNA probe (de la Monte and Wands 1993; de la Monte et al, 1995) in buffer containing 50% formamide, 5× SSPE, 1 mmol/L sodium pyrophosphate, 5× Denhardt's solution, 0.5% SDS, and 200 µg/mL sheared denatured salmon sperm DNA (Ausubel et al., 1994; Sambrook et al., 1989). The membranes were washed in stepwise dilutions of 20× SSPE containing 0.5% SDS (Ausubel et al., Sambrook et al., 1989). The final wash was in 0.1× SSPE/0.5% SDS at 65°C. To evaluate RNA content in each sample, the blots were stripped of probe and rehybridized with an excess of [32P]-γATP end-labeled synthetic 30mer corresponding to 18S ribosomal RNA (Lee et al., 1988). Nonsaturated autoradiograms were subjected to volume densitometric scanning to quantify levels of AD7c-NTP mRNA relative to 18S rRNA using ImageQuant software (Molecular Dynamics, Inc., Sunnyvale, CA, U.S.A.). To normalize the results from two distinct but identical experiments, the percentage differences in the levels of AD7c-NTP mRNA between the infarcted and uninfarcted hemispheres were calculated. The data were analyzed using Mann Whitney U-tests (t tests), two-sample sign tests for equal medians (to determine if the median of the difference between the two groups was zero), and Wilcoxon matched pairs signed rank tests (to test the difference between the paired groups).
In situ hybridization
Histological sections (8-µm thick) were dewaxed, rehydrated through graded alcohol solutions, digested with 0.2 N HCl, and acetylated with 0.025% acetic anhydride in 0.1 mol/L triethanolamine, pH 8.0 (de la Monte et al., 1995b). RNA was denatured in 50% formamide/2× SSC at 50°C, and hybridized for 12 hours at 50°C with 250 ng/mL antisense or sense [11-digoxigenin]UTP-labeled AD7c-NTP cRNA (Melton et al., 1984) probe (10 µL/cm2) in buffer containing 50% formamide, 4× SSPE, 10 mmol/L sodium pyrophosphate, 1× Denhardt's solution, 200 mmol/L DTT, 300 µg/mL denatured salmon sperm DNA, 100 µg/mL tRNA, 10% PEG 6000, and 1% SDS (de la Monte et al., 1995b). After extensive posthybridization washes, unhybridized probe was digested with 50 µg/mL RNAse A in 0.5 mol/L NaCl, 10 mmol/L Tris, 1 mmol/L EDTA, pH 8.0. The final wash was with 0.2× SSPE/0.5% Triton X-100 at 55°C for 30 minutes. Hybridized probes were detected with alkaline phosphatase-conjugated sheep anti-digoxigenin F(ab′)2 (Boehringer Mannheim, Inc., Indianapolis, IN, U.S.A.), and antibody binding was revealed with X-phosphate/5-bromo-4-chloro-3-indolyl-phosphate/nitro-blue-tetrazolium-chloride (BCIP/NBT). The sections were lightly counterstained with hematoxylin and preserved under cover-glass. The specificity of probe hybridization in tissue sections was confirmed by Northern blot analysis using identically prepared cRNA probes labeled with [32P]UTP. All specimens from each experiment were processed simultaneously and with the same reagents.
Recombinant AD7c-NTP
The AD7c-NTP cDNA was subcloned into a pTrcHis expression vector (InVitrogen Corp., San Diego, CA, U.S.A.). Recombinant protein (∼39 to 41 kD) generated in Top10 E. coli cells were affinity purified by nickel chelate chromatography (Ausubel et al., 1994). AD7c-NTP protein, cleaved from the 6-histidine fusion partner, was used to immunize rabbits to generate polyclonal antibodies (Harlow and Lane 1988). Western blot analysis showed cross-immunoreactivity of polyclonal, antihuman pancreatic thread protein (Gross et al., 1985; Gross et al., 1986) to recombinant AD7c-NTP, and absent binding with nonrelevant polyclonal anti-GAP-43 (Karns et al., 1987) (data not shown).
Generation of polyclonal antibodies
Rabbits were immunized by intramuscular injections of 0.5 mg of purified recombinant AD7c-NTP protein emulsified with complete Freund's adjuvant. Booster injections (0.25 mg) of AD7c-NTP protein emulsified in incomplete Freund's adjuvant were performed 2 and 4 weeks later, and immune sera were obtained 7 days after the final injection. Immunoglobulin fractions precipitated with ammonium sulfate and dialyzed against phosphate-buffered saline (Harlow and Lane 1988), were analyzed by Western blot analysis and immunocytochemical staining. The specificity of immunoreactivity for AD7c-NTP recombinant protein was demonstrated by the following: 1) the absence of preimmune sera binding to brain by Western blot analysis or immunocytochemistry; 2) the detection of ∼39 to 42 kD and 21 kD molecules in human brain homogenates, 14 kD and 17 kD cross-reactive species in human pancreas, and purified native PTP (Gross et al., 1985; Gross et al., 1986) by Western blot analysis; and 3) the absence of immunoreactivity in nonrelevant tissues, e.g. spleen, lung, thymus, and skeletal muscle (data not shown).
Western blot analysis
Samples containing 60 µg of cytosolic protein (Lowry et al., 1951) extracted from frozen tissue (Xu et al., 1995) were fractionated 15% SDS-PAGE gels, transferred to Immobilon-P membranes (Xu et al., 1995), and subjected to Western blot analysis (Ausubel et al., 1994; Sambrook et al., 1989; Harlow and Lane 1988) using rabbit polyclonal anti-AD7c-NTP diluted 1:400 in phosphate-buffered saline containing 0.5% BSA and 0.2% Tween-20. Antibody binding was detected with horseradish peroxidase-conjugated secondary antibody and enhanced chemiluminescence reagents (Amersham Life Science, Inc., Arlington Heights, IL, U.S.A.) according to the manufacturer's protocol. The levels of anti-AD7c-NTP immunoreactive protein were assessed by densitometric analysis of nonsaturated autoradiograms (ImageQuant, Molecular Dynamics, Inc., Sunnyvale, CA, U.S.A.). To normalize the results from two distinct but identical experiments, the percentage differences in the levels of AD7c-NTP protein between the infarcted and uninfarcted hemispheres were calculated. The data were analyzed using Mann-Whitney U-tests (t-test), two-sample sign tests for equal medians (to determine if the median of the difference between the two groups was zero), and Wilcoxon matched pairs tests (to test the difference between the paired groups).
Immunocytochemical staining
Dewaxed, rehydrated paraffin-embedded sections adjacent to those used for in situ hybridization were immunostained with rabbit polyclonal anti-AD7c-NTP diluted 1:800 in phosphate-buffered saline containing 1% bovine serum albumin. Antibody binding was detected by the avidin-biotin horseradish peroxidase method (Vector ABC Elite Kit; Vector Laboratories, Burlingame, CA) following the manufacturer's protocol, and with 3-3′ diaminobenzidine as the chromogen. The sections were counterstained with hematoxylin, dehydrated, cleared, and preserved under coverglass. Adjacent sections were immunostained with monoclonal antibody to glial fibrillary acidic protein and polyclonal antibody to the growth-associated protein, GAP-43 as positive controls, and with 5C3 monoclonal antibody to Hepatitis B surface antigen virus (Wands and Zurawski 1981), or with preimmune rabbit serum as negative controls. All specimens from each experiment were immunostained simultaneously and with the same immunodetection reagents.
RESULTS
Human studies
Patient profiles.
Among the 37 brains studied, 32 had focal cerebral infarcts and 5 were controls. There were 21 (57%) men, aged from 36 to 91 years old, and the mean age was 76.4 ± 7.9 years (mean ± SD). Thirteen brains contained solitary cerebral infarcts, and 14 contained between 2 and 11 infarcts of different ages (acute-hours to days, subacute- 1 to 2 weeks, healing-several weeks to months old, or remote->6 months). Paraffin-embedded sections of all lesions and surrounding tissue were studied by in situ hybridization and immunocytochemical staining to assess NTP gene expression. In addition, frozen tissue samples immediately adjacent to the formalin-fixed blocks were available from 14 brains, including a total of 32 blocks: 4 control; and 28 infarcts (6 acute; 8 subacute; 10 resolving; and 4 remote).
Modulation of neuronal thread protein expression shown by in situ hybridization.
AD7c-NTP mRNA transcripts were detected in neurons. In tissue surrounding acute and subacute cerebral infarcts, histologically intact neurons manifested high levels of AD7c-NTP mRNA (Fig. 1A, C), whereas regions distant from the infarcts and uninfarcted control brains showed low levels of AD7c-NTP mRNA (Fig. 1B, D). With increasing age of the infarcts, AD7c-NTP mRNA expression in peri-infarct tissue declined relative to the peak levels associated with acute lesions. In older, nearly resolved infarcts, the levels of AD7c-NTP mRNA expression were still slightly increased above normal. In remote infarcts associated with neuronal loss and fibrillary gliosis, NTP mRNA expression was low-level or undetectable both in the peri-infarct zones and within the lesions (data not shown).
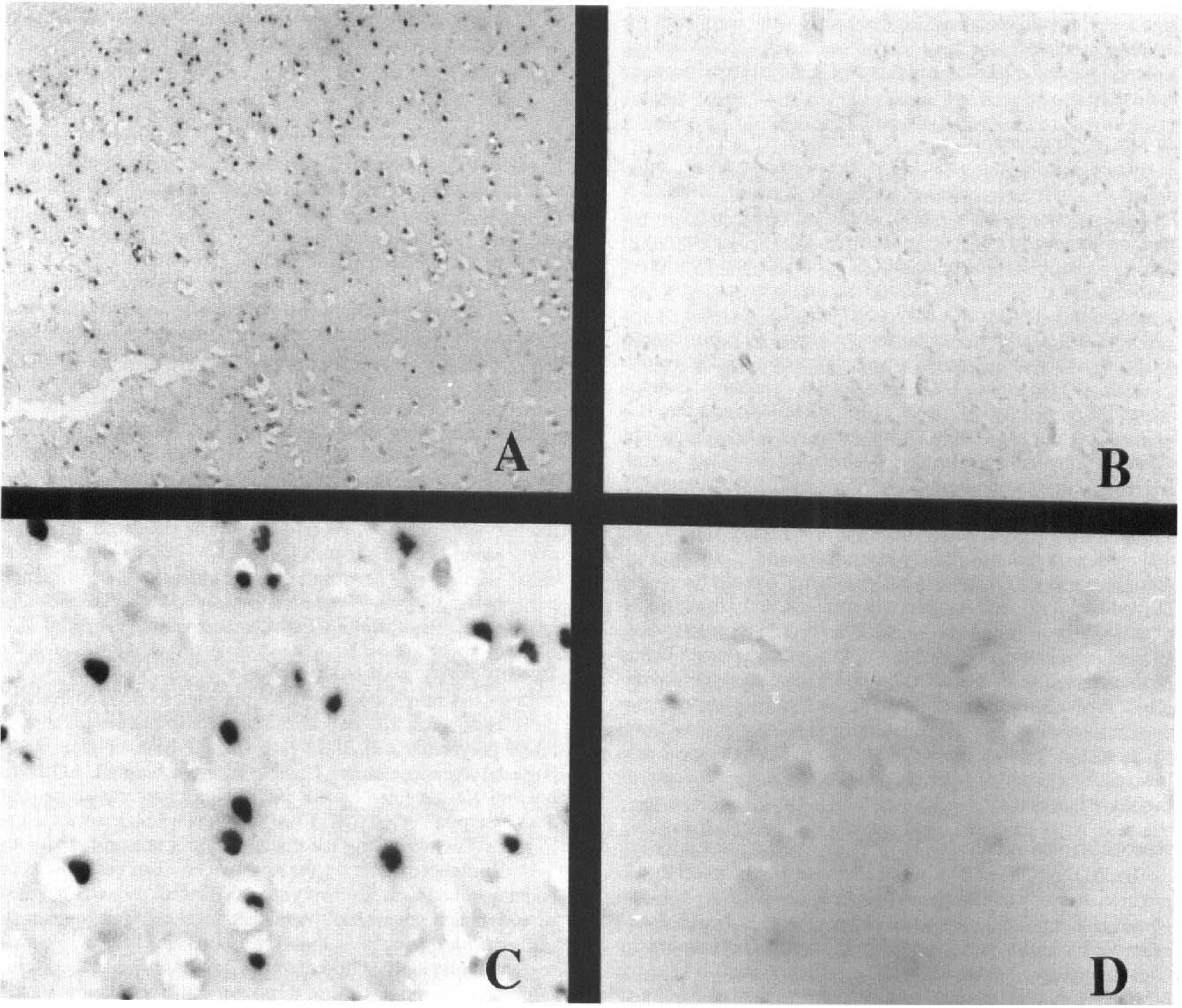
Modulation of neuronal thread protein (NTP) mRNA after cerebral infarction shown by in situ hybridization analysis of human brain. Cerebral cortex adjacent to 3 day (
Modulation of neuronal thread protein expression demonstrated by immunocytochemical staining.
In acutely infarcted cerebral tissue, AD7c-NTP immunoreactivity was low-level (Fig 2A), but in histologically intact tissue adjacent to the infarcts, AD7c-NTP immunoreactivity was strikingly increased in neuronal perikarya, neuropil fibers (Fig. 2B), and white matter fibers, relative to control tissue (Fig. 2C). The diffuse nature of the AD7c-NTP immunoreactivity in neuropil fibers precluded their further characterization as axonal, dendritic, or glial cell in origin. Acutely ischemic neurons (atrophic with condensed chromatin and eosinophilic cytoplasm, but distinct nuclear margins and cellular detail by hematoxylin and eosin staining) also showed increased AD7c-NTP immunoreactivity, but necrotic (dark, shrunken, and pyknotic) and ghost (pale, loss of nuclear margins and detail, chromatolysis) neurons did not (Fig 2A). Increased AD7c-NTP expression persisted throughout the subacute phases (2 to 4 weeks) of infarction. Infarct-associated up-regulation of NTP gradually declined, and in remote infarcts (months to years old), AD7c-NTP expression was either low-level or undetectable in neurons, white matter fibers, or within the glial scar, corresponding with the in situ hybridization results.
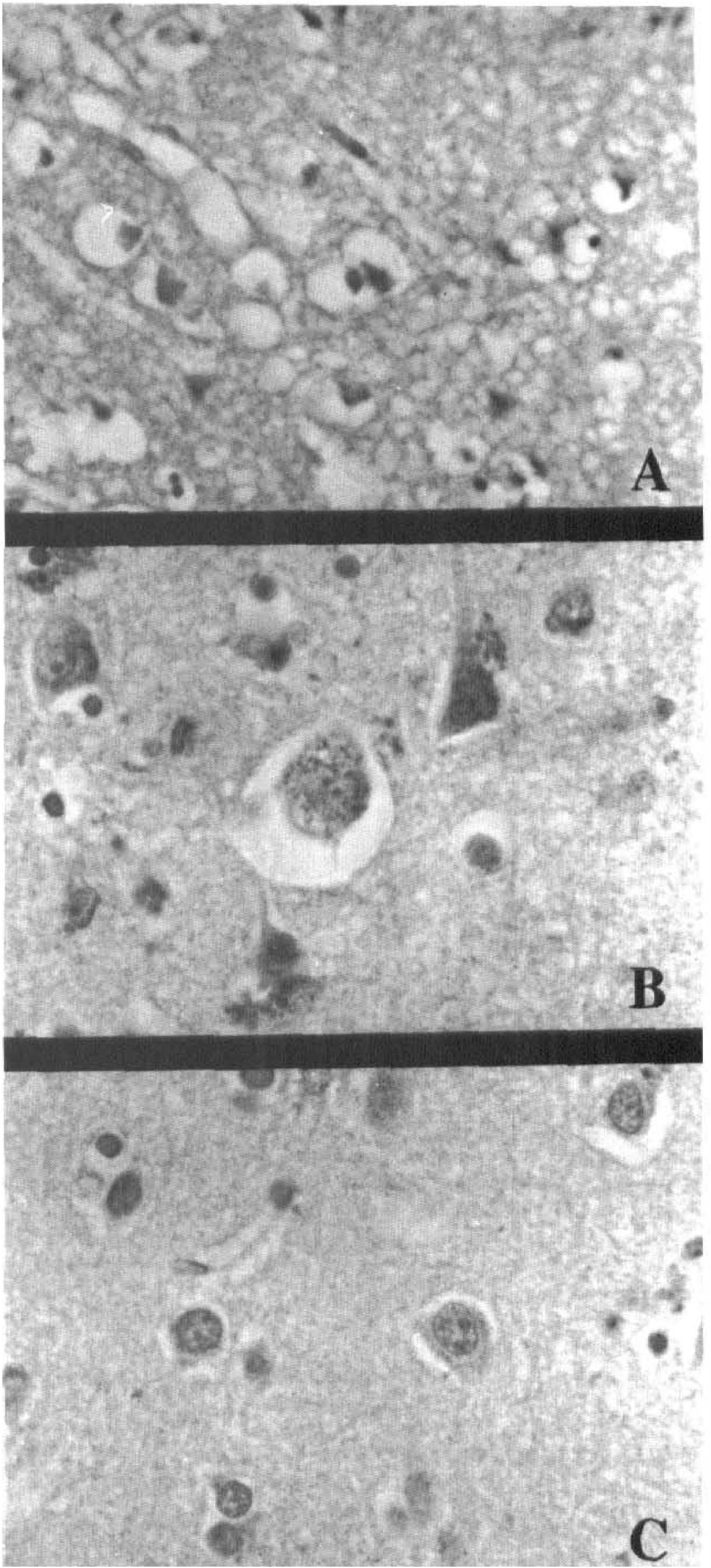
Immunocytochemical staining of human brain showing changes in AD7c-NTP expression with evolution of cerebral infarction. (
Modulation of neuronal thread proteins with cerebral infarction shown by quantitative Western blot analysis.
Western blot analysis revealed expression of 5 NTP species using AD7c-NTP polyclonal antibodies 17 to 18 kD, 21 kD, 26 kD, 39 kD, and 42 kD. Densitometric analysis of the autoradiograms revealed significant modulation of the 17 to 18 kD AD7c-NTP species in association with cerebral infarction (Fig. 3A, C). In contrast, expression of the 21 kD through 42 kD NTP species was not substantially increased by cerebral infarction, and in general, the differences between the intact and infarcted samples were less than 10%. Analysis of multiple specimens from brains with different age infarcts revealed higher levels of the 17 to 18 kD NTP species in acute or subacute peri-infarct tissue compared with remote perifocal lesions, intact tissue distant from lesions, and normal control cerebral tissue (Figs. 3B, C). The highest levels of NTP expression were observed with subacute cerebral infarcts (Fig. 3C).
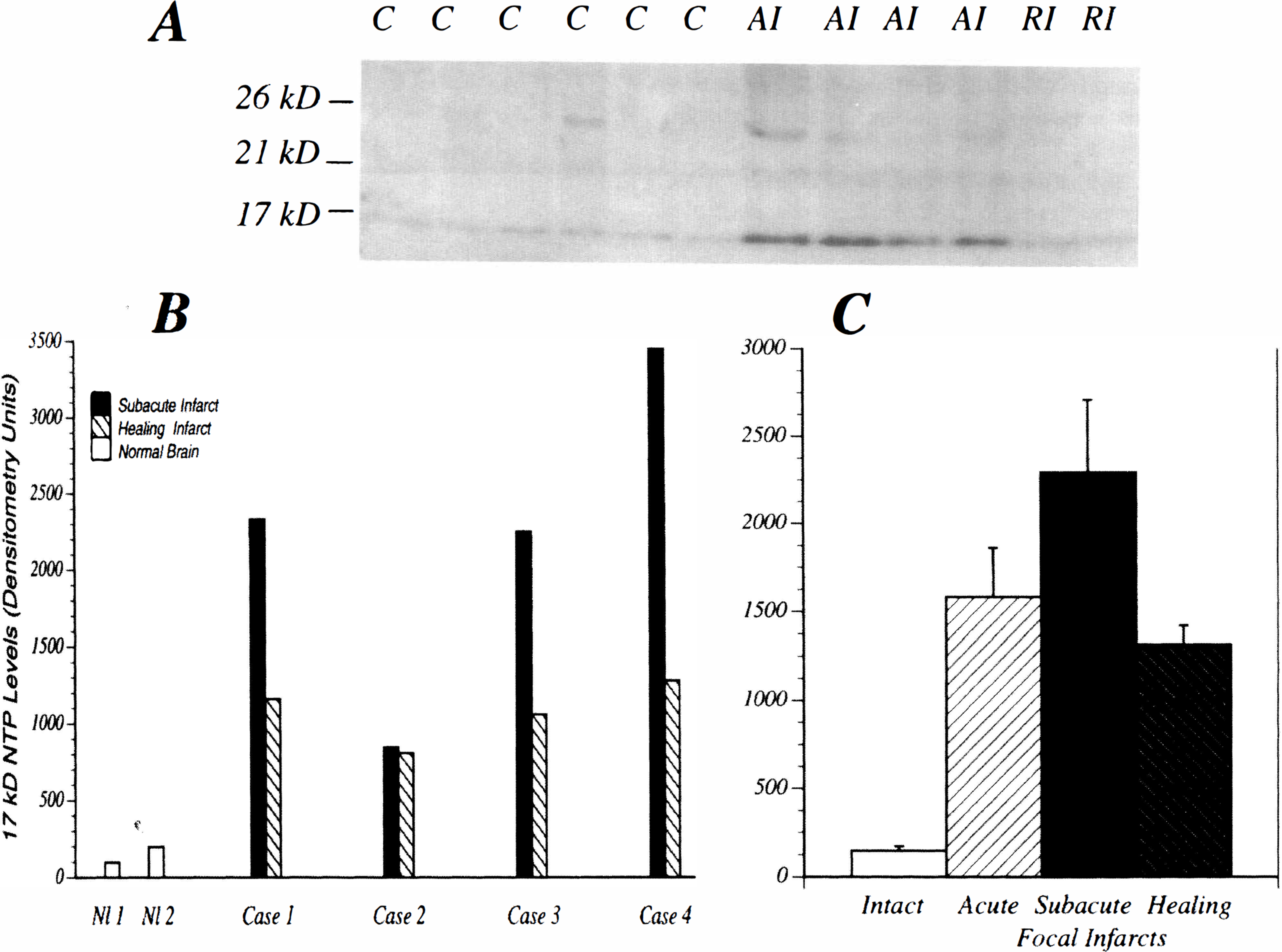
Western blot analysis and quantitative densitometric assessment of AD7c-NTP expression in intact control [C], acutely infarcted [AI], or remotely infarcted [RI] human cerebral tissue (
Experimental focal stroke model
Increased expression of a 0.8 kB neuronal thread proteins mRNA after cerebral infarction.
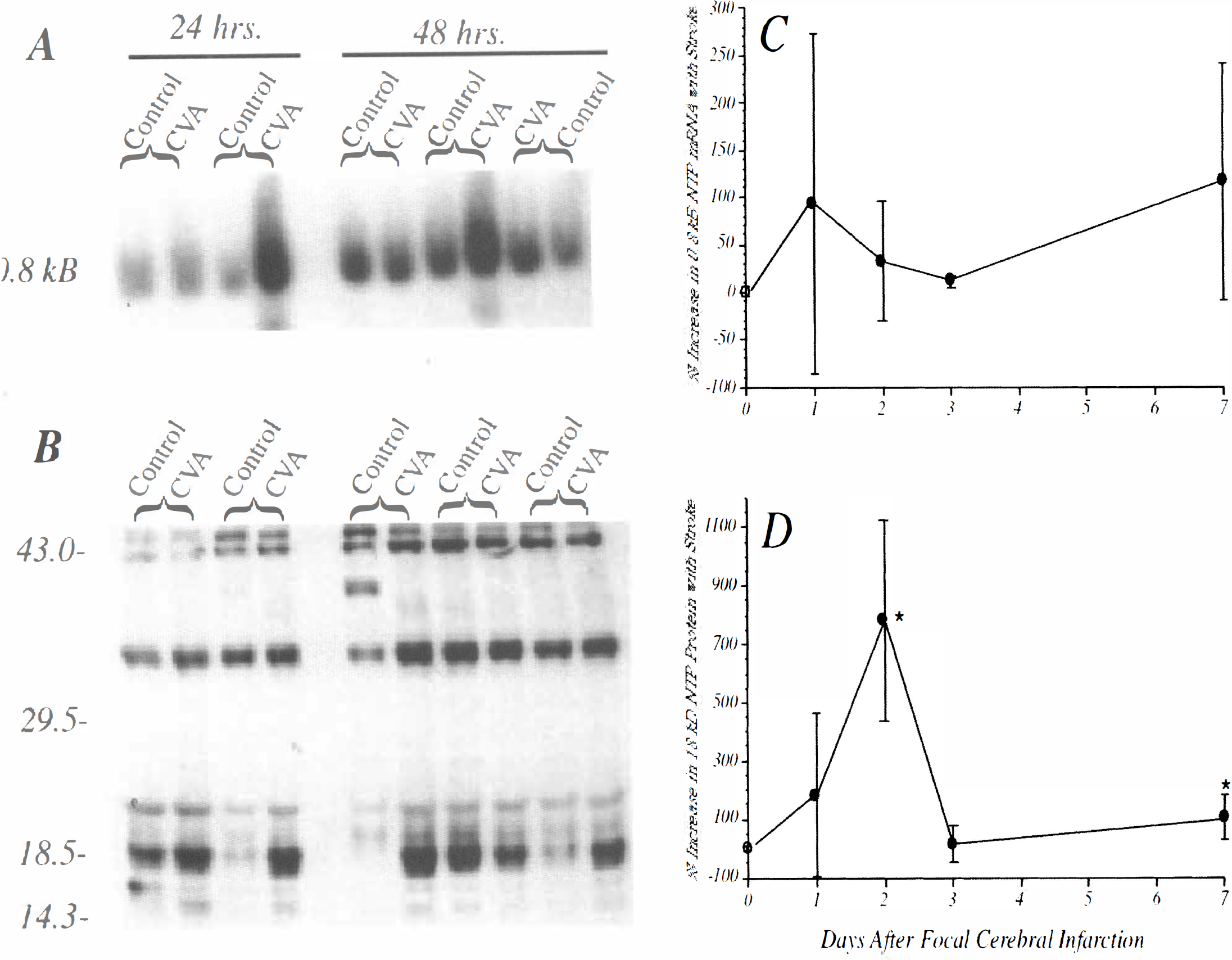
AD7c-NTP cDNA probes hybridized with predominantly 0.8 kB mRNA transcripts in rat brains (Fig. 4A). In some samples with strikingly elevated levels of AD7c-NTP mRNA expression, a related ∼0.6 kB mRNA transcript was also detected (Fig. 4A). In previous studies, −0.6 kB NTP mRNA transcripts were observed in differentiating (sprouting) neuronal cells stimulated with either insulin or retinoic acid (de la Monte and Wands, unpublished). The levels of AD7c-NTP mRNA were quantified by densitometric analysis of nonsaturated autoradiograms, with values normalized to the corresponding 18S rRNA signals. The data depicted in Table 1 reflect the mean ± SD of the percentage differences in the levels of AD7c-NTP mRNA measured between infarcted and contralateral uninfarcted tissue. The quantitative data were obtained from 2 separate experiments, each including 3 or 4 animals, i.e. N = 7 or 8 rats per time point (Table 1). In addition, Northern blot analysis was repeated twice for each experiment. In some animals, for unknown reasons, the basal levels of the 18 kD AD7c-NTP species were nonspecifically increased in the uninfarcted hemispheres (Fig. 4A, B). The net effect was to impede detection of the infarct-associated modulation of AD7c-NTP expression. To circumvent this problem, all data analyses were conducted using pairwise comparisons.
Modulation of AD7c-NTP mRNA (
AD7c-NTP gene modulation after experimental cerebral infarction in the rat model
MCA, middle cerebral artery.
Total number of animals studied.
Group trends over the entire interval of study indicated significantly increased AD7c-NTP expression in peri-infarct compared with the contralateral intact cerebral tissue by paired t-test analysis (RNA: P = 0.027; Protein: P = 0.006), Wilcoxon signed rank test (RNA: P = 0.015; Protein: P = 0.002), and two-sample test for equal medians (RNA: P = 0.003; Protein: P = 0.002). The high degree of variability was caused by differences in infarct volume and the inclusion of different proportions of infarcted tissue in the homogenates.
Within 24 hours of cerebral infarction, AD7c-NTP mRNA levels were increased relative to the contralateral uninfarcted cerebral tissue. At the 48 and 72 hour time points, the overall trend manifested infarct-associated increases in AD7c-NTP mRNA levels, but the percentage differences from control were highly variable (Table 1). However, 7 days after unilateral MCA occlusion, AD7c-NTP mRNA levels were consistently increased in the infarcted hemispheres. The sham-operative procedure did not cause AD7c-NTP mRNA levels to fluctuate more than 10% relative to the unmanipulated cerebral hemisphere (Fig. 4C). Despite considerable variability in results obtained between 24 and 72 hours, the overall percentage increases in the levels of AD7c-NTP mRNA in the infarcted compared with the contralateral uninfarcted cerebral hemispheres were statistically significant by the Wilcoxon signed rank test (Z = 2.43; P = 0.015) and the two-sample test for equal medians (P = 0.003). Mann Whitney U-tests demonstrated significantly increased AD7c-NTP mRNA levels in infarcted compared with the uninfarcted cerebral tissue at each of the time points (P < 0.05 to 0.005) (Table 1).
Increased neuronal thread protein mRNA expression in peri-infarction zones.
In situ hybridization studies demonstrated low levels of AD7c-NTP mRNA expression in control and sham-operated cerebral tissue (Fig. 5B, D). Within 24 hours of MCA occlusion, increased AD7c-NTP mRNA expression was detected in numerous cells (mainly neurons) distributed in the peri-infarct zones. In addition, scattered acutely ischemic (but not necrotic) neurons within the areas of infarction also manifested increased NTP mRNA expression. Between 2 and 7 days after MCA occlusion, perifocal histologically intact neurons showed striking increases in AD7c-NTP mRNA expression relative to corresponding regions of the contralateral hemisphere (Figs. 5A, C). Necrotic cells within the infarcted foci had no detectable AD7c-NTP in situ hybridization signals. Neurons were identified by their pyramidal shape, larger size than glial cells, presence of a single prominent nucleolus, and cortical localization.
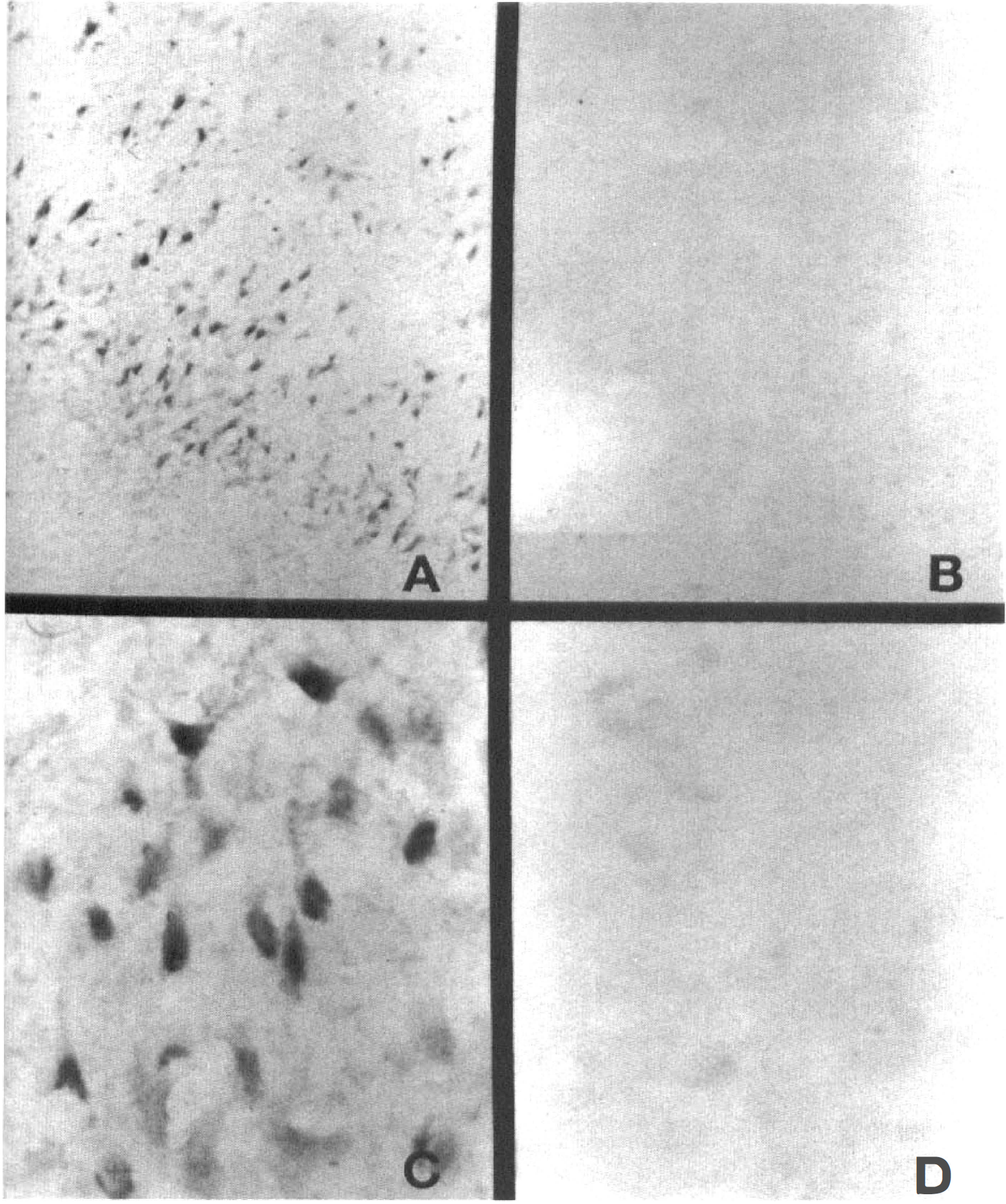
In situ hybridization study showing increased NTP mRNA expression in perifocal neurons 48 hours after permanent experimental MCA occlusion (
Selectively increased expression of the 18 kD AD7c-NTP species with cerebral infarction.
Western blot analysis detected 42 kD, 35 to 36 kD, 21 kD, 18 kD, and 15 kD AD7c-NTP-immunoreactive proteins (Fig 4B). Occasionally, an extra −39 to 40 kD band was detected, possibly resulting from either dimerization of the 18 kD species, or degradation of the 42 to 43 kD species. The 18 kD and 15 kD AD7c-NTP species were modulated with cerebral infarction, whereas the 42 kD, 35 to 36 kD, and 21 kD species were not. However, the 15 kD species was expressed only at very low levels, and previous studies demonstrated 15 kD NTP molecules to represent nonphosphorylated forms of the 18 kD NTP molecules (de la Monte et al., 1996a). Because the 15 kD and 18 kD species were similarly modulated, only data (mean ± SD) corresponding to the percentage differences in the levels of the 18 kD NTP species are depicted in Table 1. Table 1 summarizes data from two studies, each of which included three or four animals (N = 7 to 8 rats total) per time point. In addition, for each experiment, the Western blot analyses were repeated twice to verify the trends. All statistical analyses were based on paired ipsilateral and contralateral hemisphere differences to correct for unexplained fluctuations in the basal levels of AD7c-NTP expression in the rats (See Fig. 4).
In sham-operated rats (N = 3), the interhemispheric differences in the levels of AD7c-NTP expression were less than 10 percent (Fig. 4D). In contrast, 24 hours after unilateral MCA occlusion, increased levels of the 18 kD AD7c-NTP protein were detectable in the ipsilateral hemisphere, and at the 48-hour time point, the interhemispheric differences were maximum (Table 1, Fig 4B, D). At the 72-hour time point, the levels of the 18 kD AD7c-NTP protein were similar in the infarcted and uninfarcted hemispheres. However, 7 days after MCA occlusion, a small but definite increase in AD7c-NTP expression was again detected in the ipsilateral hemisphere. At the 24- and 48-hour time points, the standard deviations corresponding to the mean percentage increases in AD7c-NTP expression were large because of variability in the proportion of necrotic tissue included in the homogenates. Nonetheless, the overall mean percentage increase in the levels of the 18 kD AD7c-NTP species in the infarcted compared with the contralateral uninfarcted cerebral hemispheres was statistically significant by the Wilcoxon signed rank test (Z = 3.059; P = 0.002) and the two-sample test for equal medians (P < 0.0002). Mann Whitney U-tests demonstrated significant percentage increases in the levels of the 18 kD AD7c-NTP protein in infarcted compared with the uninfarcted cerebral tissue at the 24-hour, 48-hour, and 7-day points (all P < 0.05 to 0.005) (Table 1), coinciding with the expected period of active neuronal regeneration (David and Aguayo, 1985; Taguohi, et al., 1989; Stroemer, et al., 1995). There were no significant differences or trends detected with respect to the other AD7c-NTP species (all P > 0.1).
Increased neuronal neuronal thread protein immunoreactivity in peri-infarction tissue.
In uninfarcted cerebral tissue, neuronal perikaryal AD7c-NTP immunoreactivity was low-level (Fig. 6A, G). Within 24 hours of MCA occlusion, histologically intact neurons surrounding the foci of infarction showed increased AD7c-NTP immunoreactivity (Fig. 6B). In addition, isolated ischemic, but not necrotic cells (mainly neurons) within the infarcts, manifested transiently increased AD7c-NTP immunoreactivity (Fig. 6B, D, H), which was no longer detectable 48 hours after MCA occlusion (Fig. 6E). Between 2 and 7 days after MCA occlusion, both the number of neurons with prominent AD7c-NTP expression, and the levels of cellular and neuropil fiber immunoreactivity increased in the peri-infarct zones (Fig. 6C, F). By 7 days, increased AD7c-NTP immunoreactive was detectable in fine neuritic processes (Fig. 6I), possibly related to the regenerative response. Therefore, the findings by immunocytochemical staining confirmed the results of in situ hybridization.
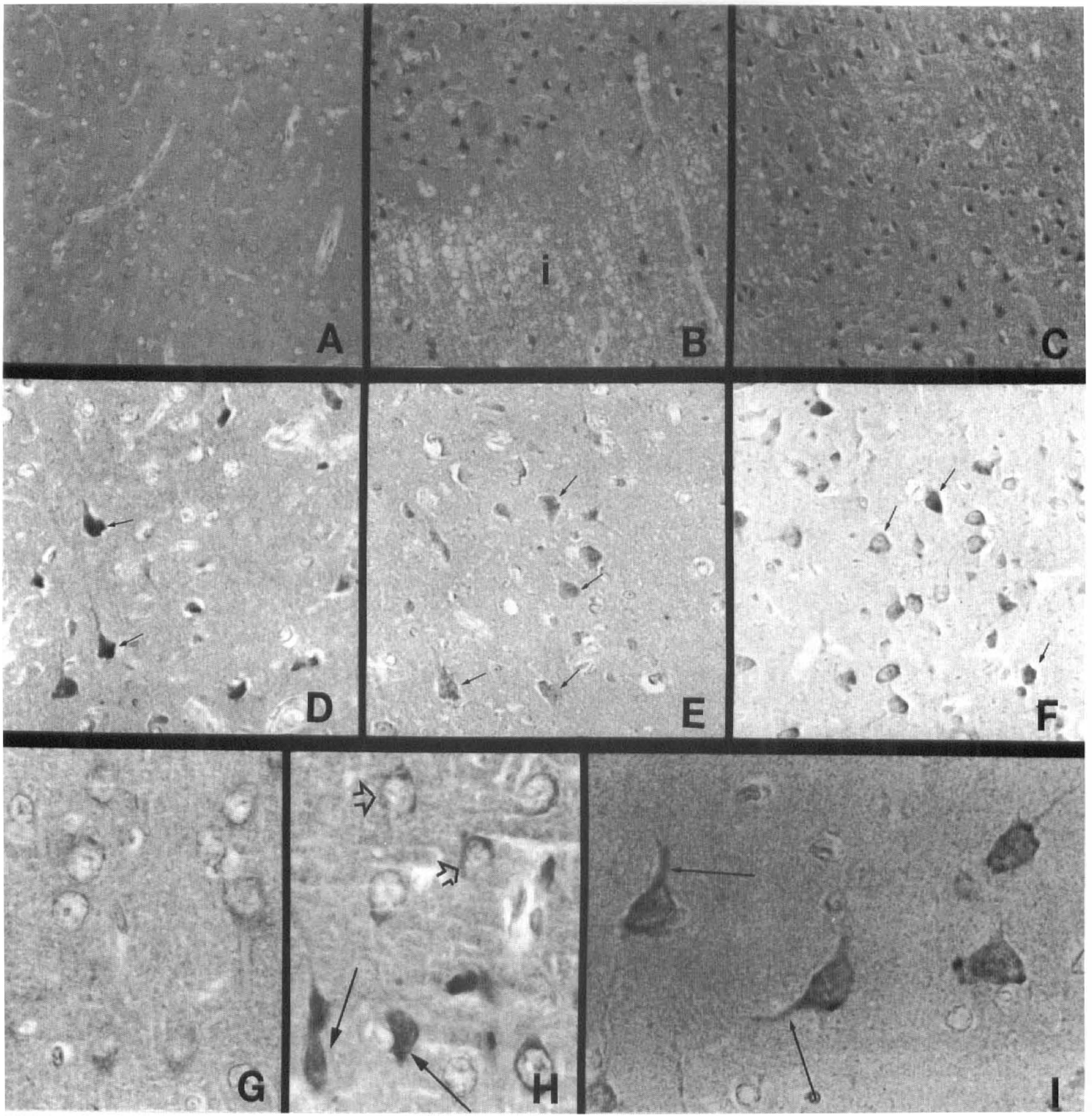
Immunocytochemical staining studies showing modulation of NTP expression in peri-infarction zone neurons after permanent experimental MCA occlusion. (
DISCUSSION
These studies showed brisk modulation of NTP gene expression after cerebral infarction in both humans and experimental animals. Northern blot analysis and in situ hybridization studies showed that modulation of NTP during the acute and subacute phases of cerebral infarction was transcriptional. In contrast, the finding of increased neuronal AD7c-NTP immunoreactivity without up-regulation of the corresponding mRNA in human brain infarcts that were several weeks or months old suggests that beyond the subacute phases of injury, NTP expression may be modulated by post-transcriptional mechanisms, e.g. prolonged protein half-life, increased phosphorylation of the 15 kD species, or altered rates of transport along regenerating axons.
Probes generated with the 1.4 kB full-length AD7c-NTP cDNA detect mainly 1.4 kB and 0.8 kB transcripts in human brains (de la Monte and Wands 1993; de la Monte et al, 1995a), and 0.8 kB transcripts in rat brains. However, the finding of several AD7c-NTP protein species by Western blot analysis of rat brains suggests that additional related mRNA transcripts were expressed but not detected under the stringent hybridization conditions employed. Although quantitative Northern and Western blot analyses helped to document these trends, the results obtained with acute lesions (24 to 48 hours) were variable because of the inconstant volumes of cerebral infarction generated in the experimental model. The inclusion of different proportions of necrotic tissue in the homogenates was also a very important factor contributing to the variable degree of increased AD7c-NTP gene expression detected by Northern or Western blot analysis. On the other hand, the in situ hybridization and immunocytochemical staining studies were consistently informative, and reproducibly demonstrated up-regulation of NTP in perifocal neurons within 24 hours of MCA occlusion. The expression of several related AD7c-NTP proteins in brain, as demonstrated by Western blot analysis, could have confounded the interpretation of the immunohistochemical staining studies. However, this interpretation is unlikely because with the antibodies diluted optimally, we were able to show minimal or absent modulation of AD7c-NTP immunoreactivity in neurons located in the contralateral hemisphere, and also ipsilateral but distant from the lesions. In addition, Western blot analysis showed minimal modulation of higher molecular weight AD7c-NTP-immunoreactive proteins after cerebral infarction. The importance of conducting in situ analysis of gene expression in conjunction with quantitative Northern and Western blot analyses is underscored by these results.
The in situ hybridization and immunocytochemical staining studies demonstrated striking up-regulation of NTP gene expression mainly in histologically intact peri-focal neurons in both humans and experimental animals. Although acutely ischemic neurons within and surrounding 24-hour-old lesions also manifested increased NTP immunoreactivity, this phenomenon was transient and probably agonal since it was no longer detectable 48 hours after MCA occlusion. Neurons were identified by their pyramidal shape, larger size than most glial cells, single prominent nucleolus, laminar distribution within the cortex, and GAP-43 immunoreactivity in adjacent histological sections. In previous studies, neuronal expression of NTP was shown by its coimmunolocalization with neuronal cytoskeletal proteins, e.g. tau-1 and neurofilament, and GAP-43 in brain (data not shown) and neuronal cell lines (Xu, et al, 1993; de la Monte, et al, 1996b). Although the majority of cells showing injury-associated increases in NTP expression were neurons, a concomitant glial cell response could not be excluded. Neuronal thread protein immunoreactivity has already been described in glial cell lines (Xu, et al, 1993), and in protoplasmic astrocytes and oligodendrocytes in brains with AD (de la Monte, et al, 1996b, de la Monte, et al., 1996c). The potential significance of increased NTP expression in glial cells has not been determined, although in vitro studies suggest a correlation with cell growth (Xu, et al, 1993).
In a recent report, increased expression of GAP-43 was shown in histologically intact perifocal neurons (Stroemer, et al., 1995), a finding confirmed herein using tissue sections adjacent to those examined for NTP expression. Therefore, the perifocal modulations of GAP-43 and NTP seem to be similar. Typically, neurons distributed in peri-lesion/infarction zones undergo regenerative sprouting (David and Aguayo, 1985; Taguohi, et al., 1989; Stroemer, et al., 1995). The time course of increased NTP expression in both human and experimental animals correlated with the expected period of neuronal regeneration and repair (Garcia et al., 1993; Chuaqui and Tapia 1993). Neuritic sprouting and axonal regeneration are associated with transcriptional modulation, or increased expression and axonal transport of the phosphoproteins GAP-43 (Meiri et al., 1986; DeGraan et al., 1985; Skene et al., 1986; Snipes et al., 1987; Ng et al., 1988), constitutive isoforms of nitric oxide synthase (ceNOS) (Wu 1993; de la Monte et al., 1995b; de la Monte and Bloch 1996;), and SNAP-25 (Hess et al., 1992; Oyler et al., 1989). The aggregate in vivo and in vitro data obtained thus far suggest that NTP gene modulation is also activity dependent and linked to neuritic sprouting associated with development (de la Monte, et al., 1996a), neuronal differentiation (de la Monte, et al., 1995b; de la Monte, et al., 1996b), neurodegeneration (de la Monte and Wands, 1992; de la Monte, et al., 1995b), and now perhaps regeneration and repair.
In contrast to the prolonged periods (days to weeks) of injury-associated NTP and GAP-43 up-regulated gene expression, transcriptional modulation c-fos, zif 268, and brain-derived neurotrophic factor (Herdegen et al., 1993; Herdegen et al., 1994; Horn et al., 1995; Herrera and Robertson 1990a) is immediate and brief, occurring within hours of cerebral infarction. Activation of these immediate early inducible genes occurs in neurons distributed throughout the lesioned cerebral hemisphere (Comelli et al., 1993; Herrera and Robertson 1990b). Therefore, unlike NTP and GAP-43, the injury-associated changes in immediate early inducible gene expression are not likely to be related to regeneration, and instead may reflect a spreading depression accompanied by marked changes in interstitial ion concentration, local blood flow, or metabolism (Comelli et al., 1993).
In previous studies, we observed abundant expression of the 15 to 18 kD NTP species in developing brains and primitive neuroectodermal tumor cell lines, and relatively low levels of these NTP species in mature brains (Xu et al., 1993; Xu et al., 1995; de la Monte et al., 1995). Instead, the 21 kD, 26 kD, and 39 to 42 kD species are the major forms of NTP expressed in the mature central nervous system (Ozturk et al., 1989; de la Monte et al., 1990; de la Monte and Wands 1992; de la Monte et al., 1992; de la Monte, et al., 1996c), and in AD, neuronal overexpression and accumulation of the 21 kD and 39 to 42 kD NTP species correlates with widespread aberrant neuritic sprouting and dementia (de la Monte et al., 1996b; de la Monte, et al., 1996c). The present study suggests that expression of the 15 to 18 kD NTP species may be strikingly modulated with cerebral infarction, and perhaps nonspecific injury as well.
One of the most important and difficult clinical distinctions is between vascular dementia and AD. However, in a recent survey, 30% of the brains with AD harvested in the Alzheimer's Disease Research Center Brain Bank at the Massachusetts General Hospital brain bank were found to contain one or more cerebral infarcts. Therefore, we suspect that some cases of dementia in elderly individuals may be caused by the synergistic effects of two independent disease processes. Whether superimposed cerebrovascular disease in AD is related to the Apoliprotein E4 genotype or β-A4 amyloid deposition (Strittmatter et al., 1993; Corder et al., 1993) has not been determined. Nonetheless, the progression of dementia may be slowed in some cases of late-onset AD by preventing or treating cerebral infarction. Previous studies using a highly sensitive immunoradiometric assay to detect NTP in cerebral spinal fluid (Ozturk et al., 1989; de la Monte et al., 1992) were designed to exclude patients with strokes because it was immediately recognized that cerebral infarction and nonspecific injury confounded the results (de la Monte, et al 1992). The present study suggests that characterization of the molecular sizes of NTP accumulated in cerebral spinal fluid or cerebral tissue in association with cognitive deterioration may facilitate clinical detection of cerebral microinfarction, and also help distinguish between vascular dementia and AD.
