Abstract
Recently we observed inter- and intrastrain differences in cortical infarct volumes after middle cerebral artery (MCA) occlusion. Variations in the anastomoses providing collateral blood supply could account for different lesion sizes. Our objectives were to compare number and internal diameters of the MCA-anterior cerebral artery (MCA-ACA) anastomoses and to determine if the lesion extended beyond branches of the MCA territory into the field of the ACA in the rat strains/lines. Sprague-Dawley rats and Wistar rats from Simonsen Laboratories (SLSD and SLWIS) and Sprague-Dawley rats from Taconic Laboratories (TLSD) and Charles River Laboratories (CRSD) were anesthetized and injected with papaverine and Vultex (white latex) for arterial visualization. Some rats were also subjected to MCA occlusion. Significantly fewer anastomoses were present in SLSD and SLWIS than in CRSD and TLSD (p < 0.05). The mean internal diameters of the anastomoses were not significantly different between the strains/lines (p > 0.05). After MCA occlusion, significantly more (p < 0.05) TLSD and CRSD than SLSD had lesions extending from the MCA field beneath the anastomoses and into the region supplied by the ACA. Neither the number, luminal diameter, nor density of MCA-ACA anastomoses appears to be the limiting factor that differentiates lesion size following MCA occlusion in these particular rat strains/lines. Therefore, factors other than anatomical variations probably account for different lesion sizes.
In rats, the middle cerebral artery (MCA) branches from the internal carotid artery (ICA) and courses laterally to the dorsal aspect of the brain where it divides into distal branches. The anterior cerebral artery (ACA) also branches from the ICA, then courses medially through the longitudinal fissure, continuing as distal branches on the dorsal surface of the brain (Greene, 1935). A majority of the ACA and MCA distal branches anastomose ∼2–3 mm from midline on the dorsal aspect of each hemisphere (Coyle and Jokelainen, 1982), thereby forming a “line of anastomoses” along the rostral-caudal axis of the brain. Following MCA occlusion, the core of the lesion located within the territory of the MCA is irreversibly damaged, while the perifocal area (the penumbra) can remain viable due to blood circulation through the collateral anastomoses (Siesjö, 1992).
In recent studies (Oliff et al., 1995a, b ), we found interstrain as well as intrastrain differences in infarct volume caused by focal cerebral ischemia induced by MCA occlusion. Cortical infarct volume was 2- through 16-fold smaller (depending on surgical manipulation of the common carotid arteries) in Simonsen Laboratories Sprague-Dawley rats (SLSD) than in Taconic Laboratories Sprague-Dawley rats (TLSD) (Oliff et al., 1995a). Variations in the number, luminal width, and location of the narrow MCA-ACA anastomoses that cause relatively high resistance to vascular flow could account for differences in cerebral infarct sizes between these strains/lines. The first goal of the present study was to compare the quantity, density, and luminal (internal) diameter of the MCA-ACA anastomoses in several rat strains and lines obtained from different vendors. Our second goal was to determine whether, after MCA occlusion, the ischemic lesion extended beyond the MCA territory and into the ACA field.
MATERIALS AND METHODS
Experiment I
Nineteen male Simonsen Laboratories Wistar rats (SLWIS) (Gilroy, CA, U.S.A.), 20 male SLSD, 24 male TLSD (Germantown, NY, U.S.A.). and 19 male Charles River Laboratories Sprague-Dawley rats (CRSD) (Hollister, CA, U.S.A.) weighing 270–310 g each were anesthetized with 2.0 vol% halothane in 70% nitrogen/30% oxygen.
The procedure followed that of Coyle and Jokelainen (1982). A lethal dose of papaverine hydrochloride (45 mg/kg in sterile water) was injected into the jugular vein to produce maximal vasodilation and to minimize cerebrovascular resistance. The blood vessels were standardized to maximal dilation before measurement because otherwise large vessels with some contraction could not be differentiated from small vessels with no contraction. A 2- to 3-mm-wide craniotomy was drilled in the skull at the level of lambda between the midline and temporal ridge so that collaterals could be visually monitored for filling with latex. The thoracic aorta was clipped at the diaphragm. Five milliliters of warmed (38°C), undiluted Vultex (a white latex; Chicago Latex Products no. 563) was injected into the ascending aorta while vessel filling was observed through the craniotomy.
Thirty to forty minutes after injection of the Vultex, the animal was decapitated and the dorsal aspects of the skull and dura were removed. The entire head was fixed in 10% formalin for several weeks before the brain was removed from the skull. The dorsal side of each brain was magnified 16 times real size and photographed. Anastomoses on the right hemisphere were localized and numbered by following the MCA and ACA from their origin to where they anastomose (Coyle and Jokelainen, 1982). The density of anastomoses was calculated by dividing the number of anastomoses by the length of the brain hemisphere (excluding the cerebellum). The diameters of the five largest anastomoses on the right hemisphere were measured with an image analysis system (Image I Program; Universal Imaging Corp., West Chester, PA, U.S.A.) and averaged to yield a mean maximal internal diameter for each brain.
Each brain was blindly evaluated on three separate occasions to ensure consistent data acquisition. Brains were excluded from the study if the latex caused vessel bursting, if latex was in the veins, or if latex incompletely filled the surface vessels. One-way analysis of variance and Tukey-Kramer post hoc analysis were used for statistical evaluation of the data. Differences were determined to be significant if p < 0.05.
Experiment II
Ten male SLWIS, 13 male SLSD, 12 male TLSD, and 9 male CRSD each weighing 280–320 g were allowed free access to food and water before and after surgery. The animals were anesthetized with 3.5 vol% halothane in 70% nitrogen/30% oxygen and then intubated and connected to a Harvard ventilator. Anesthesia was maintained throughout the surgery with 2.0 vol% halothane. Aseptic animal surgical preparation was conducted and procedures were in accordance with institutional guidelines. Body temperature was maintained at 37.5°C throughout surgery by means of a rectal thermometer and heating pad beneath the animal that was connected to a servo-control unit. The femoral artery was cannulated (PE-50) to monitor the blood pressure and the blood gases.
Modifying the technique originally described by Koizumi et al. (1986), the right common carotid artery (RCCA) was isolated after a ventral midline incision on the neck. Permanent 3-0 silk ligatures were placed around the external carotid artery (ECA) and the proximal portion of the RCCA. A vessel clip was placed temporarily (it was removed just prior to the insertion of the filament) on the RCCA, proximal to the bifurcation of the ICA and the ECA. A small incision was made in the RCCA, and a 0.30-mm-diameter filament (0.30-mm nylon fishing line, the tip of which was rounded with fine sandpaper and darkened with ink), was inserted and secured with 3-0 silk. After the RCCA vessel clip was removed, the filament was advanced into the intracranial ICA 18 mm from the ICA/ECA bifurcation, thereby occluding the MCA. The contralateral CCA was occluded with a vessel clip for 60 min after the filament had been advanced. The animals were routinely checked after the completion of the surgery. Rectal temperatures were measured at 4 h and at 24 h postsurgery. Blood gases, serum glucose, and hematocrit were measured prior to and just after MCA occlusion. Twenty-four hours after occlusion of the MCA, the animals were reanesthetized with 2.0 vol% halothane in 70% nitrogen/30% oxygen delivered via a nose cone. The cerebral vessels were perfused with Vultex as described in Experiment I.
Ten minutes after injection of the Vultex, the neck incision was reopened, and the filament was removed. Then the animal was decapitated and the brain carefully removed. The whole brain was stained with 1% 2,3,5-triphenyltetrazolium chloride (TTC), incubated at 37.5°C for 30 min, and then placed in 10% formalin. The brains could not be sectioned due to the elastic properties of Vultex. The brains were inspected to ensure that the filament had passed the MCA (residual black ink from the tip of the filament could be visualized).
The brains were magnified six times real size and photographed. The anastomoses were localized by visually following MCA and ACA arterioles from branch angles to meeting points as described earlier (Coyle and Jokelainen, 1982). An anastomosis was at the narrowest vessel width or one-half the distance between adjacent branch points of corresponding ACA and MCA branches (Coyle and Jokelainen, 1982). Adjacent anastomoses form a broken line of anastomoses along the rostral-caudal axis of the brain (Coyle and Jokelainen, 1982) (Fig. 1). Thus, the “line of anastomoses” is made up of all pial surface arterial interconnections between distal ACA and MCA branches on the dorsal surface of the brain and the “line” defines the anatomical boundary between the ACA territory and the MCA territory. The distance from the line of anastomoses to the lesion border was measured at three locations (Fig. 1). Measurements from the line medialward into the ACA field were assigned negative values. Measurements from the line lateralward into the MCA field were assigned positive values. The values were averaged separately for rostral, middle, and caudal locations. Lesion frequencies were evaluated with Fisher's exact test (two tailed). Differences were considered significant if p < 0.05.
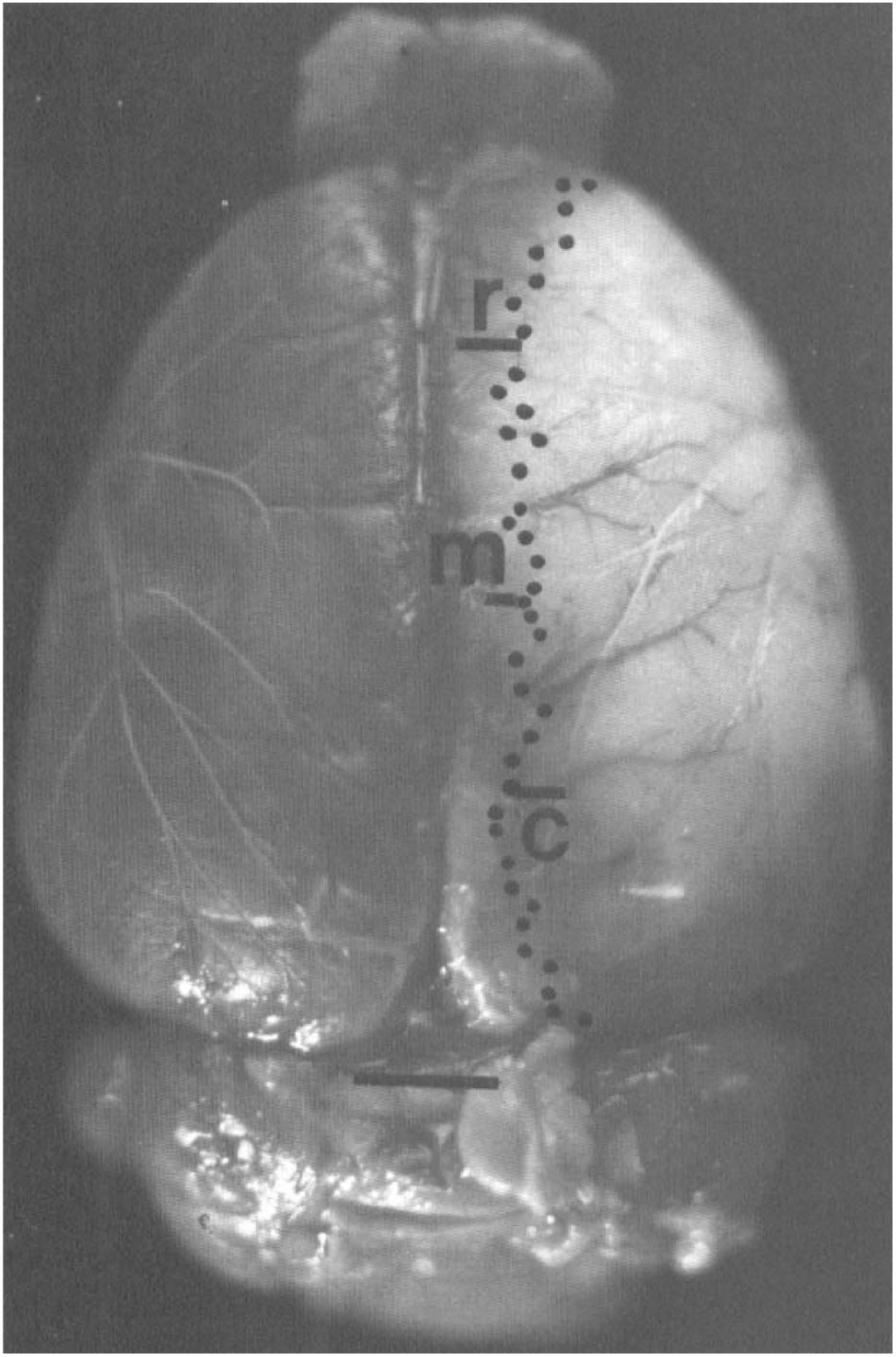
Taconic Laboratories Sprague-Dawley rat brain stained with 2,3,5-triphenyltetrazolium chloride and injected with Vultex. Normal tissue is stained red and the lesioned tissue is white. The lesion in the right hemisphere extends past the middle cerebral artery territory into the anterior cerebral artery territory. R, rostral location; M, middle location; C, caudal location, where the distance from the border of the lesion to the line of anastomoses was measured. The dotted line is the location of the “line of anastomoses.” Distance measurements and determination of the line of anastomoses were made at a much higher magnification (six times real size) to properly visualize the branch angles and lumen diameters. Scale bar = 3 mm.
RESULTS
Experiment I
Significant differences were found in the number of MCA-ACA anastomoses between rats from different vendors and between rats from different strains (Table 1). Significantly fewer MCA-ACA anastomoses were present in SLSD and SLWIS than in TLSD and CRSD (p < 0.05; Table 1). However, there was no significant difference in the number or luminal (internal) width of MCA-ACA anastomoses between SLSD and SLWIS or between TLSD and CRSD (p > 0.05; Table 1).
Mean quantity and internal diameter of MCA-ACA anastomoses of each strain/vendor
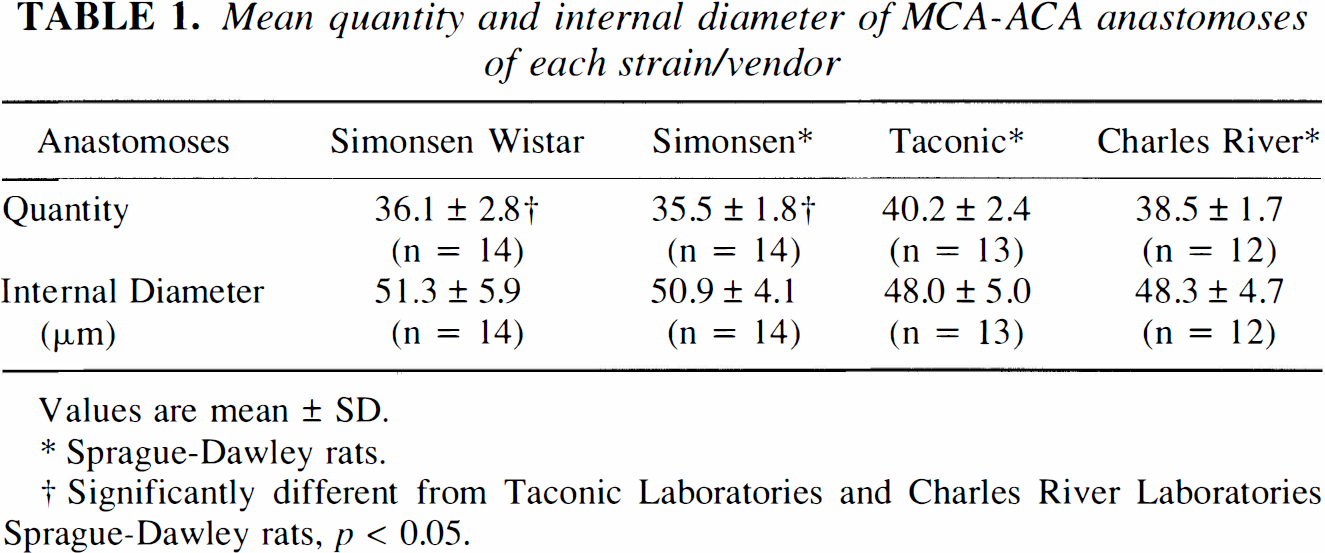
Values are mean ± SD.
Sprague-Dawley rats.
Significantly different from Taconic Laboratories and Charles River Laboratories Sprague-Dawley rats, p < 0.05.
The number of anastomoses per brain could be proportional to brain length with fewer anastomoses in shorter brains and more in longer ones, assuming equal density. The mean cortical length was significantly less in SLSD (12.6 ± 0.31 mm) and SLWIS (12.6 ± 0.21 mm) than in TLSD (13.1 ± 0.43 mm; p < 0.05), but not appreciably different from CRSD (12.9 ± 0.38 mm). Density of anastomoses was significantly lower in SLSD (2.84 ± 0.18/mm) than in TLSD (3.06 ± 0.23/mm; p < 0.05). No significant differences in anastomotic density were found in other comparisons (p > 0.05; SLWIS 2.87 ± 0.24/mm, CRSD 3.01 ± 0.16/mm). Body weights were similar for the rat groupings (p > 0.05, data not shown).
Experiment II
Monitored physiological variables are listed in Table 2. Temperature hematocrit and serum glucose were not significantly different among the rat groups. There were some significant differences in pH, P
Physiological variables of rats subjected to middle cerebral artery occlusion
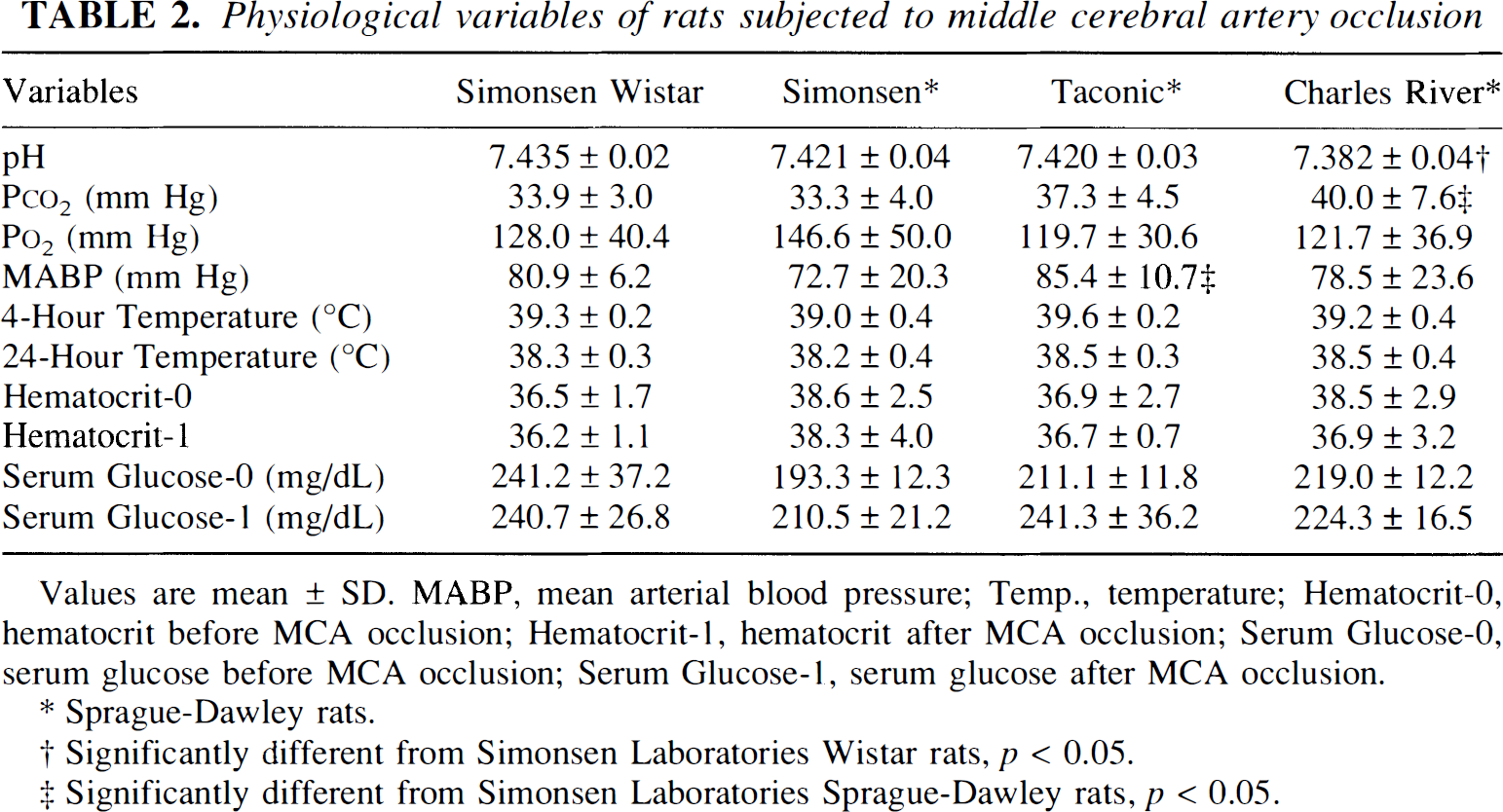
Values are mean ± SD. MABP, mean arterial blood pressure; Temp., temperature; Hematocrit-0, hematocrit before MCA occlusion; Hematocrit-1, hematocrit after MCA occlusion; Serum Glucose-0, serum glucose before MCA occlusion; Serum Glucose-1, serum glucose after MCA occlusion.
Sprague-Dawley rats.
Significantly different from Simonsen Laboratories Wistar rats, p < 0.05.
Significantly different from Simonsen Laboratories Sprague-Dawley rats, p < 0.05.
Reduced TTC staining was observed either in the MCA field or in both the MCA and the ACA fields (Table 3). The lesion, when observed in both fields, was present as a single lesion that extended from the MCA territory beneath the anastomoses into the ACA territory. Lesion frequency (presence) in the rostral ACA field was not uniform among strainsAines (p < 0.05). Lesion frequency in the rostral ACA field varied from greatest to least in the following order: TLSD (83%) > CRSD (77%) > SLWIS (40%) > SLSD (8%). Within the ACA field of TLSD and CRSD, lesion frequency was significantly greater at the rostral location than at the middle site (p < 0.05) and was least at the caudal location (Table 3; Fig. 1). There was no observed gross reduction of TTC staining on the pial surface of the brain in 23% of SLSD rats. In contrast, in the other strainsAines, gross reduction of TTC staining was observed in every rat.
Lesion frequencies at specific brain locations after middle cerebral artery occlusion
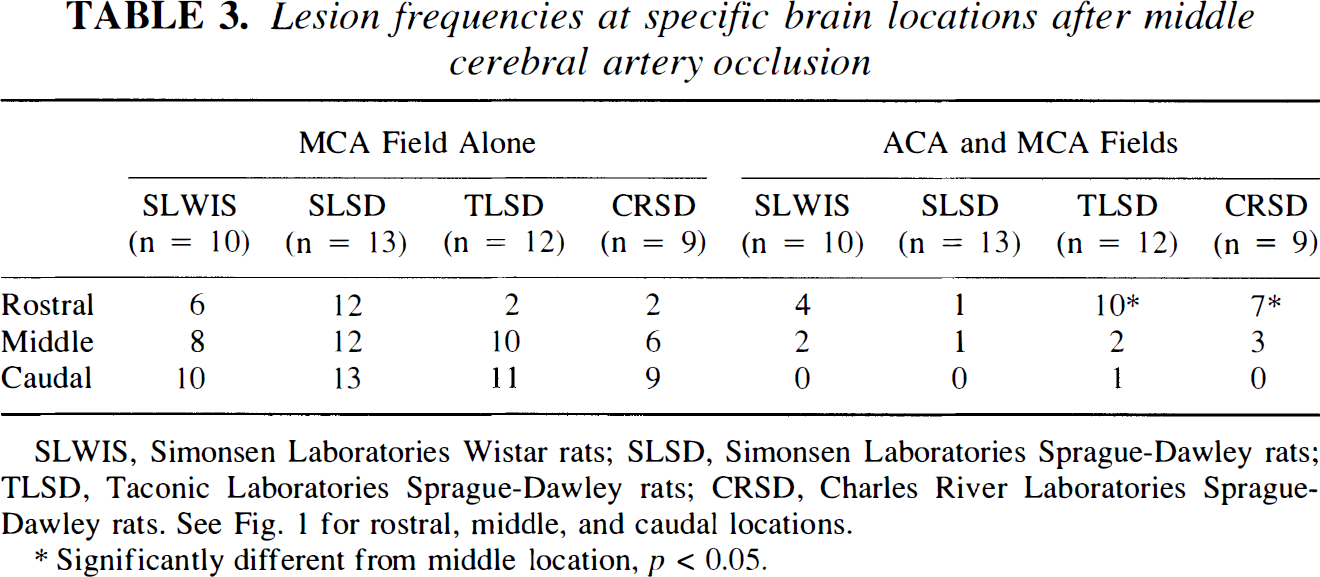
SLWIS, Simonsen Laboratories Wistar rats; SLSD, Simonsen Laboratories Sprague-Dawley rats; TLSD, Taconic Laboratories Sprague-Dawley rats; CRSD, Charles River Laboratories Sprague-Dawley rats. See Fig. 1 for rostral, middle, and caudal locations.
Significantly different from middle location, p < 0.05.
To determine the relationship of the lesion border to the line of anastomoses, we examined the medial-lateral location of the lesion border in comparison to the line of anastomoses (Figs. 1 and 2). In the rostral location, the mean lesion border was located medial (beyond) to the anastomoses and into the ACA field in the TLSD (0.72 ± 0.52 mm) and the CRSD (0.34 ± 0.57 mm). At middle and caudal sites, the mean lesion border was located lateral to the anastomoses, within the MCA field, in all rat groupings. At all sites the lesion border was significantly further lateral to the anastomoses (deeper within the MCA field) in SLSD compared with the other strains/lines (p < 0.05; Fig. 2).
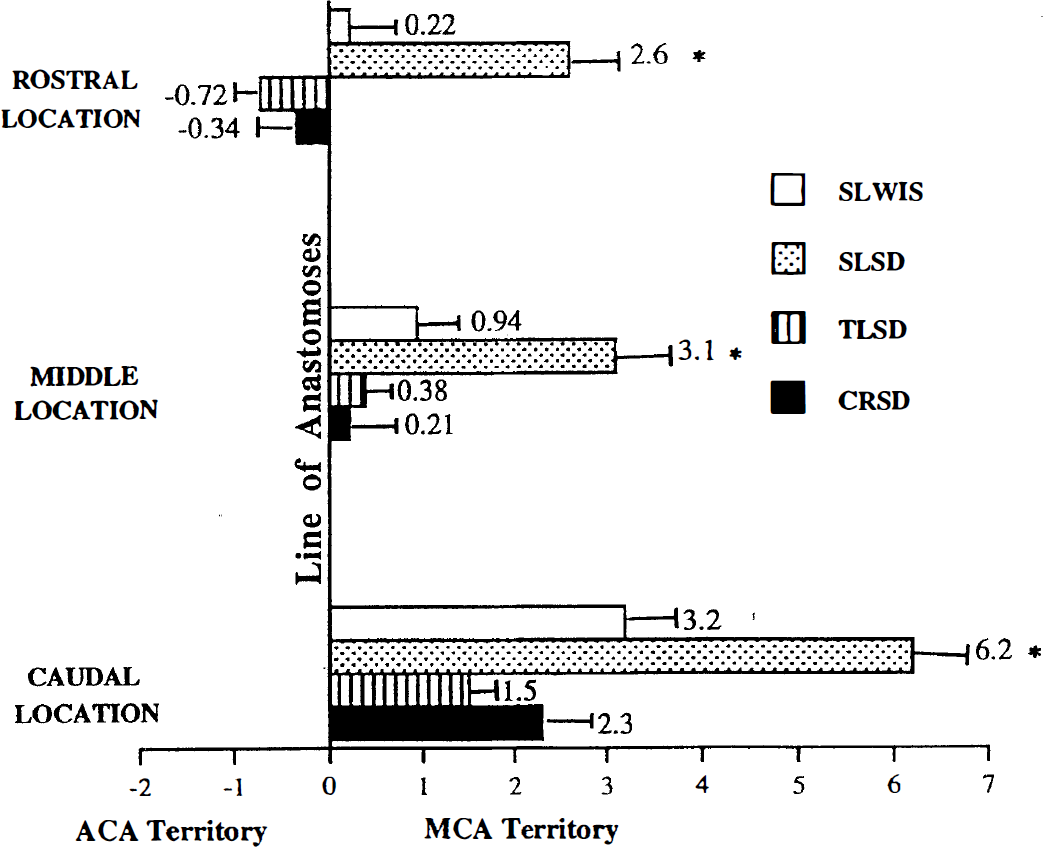
Mean distance between the anastomoses and the border of the lesion in each strain/vendor. Negative values indicate distance the lesion extends beyond the anastomoses (0 reference point) into the area of the anterior cerebral artery. Positive values indicate the distance from the anastomoses lateral to the border of the lesion. Mean ± SD. SLWIS, Simonsen Laboratories Wistar rats; SLSD, Simonsen Laboratories Sprague-Dawley rats; TLSD, Taconic Laboratories Sprague-Dawley rats; CRSD, Charles River Laboratories Sprague-Dawley rats. Refer to Fig. 1 for rostral, middle, and caudal locations. *p < 0.05.
DISCUSSION
The major new findings of this study are that first, the numbers of MCA-ACA anastomoses differ among rat strainsAines independently of body weight or cortical length. This allows one to hypothesize that after MCA occlusion the lesion is smaller in rats with more anastomoses. This hypothesis is rejected as discussed below. Second, the mean luminal diameters of the widest anastomoses are not significantly different among the rat strains/lines. This suggests that high vascular resistance of the MCA-ACA anastomoses to collateral blood flow is not the major determinant of significantly different lesion sizes in these particular strainsAines. Third, the finding that the lesion extended beyond the anatomically defined territory of the MCA into the field beneath branches of the ACA suggests that the MCA-ACA anastomoses were not the limiting factor responsible for the lesion in the ACA field. This discussion focuses on these issues and the possible effects that the intraluminal thread method of MCA occlusion has on collateral circulation.
Lesion volumes were not examined because TTC does not penetrate the entire whole brain following immersion (even after 3 h) and because the elastic properties of latex preclude routine gross sectioning of the brain. Previously we reported (Oliff et al., 1995a) that cortical lesion volumes (mean ± SEM, mm3) in SLWIS (152.4 ± 22.9) and in SLSD (113.0 ± 18.8) were significantly smaller (p < 0.05) than for TLSD (227.8 ± 19.3) and CRSD (248.4 ± 24.0) following MCA occlusion using identical procedures to those reported here, except latex was not injected into the vessels of those brains. The hypothesis that, after MCA occlusion, the lesion is smaller in rats with significantly more or larger anastomoses is rejected. This is rejected because the anastomoses are fewer, not greater, in number and the lesions are smaller, not larger, in volume in SLSD and SLWIS than in TLSD and CRSD. Furthermore, the issue could be one of anastomotic density because of different brain lengths, but that was not the case. A low density of anastomoses (as in SLSD) arranged in parallel, as are these, presents greater, not less, total resistance to collateral blood flow than does a higher density of anastomoses of equal size (Berne and Levy, 1977). Any difference in resistance to collateral blood flow resulting from a low density of anastomoses was insufficient to produce a larger lesion in SLSD than in rats from the other two vendors. Thus, the number or density of MCA-ACA anastomoses in these particular strainsAines from different vendors is not the major determinant of different lesion sizes.
Studies of spontaneously hypertensive stroke-prone rats have demonstrated narrower MCA-ACA anastomoses (Coyle, 1987a), less than maximal blood flow through the anastomoses after MCA occlusion (Coyle and Heistad, 1987a, b ), and larger lesions confined to the MCA territory as compared with normotensive control rats (Coyle and Jokelainen, 1982; Coyle et al., 1984). Appreciably narrower MCA-ACA anastomoses present greater vascular resistance that predisposes stroke-prone rats to less collateral blood flow and larger lesions, with the lesion border located at a distance inside the anatomically defined MCA risk area that is proportional to the anastomosis width (Coyle, 1987b; Coyle and Feng, 1993).
The finding here of MCA-ACA anastomoses of nearly equal width for all the strains/lines suggests that these anastomoses are not the limiting factor responsible for significantly different lesion sizes. First, the lesion was not confined to the MCA field. The extension of the lesion from the MCA field beneath the MCA-ACA anastomoses into the ACA field suggests inadequate perfusion from branches of the ACA with blood that never flowed through the MCA-ACA anastomoses. Second, the present study did not test uncompromised ACA flow with a normal circulation potential. Both the filament used to occlude the MCA and bilateral carotid artery occlusion could be expected to profoundly reduce pressure and flow in preanastomotic branches supplying the rostral ACA tissue that develops the lesion in TLSD and CRSD.
The intraluminal thread occlusion method imposes considerable risk to the cerebral circulation that is far greater than point occlusion of the MCA with a ligature or segmental occlusion of the MCA by thermocoagulation (Benderson et al., 1986). First, entry of the thread into the azygos ACA (Scremin, 1995) would be expected to reduce flow and pressure in the more distal ACA, thereby increasing the risk of a lesion occurring in the ACA field. Second, even if the thread passes the MCA but does not enter the azygos ACA, the thread reduces or totally blocks blood flow to the posterior cerebral artery. In the rat, the major blood source for the posterior cerebral artery is the ICA, not the basilar artery as in humans (Scremin, 1995). Thus, collateral blood flow from the basilar artery via the posterior communicating artery must be necessary, at least in part, for preservation of the territory of the posterior cerebral artery in the intraluminal thread model of MCA occlusion. Other investigators suggested that differences in lesion volumes could be partially explained by differences in the functional integrity of the circle of Willis (Barone et al., 1993) or by the variability in the number and type of arterial branches (Fox et al., 1993), but differences in lesion volumes in different rat strains do not appear to depend on the density of capillaries within the neocortex (Grabowski et al., 1993).
In summary, there are differences in the number of MCA-ACA anastomoses, but not in the luminal width of the widest anastomoses of rats from different vendors and between rats of different strains. Anastomotic density was equal to or significantly less in the SLSD rats compared with the other rat strains/lines whose mean lesion borders were significantly closer to the anastomoses after occlusion of the MCA. In TLSD rats and CRSD rats, the mean lesion in the rostral region extended from the MCA field beneath the MCA-ACA anastomoses into the ACA field. Because normal blood flow to the ACA field is not required to flow through the MCA-ACA anastomoses, high vascular resistance of these anastomoses most likely was not the determinant of lesion size differences. Possibly, the lesion size differences may be caused by the tissue's response to ischemia rather than microvascular variations. We can conclude that the differences observed in number and size of anastomoses were due to the vendor and strain rather than variance between different shipments within the same vendor and strain because similar infarct volumes (Oliff et al., 1995a, b ), anastomosis size, and quantity were repeatedly found between different rat shipments.
