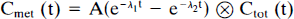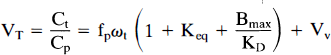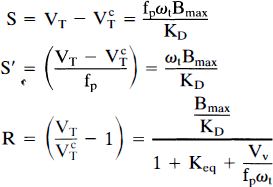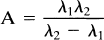Abstract
Positron emission tomography and single-photon emission computer tomography receptor-binding ligands can be used to measure changes in neurotransmitter levels. In particular, amphetamine-induced dopamine release has been assessed with [11C]raclopride by paired bolus injections and with [123I]iodobenzamide by using a single bolus plus infusion (B/I) study. Here, we measured the change in [11C]raclopride-specifìc binding in rhesus monkeys after i.v. administration of 0.4 mg/kg amphetamine by using both the bolus and B/I paradigms. Paired bolus studies (control and postamphetamine) were analyzed using compartment modeling and graphical analysis with a new plasma metabolite model to measure the total distribution volume (VT). Specific binding, calculated with three measures linearly proportional to the binding potential, demonstrated a 22–42% reduction in the postamphetamine study. VT values from B/I studies were determined by the tissue-to-plasma ratio at equilibrium, in addition to the bolus methods. There was good agreement between the control VT values between bolus and B/I studies. The amphetamine-induced change in specific binding in B/I studies was 19 ± 16%, measured directly from tissue radioactivity levels. This study demonstrates that stimulus-induced changes in specific binding can be measured with a single [11C]raclopride study using the B/I method.
The tissue concentration of receptor-binding radiopharmaceuticals depends on many physiological parameters. A wide variety of approaches have been developed to determine parameters of neuroreceptor binding that are ideally equal or at least linearly proportional to the free receptor concentration. These techniques include full kinetic modeling with multiple-parameter estimation, graphical approaches that estimate one or two combined model parameters such as the total volume of distribution (VT) and simple tissue concentration ratios. With such techniques, within-subject changes in free receptor concentration—for example, caused by an intervention that alters endogenous ligand concentration or occupancy by competing drugs—can be detected.
Two study designs have been used to estimate receptor-binding changes in such an intervention paradigm. In the first approach, two studies are performed, each with a bolus injection of high-specific-activity tracer. In the first study, control levels of binding are measured, for example, by determining VT by compartment modeling (Koeppe et al., 1991) or graphical analysis (Logan et al., 1990). Then, following the pharmacological intervention, total binding is measured again with a second injection of tracer. This approach has been used successfully with the D2 ligand [11C]raclopride (Farde et al., 1986, 1989) as well as with a number of other tracers. For example, Dewey et al. have demonstrated the effects of changes in synaptic dopamine by direct effects on the dopamine system itself (Dewey et al., 1993) and by indirect pharmacological interventions (Dewey et al., 1992; Dewey et al., 1995). In humans, this paired-study approach has been used to measure drug occupancy of the D2 receptor (Farde et al., 1992; Nordstrom et al., 1992; Nyberg et al., 1993).
The second study design is to administer the tracer as a combined bolus plus continuous infusion (B/I). Infusion has been used for absolute quantification of receptor parameters for tracers with reversible binding characteristics (Carson et al., 1993a; Abi-Dargham et al., 1994; Laruelle et al., 1994; Minoshima et al., 1994; Kapur et al., 1995). This approach can be extended to the measurement of short-term changes in free receptor concentration. First, the B/I administration of tracer is performed to achieve constant radioactivity levels in blood and all brain regions. Once equilibrium is achieved, control binding levels can be determined. For example, VT can be measured directly from the tissue-to-plasma concentration ratio. Then, a stimulus is administered while the infusion of radiotracer continues, and the change in specific binding of the tracer can be monitored. This study design enables the measurement of pre- and postintervention binding levels from a single administration of tracer and has previously been used with single-photon emission computed tomography (SPECT) to measure the effect of an amphetamine challenge (Laruelle et al., 1995) and drug occupancy (Seibyl et al., 1996) with [123I]iodobenzamide.
In this study, we evaluated the ability of the B/I method to measure stimulus-induced changes in specific binding with a short-lived tracer by using a single positron emission tomography (PET) study. We used both bolus and B/I studies in rhesus monkeys and assessed the impact of dopamine release from an amphetamine challenge on [11C]raclopride-specific binding. A preliminary report of this work has been published (Carson et al., 1995).
MATERIALS AND METHODS
Radiochemistry
[11C]raclopride was synthesized by using an adaptation of the method of Ehrin et al. (1987). The product was separated using a semiprep HPLC column [YMC Basic, YMC Inc, Wilmington, NC, U.S.A.; C18, 5 μm (10 × 250 mm)] eluted with a mobile phase of 30 ml CH3CN, 70 ml H2O, 0.5 ml acetic acid, and 0.63 g ammonium formate at 8 ml/min. This system separates the product from precursor. The time of synthesis was ∼40 min with a radiochemical yield of ∼25% at end of bombardment, Quality control was completed before injection, and the radiopurity was usually >98%. The specific activity was 2,098 ± 882 Ci/mmol at end of bombardment, 507 ± 249 Ci/mmol at end of synthesis, and 363 ± 181 Ci/mmol at time of injection.
Animal Studies
Eighteen PET studies were performed in eight rhesus monkeys (∼10 kg) on 13 scanning days. When two studies were performed in the same monkey on one day, the [11C]raclopride injections were separated by ∼3 h. The monkeys were initially anesthetized with ketamine. Endotracheal intubation was performed for control of respiration, an intravenous line was inserted in a distal lower extremity, and a femoral artery catheter was inserted on the contralateral side by either percutaneous puncture or cutdown, and sutured in place. The monkeys were transported to the PET suite, placed under isoflurane anesthesia, and positioned on the scanning table. Blood pressure, ECG, temperature, and end-tidal P
Administration of raclopride and amphetamine
Eight studies were performed with bolus administration, and 10 studies used B/I. For bolus studies, 2–5 mCi of [11C]raclopride was administered i.v. over a 1-min period by infusion pump. When amphetamine was administered in conjunction with bolus studies (n = 3), 0.4 mg/kg was given as an i.v. bolus 5 min before the raclopride injection (Dewey et al., 1993).
In the B/I studies, a computer-controlled pump (Harvard model 22; Harvard Instruments, South Natick, MA, U.S.A.) was used to administer the radioactivity. First a priming volume was delivered at the highest pump speed (14 ml/min) to fill the catheter dead space. The bolus portion of the dose was administered over 1 min. Pump speed was changed by computer at that time to administer the remaining dose uniformly until 90 min. The monkeys received 2–6 mCi from the B/I administrations. In five studies, 0.4 mg/kg of amphetamine was administered 40 min after the beginning of B/I administration. In one study, 400 μg (∼1.2 μmol) of unlabeled raclopride was administered i.v. at 40 min to demonstrate a near-maximal displacement of radiotracer.
The B/I protocol requires the definition of one parameter, Kbol, the magnitude of the bolus component of the dose (Carson et al., 1993a). This parameter has units of time and defines the bolus portion of the dose to be equal to Kbol min worth of infusate. The selection of the Kbol value was based on analysis of the tissue response curves from the bolus experiments. The predicted tissue response for B/I administration was calculated for basal ganglia and cerebellum as a function of Kbol. The optimal Kbol value of 60 min was chosen to achieve constant radioactivity levels in both regions in the shortest time.
Blood sampling and metabolite determination
Following [11C]raclopride administration, serial arterial blood samples were withdrawn over 90 min (blood data were not available in three of 18 studies). Samples were centrifuged, and 0.1 ml of plasma was counted in a calibrated gamma counter. Five to six samples were analyzed for metabolite fraction by HPLC (de Bartolomeis et al., 1996). Briefly, 0.3 ml of plasma was mixed with 0.2 ml of acetonitrile and 50 μl of cold raclopride. These samples were vortexed (>15 s), centrifuged at 26,000 g for 3 min, and an 80-μl sample was injected onto the HPLC. Twenty fractions were collected, counted with a gamma counter, and corrected for background and decay. The location of the raclopride peak was determined by processing a reference sample consisting of ∼5 μCi of [11C]raclopride added to 5 ml of nonradioactive blood. The location of this peak was confirmed by UV detection of the cold raclopride in the HPLC analysis. The fraction of unmetabolized [11C]raclopride for each time point was calculated from the ratio of the HPLC counts in the raclopride peak divided by the total counts in the HPLC samples. These fractions were then normalized to the value obtained from the reference sample.
To perform the compartment-modeling analysis, a continuous input function is required. To generate a continuous curve from the 5–6 HPLC measurements, we used a method based on that of Huang et al. (1991). Figure 1 shows the whole-body model used to describe raclopride metabolites. The first compartment represents the unmetabolized raclopride in plasma, Cp. This activity can enter a single-body compartment (Ctis) with rate constant K1. From this compartment, unmetabolized raclopride can return to plasma (rate constant k2) or metabolites (Cmet) can enter the plasma (rate constant k3) and be subsequently cleared (rate constant k4). The measured total radioactivity in the plasma Ctot is assumed equal to Cp + Cmet.
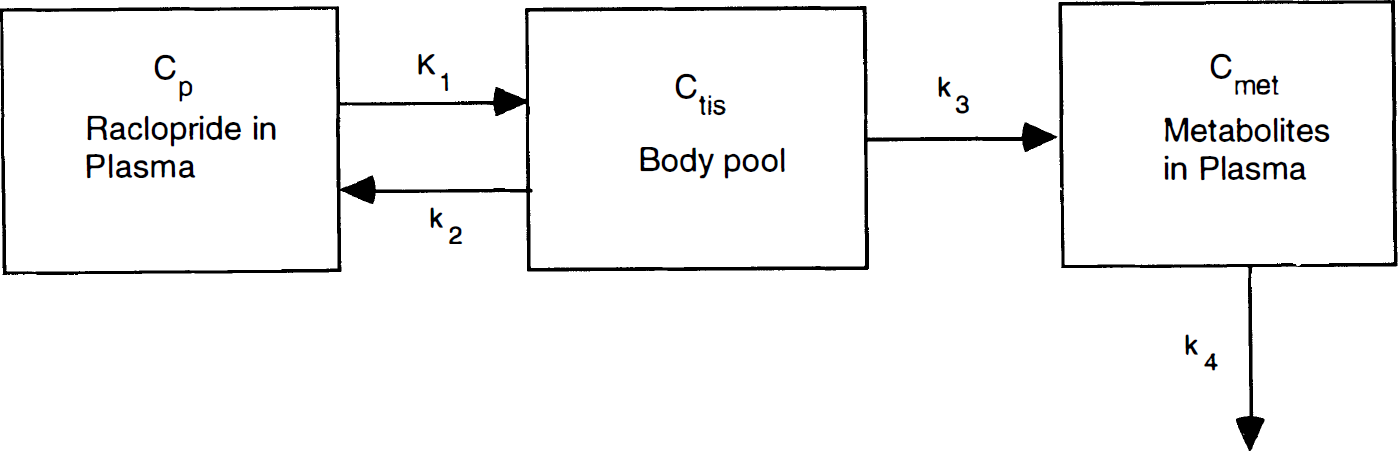
Model used for analysis of raclopride plasma metabolite data. [11C]Raclopride in plasma (Cp) exchanges with raclopride in the body pool (Ctis) from which metabolites in plasma (Cmet) are produced.
To fit the model of Fig. 1 to the HPLC data, the differential equations for Ctis and Cmet were derived. Since the input function Cp is unknown, this term is replaced by (Ctot − Cmet) and the system is solved analytically. The metabolite term has the form
The HPLC fractions of unmetabolized raclopride were fit to (1 − Cmet/Ctot) with a three-parameter fit (A, λ1, λ2), and this fitted fraction curve was then used to correct the plasma radioactivity data. Note that since there was no observation of the body pool, all four model parameters were not identifiable.
Plasma-free fraction determination
The fraction of [11C]raclopride in plasma bound to plasma proteins was determined by ultrafiltration (Sawada et al., 1990); 300 μl of plasma from the reference sample was applied to a Centrifree micropartition membrane (Amicon, Danvers, MA, U.S.A.) and centrifuged for 15 min at 2,000 g. The free fraction in plasma fp, was calculated from Cu/Cp, where Cu is the concentration in the ultrafiltrate (nCi/ml plasma water). Measured fp values were 0.124 ± .043 ml plasma/ml plasma water (n = 15), much higher than values measured in humans of 0.043–0.06 (Farde et al., 1989).
PET scanning procedure
Scans were performed with the Scanditronix PC2048-15B brain tomograph (Litton et al., 1990), which acquires 15 simultaneous slices, 6.5 mm apart. The reconstructed in-plane resolution is 7 mm, and the axial slice width is 6 mm. Transmission scans were acquired with a rotating rod source. Dynamic scans were acquired beginning with tracer injection. Image reconstruction included corrections for attenuation, scatter, randoms, and dead time. Pixel values were calibrated in nCi/ml with a uniform phantom filled with 18F. Scan frames were summed from 0 to 6 min after injection and from 20 to 40 min to provide high-quality images for the purpose of identifying regions of interest (ROIs). Circular ROIs (84 mm2) were placed on the left and right caudate and putamen. Irregular ROIs were drawn on the cerebellum on two adjacent slices. Decay corrected time–activity curves were generated and averaged curves for basal ganglia and cerebellum were produced.
Data analysis
The primary parameter extracted from the data in this work is the total volume of distribution (VT), that is, the equilibrium concentration ratio of tissue to metabolite-corrected plasma. For bolus studies, estimates of VT were derived in three ways: (a) The tissue data Ct from 0 to 60 min was fit to a two-parameter, one-compartment model producing estimates of K1 (ml/min/ml) and k2 (min–1). VT was calculated as K1/k2. (b) Fits were performed to a four-parameter, two-compartment model (Farde et al., 1989) for K1, k2, k3, and k4, and VT was calculated from K1/k2 (1 + k3/k4). (c) Estimates of VT were obtained by graphical analysis (Logan et al., 1990), that is, the slope of (∫Ct)/Ct versus (∫Ct)/Cp using data obtained from 10 to 40 min after injection. For nonlinear parameter estimation, the tissue model included integration over each scan interval, and data were weighted by the inverse of the estimate of ROI variance (Carson et al., 1993b). No correction for vascular radioactivity was made in the model fits so that the estimates of VT could be directly compared to those from graphical analysis or tissue-to-plasma ratios in the B/I studies (see Discussion). These methods were applied to control (n = 5) and postamphetamine (n = 3) bolus studies.
For B/I studies, two types of analysis were performed. First, VT was estimated in the same manner as bolus data, except that only 40 min of data were used so that the preintervention portion of the amphetamine studies as well as the control studies could be used to estimate baseline binding levels. In addition, based on the near constant radioactivity levels achieved by the B/I administration method, the data were analyzed assuming that equilibrium was attained, so that VT was measured directly from the concentration ratio of tissue to metabolite-corrected plasma. In all studies, a control value was obtained by the concentration ratio obtained from 20 to 40, 25 to 40, and 30 to 40 min into the study. For studies where amphetamine was administered at 40 min, postamphetamine VT values were determined by averaging data collected from 70 to 90 min. For control studies, VT values from 70 to 90 min were compared to pre-amphetamine levels as a measure of test–retest variability.
Specific Binding Measures
Total ROI radioactivity is composed of free, nonspecifically bound, specifically bound, and intravascular tracer. Following the nomenclature in Carson et al. (1993a), the physiological interpretatoin of VT, the equilibrium tissue-to-plasma concentration ratio, for a receptor-binding radioligand is
where fp is the plasma-free fraction, ωt is the tissue water content, Keq is the association equilibrium constant for nonspecific binding (k5/k6 in other nomenclature), Bmax is the free receptor concentration (nM), KD is the receptor disassociation equilibrium constant (nM), and Vv is the vascular volume per unit of tissue. If the volume of distribution in a region with no specific binding (VcT) is also determined, then three direct measures of specific binding can be derived.
The three measures S, S′, and R are all linearly proportional to the binding potential, Bmax/KD. The physiological intepretations of these measures, given on the right-hand side of Eq. 3, assume that both the nonspecific binding (Keq) and the vascular volume (Vv) are the same in specific and nonspecific regions. The relative utility of the three measures depends on the variability of their measurement and the population variability of extraneous parameters (for example, fp, Keq) that contribute to their values. The measure S makes no correction for plasma-free fraction and is valuable if the population variability in fp is small. S′ corrects for free fraction and is valuable if this measurement can be made reliably. The ratio measure R is most valuable if nonspecific binding and vascular uptake are similar across all subjects. It is also the least sensitive to errors in the measurement of the input function since such errors will have similar effects on the estimates of VT and VcT. Note that when VT is measured from the tissue-to-plasma ratio in the B/I protocol, no measurement of plasma radioactivity is required to calculate R. The three measures S, S′, and R have different units. The magnitude of the amphetamine effect was determined by calculating the percent reduction in S, S′, and R for bolus and B/I studies.
RESULTS
Metabolite data
Figure 2A shows an example of typical plasma unmetabolized fraction from the HPLC analysis for a bolus injection of [11C]raclopride. The appearance of plasma metabolites is substantially more rapid in rhesus monkeys than that reported for humans (Farde et al., 1989). The unmetabolized fractions at 10 and 40 min after injection were 0.67 ± 0.08 and 0.21 ± 0.09, respectively. In three monkeys in which paired-control and amphetamine studies were performed on the same day, there was a trend toward more rapid metabolism following amphetamine administration, with unmetabolized fractions reduced by 16% from 10 to 40 min after injection (p = 0.09). Note that since the input function and metabolite correction were measured for every study, any changes in brain uptake caused by amphetamine effects on the input function are appropriately corrected by the model.
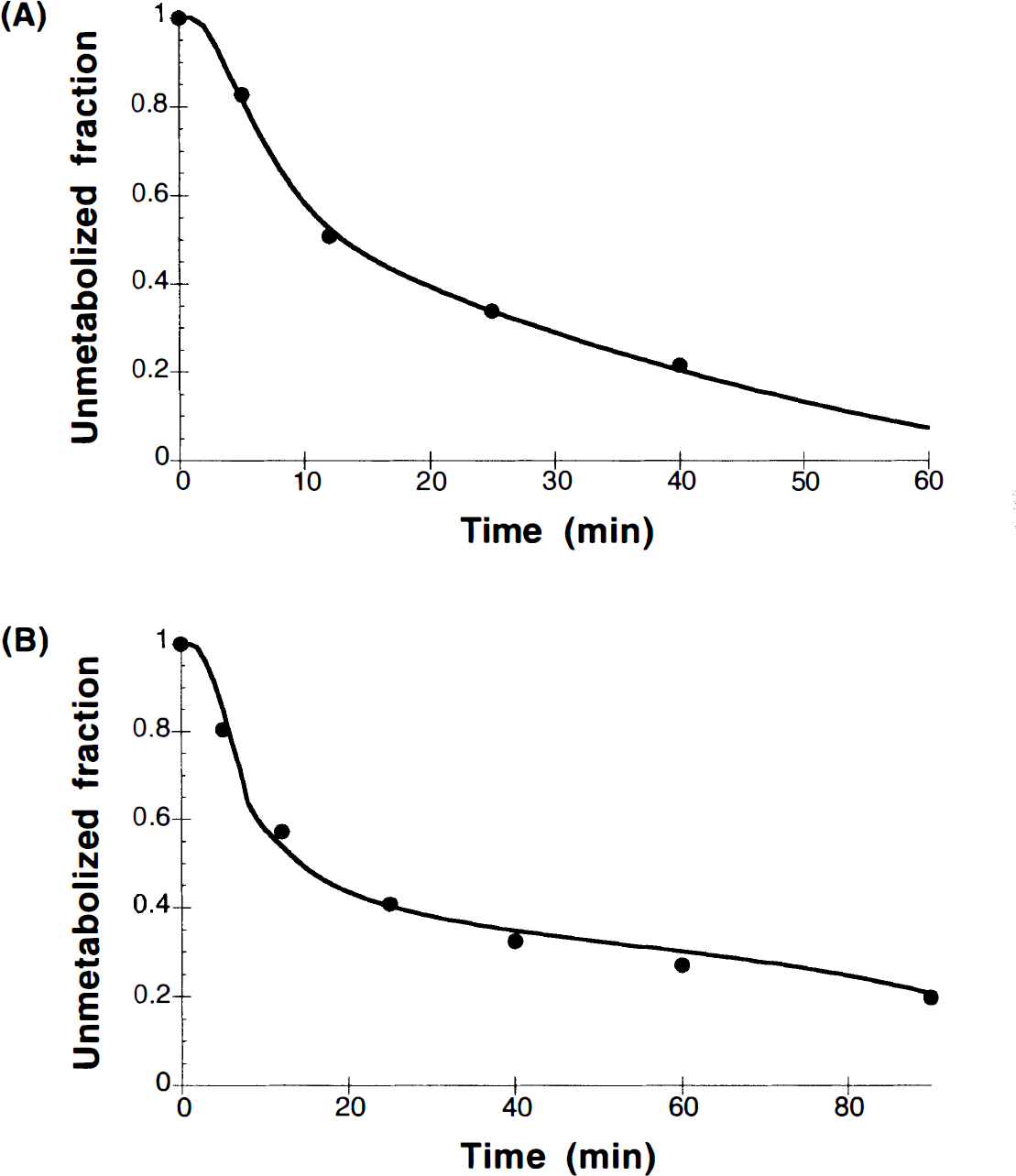
The metabolite model based on Eq. 1 was applied to the HPLC data to produce a continuous input function. This model was more than adequate to describe the data. In fact, the three-parameter metabolite model fits showed very high correlation between the parameters with difficulty in achieving convergence. This is not surprising due to the limited number of data points and the relatively poor statistical quality of the late HPLC data. To simplify the model further, we tested whether we could ignore the clearance of metabolites from blood. Applying the assumption of k4 = 0 to the metabolite model allowed a reduction from three-parameters to two-parameters, in that the leading constant A in Eq. 1 can be expressed in terms of the eigenvalues (λ1, λ2) of the system:
This two-parameter metabolite model was sufficient to describe the data, and an example of the fit is shown in Fig. 2A.
Not surprisingly, in the B/I studies, the unmetabolized fraction in plasma was higher at later times than in the bolus studies because of the continuous delivery of tracer. At 10 and 40 min, the unmetabolized fractions for B/I administrations were 0.70 ± 0.15 and 0.42 ± 0.13, respectively. In the B/I studies, beyond 10–15 min, the total radioactivity in plasma was increasing while the unmetabolized fraction was decreasing. The same metabolite model was able to describe the B/I metabolite data, and an example is shown in Fig. 2B. The ability of this model to describe both bolus and B/I metabolite data is not unexpected since the metabolite model explicitly includes the measured total radioactivity curve.
Bolus studies
Figure 3 shows an example of model fits for basal ganglia and cerebellar regions to the one-compartment, two-parameter model (dashed line) and the two-compartment, four-parameter model (solid line). As in this figure, it was evident that the simpler model did not adequately describe the time–activity data. Mean and percent standard deviation values for VT in the basal ganglia and cerebellum are listed in Table 1. For both regions, the estimates for the four-parameter model were larger than those for the two-parameter model (p < 0.01). VT estimates based on Logan's graphical analysis are also presented in the table. The graphical estimate for the basal ganglia was in good agreement with the two-parameter model and significantly lower than the four-parameter values. Conversely, the graphical estimate for the cerebellum was in good agreement with the four-parameter model and significantly higher than the two-parameter model. The variability of the estimates was ∼25% and was similar across the different methods.
Raclopride control binding: bolus studies a

Mean values (n = 5) with percent standard deviations in parentheses.
Rows 1 and 2: two-parameter and four-parameter model fits to data from 0 to 60 min. Row 3: volumes of distribution from graphical analysis of data from 10 to 40 min.
Specific binding measures S (ml tissue/ml plasma), S′ (ml tissue/ml plasma water), and R (dimensionless) are defined in Eq. 3.
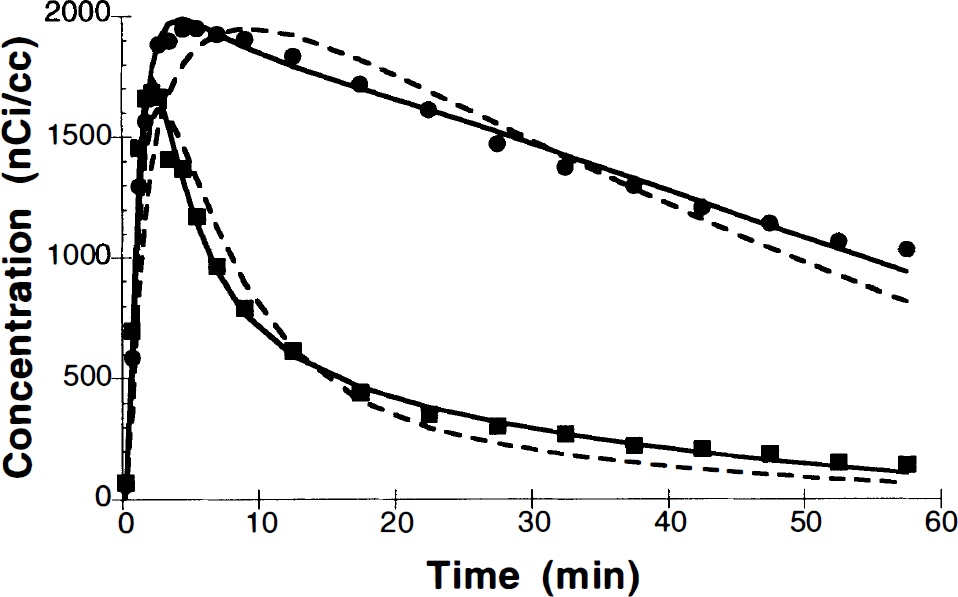
Region-of-interest data from basal ganglia (•) and cerebellum (▪) from a bolus study (4.7-mCi injection). The dashed line is fit to the two-parameter model, and the solid line is the best fit to the four-parameter model.
The three measures of specific binding derived from the VT estimates (Eq. 3) are also listed in Table 1. Theoretically, all of these measures are linearly proportional to Bmax/KD. Note that due to the large radioactive dose given in these monkeys, a fraction of the receptors was occupied by unlabeled raclopride. Assuming a KD value of 10 nM (Farde et al., 1989), and using the cerebellum activity from 20 to 40 min and the specific activity to estimate unbound tracer concentration, we calculate that unlabeled raclopride occupied 10 ± 4% of the receptors, resulting in somewhat smaller values for specific binding. The choice of model had a relatively small effect on the mean values of each measure, with only a 10% range in the specific binding estimates that use plasma directly (S and S′) and a 20% range in the ratio measure R. Interanimal variability was highest for the four-parameter model. For bolus data, as shown previously (Logan et al., 1994), variability was lowest for the ratio measure R. Including a correction for plasma-free fraction (S′) did not reduce the interanimal variability compared to S for the bolus data. Note that there was a weak positive correlation between the measured free fraction and VcT across bolus and B/I studies (r = 0.34, n = 15, p = 0.10). Previously, a linear relationship between VcT and fp had been demonstrated in human studies (Farde et al., 1989).
In three monkeys, paired-control and amphetamine bolus studies were performed on the same day, with injections separated by 3 h. Table 2 shows the calculated percent reductions in basal ganglia specific binding due to the amphetamine-induced increase in synaptic dopamine. Depending on the method chosen, the 0.4-mg/kg dose of amphetamine reduced specific binding by 22–42%. These results are in reasonable agreement with specific binding reductions of ∼24% found by Dewey et al. (1993) (reported as a 16% reduction in the basal ganglia-to-cerebellum ratio) although the amphetamine dose in our study was lower (0.4 vs. 1.0 mg/kg). The interanimal variability in the response is large, so with this small sample it is difficult to compare methods. Overall, the ratio measure R showed the smallest interanimal variability.
Amphetamine-induced percent reductions in raclopride specific binding: bolus studies a
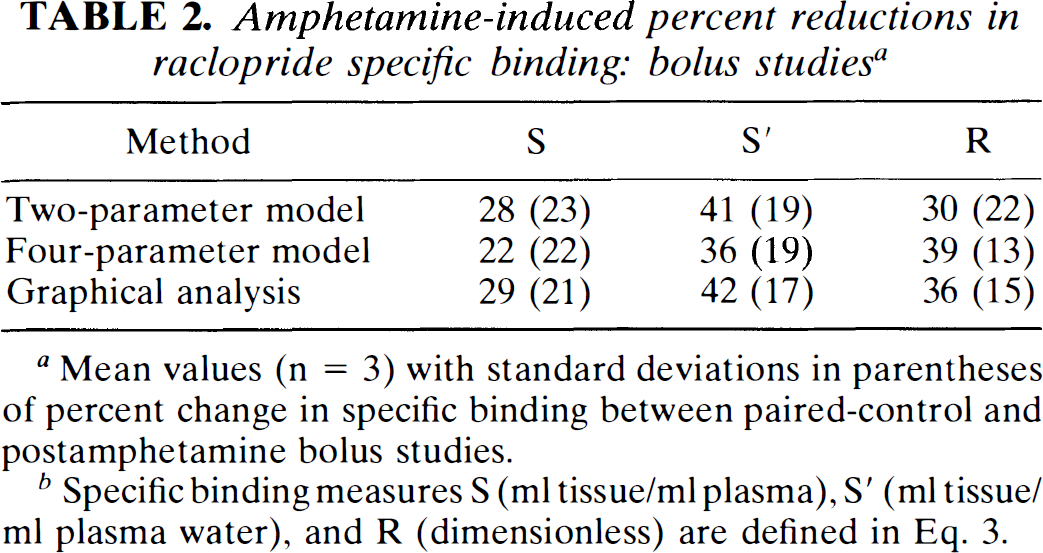
Mean values (n = 3) with standard deviations in parentheses of percent change in specific binding between paired-control and postamphetamine bolus studies.
Specific binding measures S (ml tissue/ml plasma), S′ (ml tissue/ml plasma water), and R (dimensionless) are defined in Eq. 3.
B/I studies
Four control B/I studies were performed. Figure 4 shows an example of the time–activity data from basal ganglia and cerebellum for a B/I study. This figure shows an ideal behavior of the B/I method where the bolus phase rapidly achieves tissue concentration levels in basal ganglia and cerebellum between the eventual equilibrium levels for these regions. Ideally, this initial level is closer to the equilibrium level for the basal ganglia since, in general, tissue kinetics are faster for the “nonspecific” regions. To examine the level of equilibrium, time–activity data from 30 to 90 min were fit to a single exponential function and the “clearance” rate (ideally 0) was calculated. For cerebellum and basal ganglia, the rates were −0.01 ± 0.21%/min and −0.03 ± 0.15%/min, respectively.
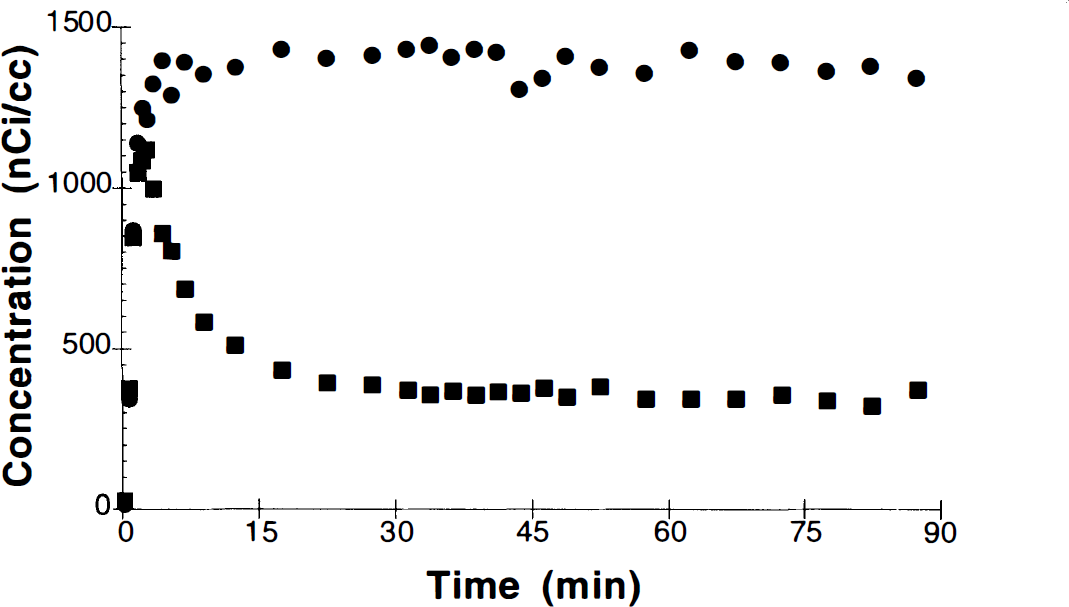
Region-of-interest data from basal ganglia (•) and cerebellum (▪) following bolus plus infusion administration of [11C]raclopride (administered dose, 6.1 mCi). The bolus portion of the dose (Kbol) was equal to the volume administered during 60 min of infusion.
Figure 5 shows the results of a single study used to determine the magnitude of displaceable binding by using the B/I protocol. At 40 min, 400 μg of unlabeled raclopride was injected as an i.v. bolus. By 60–75 min, at least 75% of the specific binding (basal ganglia − cerebellum) was displaced. The apparent rise in the basal ganglia curve beyond this point is due to continued infusion of [11C]raclopride during the clearance of the unlabeled raclopride.
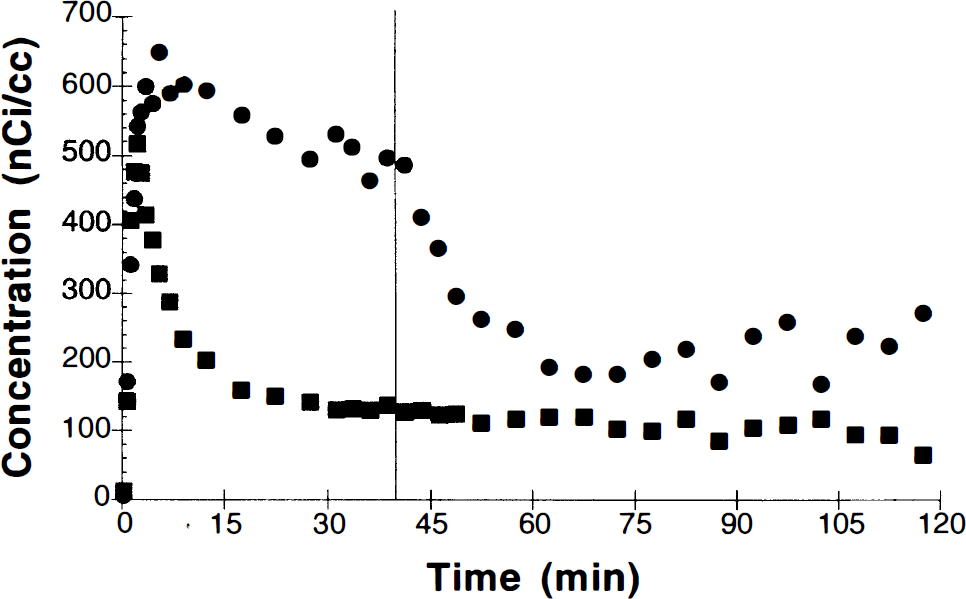
Region-of-interest data from basal ganglia (•) and cerebellum (▪) following bolus plus infusion administration of [11C]raclopride (administered dose, 2.0 mCi). At 40 min, 400 μg of unlabeled raclopride were administered as an i.v. bolus.
Control values for volume of distribution from B/I studies are presented in Table 3. Data are tabulated from seven studies (three control and four amphetamine studies) with two other studies not used due to lack of arterial blood data. VT values were derived from model fits, graphical analysis, and the tissue-to-plasma ratios. There was generally good agreement between the bolus and B/I data in the values of VT both in mean and in percent variability. As in the bolus case, it was clear that the two-parameter model did not adequately describe the time–activity curves (data not shown). For both basal ganglia and cerebellum, the VT estimates for the four-parameter model and graphical analysis were larger than those for the two-parameter model (p < 0.01). There was no statistically significant difference between the four-parameter and graphical analysis results. Furthermore, there were no statistically significant differences between the VT estimates between bolus (Table 1) and B/I (Table 3) control studies for model fits or graphical analysis.
Raclopride control binding: bolus plus infusion studies a
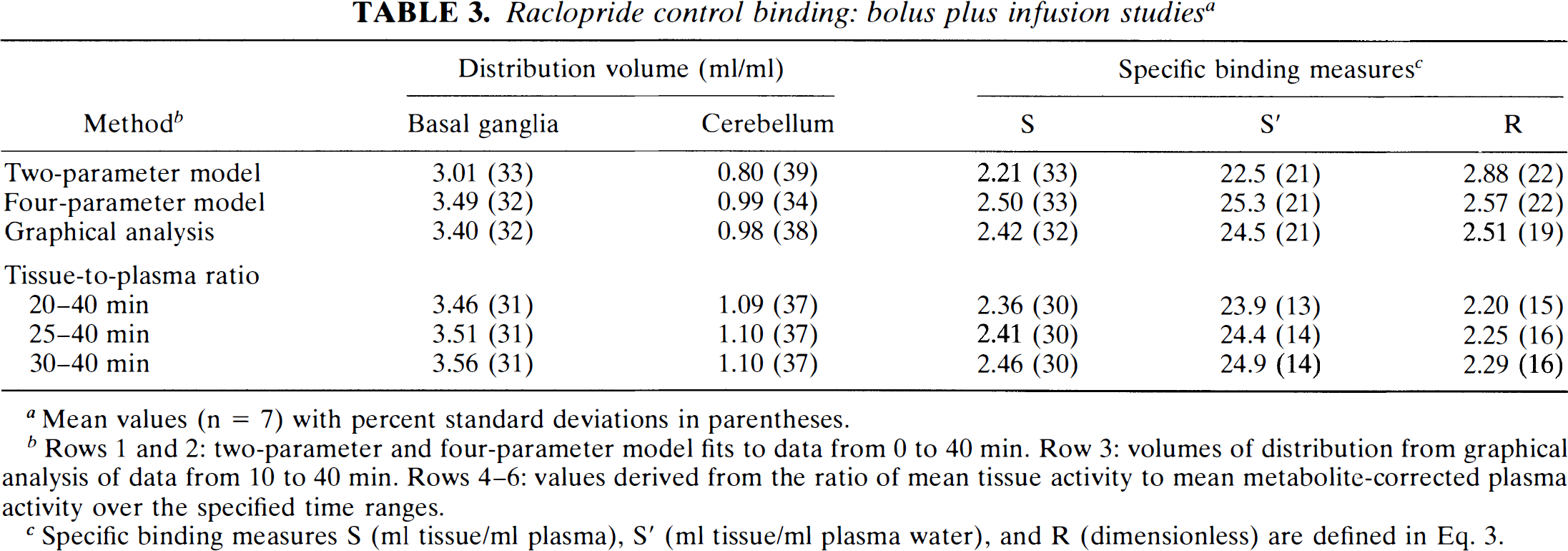
Mean values (n = 7) with percent standard deviations in parentheses.
Rows 1 and 2: two-parameter and four-parameter model fits to data from 0 to 40 min. Row 3: volumes of distribution from graphical analysis of data from 10 to 40 min. Rows 4–6: values derived from the ratio of mean tissue activity to mean metabolite-corrected plasma activity over the specified time ranges.
Specific binding measures S (ml tissue/ml plasma), S′ (ml tissue/ml plasma water), and R (dimensionless) are defined in Eq. 3.
Distribution volume estimates from the ratio of mean tissue activity to mean metabolite-corrected plasma activity from 20 to 40, 25 to 40, and 30 to 40 min after injection are also shown in Table 3. For basal ganglia, VT values increased slightly when the earlier data (20–30 min) were excluded, probably because the specific binding regions had not reached equilibrium by 20 min. For quantification of control binding, the time period 25–40 min was chosen as a compromise between achieving equilibrium and maintaining statistical quality. Using the four-parameter model values for comparison, there was no statistically significant difference for the basal ganglia tissue-to-plasma ratios. However, the cerebellum VT values were significantly higher (p < 0.05, paired t test), although the magnitude of this difference was only 10%. Some of this difference may be due to the effects of intravascular radioactivity (see Discussion).
For the specific binding measures based on fits or graphical analysis, there were no statistically significant differences of corresponding values (for example, S measures from four-parameter fit) between bolus and B/I studies. There was a trend for differences in the R measures (p < 0.10). When comparing specific binding measures in B/I studies, there were no significant differences for S or S′ from tissue-to-plasma ratios (25–40 min) to the fitted values. However, the R measure based on tissue-to-plasma ratios was lower than those based on model values (p < 0.05, paired t test). We attribute these differences with the R specific binding ratio to a combination of incomplete equilibrium and the effects of intravascular radioactivity (see Discussion). For the B/I data, the R and S′ measures had comparable intersubject variability, smaller than the S measure, suggesting that the correction for free fraction in the S′ measure was useful.
In five B/I studies, 0.4 mg/kg amphetamine was administered at 40 min. An example of the time–activity curves is shown in Fig. 6. A break in the basal ganglia curve is evident, consistent with displacement of [11C]raclopride by increased synaptic dopamine. The radioactivity concentration in basal ganglia appears to approach a new equilibrium level by 60–75 min. Postamphetamine VT values were calculated from the ratio of mean tissue activity to mean metabolite-corrected plasma activity from 70 to 90 min. From these values, measures of specific binding and the percent change in specific binding with respect to control (25–40 min) were determined. The receptor measures that require plasma activity (S and S′) were extremely noisy, and estimates of the percent reduction in specific binding had >100% uncertainty. However, the ratio measure R, which for B/I studies is independent of plasma data, showed smaller variability. For amphetamine studies (n = 5), specific binding was reduced by 19 ± 16%. For control B/I studies (n = 4), the mean R measure slightly increased (between 25–40 min and 70–90 min) by 5 ± 13%. These amphetamine reductions are significantly different than control (p < 0.05, unpaired t test). The amphetamine-induced changes in specific binding for both the bolus and B/I studies using the R measure are shown in Fig. 7. There was no statistically significant difference between the amphetamine-induced specific binding reductions in the bolus studies (22–42%) and B/I studies (19%), although the number of studies was small and the interanimal variability was large. The intersubject variability was comparable for the R measure between bolus and B/I studies.
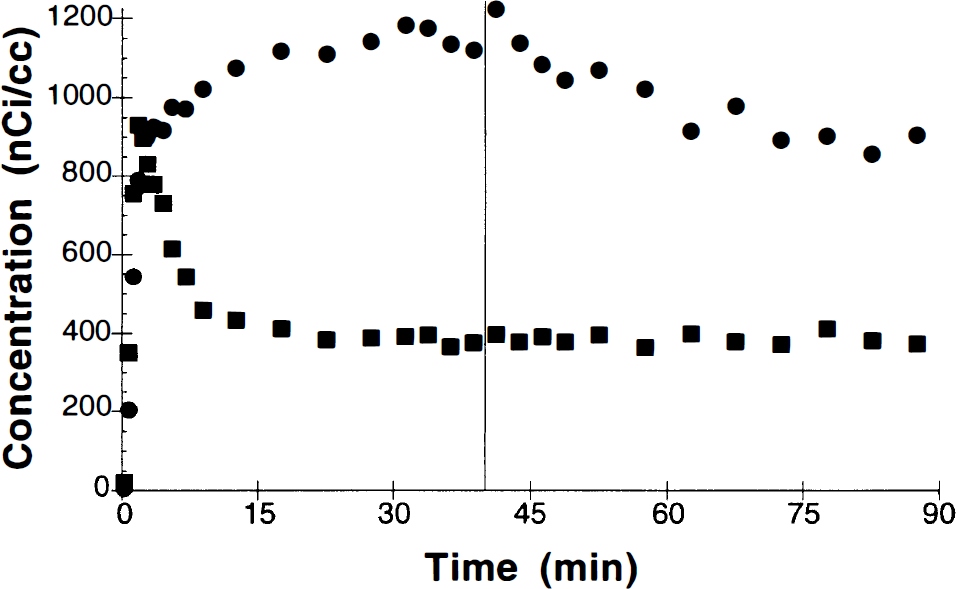
Region-of-interest data from basal ganglia (•) and cerebellum (▪) single bolus plus infusion administration of [11C]raclopride (administered dose, 6.0 mCi). At 40 min, 0.4 mg/kg of amphetamine was administered i.v., producing displacement of raclopride due to competition with increased dopamine.
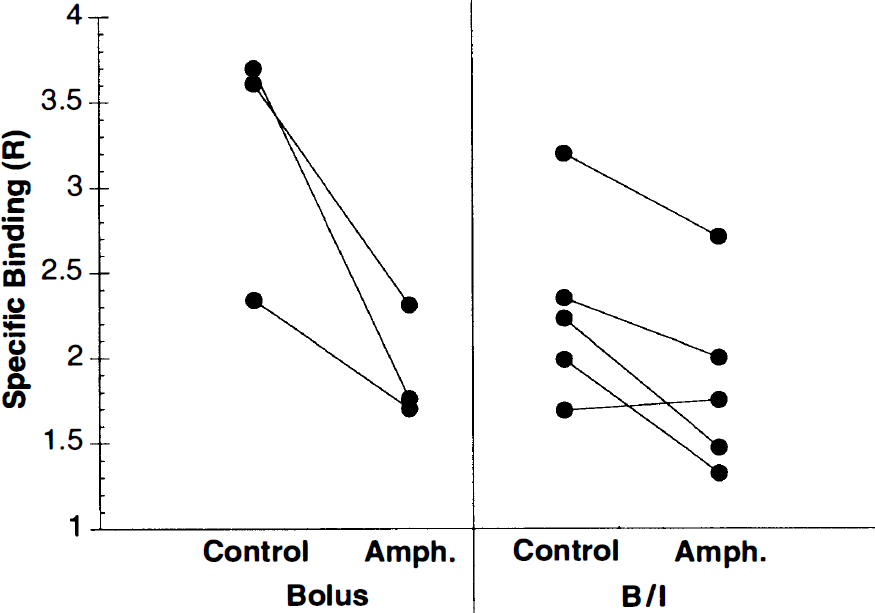
Amphetamine-induced changes in specific binding using the R measure (Eq. 3). The solid lines connect paired studies from the same monkeys. Bolus results were obtained from the four-parameter model fits using two [11C]raclopride injections. Results of single bolus plus infusion (B/I) were based on tissue-to-plasma ratios from a single study.
Plasma amphetamine levels were measured in the B/I studies. The correlation coefficients between the percent change in the specific binding measure R and plasma amphetamine levels at 60 and 90 min (20 and 50 min after amphetamine, respectively) were 0.67 and 0.52. Although these coefficients were not statistically significant (n = 5), there is the suggestion that higher availability of amphetamine produces a larger dopamine pulse and a larger reduction in [11C]raclopride-specific binding.
DISCUSSION
In vivo imaging studies with receptor-binding radiopharmaceuticals can be used to assess changes in neurotransmitter levels or drug occupancy of receptors. As one example, amphetamine-induced increases in extracellular dopamine have been extensively studied (Young et al., 1991; Innis et al., 1992; Hume et al., 1992; Wong et al., 1992; Dewey et al., 1993; Laruelle et al., 1995). This study compared two tracer administration schemes—bolus and B/I—for the purpose of quantification of changes in free reactor tor concentration with PET. The results single factory agreement between the methods, with the B/I approach enabling the measurement of changes in specific binding from a single study without blood sampling.
[11C] Raclopride metabolite model
A new feature presented in this analysis was a model for the plasma metabolite profile for [11C]raclopride. We consider the use of a metabolite model in this study important because (a) a simple empirical function, such as a multiexponential fit, might not describe both the bolus and B/I metabolite data; and (b) the late sample data have poor statistical quality due to 11C decay. A two-parameter fit of the HPLC fractions (Fig. 2) describes the data well for both bolus and B/I studies, although given the number of data points and their statistical quality, a careful evaluation of this model was not possible. Note that this metabolite model is sensitive to noise or sampling errors in the total plasma radioactivity curve, that is, breaks in the measured plasma curve are enhanced. In some cases, preliminary smoothing of the total plasma radioactivity data was performed to minimize this effect.
Volume of distribution
Several different quantification approaches have been employed for [11C]raclopride data (Farde et al., 1989; Logan et al., 1994; Lammertsma et al., 1996). Generally, compartment model fits using metabolite-corrected input functions have 45 two-tissue compartments and the estimation of 4–5 parameters. In both bolus and B/I studies, the model with two tissue compartments produced consistently higher values for VT than did the model with one tissue compartment, similar to the results of Lammertsma et al. (1996). The absolute values of VT and VcT in these monkey studies are similar to those in baboons (Dewey et al., 1993) and higher than those in humans (Farde et al., 1989; Lammertsma et al., 1996), most likely due to the higher plasma-free fraction in nonhuman primates (Eq. 2).
As has also been shown for other ligands (Carson et al., 1993a), there was generally good agreement across all of the different techniques to measure VT and VcT, that is, model fits and graphical analysis for bolus and B/I studies as well as tissue-to-plasma ratios for B/I. Any substantial differences would, in fact, have been quite surprising, since these approaches were designed to estimate the same parameter. Nevertheless, this good correspondence of results lends support to the modeling approaches and the accuracy of the input function measurements. Note that there was a slight lack of equilibrium in the basal ganglia B/I data so that extending the time period for measures of control binding levels (or slightly increasing Kbol) may be appropriate.
No attempt was made to correct for intravascular activity in order to enable direct comparison between all VT values. Unlike tracers with higher distribution volumes, intravascular activity does make a nontrivial contribution to VT estimates for [11C]raclopride, particularly in the cerebellum. For example, at 40 min into a B/I study, where the unmetabolized fraction is ∼ 42%, a 5% blood volume would correspond to an increase in VT and VcT values of 0.12, or 10–15% of the cerebellum value. Simulations of the B/I studies (data not shown) suggest that this bias caused by intravascular activity should be larger for the tissue-to-plasma ratios than the fitted VT values since the bias is distributed into all the parameter values for the fitting method. Also, since at late times intravascular activity is higher for B/I studies than for bolus studies, the bias may be larger for B/I, contributing to the trend toward differences in the R measure due to overestimation of VcT values. In human studies, this bias will be of comparable magnitude even with lower tracer metabolism rate because of the lower tissue uptake due to higher plasma protein binding. For measures of specific binding, however, this issue is of little importance. If the intravascular volumes for basal ganglia and cerebellum are identical, this bias in VT values would produce no error in the S or S′ specific binding measures and, in fact, no differences were detected with these measures between methods. This bias will affect the ratio measure R (see Eq. 3), but such an error will be unimportant to the extent that blood volume (and nonspecific binding) is uniform across subjects.
Measures of specific binding
We used three different mathematical functions (Eq. 3) to derive measures that are linearly proportional to the binding potential, Bmax/KD. As in most medical imaging procedures, using the measure with the “purest” physiological interpretation (in this case S′) does not always produce the most useful biological measure, that is, the one with the best signal-to-noise characteristics. As shown previously (Logan et al., 1994), the smallest coefficient of variation was obtained with the R measure. Comparable variability was seen for the S′ measure in the B/I control studies, but this did not occur in the bolus data. Furthermore, the statistical quality of the late metabolite samples caused large variation in measures dependent on the blood data for B/I studies. For other tracers, particularly with longer half-lives or less metabolism, these other measures (S and S′) may have less variability.
Amphetamine-induced reduction in specific binding
Using the R measure of specific binding, we found a 22–42% reduction in specific binding in bolus studies and a 19% reduction in the B/I studies. Although there was no statistically significant difference between these values, we expected that the bolus and B/I protocols may have different sensitivity to the dopamine pulse because of the difference in timing of the amphetamine administration with respect to [11C]raclopride delivery. The interpretation of VT estimates from bolus or B/I studies is based on the assumption that all kinetic parameters are constant during the study. Thus, exact agreement between bolus and B/I methods might be expected only if amphetamine produced a prolonged near-constant synaptic dopamine level. Microdialysis studies in nonhuman primates have shown a time-varying extracellular dopamine concentration following i.v. administration of 1 mg/kg amphetamine (Moghaddam et al., 1993). In a parallel set of studies, we performed microdialysis measurements of extracellular dopamine in rhesus monkeys simultaneously with [11C]raclopride–PET during amphetamine challenge (Endres et al., 1996) and showed that extracellular dopamine rose rapidly and returned to near baseline levels within 40 min. This changing dopamine concentration will produce a time-dependent change in free receptor concentration. Since the free receptor concentration is not constant in postamphetamine studies, it is likely that VT estimates represent a time-weighted average of the free receptor concentration (Eq. 2), similar to the manner in which a time-varying glucose metabolic rate affects estimates using fluorodeoxyglucose (Huang et al., 1981). This weighted average will be different for bolus and B/I studies.
In the B/I studies, the time–activity curves (Fig. 6) show more directly the effects of increased synaptic dopamine on specific binding. The break in the basal ganglia curve is due to the displacement of [11C]raclopride by dopamine. Ideally, the tracer curve would be a mirror image of the endogenous dopamine curve. However, the tissue radioactivity curve can only change if [11C]raclopride molecules exit the brain; thus, the kinetics of raclopride clearance as well as the synaptic dopamine curve contribute to the shape of the PET time–activity data. For example, in the B/I studies, the tissue concentration in the basal ganglia does not begin to return to its baseline value within the 50 min of data collected after amphetamine administration despite the rapid kinetics of the amphetamine-induced dopamine pulse. Similar effects over a longer time scale have been seen with amphetamine displacement of [123I]iodobenzanide and SPECT (Laruelle et al., 1995). A modeling approach for B/I studies incorporating the time course of tissue dopamine (as measured by microdialysis) has been developed by Endres et al. (1996) and showed that an extension of the conventional [11C]raclopride model can account for the measured time course found in the B/I studies. These modeling studies should contribute to the interpretation of measured changes in specific binding in human studies. In addition, they may be helpful in determining the characteristics of neuroreceptor tracers that would be best suited for the measurement of changes in endogenous ligand concentration.
B/I versus bolus administration
Most neuroreceptor measurements have been performed using one or more bolus injections rather than the B/I method. There are a number of considerations in choosing between these two approaches. Quantification of VT for the B/I protocol using the tissue-to-plasma ratio is simpler than for bolus techniques, although the graphical analysis method (Logan et al., 1990) is straightforward. The accuracy of the VT estimates from B/I studies depends on achieving constant radioactivity levels. Although this will never be achieved for all subjects in all tissues with a single infusion protocol, by minimizing the clearance rate with an infusion, the error in the tissue-to-plasma ratios is minimized (Carson et al., 1993a). To estimate specific binding with the R measure, no blood measurements are required for the B/I protocol. Recently, techniques have been developed to calculate specific binding for bolus studies without blood data (Ichise et al., 1996).
In this paradigm, the goal is to measure the change in specific binding between two states. The primary advantage of the B/I methodology is that it enables direct measurement of receptor occupancy changes in vivo in a single study, that is, requiring a single chemical synthesis of a short-lived radiopharmaceutical. By maintaining constant plasma levels during the stimulus, any compounding effects of dynamic flow changes can be minimized, although these effects are likely to be small in [11C]raclopride bolus studies as well (Logan et al., 1994). Also, it may be possible to extract information about the time course of synaptic neurotransmitter changes from the shape of the displacement curve, although this may require the use of a tracer with faster clearance than [11C]raclopride. A disadvantage of the B/I scheme for 11C studies is the poor statistical quality of the late data, particularly when metabolite correction of the plasma is necessary. However, the use of three-dimensional PET acquisition improves the statistical quality of the images and can provide good-quality data with [11C]raclopride in human studies (Breier et al., 1996). In this way, changes in specific binding can be quantified by using the B/I approach and a single radiochemical synthesis of [11C]raclopride in a study of ∼2-h duration.
Footnotes
Acknowledgment:
The authors express their appreciation for the excellent technical assistance of the National Institute of Health PET Department cyclotron, radiochemistry, and technologist staff. The helpful suggestions of Drs. Christopher Endres and Peter Herscovitch are gratefully acknowledged.