Abstract
Reactive oxygen species (ROS) are normally generated in the brain during metabolism, and their production is enhanced by various insults. Low molecular weight antioxidants (LMWA) are one of the defense mechanisms of the living cell against ROS. The reducing capacity of brain tissue (total LMWA) was measured by cyclic voltammetry (CV), which records biological oxidation potential specific to the type of scavenger(s) present and anodic current intensity (Ia), which depends on scavenger concentration. In the present study, the reducing capacity of rat brain following closed head injury (CHI) was measured. In addition, CV of heat-acclimated traumatized rats was used to correlate endogenous cerebroprotection after CHI with LMWA activity. Sham-injured rat brains displayed two anodic potentials: at 350 ± 50 mV (Ia = 0.75 ± 0.06 μA/mg protein) and at 750 ± 50 mV (Ia = 1.00 ± 0.05 μA/mg protein). Following CHI, the anodic waves appeared at the same potentials as in the sham animals. However, within 5 min of CHI, the total reducing capacity was transiently decreased by 40% (p < 0.01). A second dip was detected at 24 h (60%, p < 0.005). By 48 h and at 7 days, the Ia levels normalized. The acclimated rats displayed anodic potentials identical to those of normothermic rats. However, the Ia of both potentials was lower (60% of control, p < 0.001). The Ia profile after CHI was the direct opposite of the normothermic Ia profile: no immediate decrease of Ia and an increase from 4 h and up to 7 days (40–50%, p < 0.001). We suggest that the lowered levels of LMWA in the post-CHI period reflect their consumption due to overproduction of free radicals. The augmented concentration of LMWA found in the brain of the heat-acclimated rats suggests that these rats are better able to cope with these harmful radicals, resulting in a more favorable outcome following CHI.
Traumatic injury to the brain triggers a cascade of events that may result in delayed tissue edema, necrosis, and impaired function. These effects are associated with accumulation of harmful mediators (Siesjö, 1988; Faden and Salzman, 1992; Phillis, 1994). Recently, reactive oxygen species (ROS) have been gaining increased interest as the major candidates responsible for various pathological responses in the pathogenesis of trauma (Demopoulos et al., 1982; Baethmann, 1989; Chan, 1989; Hall and Braughler, 1989; Kontos, 1989; Siesjö et al., 1989; Yukido and Long, 1990). ROS are normally produced in the brain during mitochondrial metabolism. They can also be derived from enzyme activity, either as a result of their catalytic role or as a by-product of their activity (Kontos, 1989). Various insults such as ischemia, degenerative diseases, inflammation, and trauma result in enhanced ROS production. ROS may also be released from activated neutrophils in inflammatory processes or as the result of mobilization of transition metals during and following these injurious events in the brain. It is well documented that exposure of the brain to high levels of ROS may result in irreversible damage to membranes, proteins, and DNA (Halliwel, 1990). To cope with the continuous exposure to ROS, living cells have developed several mechanisms. These include antioxidant enzymes and low molecular weight antioxidants (LMWA; e.g., ascorbate, carnosine, tocopherol, glutathione, NADH, Q10, etc.). While the enzymes remove superoxide radicals and hydrogen peroxide, the LMWA protect essential biological targets from the damage induced by reactive species, such as hydroxyl radicals, peroxyl radicals, nitric oxide radicals, and other activated forms of oxygen, by reducing these active species.
It has been suggested that ROS may be produced in a rat model of closed head injury (CHI). We have demonstrated the activation of the arachidonic acid metabolic cascade (Shohami et al., 1987, 1989) in the posttraumatic period. This metabolic pathway is known to produce free radicals. In addition, stable nitroxide radicals, known to scavenge for endogenous radicals, were injected to rats following CHI and were found to improve the outcome markedly (Beit-Yannai et al., 1996). These data supported the suggestion that ROS produced after CHI are involved in the pathophysiology of this model.
Rats that were acclimated to heat for 30 days showed a better outcome following CHI than matched controls subjected to a similarly severe injury (Shohami et al., 1994). They suffered to a lesser extent from blood–brain barrier disruption and edema formation than controls, and their clinical recovery was faster. Similar protection was also found in the ischemic heart of heat-acclimated rats that showed better endurance accompanied by energy sparing during ischemia and better recovery of contractile function upon reperfusion that coincided with enhanced recovery of the energy pool (Horowitz et al., 1985, 1986; Levi et al., 1993). We thus hypothesize that during the period of acclimation, some endogenous mechanisms develop that later offer protection against the sequelae of trauma. The multiorgan-protective effect achieved in different organs in the acclimated rats suggests that it is mediated by both centrally and peripherally acting factors.
Most of the methods used to evaluate antioxidant activity are related to specific scavengers or are specific for the reactive species involved and are insufficient to indicate the overall antioxidant status of the tissue (Rice-Evans and Miller, 1994). Attempts to develop methods for assessing the total antioxidant activity of biological samples have met with only partial success. Recently, Dirnagl et al. (1995) used lucigenin, which is particularly sensitive to superoxide radical, for in vivo monitoring of ROS production. Most of these methods were designed to determine the antioxidant activity of biological fluids such as plasma, and they indicate the level of a limited number of antioxidant compounds. A different approach to LMWA activity measurements in tissues and fluids was recently developed by Kohen and colleagues. They showed that the antioxidant activity of biological tissues and fluids correlates with their biological oxidation potential and level (anodic current) (Kohen et al., 1992; Kohen, 1993). Therefore, LMWA activity of biological samples can be evaluated by measuring the reducing power of the sample (expressed by both parameters, biological oxidation potential and anodic current). A convenient method of measuring the overall reducing power of biological fluids or tissue homogenate is cyclic voltammetry (CV) (Bard and Faulkner, 1980). The CV tracing records the biological oxidation potential, specific to the type of scavenger(s) present and the anodic current, which depends on the scavenger concentration(s).
The present study was designed to measure the reducing capacity of rat brain following CHI and to assess changes in anodic current during the posttraumatic period. In addition, we compared the reducing ability of control normothermic (CON) and heat-acclimated (ACC) rats in an attempt to correlate endogenous cerebroprotection after CHI with LMWA activity.
MATERIAL AND METHODS
Animals and maintenance
Male Sabra rats (Hebrew University strain) weighing 200–240 g (6–7 weeks old) were used in this study. Animals were maintained according to the regulations of the Animal Care Committee of the Hebrew University, with a 12-h/12-h light/dark reversed light cycle, and food and water were provided ad libitum. The animals were divided into two groups: CON, maintained at an ambient temperature of 24 ± 1°C; and ACC, held in a climatic chamber at 34 ± 1°C for 30 days. This period was found to be long enough for the achievement of a stable acclimated state, characterized by a lower basal metabolic rate, lower heart rate, and improved heat tolerance (Levi et al., 1993).
Trauma model
CHI was induced under ether anesthesia, confirmed by testing corneal and pupillary reflexes as described by Shapira et al. (1988). In brief, CHI was caused by a calibrated weight drop device that was allowed to fall over the exposed skull, covering the left cerebral hemisphere 1–2 mm lateral to the midline, in the midcoronal plane. After recovering from anesthesia, the rats were returned to their home cages with postoperative care and free access to food and water. Sham rats were anesthetized with ether, their skulls were incised, but trauma was not induced.
Experimental protocol
CON and ACC rats were subjected to sham operations or CHI, and at various intervals after CHI or sham operation (5 min to 7 days, n = 8 rats/time point), the rats were killed. Their brains were removed and the hemispheres were separated and homogenized in 1 ml 0.1 M phosphate-buffered saline buffer (pH 7.4). An aliquot was taken for protein determination according to Lowry et al. (1951). To determine changes in reducing capacity in peripheral organs in the post-CHI period, the hearts were removed and a segment of 150 ± 10 mg was homogenized and assayed in the CV, like the brain tissue.
CV analysis
Brain tissue extracts were prepared in 0.1 M phosphate buffer (pH 7.4). CV measurements were carried out using the BAS model CV-1B CV apparatus, modified for a 250-μl cell volume (West Lafayette, IN, U.S.A.). CV tracings were recorded at a range of 0–1.3 V and at a rate of 100 mV/s vs. an Ag/AgCl reference electrode. A three-electrode system was used throughout the study. The working electrode was a glassy carbon disk (BAS MF-2012) of 3.2-mm diameter. Platinum wire served as the counterelectrode. The working electrode was polished prior to each measurement with a polishing kit (BAS-PK-1). Analysis of CV tracings, namely, determinations of oxidation potential (E1/2) and the detector anodic current (Ia), were carried out as described in Fig. 1. A change of ≥50 mV in the potential was considered significant.
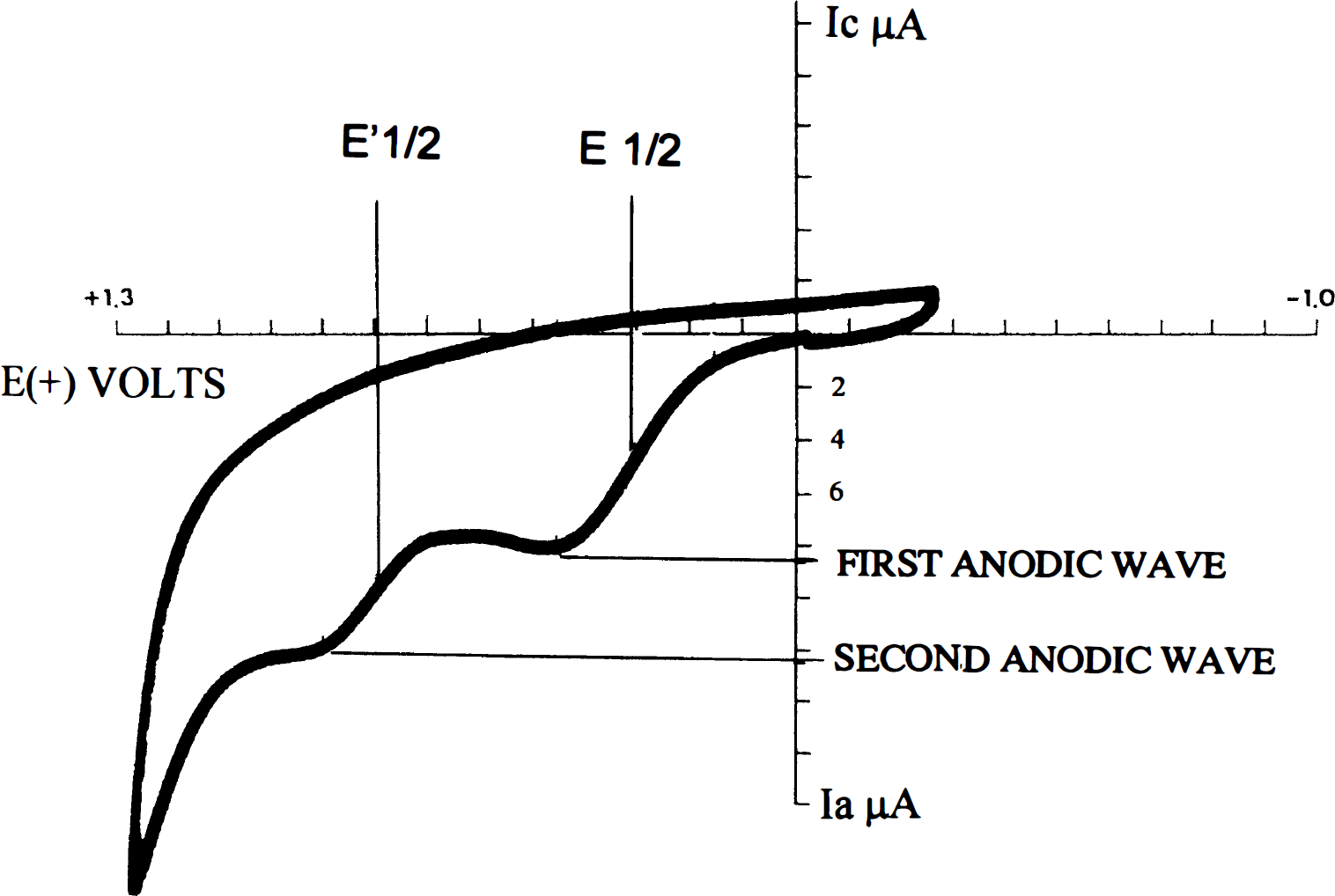
A typical tracing of brain cyclic voltammetry. E1/2 denotes the oxidation potential (mV), and the anodic current (Ia) denotes the total concentrations of the molecules being oxidized at the particular potential.
Nature of anodic wave components
To characterize the chemical nature of the reducing agents that yield the first and second anodic waves, brain homogenates were supplemented with LMWA abundant in the brain (ascorbate, urate, carnosine, l-tryptophan, and melatonin) and their CV tracings were evaluated. An increase in Ia at one of the two potentials suggests that the tested compound contributes to the pool of components undergoing oxidation at that particular potential.
Statistics
Values of anodic potential and anodic current are expressed as mean ± SEM E1/2 (mV) of at least n = 8 rats/group. One-way analysis of variance was performed to compare the different anodic currents at the various time points within a particular group (CON or ACC). The Student t test was used for between-group comparison at specific time points. The level of significance was p < 0.05.
RESULTS
CV system and its components in sham rats
A normal pattern of CV tracing of the rat brain is presented in Fig. 1. It is evident that there are two anodic waves, the first appearing at 350 ± 50 mV and the second at 750 ± 50 mV. These waves reflect the oxidation potential (E1/2) of the tissue, and they are related to the nature of the components undergoing oxidation during the cycle. Wave current height Ia (intensity) is proportional to the concentration of the component(s) being oxidized at the specific potential as expressed by the Randles–Sevcik equation (Greef et al., 1990). At 350 mV, sham rats had an Ia of 0.75 ± 0.06 pA/mg protein, and at the second anodic potential (750 ± 50 mV), the Ia was 1.00 ± 0.05 μA/mg protein.
To identify the LMWA at each E1/2, a number of major reductants (antioxidants) were examined as putative candidates. Addition of ascorbate and urate to brain homogenates generated anodic waves characterized by the same E1/2 as the first wave observed in the brain (350 mV). Tryptophan, carnosine, and melatonin yielded E1/2 similar to that of the second wave (750 mV). Examples of CV tracings of brain extracts supplemented with ascorbate and tryptophan are shown in Fig. 2. It is clear that the Ia at both anodic potentials increased upon the addition of the various tested reducing agents and that the increment was dose dependent (Chevion et al., 1996).
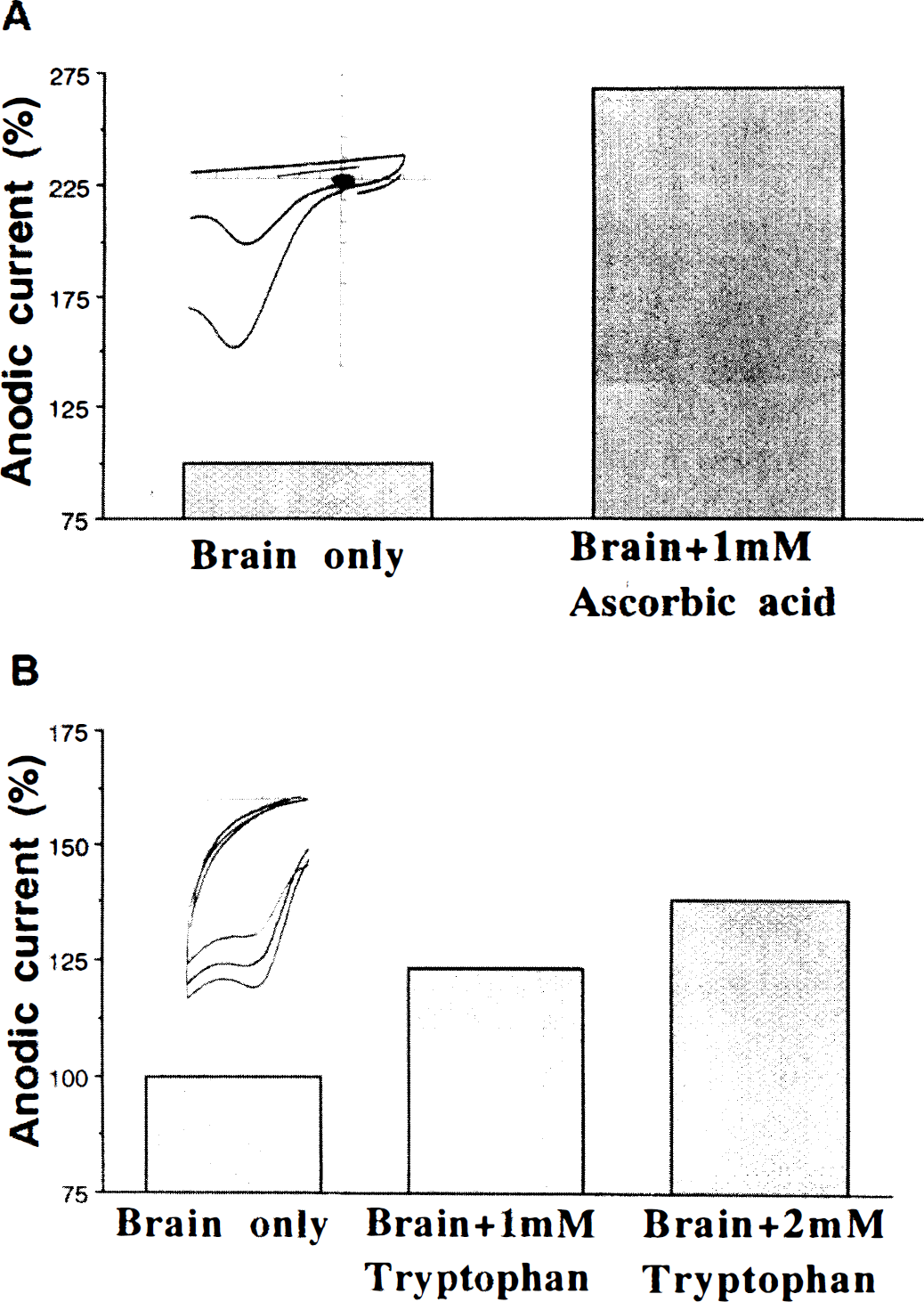
Changes in anodic current upon the addition of ascorbate (
CHI-induced changes in Ia measured at both anodic potentials
Following CHI, CV tracings of brain extracts revealed two anodic waves that appeared at the same potentials as those in the sham rat. However, there were significant changes in the levels of Ia at both potentials. Figure 3 presents the pattern of temporal changes in Ia values at the first (Fig. 3A) and second (Fig. 3B) anodic potentials of rat brain after CHI. Within 5 min of CHI, the anodic current of the left (injured) hemisphere decreased by 40% (p < 0.01 vs. sham), indicating a fall in the concentration of the reducing equivalents (antioxidants). At 4 h post-CHI, they were similar to those of the sham rats, whereas at 24 h, a further fall was observed (60%, p < 0.005 vs. sham). By 48 h and 7 days, the concentrations had returned to the preinjury levels. Similar biphasic changes in brain reducing capacity levels were found in the right contralateral hemisphere as well, suggesting that the trauma exerted global effect on brain reducing capacity.
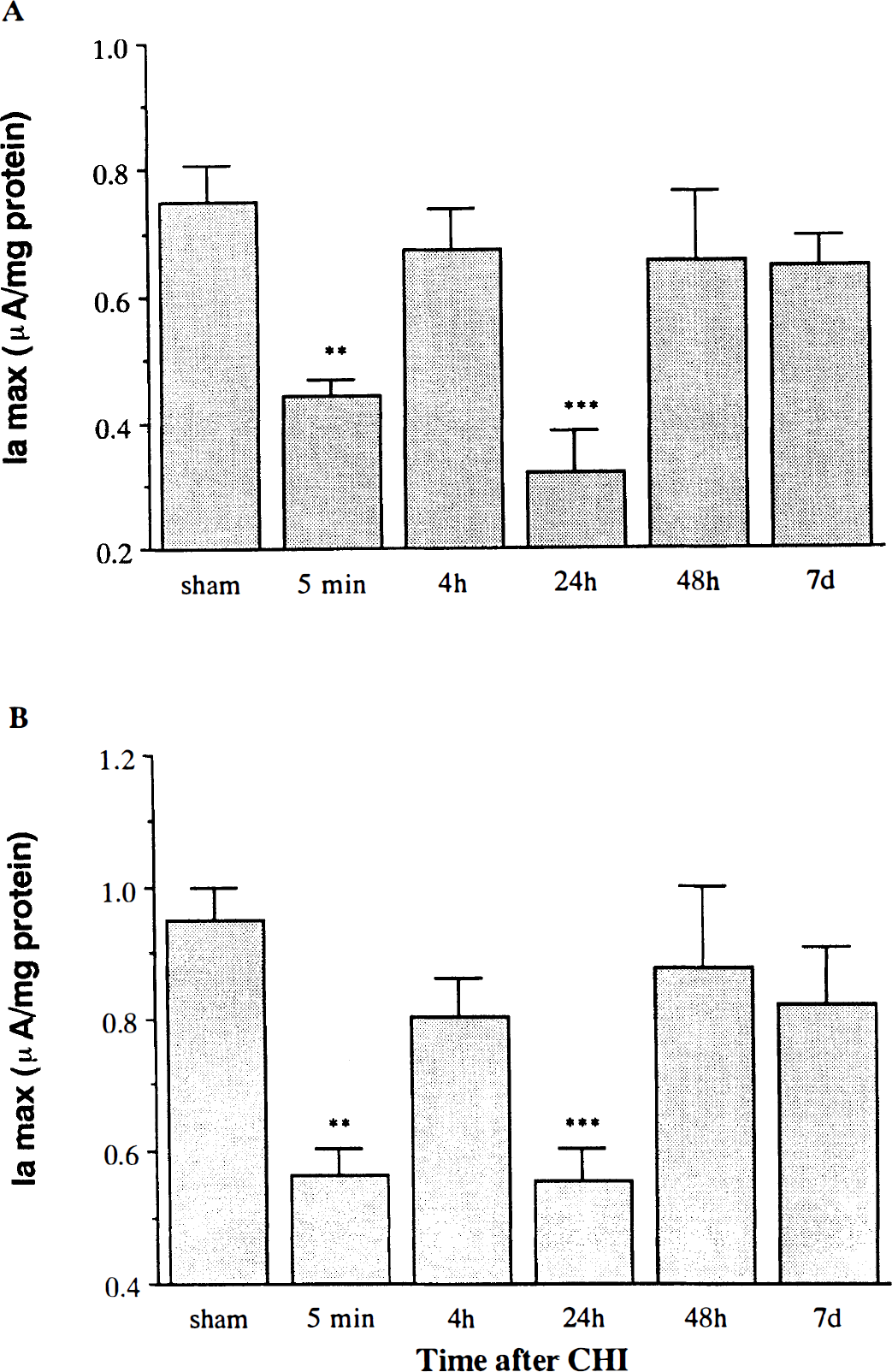
The effect of closed head injury (CHI) on the anodic currents (Ia) at 350 mV (
Effect of heat acclimation on brain CV after CHI
Brains of acclimated rats differed from those of nonacclimated (sham) rats both in basal Ia and in their temporal response to CHI. Prior to CHI, brains of sham ACC rats displayed the same two anodic potentials as in the sham controls (350 ± 50 and 750 ± 50 mV). However, the levels of the reducing equivalents (Ia) at both potentials were significantly lower than those of the controls (56 and 60% of control, respectively; p < 0.0001) (Fig. 4). In contrast to the CON rats, there was no immediate change (5 min following CHI) in reducing equivalents at 350 mV after CHI in the ACC rats, while some increase was observed at the second anodic wave (750 mV). Moreover, at 4 h and for up to 7 days, the Ia was 40–50% (p < 0.001) higher than that of the sham rats at both oxidation potentials.
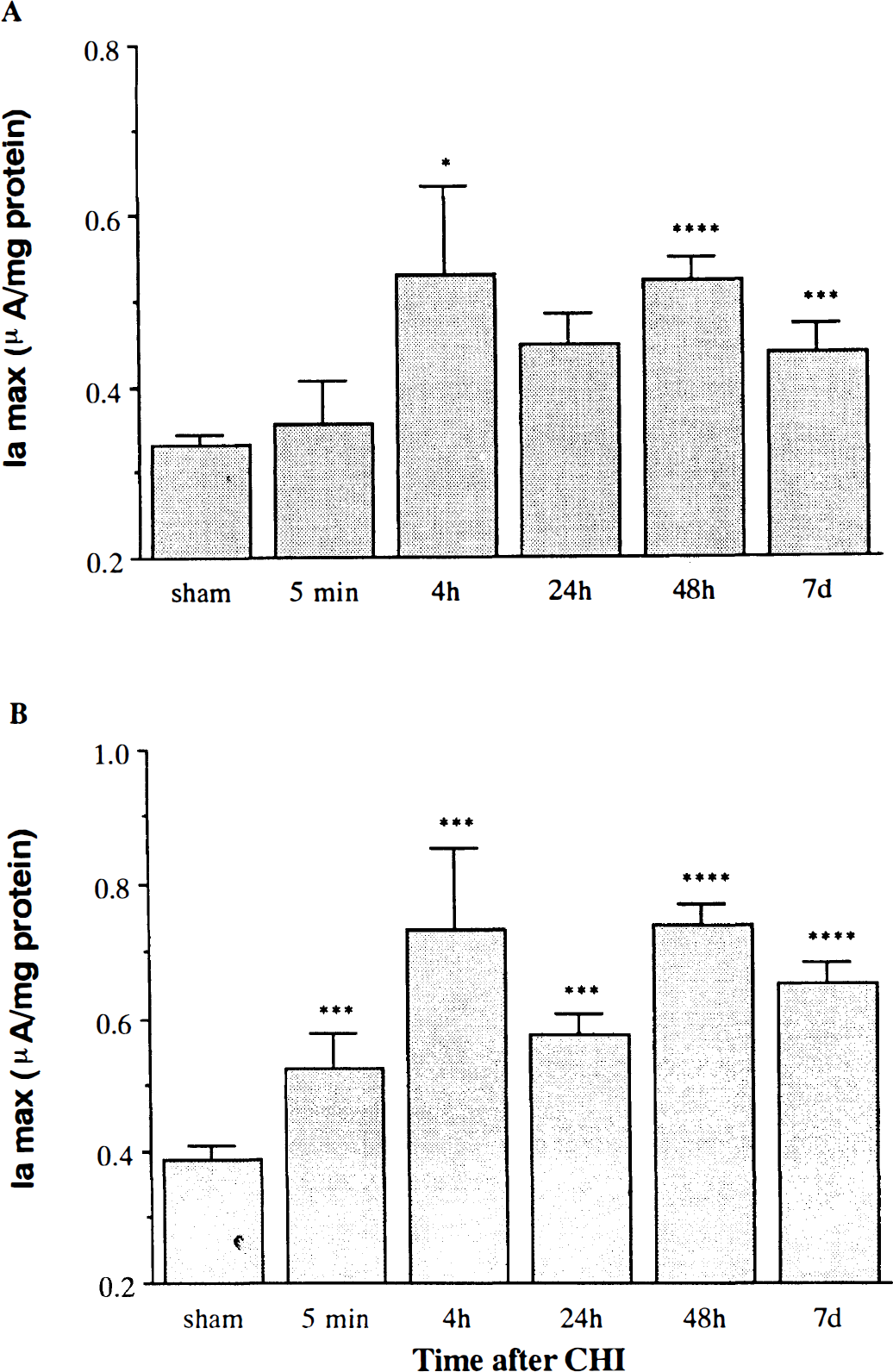
The effect of closed head injury (CHI) on the anodic current (Ia) at 350 mV (
At each time interval after CHI, the ratio between the anodic current at that time and before injury (sham) was calculated. The relative changes in fa during the post-CHI period in both CON and ACC rats are shown in Fig. 5. In the CON rats, it is clear that at any time postinjury, this ratio was <1; namely, the level of LMWA was lower than in the sham rats. In contrast, the ACC rats displayed markedly higher levels during the entire posttraumatic period, maintaining ratios of > 1 for up to 7 days.
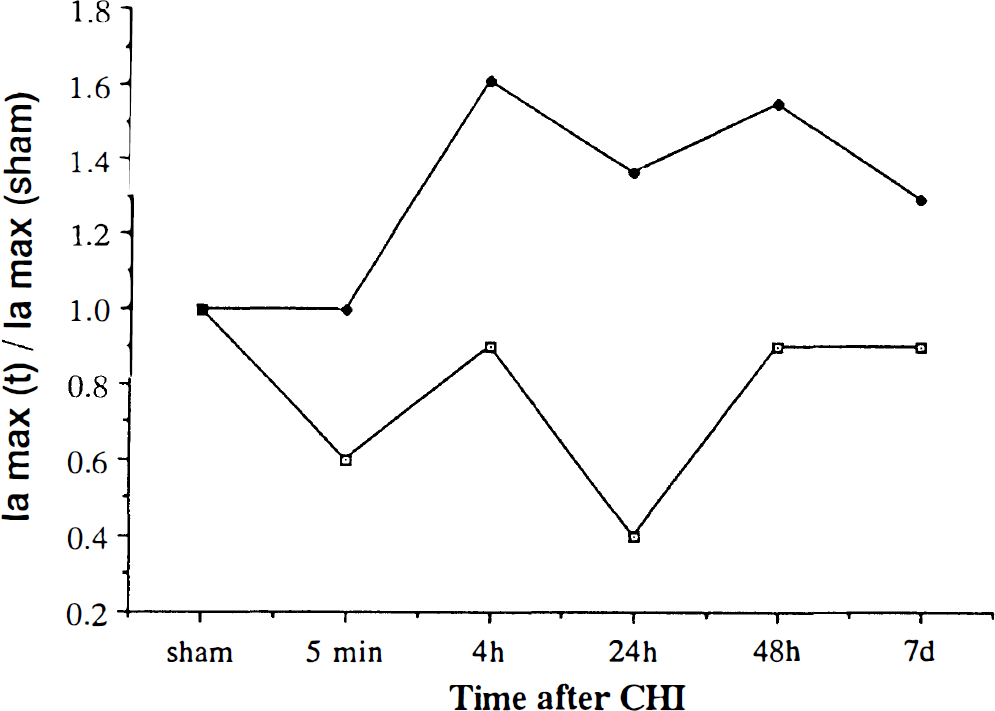
At each time interval after closed head injury (CHI), the ratio between the anodic current (at 350 mV) at that time and before injury (sham) was calculated. The relative changes in Ia during the post-CHI period in both control (open squares) and heat-acclimated rats (solid diamonds) are shown. At each time point, the mean Ia was divided by the Ia of sham rats at the same anodic potential.
Effect of CHI on heart CV in normothermic and acclimated rats
The CV of the heart of rats subjected to CHI revealed the same two anodic potentials as the brain (350 and 750 mV); however, the levels of total LMWA in these potentials were lower than in the brain (0.175 ± 0.08 and 0.570 ± 0.08 μA/mg protein, respectively). No changes in these levels were noticed after CHI in the normothermic rats, up to 7 days postinjury. In contrast, ACC rats showed increase in Ia that paralleled that found in the brain of the same rats. Figure 6 depicts the ratio of Ia in the heart at various intervals after CHI to Ia before injury at both anodic potentials. A marked increase of the ratio was found in the ACC rats, at 5 min and 24 h, in contrast to the constant ratio of 1 in CON rats.
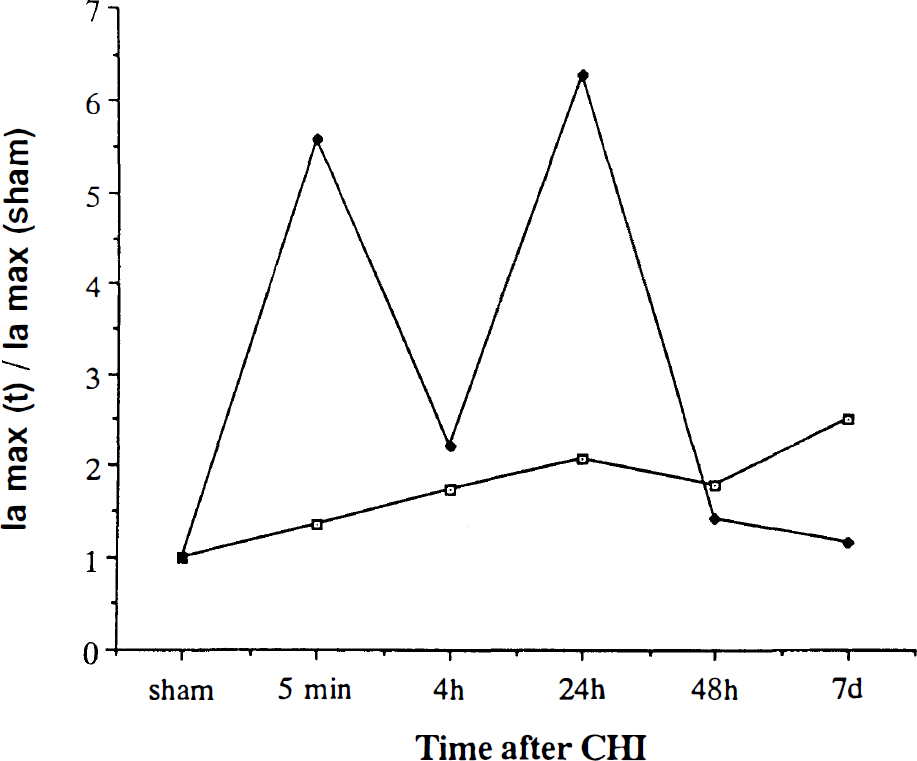
The response of heart tissue from control (open squares) and heat-acclimated rats (solid diamonds) after closed head injury (CHI), as expressed by the ratio between Ia at various times after CHI and that of sham. For details see Fig. 5.
DISCUSSION
To the best of our knowledge, this is the first time that CV has been used to measure endogenous reducing equivalents in normal and injured brain tissue. Using this technique, we have characterized the temporal profile of changes in the reducing power of the brain for up to 1 week following CHI and shown that the immediate response to injury is a decrease in brain levels of endogenous LMWA. We also demonstrated that ACC rats, which have a better clinical outcome after CHI (Shohami et al., 1994), show markedly elevated brain levels of reducing equivalents in response to injury, despite the fact that they have a lower basal levels of these compounds than CON rats. A similar increase in LMWA was also noted in the hearts of ACC rats following CHI, but not in the CON rats. This finding suggests that the augmented concentrations of endogenous antioxidants are part of a systemic response to CHI that occurs only in the ACC rat and provides protection.
To date, the CV technique has been used in chemistry and biochemistry for measuring overall antioxidant capacity. Kissinger et al. (1973) showed that the introduction of a carbon electrode into the brain enabled a CV tracing to be made and suggested that this technique might be suitable for measuring changes in biological oxidation potential under pathological conditions. Several investigators have applied this technique to evaluate the antioxidant capacity of saliva, of tissue homogenates after oxidative damage, and of plasma (Kissinger et al., 1973; Kohen et al., 1992; Kohen, 1993). The present study, however, is the first demonstration of brain LMWA under pathological conditions. In the CV tracing, two component-specific anodic waves were identified at E1/2 of 350 ± 50 and 750 ± 50 mV (Fig. 1), which represent two classes of reducing equivalents. The first class consists of ascorbate and urate (identified with HPLC equipped with an electrochemical detector; unpublished data), and the second most likely includes imidazole-containing molecules such as histidine, NADH, melatonin, carnosine, and l-tryptophan. During the post-CHI period, the two anodic potentials were stable, indicating that the nature of the LMWA was not altered. However, the anodic current Ia was markedly affected by CHI. As summarized in Fig. 3, a biphasic response to CHI was recorded in the CON group, with an initial decrease (at 5 min), followed by a return to normal pre-CHI levels at 48 h, which persisted for up to 1 week. The significant decrease in the concentration of the reducing equivalents (Ia) 5 min after CHI could be accounted for by their immediate utilization. This may further support the observation that ROS are being formed in the early stages following CHI (Beit-Yannai et al., 1996). Four hours post-CHI, the levels of LMWA increased. Since at 4 h, blood–brain barrier disruption is maximal (Shapira et al., 1993), high levels of LMWA originating in the plasma may flood the damaged area. These compounds could then react with the ROS, which are continuously being produced via pathways such as inflammation, activated in the posttraumatic period, whose levels decrease again 24 h following CHI. It is also possible that these compounds are being endogenously synthesized by resident brain cells following CHI. We have indeed demonstrated that at 24 h post-CHI, the levels of prostaglandin E2 are sevenfold higher in the contused hemisphere than in the brains of sham-injured rats (Shohami et al., 1987).
The CV tracing of the ACC rats was similar to that of the CON rats: The appearance of the two anodic waves at the same potentials indicates that the nature of the LMWA in the two groups was similar. However, the level of these compounds, as indicated by the Ia at both potentials, was significantly lower (by 50%) in the ACC rats. Moreover, there was a significant difference in the brain's response to trauma in the two groups. In contrast to the decreased levels of LMWA in the CON rats, the ACC rats displayed an increase in LMWA at all time points after CHI (Fig. 4). This reversed pattern of changes in fa in the ACC rats may point to a difference in their ability to cope with the burst of ROS produced and accumulated after CHI. It is probably not due to the higher water content found in CON rats (84%) than in ACC rats (81%) after CHI (Shohami et al., 1994), since fa levels were normalized to the protein content of the tissue. Furthermore, the difference in fa between the groups is much larger than can be accounted for by variations in protein content resulting from edema.
Reducing equivalents may serve as both pro-oxidants and anti-oxidant compounds. For example, it has been shown that ascorbic acid may react with transition metals such as copper or iron to produce the highly reactive hydroxyl radical (Shinar et al., 1983; Higson et al., 1988). Therefore, following CHI or postischemia, high levels of reducing equivalents may act as pro-oxidants rather than anti-oxidants, while interacting with the redox-active metals, released within the damaged area. In contrast, in the later stage, when ROS are being continuously produced (e.g., inflammation), high levels of reducing equivalents may be advantageous since they can act as scavengers, neutralizing the damaging species. Thus, a possible explanation for the relative resistance of the ACC rats to CHI may be concerted biological adaptation, reflected in a low basal level of the reducing equivalents, which rules out their role as pro-oxidants, and production of sufficient LMWA to enable the brain to cope more effectively with the ROS produced via other pathways.
The biphasic pattern of changes in fa in both the CON and the ACC rats (Figs. 3 and 4) supports the hypothesis that rats indeed utilize their stores of LMWA in an attempt to neutralize the ROS and to protect the injured tissue. In fact, the lower level of LMWA in the posttraumatic period in CON rats may suggest that LMWA are consumed at a faster rate than they are produced. In contrast, ACC rats, which prior to injury display lower levels of LMWA, have the capacity to produce (or recruit) high levels of these compounds in response to a growing demand for neutralization of harmful radicals. The reported lower basal metabolic rate following heat acclimation (Horowitz and Samueloff, 1989) lends support to this notion, and we may speculate that less ROS are produced. The notion of altered metabolic balance agrees with the findings of Hadas et al. (1989) who showed a reduced rate of prostaglandin synthesis under basal conditions in ACC as compared with CON rats. This and the present results indicate that, indeed, some basic metabolic pathways are affected in the course of the acclimatization process. Although CHI involves the brain, the ACC rats, but not the CON animals, displayed a significant biphasic increase in heart LMWA. The pattern of changes was similar to that found in the brain of the same rats, although there was a delay (Fig. 6). This may suggest that heat acclimation potentiates a systemic mechanism that enables the rats to cope better with additional acute stress. Indeed, it has been documented that the heart of ACC rats is better protected than that of CON animals during cardiac ischemia and reperfusion (Levi et al., 1993). Based on our present data, we suggest that, at least in part, long-term exposure to high ambient temperature induces more efficient coping with ROS. This global effect is specific to heat acclimation, since both brain and heart share a similar response to CHI.
Footnotes
Acknowledgment:
This study was partly supported by the Israel Ministry of the Sciences and Arts.
