Abstract
The effect of preload on myofilament Ca2+ sensitivity was examined using α-toxin permeabilization and fura-2 fluorometry in rabbit cerebral arteries. The [Ca2+]i-force curves shifted leftward at a high preload, with a decrease in median effective concentration of Ca2+ in the permeabilized artery. In the fura-2-loaded artery, the preload modulated the force without affecting [Ca2+]i levels during K+ depolarization, and a high preload moved the [Ca2+]i-force curve upward and to the left. It is thus concluded that the preload regulates the Ca2+ sensitivity of the myofilament and, therefore, may play a role in the regulation of cerebral arterial tonus and blood flow.
A rise in intravascular pressure increases the tone of the cerebrovascular smooth muscle and contracts the diameter of the artery, which has been observed as a myogenic response (Vinall and Simeone, 1981). It was reported that the preload or initial length at which the muscle was set (Tallarida et al., 1974) or the intravascular pressure (Harder, 1984) regulates the sensitivity of vascular smooth muscle to vasoactive agents. This effect of preload on the vascular sensitivity was observed in the contractions induced by both K+ depolarization and various vasoactive agents (Price et al., 1981).
In smooth muscle, cytosolic Ca2+ ([Ca2+]i) and, hence, Ca2+-dependent phosphorylation of the myosin light chain (MLC) is the major regulatory system of force generation (Rembold and Murphy, 1988). It has been reported that muscle stretching could induce both MLC phosphorylation and force generation (Bárány et al., 1985). It has also been shown that the stretch of the vascular wall increases membrane depolarization and Ca2+ influx of cerebral arterial smooth muscle (Harder, 1984). Rembold and Murphy (1990) reported that decreased vascular sensitivity to agonists at a lower preload could be explained by reduced [Ca2+]i mobilization. On the other hand, it has also been reported that the shortening of muscle length enhanced the increase in [Ca2+]i elevation (Gunst, 1986), while, in addition, the alteration in the influx of extracellular Ca2+ was not sufficient to explain the effects of preload on vascular sensitivity (Herlihy and Berardo, 1986). Therefore, the underlying mechanism of the effect of preload on vascular sensitivity remains a subject of controversy.
We recently showed, using fura-2 fluorometry, that the resting load regulates the contractility of cerebral arterial smooth muscle, mainly by modulating the [Ca2+]i-force relationship in cerebral vascular smooth muscle, without affecting [Ca2+]i elevation (Miyagi et al., 1995a). In the present study, we determined the effect of the resting load on the relationship between Ca2+ and force by using α-toxin-permeabilized smooth muscle and fura-2 microfluorometry of intact smooth muscle of the rabbit cerebral artery. The permeabilization of smooth muscle allowed for the direct operation of [Ca2+]i and thus enabled us to make a detailed investigation of the contractility-regulating mechanism downstream from [Ca2+]i elevation to the force generation in smooth muscle (Nishimura et al., 1988).
MATERIALS AND METHODS
Permeabilized arterial smooth muscle
Preparations of arterial rings.
Adult male Japanese white rabbits (2.5–3.0 kg) were killed by intravenous injection of heparin (1,000 U) and pentobarbital sodium (400 mg). Cerebral and ear arteries were rapidly excised and put into ice-cold physiological salt solution (PSS). The cerebral arteries were sampled at three different parts: the basilar artery (BA) as the main large cerebral artery and the posterior cerebral artery (PCA), and the middle cerebral artery (MCA) as more distal cerebral arteries. In addition, the ear arteries were sampled at 5 cm from their origin. The diameters of the BA, PCA, MCA, and ear artery were approximately 750, 400, 300, and 800 μm, respectively. The connective tissue around the arteries was removed, and the arteries were cut for use into small rings measuring 250 μm in width. This width of the arterial rings was so small that the contribution of restoring force due to ring shape to its resting force could not be detected and thus was negligible.
α-Toxin pretreatment and force measurement.
Permeabilization of the smooth muscle with α-toxin was carried out as previously reported (Nishimura et al., 1988). In brief, the small rings were permeabilized with staphylococcal α-toxin (5,000 U/ml) in cytoplasmic substitution solution (CSS) supplemented with 2 mmol/L ethyleneglycol-bis (β-amino-ethylether)-N,N,N′,N′-tetraacetic acid (EGTA) and no Ca2+. After 30–40 min of treatment with α-toxin, the small ring was mounted between two tungsten wires. One wire was fixed, and the other was attached to a force transducer (U Gauge; Minebea Co., Ltd., Japan). The tissue was stretched to a high resting load (the preliminarily determined optimal resting load at which a maximal contraction was obtained with increasing Ca2+ concentration) or to a low resting load (the preliminarily determined resting load at which the resting load of the rings were reduced without slack) (Miyagi et al., 1995b). After 1 h of equilibration at the given resting load, the Ca2+-dependent force of the tissue was induced by exchanging CSS, which contained increasing concentrations of Ca2+. All experiments on α-toxin-permeabilized fibers were carried out at 25°C.
Simultaneous measurements of [Ca2+]i and tension of the intact arterial strips using fluorescence microscope (fura-2 microfluorometry of intact arterial smooth muscle)
Fura-2 loading.
Rabbit basilar arterial strips were loaded with fura-2 by incubating in Dulbecco's Modified Eagle's Medium containing 10 μmol/L fura-2/AM (an acetoxymethyl ester form) and 5% fetal bovine serum for 3 h at 37°C, under an aeration with 95% oxygen and 5% carbon dioxide.
Measurement of force generation and microfluorometry of fura-2.
The strips were mounted horizontally between two tungsten wires for force measurement, similar to those used in a skinned preparation, in an organ bath specially designed for an inverted fluorescent microscope (TMD 56; Nikon, Japan) equipped with a spectrophotometer (CAM 220; Japan Spectroscope Co., Japan). The fura-2-microfluorometry and force measurement were simultaneously carried out at 37°C, as previously described in detail (Miyagi et al., 1995a, b ). In brief, during the equilibration period after the fura-2 loading, the rings were stimulated with 118 mmol/L K+-PSS every 10 min while monitoring the resting load, and the optimal resting load was thus determined in each strip (high resting load, 100–200 mg). In the experiments at a low resting load (5–10 mg), at first the control response to 118 mmol/L K+-PSS was obtained at a high resting load, and thereafter the resting load was gradually decreased. The ratio of the fluorescence intensities (500-nm emission) at 348-nm excitation to those at 380-nm excitation was monitored. The fluorescence ratio was expressed as the percentage (% ratio), assigning the values in normal (5.9 mmol/L K+) and 118 mmol/L K+-PS S at each resting load to be 0% and 100%, respectively. A statistical analysis of the [Ca2+]i signals was performed using the % ratio.
Drugs and materials.
The composition of PSS (in mmol/L) was NaCl, 123; KCl, 4.7; NaHCO3, 15.5; KH2PO4, 1.2; MgCl2, 1.2; CaCl2, 1.25; and
Statistical analysis.
The half maximally effective concentrations (EC50) were calculated using the four-parameter logistic equation (De Lean et al., 1978). All values were expressed as the mean ± standard deviation (SD). The EC50 values were compared when analyzing the right-left shift of the dose-response curves at different resting loads. The two-tailed Student's t test was used to determine the statistical significance. A value of p < 0.05 was considered to be significant. An analysis of covariance was used to determine the nonoverlapping (or shift) of the [Ca2+]i-force relationship (Fig. 2B).
RESULTS
Figure 1 (A–C) shows the [Ca2+]i-relative force curves of α-toxin-permeabilized cerebral arteries at high and low resting loads. Each data point was plotted by assigning the forces at the resting state and at the maximal response to the increase in Ca2+ concentration to be 0% and 100%, respectively. The [Ca2+]i-force curves at high resting load thus moved to the left from those at low resting load. The EC50 values of Ca2+ for force generation at lower resting loads of BA, PCA, and MCA were 281 ± 42, 186 ± 32, 148 ± 22 nmol/L, respectively, whereas those obtained at higher resting loads were 190 ± 36, 78 ± 29, 104 ± 17 nmol/L, respectively (p < 0.01 compared with those at lower resting loads in each artery). These data indicated that the contractile apparatus of each artery at a high resting load was more sensitive to the Ca2+ concentration than that at a low resting load. Regarding the EC50 values among cerebral arteries (BA, PCA, MCA), it seems plausible that the greater the distance to the cortical artery, the smaller the EC50 value.
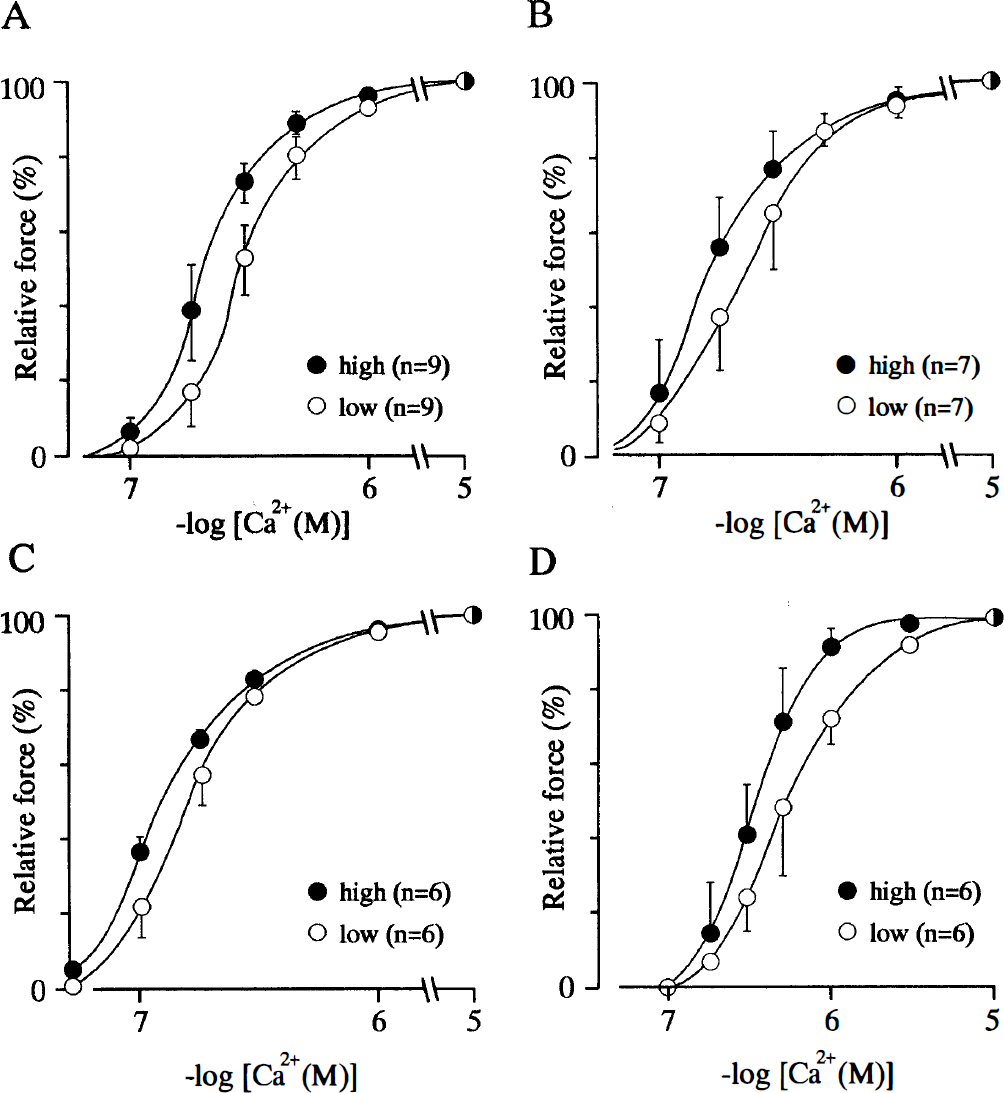
The effects of resting loads on the Ca2+ sensitivity of the contractile apparatus of α-toxin-permeabilized cerebral and ear arteries. The rings of cerebral (
In order to determine whether the resting load regulation of Ca2+ sensitivity observed is a characteristic feature of the cerebral arteries, the effect of the resting load on the Ca2+ sensitivity of the contractile apparatus was also determined in smooth muscle of the ear artery. As shown in Fig. 1D, the [Ca2+]i-relative force curve at a high resting load was located to the left of that at a low resting load. The EC50 value of Ca2+ for force generation of the ear artery was 326 ± 7 nmol/L at a higher resting load, whereas that obtained at a lower resting load was 482 ± 85 nmol/L (p < 0.01 compared with that at lower resting load in the ear artery; Fig. 1D). EC50 values for the ear artery were much greater than those of the cerebral arteries (p < 0.01 compared with those at higher and lower resting loads in BA, PCA, MCA), thus indicating that the Ca2+ sensitivity of the ear artery is smaller than that of the cerebral arteries.
To confirm the enhancing effect of a high resting load on the Ca2+ sensitivity of the contractile apparatus in the intact smooth muscle, we measured the changes in the [Ca2+]i and force generation in the rabbit basilar artery by the use of fura-2 microfluorometry. Figure 2 shows the effects of high and low resting loads on increases in the % ratio and force induced by increasing extracellular K+ concentrations ([K+]o). As shown in Fig. 2A, the force generations to 118 mmol/L K+ were attenuated by the decrease in resting load, whereas the associated [Ca2+]i elevations were not affected. Figure 2B shows the results of the changes in [Ca2+]i and force induced by a stepwise increase in [K+]o at low and high resting loads. The decrease in the resting load shifted the [Ca2+]i-force relationship during high [K+]o contraction both downward and to the right (p < 0.05 by analysis of covariance). To determine the sensitivity (EC50) of the responses to stimuli, the [K+]o-% ratio ([Ca2+]i) and [K+]o-force curves at high and low resting loads were normalized by assigning the values at rest (normal PSS) and at a maximal response at each resting load to be 0% and 100%, respectively (Figs. 2C and 2D). There was no significant difference in the EC50 value of the % ratio elevation between the high and low resting loads (high load, 20.6 ± 2.2 mmol/L; low load, 20.2 ± 2.2 mmol/L; p > 0.05). However, the EC50 value of the force at a low resting load was significantly greater than that at a high resting load (high load, 27.6 ±3.5 mmol/L; low load, 23.7 ± 4.5 mmol/L; p < 0.05).
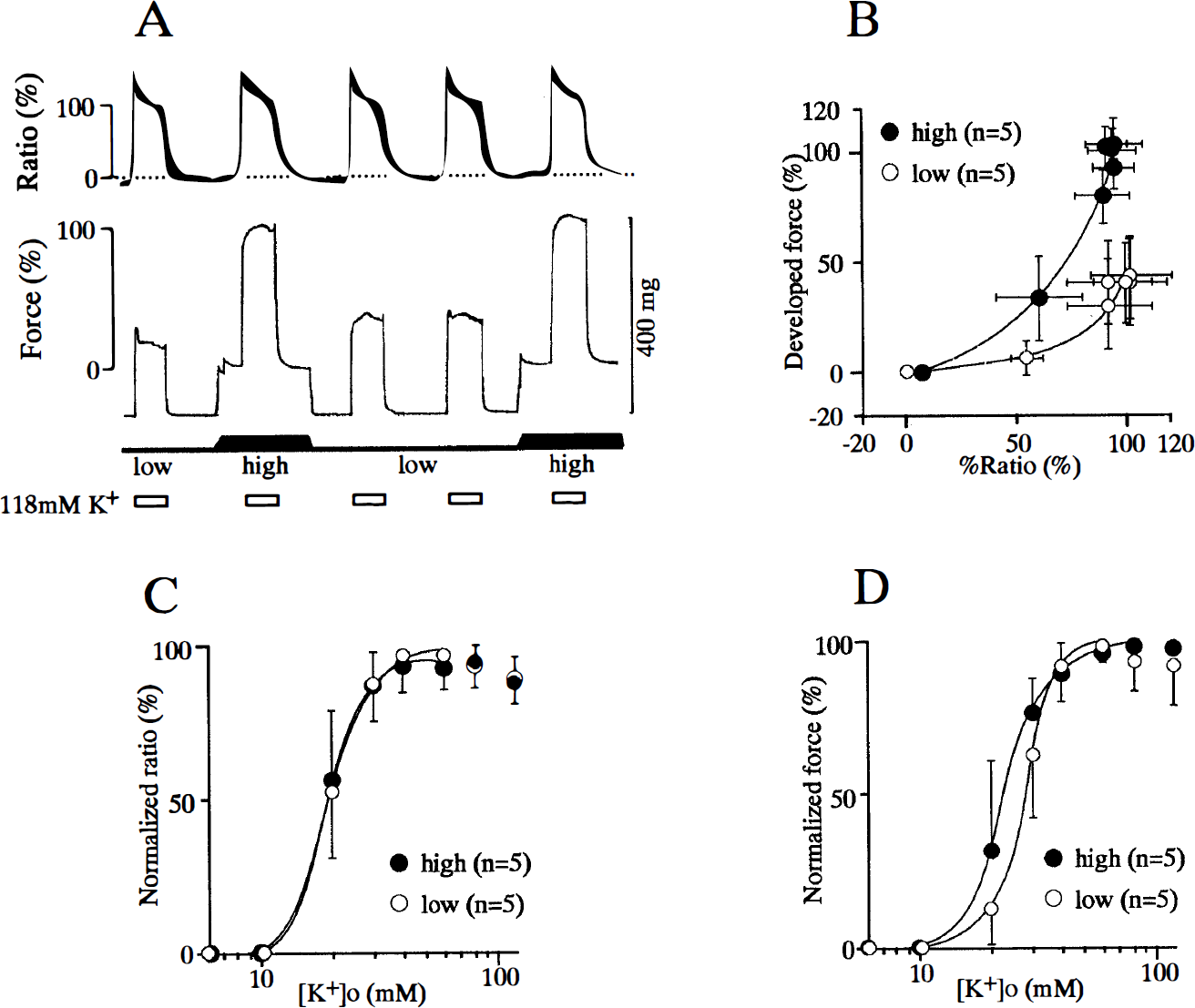
The effect of changes in the resting load on the increases in [Ca2+]i and force induced by various concentrations of extracellular K+ ([K+]o).
DISCUSSION
It is well known that the contractility or sensitivity of smooth muscle depends on the preload or the initial length loaded on muscle (Murphy, 1976); however, the related mechanisms at the cellular level have been poorly understood. We demonstrated for the first time that the resting load regulates the Ca2+ sensitivity of the contractile apparatus of vascular smooth muscle permeabilized with staphylococcal α-toxin. In permeabilized cells, [Ca2+]i can be directly operated, because [Ca2+]i is equal to the free Ca2+ concentration of the bathing solution. In the present study, the [Ca2+]i-force curves at a high resting load were clearly located to the left of those seen at a low resting load in cerebral and ear arteries. It was thus apparent that the resting load regulates the EC50 of Ca2+ (Ca2+ sensitivity of the contractile apparatus). We also established the regulation of Ca2+ sensitivity of the contractile apparatus by the resting load in intact smooth muscle by measuring [Ca2+]i and the force with fura-2 microfluorometry. A decrease in the resting load did not affect the [Ca2+]i elevation by high [K+]o, but it did attenuate the force generation and EC50 value for force generation in the intact smooth muscle. These data are in agreement with our previous findings on the intact bovine cerebral artery, in which the resting load regulates the contractility and sensitivity of smooth muscle by modulating the [Ca2+]i-force relationship without affecting the [Ca2+]i elevation induced by high K+ or agonist (Miyagi et al., 1995a, b ).
At rest, the stretch of the smooth muscle can induce [Ca2+]i mobilization, which may regulate vascular tone (Bárány et al., 1985). We also reported that the resting level of [Ca2+]i measured with fura-2 depends on the resting load imposed on unstimulated smooth muscle (Miyagi et al., 1995a, b ). It has been reported that stretching itself induces the activation of the mechano-sensitive cation channel (Davis et al., 1992), thus leading to membrane depolarization and Ca2+ entry (Harder, 1984). However, when aequorin was used as the [Ca2+]i indicator, there was no increase in the [Ca2+]i levels at rest (Gunst, 1986; Rembold and Murphy, 1990).
The results obtained with permeabilized smooth muscle in the present study clearly indicate that some mechanotransduction pathway other than the mechano-sensitive cation channel is present, which is responsible for the resting load regulation of the Ca2+ sensitivity of the contractile apparatus in smooth muscle. In striated muscle, it has been reported that troponin C, a Ca2+-binding subunit of troponin complex, regulates the activation of thin filaments in a sarcomere length–dependent manner (Hofmann and Fuchs, 1987; Babu et al., 1988). In smooth muscle, too, there is also a possibility that some filaments (or messengers) in the cytoskeletal matrix connect to the myofilaments and thus regulate Ca2+ sensitivity in a preload-dependent manner. At present, we are not sure whether or not the underlying mechanotransduction pathway is similar to that seen in striated muscle.
The EC50 values of Ca2+ for the force generation of the cerebral arteries at high and low resting loads (78–281 nmol/L; Figs. 1A–C) are around the physiological range (145–212 nmol/L; Miyagi et al., 1995a, b ) and are also much smaller than those observed in the ear artery (326–482 nmol/L; Fig. 1D). In the permeabilized rat mesenteric artery, a 10–7 mol/L Ca2+ concentration alone did not induce any contraction, but it did induce a significant contraction in the G-protein-activated state (Nishimura et al., 1990). These observations thus suggest that in the case of the cerebral artery, a Ca2+-sensitizing mechanism may be involved in the regulation of vascular tone, and the contribution of this mechanism is considered to be more significant in the distal artery closer to the cerebral cortex. It is thus suggested that at levels near that of physiological [Ca2+]i, there may be a more pronounced effect of the resting load on the tone of the cerebral arteries than that of the extracranial arteries; the resting load might thus contribute to a more sophisticated autoregulation of the cerebral blood flow than of other local blood flows (Harder, 1984; 1988). However, further investigation using vessels smaller than those used in the present study would be needed to evaluate the role of the resting load in autoregulation, because autoregulation certainly involves resistance arteries.
In summary, at a high resting load, the Ca2+ sensitivity of the contractile apparatus, both in the intact and in α-toxin-permeabilized smooth muscle, was enhanced in comparison to that at a low resting load. The effect of the resting load on the contractility of smooth muscle was also observed at a relatively lower [Ca2+]i level (around the physiological level of [Ca2+]i) in the cerebral artery than in the ear artery.
Footnotes
Acknowledgment:
We thank Mr. B. T. Quinn for critical comments. This study was supported in part by Grants-in-Aid for Developmental Scientific Research (no. 06557045), for General Scientific Research (nos. 07407022, 07833008), and for Creative Basic Research “Studies of Intracellular Signaling Network” from the Ministry of Education, Science, and Culture, Japan; by a grant from the Japan Research Foundation of Clinical Pharmacology; and by a grant from the Kimura Memorial Heart Foundation.
