Abstract
We tested the hypothesis that stimulation of metabotropic glutamate receptors (mGluRs) increases nitric oxide (NO) production in the hippocampus in vivo. Microdialysis probes were placed bilaterally into the CA3 region of the hippocampus of adult Sprague–Dawley rats under pentobarbital anesthesia. Probes were perfused for 5 h with artificial cerebrospinal fluid (CSF) containing 3 μM [14C]-L-arginine. Recovery of [14C]-L-citrulline in the effluent was used as a marker of NO production. In nine groups of rats, increases in [14C]-L-citrulline recovery were compared between right- and left-sided probes perfused with various combinations of the selective mGluR agonist, trans-(1S,3R)-1-amino-1,3-cyclopentanedicarboxylic acid (ACPD); the mGluR antagonist, (±)-α-methyl-4-carboxyphenylglycine (MCPG); the NO synthase inhibitor, N-nitro-L-arginine (LNNA); the ryanodine sensitive calcium-release channel inhibitor dantrolene, the non-N-methyl-D-aspartate (NMDA); receptor antagonist 6-cyano-7-nitroquinoxaline-2,3-dione (CNQX); the NMDA receptor antagonist (+)-5-methyl-10,11-dihydro-5H-dibenzo[a,d] cyclohepten-5,10-imine (MK-801); and the Na+ channel blocker, tetrodotoxin. Recovery of [14C]-L-citrulline during perfusion with artificial CSF progressively increased to 90 ± 21 fmol/min (± SD) over 5 h. Perfusion in the contralateral hippocampus with 1 mM ACPD augmented [14C]-L-citrulline recovery to 250 ± 81 fmol/min. Perfusion of 1 mM nitroarginine + ACPD inhibited [14C]-L-citrulline recovery compared to that with ACPD alone. Perfusion with 1 mM MCPG + ACPD attenuated ACPD enhanced [14C]-L-citrulline recovery. Perfusion of 1 mM dantrolene + ACPD inhibited the ACPD-evoked increase in [14C]-L-citrulline recovery. Perfusion of 1 mM MCPG or dantrolene without ACPD did not decrease [14C]-L-citrulline recovery as compared to CSF alone. ACPD-enhanced [14C]-L-citrulline recovery was not attenuated by CNQX, MK-801, or tetrodotoxin (TTX). Using an indirect method of assessing NO production in vivo, these data demonstrate that mGluR stimulation enhances NO production in rat hippocampus. Inhibition with dantrolene suggests that calcium-induced calcium release amplifies the inositol triphosphate-mediated calcium signal associated with mGluR stimulation, thereby resulting in augmented calcium-dependent NO production.
Constitutive nitric oxide synthase (NOS), present in selective neurons, is regulated by intracellular calcium (Bredt and Snyder, 1990; Bredt et al., 1991). Ionotropic glutamate receptors, notably N-methyl-
The role of metabotropic glutamate receptors (mGluRs) in regulating NOS activity has received less attention. This family of receptors are thought to be G-protein-linked to phospholipase C, adenyl cyclase, and, possibly, phospholipase D (Boss and Conn, 1992; Holler et al., 1993; Schoepp and Conn, 1993; Winder and Conn, 1992). Stimulation of phospholipase C results in increased inositol triphosphate (IP3) formation and mobilization of intracellular calcium stores, which, in cells containing NOS, could stimulate NO production. Application of the mGluR agonisttrans-(1S,3R)-1-amino-1,3-cyclopentanedicarboxylic acid (ACPD) to cerebellar slices increases cyclic GMP (Okada, 1992). Elevations in cyclic guanine monophosphate (GMP) in the extracellular fluid utilizing the technique of cerebral microdialysis have been demonstrated with local application of NMDA, 2-amino-3-hydroxy-5-methylisoxazole-4-propionic acid (AMPA), and ACPD in vivo (Luo et al., 1994; Vallebuona and Raiteri, 1994).
Direct measurements of NO in vivo is difficult due to the lability of the gas, although a porphyrinic microsensor has had limited use in the brain (Malinski et al., 1993). Formation of methemoglobin in microdialysis effluent when kainate is administered systemically has been used as a marker of NO in vivo (Bilcioglu and Maher, 1993). Catalytic activity of the enzyme in homogenized tissue is usually measured from the accumulation of radiolabeled citrulline when the radiolabeled substrate L-arginine is added. We adapted this technique in vivo by perfusing microdialysis probes with radiolabeled arginine, and measuring radiolabeled citrulline in the dialysate effluent from both rat hippocampus (Bhardwaj et al., 1995b) and lamb cortex (Northington et al., 1995). This measure is an indirect marker of NO production in vivo; nevertheless, we were able to show that addition of NMDA to the microdialysis perfusate increased citrulline recovery, which was attenuated by adenosine A1 receptor agonists and blocked by N-nitro-L-arginine (LNNA) methyl ester, but not by its stereoisomer. Therefore, the technique has adequate sensitivity for detecting NMDA-evoked increases in NO production as well as modulation of NO production by other drugs in vivo.
In the present study, we used ACPD (Palmer et al., 1989; Schoepp et al., 1991) to investigate the role of mGluRs in regulating NO production in vivo in rat hippocampus dialysed with labeled arginine. We tested the hypotheses that ACPD increases labeled citrulline recovery and that this increase is attenuated by the mGluR antagonist (±)-α-methyl-4-carboxyphenylglycine (MCPG) (Eaton et al., 1993) but not by the NMDA receptor antagonist (+)-5-methyl-10,11-dihydro-5H-dibenzo [a,d] cyclohepten-5,10-imine (MK-801) or by the non-NMDA receptor antagonist 6-cyano-7-nitroquinoxaline-2,3-dione (CNQX). We further tested the hypothesis that ACPD does not enhance citrulline recovery via neuronal activation secondary to epileptiform activity by perfusion with tetrodotoxin (TTX).
To our knowledge, no selective IP3 receptor agonists presently exist. However, calcium-induced calcium release, mediated by intracellular ryanodine-sensitive calcium channels shown to be present in brain (Sharp et al., 1993), may amplify NO production. Dantrolene is an antagonist of ryanodine-sensitive channels in muscle, and has been found to reduce intracellular calcium increases evoked by glutamate and NMDA (Bouchelouche et al., 1989; Fransden and Schousboe, 1991; Simpson et al., 1993), as well as cytotoxicity from glutamate, NMDA and quisqualate (Fransden and Schousboe, 1992; Fransden and Schousboe, 1993). Therefore, we also tested the hypothesis that dantrolene reduces the ACPD-evoked increase in citrulline recovery in the dialysate.
MATERIALS AND METHODS
The experimental protocol was approved by the institutional animal care and use committee of the Johns Hopkins University and conforms to National Institutes of Health guidelines for the care and use of animals in research. Methods for surgical preparation, cerebral microdialysis, and measurement of [14C]-L-citrulline measurements have been described in detail in previous publications (Northington et al., 1995; Bhardwaj et al., 1995b) and will only be summarized here.
General surgical procedures
Adult male Sprague–Dawley rats (300–450 g) were anesthetized with intraperitoneal (i.p.) pentobarbital (60 mg/kg). Rats were tracheotomized and mechanically ventilated to maintain normoxia and normocarbia. Arterial blood pressure, heart rate, and arterial blood gases were monitored using tail artery cannulation throughout the course of the experiments. Rectal temperature was maintained at 37–38°C with a warming blanket. Care was taken to avoid direct heating of the animal's head, since this would alter the normal thermal gradients between the brain and the body core. Bilateral microdialysis cannulae were placed into CA3 region of the hippocampus by stereotaxic surgery and fixed in position with dental cement. Animals were then removed from the stereotaxic apparatus and allowed a 60-min postsurgical equilibration period prior to beginning the experiment.
Microdialysis
Dialysis probes used in these studies were made with modifications (Northington et al., 1995) according to the description of Johnson and Justice (1983). Probes consisted of a single, hollow dialysis fiber, one end of which was sealed with epoxy. The dialysis membrane diameter was 300 μm, and had a molecular mass cut-off of 5 kDa. Two hollow silica perfusion tubes were inserted into the dialysis fiber so that their ends were 5 mm apart. The distance between the tips comprised the effective dialyzing area of the cannula, as verified by perfusion with dye (Northington et al., 1995; Bhardwaj et al., 1995b). After insertion, the cannulae were perfused at a rate of 1 μl/min.
Estimation of NO production was performed using modifications of the assay described previously by Bredt et al. (1990). Arginine is converted to equimolar concentrations of citrulline and NO by the action of NO synthase. During continuous infusion of artificial cerebrospinal fluid (CSF) (containing 3 μM [14C]-L-arginine), 60 μl of effluent dialysate samples were collected over 1-h periods and assayed for [14C]-L-citrulline content. Samples were diluted with 200 μl of water and poured over 0.5 ml resin AG-50WX8 (Na+ form, pH 7.0) 400 mesh columns. Columns were washed with 2 ml buffer containing 30 mM N-2-hydroxymethylpiperazine-2-ethyl ethane sulfonic acid (HEPES) (pH 5.2), 3 mM ethylene diamine tetraacetic acid (EDTA), and 1 ml of water. To determine resin efficiency of arginine trapping, 60 μl of artificial CSF containing 3 μM [14C]-L-arginine, (not used for dialysis) was diluted in 200 μl of water, poured over a column, and washed as above. Radioactivity of the flow-through was quantified by liquid scintillation spectroscopy. Flow-through radioactivity of the non-dialyzed CSF was assumed to be arginine, presuming that no conversion of arginine to citrulline occurred in the CSF not exposed to any source of enzyme activity. Specific activity was corrected for counting efficiency and background activity, and expressed as femtomoles/min of perfusion. As an internal control, 100 μl of CSF not used for dialysis and not exposed to resin was directly assayed for activity to ensure that consistent concentrations of [14C]-L-arginine were added to the CSF. Aliquots of CSF containing known amounts of radiolabeled arginine and citrulline were poured over columns to determine trapping and flow-through efficiency. The efficiency of arginine trapping was >99% and that of citrulline flow-through was >86%. The detection limit was 16 femtomoles/min.
Experimental groups
Rats were divided into nine treatment groups to receive perfusates containing various combinations of ACPD, LNNA, MCPG, dantrolene, MK-801, CNQX, and TTX in artificial CSF with 3 μM [14C]-L-arginine. Within each group, the particular drug combination was randomly assigned to either right or left hippocampus, and the effluent concentrations of [14C]-L-citrulline were compared on a paired basis. Experimental groups were as follows: group 1, control CSF versus 1 mM ACPD (n = 8); group 2, 1 mM ACPD versus 1 mM ACPD + 1 mM LNNA (n = 7); group 3, 1 mM ACPD versus 1 mM ACPD 4 1 mM MCPG (n = 8); group 4, 1 mM ACPD versus 1 mM ACPD 4 1 mM dantrolene (n = 8); group 5, control CSF versus 1 mM MCPG (n = 7); group 6, control CSF versus 1 mM dantrolene (n = 8); group 7, 1 mM ACPD versus 1 mM ACPD + 1 mM MK-801 (n = 8); group 8, 1 mM ACPD versus 1 mM ACPD + 1 mM CNQX (n = 7); and group 9, 1 mM ACPD versus 1 mM ACPD + 1 mM TTX (n = 5).
In preliminary experiments using a similar experimental paradigm, we found that a 1-mM concentration of MK-801 and CNQX attenuates the NMDA and 2-amino-3-hydroxy-5-methylisoxazole-4-propionic acid (AMPA)-enhanced citrulline recovery (Bhardwaj et al., 1994, 1995a, b ). The 1-mM concentration of ACPD that we used in the infusate is relatively high. However, citrulline recovery with these microdialysis probes is <10% (Bhardwaj et al., 1995b), and the concentration of ACPD immediately outside the dialysis membrane is probably <10% that of the infusate.
The dialysate effluent was collected hourly for 5 h starting 1 h after probe insertion. After 5 h, rats were killed with an intravenous (i.v.) injection of potassium chloride while still anesthetized, and dialysis probes were perfused with methylene blue. The animal was decapitated, and the brain removed and stored in 4% paraformaldehyde for 48 h. Brains were then dissected to visualize the probe tracts. The recovery of [14C]-L-citrulline measured in vitro on 14 previously-tested probes (Bhardwaj et al, 1995b) was 9.5 ± 4.5% before implantation and 5.4 ± 3.4% after removal at the end of the experiment. Additional experiments performed at various flow rates (0.5–10 μl/min) indicated that the recovery of [14C]-L-citrulline varied inversely with flow rate. Relative recovery appeared optimal at 1 μl/min.
Materials
[14C]-L-arginine (292 mCi/mmol) was obtained from Amersham Life Sciences Inc. (Arlington, IL, U.S.A.), and LNNA was obtained from Sigma Chemical Co. (St. Louis, MO, U.S.A.). MK-801, CNQX, ACPD, MCPG, and TTX were obtained from Research Biochemical International (Natick, MA, U.S.A.).
Statistical analysis
Within each group, effluent citrulline data were analyzed by two-way analysis of variance (ANOVA) where the two treatments delivered to the two hippocampi was one within-subject factor, and the five hourly collections were a second within-subject factor. If the overall effect of treatment or the treatment × time interaction was significant, comparisons of mean values between the two treatments at individual time points were made by Newman-Keuls multiple range test; p < 0.05 was considered significant. Within each group, time-dependent changes in hemodynamic and blood gas data were determined with one-way repeated measures ANOVA. Data are presented as means ± SD.
RESULTS
In group 1, perfusion with [14C]-L-arginine in artificial CSF resulted in increases in [14C]-L-citrulline in the effluent. The nature of these time-dependent increases represent the time required for diffusion of labeled arginine into the tissue and the time required for diffusion of labeled citrulline back into the probe. In the side perfused with 1 mM ACPD, recovery of labeled citrulline was increased from the third through the fifth hour of perfusion compared to the control side (Fig. 1A). In group 2, combined perfusion of 1 mM ACPD + 1 mM LNNA reduced labeled citrulline recovery from the third to the fifth hour compared to perfusion with 1 mM ACPD alone (Fig. 1B).
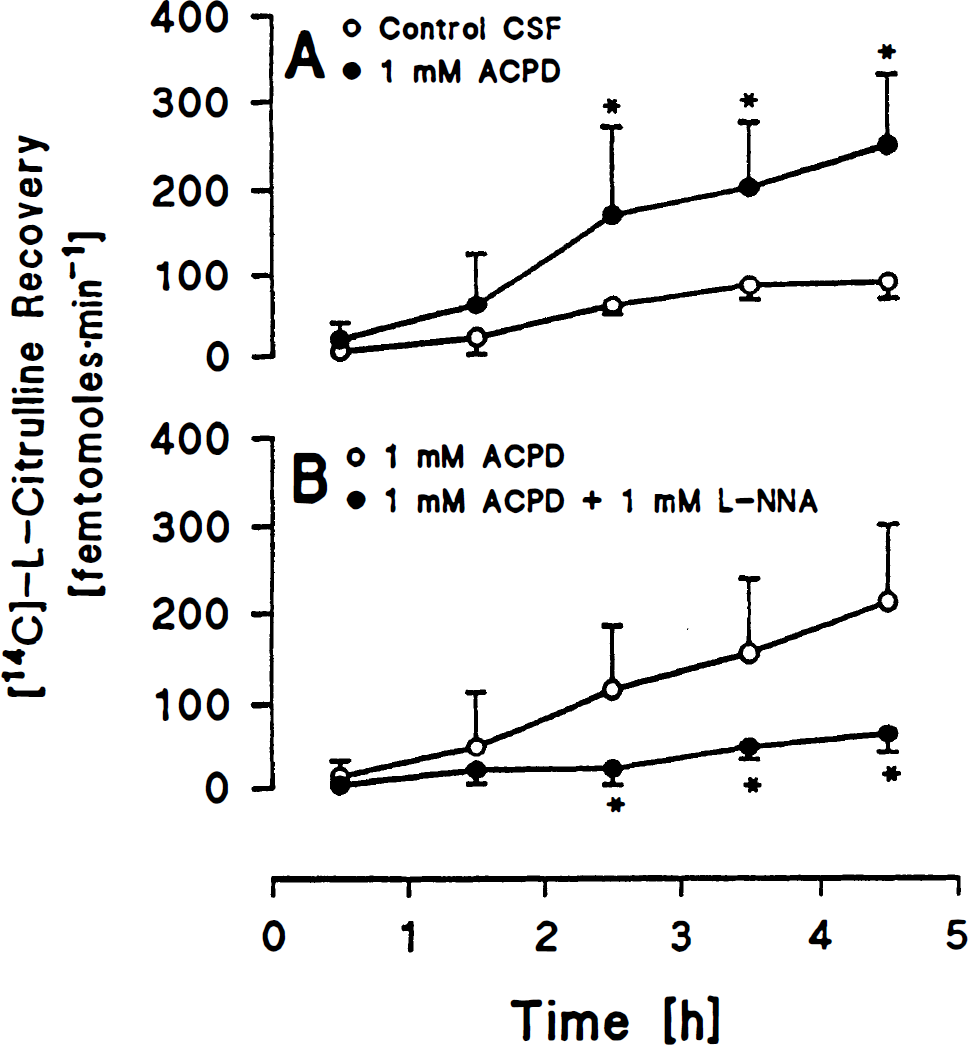
In group 3, combined perfusion of 1 mM ACPD + 1 mM MCPG attenuated labeled citrulline recovery from the third to the fifth hour compared to 1 mM ACPD alone (Fig. 2A). In group 4, combined perfusion of 1 mM ACPD + 1 mM dantrolene attenuated labeled citrulline recovery from the third to the fifth hour compared to perfusion with 1 mM ACPD alone (Fig. 2B).
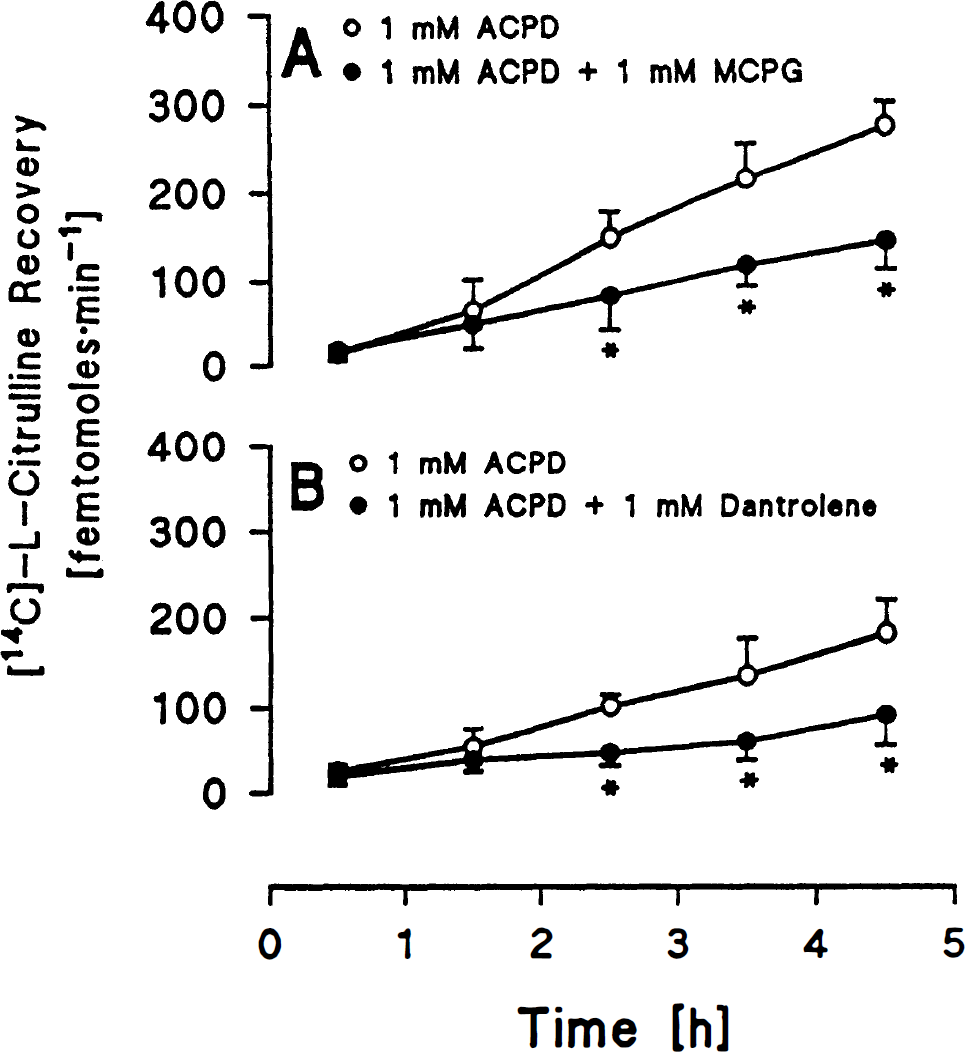
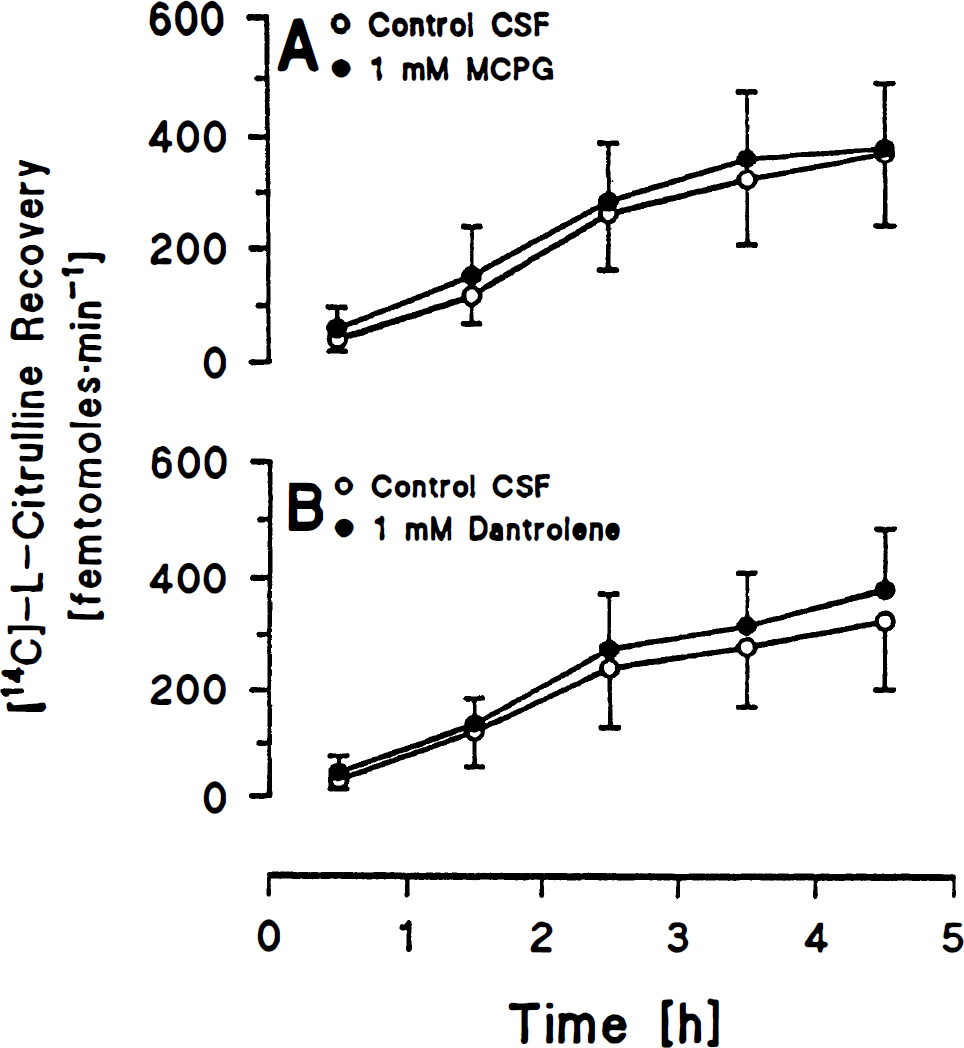
In group 5, perfusion of 1 mM MCPG did not change labeled citrulline recovery as compared to control CSF alone (Fig. 3A). In group 6, perfusion of 1 mM dantrolene did not change labeled citrulline recovery compared to control CSF alone (Fig. 3B).
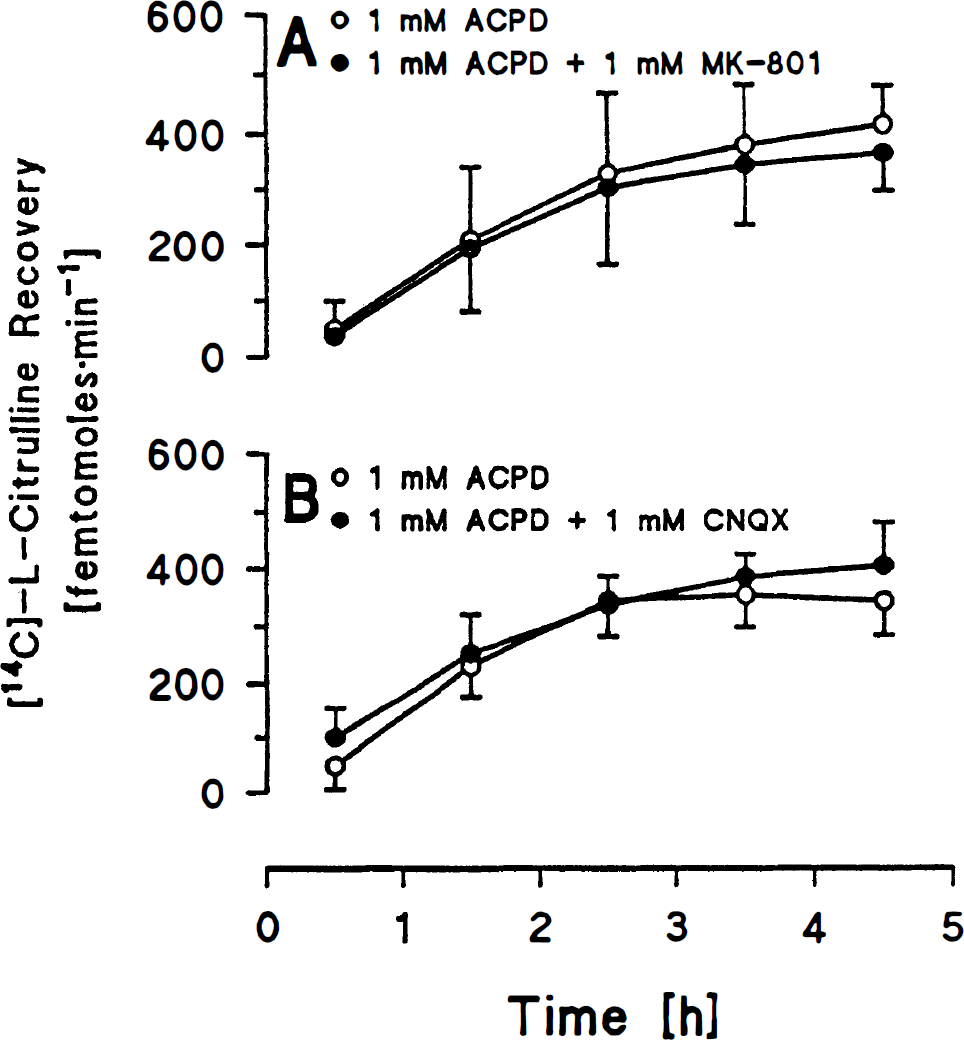
In group 7, combined perfusion of 1 mM ACPD + 1 mM MK-801 did not change labeled citrulline recovery as compared to perfusion with 1 mM ACPD alone throughout the course of the experiment (Fig. 4A). In group 8, combined perfusion of 1 mM ACPD + 1 mM CNQX did not change labeled citrulline recovery as compared to perfusion with 1 mM ACPD alone throughout the course of the experiment (Fig. 4B).
In group 9, combined perfusion of 1 mM ACPD + 1 mM TTX did not change labeled citrulline recovery as compared to perfusion with 1 mM ACPD alone throughout the course of the experiment (Fig. 5).
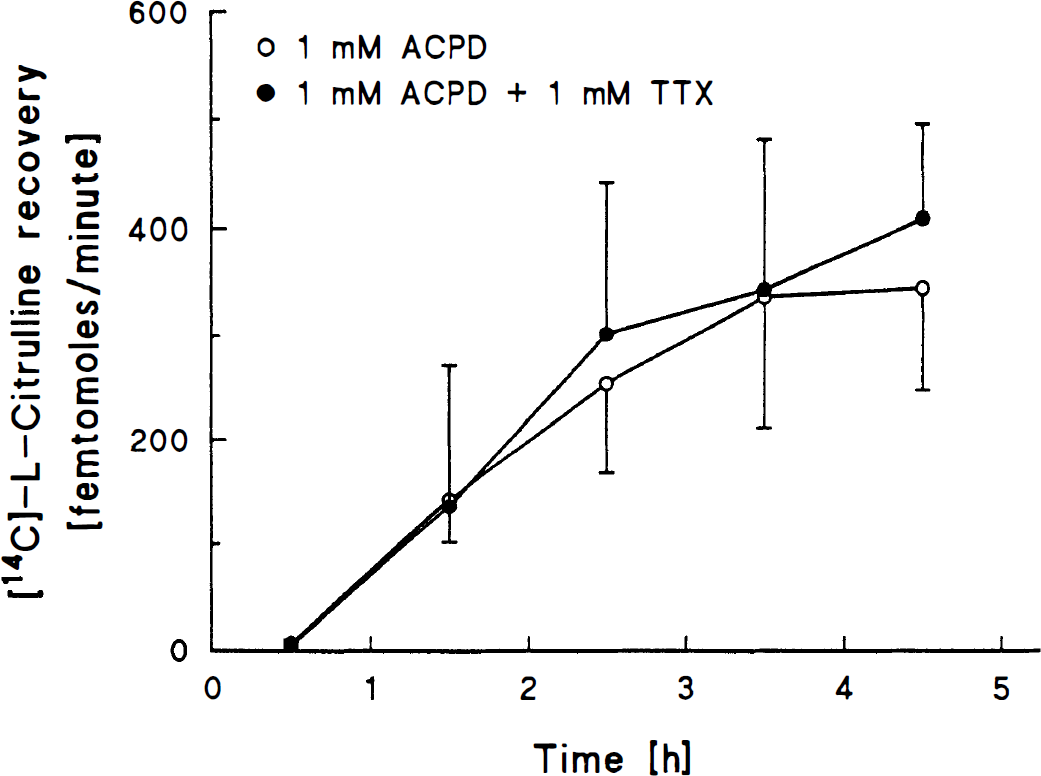
Line graph depicts [14C]-L-citrulline recovery in dialysates from probes perfused with 1 mM trans-ACPD in one hippocampus and 1 mM trans-ACPD + 1 mM TTX in the contralateral hippocampus (n = 5 rats). Values are mean ± SD.
Mean arterial pressure, arterial blood gases, pH, and rectal temperatures were stable throughout the experiment. Values obtained during the fifth hour of perfusion are summarized in Table 1.
Physiological data at 5-h of perfusion
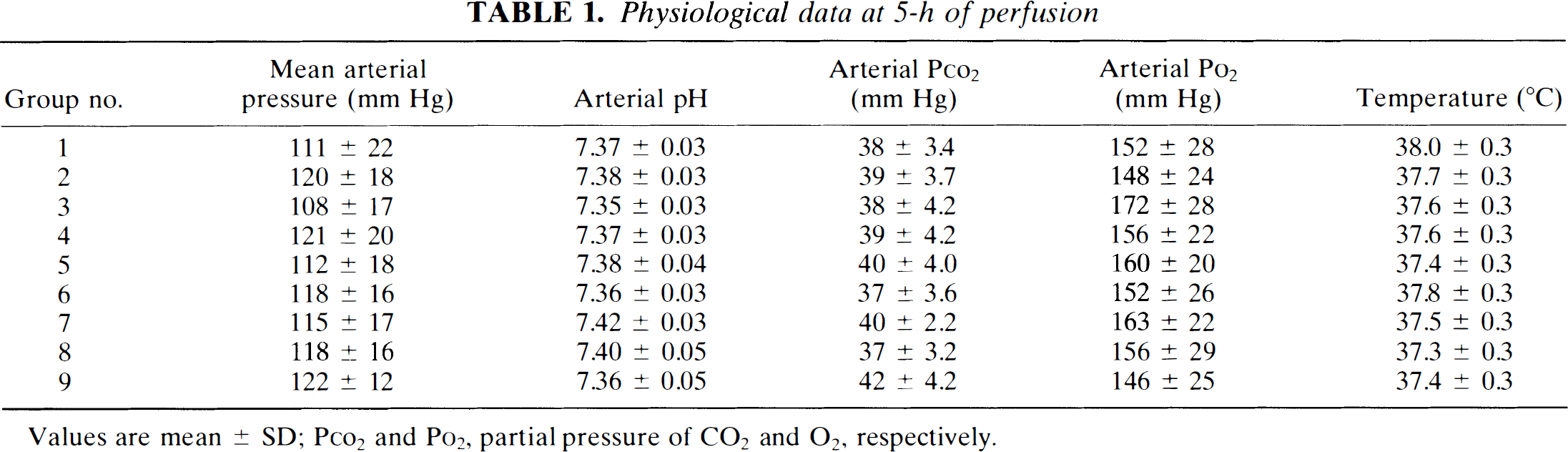
Values are mean ± SD; PCO2 and PO2, partial pressure of CO2 and O2, respectively.
DISCUSSION
The significant finding of this study is that local administration of the mGluR agonist ACPD enhances NO production in the hippocampus in vivo as measured indirectly by conversion of arginine to citrulline. This ACPD-enhanced NO production was attenuated by the mGluR antagonist MCPG but not by MK-801 or CNQX, indicating that the action of ACPD was mediated by mGluR rather than by downstream activation of NMDA or AMPA receptors. Furthermore, the ACPD-evoked increase in citrulline recovery was inhibited by dantrolene, which suggests that calcium-induced calcium release mediated by intracellular ryanodine-sensitive channels plays a role in amplifying the mGluR response.
The tip of the dialysis probes were localized in the CA3 region of the hippocampus, as confirmed by postmortem infusion of methylene blue dye. Neurons containing NOS have been identified in the hippocampus (Bredt et al., 1991; Valtschanoff et al., 1993). We have further confirmed the presence of these neurons immunohistochemically with an antibody to the neuronal isoform of NOS in the pyramidal area of the CA3 hippocampal region (Bhardwaj et al, 1995b; Duneral, 1994). In addition, the endothelial isoform of NOS appears to be present in hippocampal pyramidal neurons (Dinerman et al., 1994); thus, the necessary NO synthetic mechanisms are present. In addition, cellular localization of phosphoinositide-linked mGluR has been demonstrated in the rat hippocampus using antibodies that recognize the C-terminus of the mGluR (Martin et al., 1992).
The present results extend our previous studies with NMDA (Bhardwaj et al., 1994, 1995a, b ; Northington et al., 1995) and indicate that glutamate may regulate NO production in vivo not only via NMDA receptors but also via mGluRs. ACPD may stimulate presynaptic adenyl cyclase-coupled mGluR (Adamson et al., 1990) to facilitate release of glutamate, which could act postsynaptically by stimulating NMDA and nonNMDA receptors on NOS neurons. However, this mechanism seems less likely since MK-801 and CNQX failed to inhibit ACPD-evoked increases in citrulline recovery. Stimulation of AMPA receptors has been reported to inhibit ACPD-evoked increases in inositol triphosphate, and the inhibition was reversed by CNQX (Lonart et al., 1993). We did not find that CNQX augmented ACPD-evoked citrulline release, suggesting that any tonic effect of AMPA receptor stimulation was too low to inhibit an effect of ACPD on NO production. Therefore, the ACPD-stimulated NO production appears to be a response mediated directly by postsynaptic mGluRs. The simplest model to explain these results is that phosphoinositide-coupled mGluR on NOS-containing neurons are directly stimulated by ACPD, resulting in inositol triphosphate receptor-mediated mobilization of intracellular calcium, with further calcium amplification by ryanodine receptors sufficient to stimulate NOS. This interpretation is supported by observations showing that nonpyramidal neurons in CA subfields postsynaptically express the phosphoinositide-linked mGluR1 α (Martin et al., 1992) and that subsets of these neurons are NOS-positive (Dun et al., 1994). Alternatively, the phosphoinositide-linked mGluR5 is the dominant mGluR subtype in the hippocampus (Abe et al., 1992). Activation of this receptor on pyramidal neurons may lead to feed-forward activation (Lacaille et al., 1987) of nonpyramidal neurons, some of which are likely to be NOS-positive. Further support for these observations is derived from immunocytochemical studies showing that inositol triphosphate receptor and ryanodine-sensitive calcium channels are present in the hippocampus (Sharp et al., 1993). However, spatial heterogeneity between these two intracellular calcium channels exists in cell bodies, axons, and dendrites. Thus, regulation of NOS activity by calcium may be dependent upon the loci within the cell, and this spatial regulation may differ in different brain regions.
Another possibility is that ACPD may act via multisynáptic pathways rather than directly on NOS neurons. For example, ACPD activation of mGluR5 on CA3 pyramidal neurons may facilitate hippocampal–septal and septal–hippocampal pathways (Zheng and Gallagher, 1992), which could then evoke the release of acetylcholine in the hippocampus. Lack of inhibition by MK-801 and CNQX argues against a multisynaptic pathway. Moreover, ACPD augments long-term depression and decreases excitatory postsynaptic currents in hippocampal slices (Bolshakov and Siegelbaum, 1994), thereby decreasing efficiency of neurotransmission in specific pathways. However, other reports show that a long-lasting enhancement of synaptic excitability is produced by activation of mGluRs (Liu et al., 1993). Local perfusion with TTX failed to attenuate ACPD-enhanced citrulline recovery, thereby further suggesting that the effect of ACPD is not via neuronal activation caused by epileptiform activity. Although the interstitial concentration of TTX may be an order of magnitude less than the microdialysis perfusate concentration of 1 mmol/L, the local interstitial concentration should be adequate to inhibit epileptiform activity, since only micromolar concentrations of TTX are sufficient for blocking Na+ channels (Katz and Miledi, 1967).
In the absence of ACPD, MCPG and dantrolene did not decrease citrulline recovery below basal levels. These results suggest that any tonic effect of mGluR activity on NO production is relatively small. The lack of a significant decrease below basal levels is unlikely to be due to an inability of the technique to detect significant decreases, since we have previously detected decreases in citrulline recovery below basal levels using 2-chloroadenosine or nitroarginine methyl ester administration (Bhardwaj et al., 1995b; Northington et al., 1995). Alternatively, use of pentobarbital anesthesia may have suppressed tonic mGluR activity in the present study.
Cerebral microdialysis measures neurochemical changes occurring in the brain extracellular fluid, a microenvironment that serves as a communication between neurons, glia, and the vasculature. [14C]-L-citrulline recovery increased over time in controls. This is attributable to (a) diffusion of [14C]-L-arginine away from the dialysis probe over time and recruitment of distant cells capable of producing NO, and (b) time-dependent extrusion of radiolabeled citrulline from the cell, and diffusion back to and across the dialysis membrane. Others have shown increases in cyclic GMP in microdialysis effluent from rat cerebellum within 20 min of adding ACPD to the perfusate (Luo et al., 1994) and in effluent from rat hippocampus within 20 min after adding NMDA to the perfusate (Vallebuona and Raiteri, 1994, 1995). Therefore, a disadvantage of our technique is poor temporal resolution.
Also, a significant fraction of labeled citrulline may be recycled back to arginine, leading to an underestimation of NO production. We assume that this fraction remains relatively constant with ACPD administration. Inhibition of the response with LNNA is used to infer that increased citrulline recovery is mediated by increased NOS activity. However, we cannot exclude that LNNA might also inhibit [14C]-L-arginine uptake (Westergaard et al., 1993). There was also interanimal variability, which we attempted to minimize by using a paired experimental design between the two hippocampi. All drugs were administered locally to reduce the likelihood of systemic or transcallosal interactions. As with all studies using microdialysis, one has to consider the effect of local trauma and consequent astrocytosis. The immunologic NOS (iNOS) isoform can be induced in astrocytes (Endoh et al., 1994; Simmons and Murphy, 1992), and subsets of astrocytes express adenyl cyclase-coupled mGluR (Ohishi et al., 1993). However, we detected significant increases with ACPD by the 2–3-h collection period (3–4 h after probe insertion), and we consider it unlikely that substantial amounts of iNOS are expressed by this time. Moreover, activation of iNOS is calcium independent and we would not expect the response to be blocked with dantrolene, as in this case.
In conclusion, this study demonstrates that mGluR stimulation enhances NO production in the hippocampus as measured in vivo by labeled citrulline recovery following labeled arginine infusion. Inhibition of the response with dantrolene suggests that ryanodine-sensitive calcium channels sufficiently amplify the inositol triphosphate-induced calcium transients to increase calcium-dependent NOS activity. Therefore, mGluRs as well as ionotropic glutamate receptors need to be considered in the regulation of NO production in vivo.
Footnotes
Acknowledgment:
This work was supported, in part, by a grant from the U.S. Public Health National Institute of Neurological Disorders and Stroke (USPHS NINDS) P01-20020. Dr. Bhardwaj is supported, in part, by a National Stroke Association Fellowship Career Development Award and a Richard S. Ross Clinician Scientist Award from the Johns Hopkins University School of Medicine. Dr. Northington is supported by a Merck Clinician Scientist Award from the Johns Hopkins University School of Medicine and by USPHS NINDS K08-1742. Dr. Martin is a recipient of a Leadership and Excellence in Alzheimer's (LEAD) Award from the National Institute on Aging, no. NIA AG 07914. Partial support for these studies were provided by a grant-in-aid from the American Heart Association with funds contributed, in part, by the American Heart Association, Maryland Affiliate.
