Abstract
We tested the hypothesis that increasing durations of focal ischemia that have been shown to result in enlargement of cortical infarct will be associated with progression of behavioral dysfunction that can be measured by a battery of tests sufficiently sensitive and reproducible to detect a positive effect of pharmacotherapy. Untreated or N-methyl-D-aspartate receptor antagonist (CNS-1102)-treated spontaneously hypertensive rats underwent 45, 60, 90, or 120 min of tandem middle cerebral and common carotid artery occlusion followed by reperfusion. We then evaluated the extent of damage and its recovery for up to 21 days using nine behavioral tests aimed at analyzing strength, coordination, and bilateral asymmetry. Also using a graded bioassay that employs a curve-fitting computer program (ALLFIT) to correlate duration of ischemia with degree of behavioral dysfunction, we calculated the average maximal behavioral dysfunction and duration of ischemia required to produce half-maximal behavioral dysfunction and compared these values in untreated controls with analogous values obtained from animals treated with CNS-1102. Three behavioral tests, forearm flex, tape (somatosensory neutralization), and foot-fault placing, were each separately and combined able to distinguish between the degrees of damage produced by increasing durations of ischemia. The behavioral abnormalities assessed using the tape test were reversible within a week, whereas those using forearm flex or foot-fault tests persisted for at least 21 days. CNS-1102 significantly reduced behavioral dysfunction measured by all three tests. This analysis of behavioral dysfunction represents a useful experimental model to grade efficacy of therapies aimed at protecting the brain from damage produced by acute stroke and might also be used to assess recovery from preexisting ischemic damage.
Keywords
Most preclinical evaluations of outcome from stroke have used morphometric assessment of infarct volume as their primary measure. It is logical to assume that the volume of infarction will be closely correlated with outcome. However, several studies of neuroprotective drugs have demonstrated improved memory in the absence of histologic protection after global forebrain ischemia (Grotta et al., 1988, 1990), and behavioral measures are the most reliable outcome in head trauma models (Dixon et al., 1991). Impairment or preservation of complex behavior may reflect changes at a subcellular level or changes in synaptic or electrophysiologic function or may reflect diffuse morphologic changes that are not quantitated by counting dead neurons or drawing a cursor around a contiguous infarcted region. Furthermore, it is important that preclinical drug trials be relevant to help human stroke. In stroke patients, functional (primarily motor) outcome rather than infarct volume is most important. For these reasons, complementation of histological analyses with behavioral testing in the preclinical evaluation of potential stroke therapies is important. To do this, a convenient, predictable, and reliable method of analyzing the nature and extent of behavioral dysfunction that results from ischemia must be established.
Several animal models of cerebral ischemia that attempt to reproduce many of the conditions of human ischemic stroke exist and are commonly used. One such model is the tandem middle cerebral artery/common carotid artery (MCA/CCA) occlusion model in the spontaneously hypertensive rat (SHR) (Coyle, 1986; Brint et al., 1988) which produces ischemia analogous to MCA branch occlusion in humans. This SHR model results in neocortical infarction of more reproducible size and location when histologically evaluated as compared with other strains of rat (Jacewicz, 1992; Aronowski et al., 1994). Although the positive relationship between duration of MCA/CCA occlusion and volume of infarction has been well documented in our laboratory and others (Kaplan et al., 1991; Aronowski et al., 1994), a comparable positive relationship between duration of ischemia and behavioral outcome has not yet been established. Moreover, since many N-methyl-D-aspartate (NMDA) receptor blockers including the noncompetitive antagonist CNS-1102 have been shown to reduce infarct size following focal ischemia, measured either histologically (Aronowski et al., 1994)or with magnetic resonance imaging (Minematsu et al., 1993), a comparison between the profile and magnitude of infarct size reduction and behavioral protection due to this treatment might be used to further characterize the correlation between morphological and behavioral improvement.
Various neurological and behavioral examinations have been employed to evaluate functional impairment of rats after focal cerebral ischemia. In the early search for assessment of neurological deficit following permanent occlusion of different portions of the MCA producing various infarct sizes, Bederson et al. (1986) introduced a crude scale evaluating changes in forelimb flexure, resistance to lateral push, and circling behavior. With use of this crude scale, correlation between histological damage and behavioral deficit could only be seen in rats with both basal ganglia and cerebral cortex infarction. The same model of focal ischemia was further refined by Menzies et al. (1992) who, to improve neurological assessment, added two additional behavioral tests. Many other behavioral and neurological tests to evaluate impairment and recovery after irreversible focal cerebral ischemia have been studied during recent years (Persson et al., 1989; Bharucha et al., 1991; Markgraf et al., 1992; Wahl et al., 1992; Grabowski et al., 1993; DeGraba et al., 1994). In the present study, we evaluated a battery of motor and sensorimotor tasks to select those with the highest potential to discriminate between various degrees of damage as produced by varying durations of MCA/CCA occlusion of SHRs. We found three convenient tests that individually and in combination are able to distinguish between the amount of behavioral dysfunction produced by increasing durations of ischemia. We also observed that an NMDA receptor antagonist was able to ameliorate this dysfunction in a similar fashion to its ability to reduce infarct size as we reported recently (Aronowski et al., 1994).
MATERIALS AND METHODS
Production of ischemia
Male SHRs weighing 250–300 g underwent unilateral reversible focal ischemia of variable duration as previously described (DeGraba et al., 1993; Aronowski et al., 1994). Briefly, with the animal under chloral hydrate anesthesia (single 0.5 g/kg i.p. bolus in 1 ml of saline provided anesthesia lasting at least 2 h), a 0.005-in-diameter stainless-steel wire (Small Parts, Miami, FL, U.S.A.) was placed underneath the left MCA rostral to the rhinal fissure, proximal to the major bifurcation of the MCA, and distal to the lenticulostriate arteries. The artery was then lifted, and the wire rotated clockwise. The left CCA was then occluded using two atraumatic Heifetz aneurysm clips, resulting in a reduction of blood flow in the core of the infarct (4 mm dorsal to MCA occlusion) to 4–8% of the preischemic baseline value throughout the entire period of ischemia, as measured by a Vasamedics Laserflo Blood Perfusion Monitor. After a predetermined period of CCA/MCA occlusion ranging from 0 to 120 min, reperfusion was established by first removing the aneurysm clips from the CCA and then rotating the wire counterclockwise and removing it from beneath the MCA. Temporalis muscle temperature was maintained at 36.5 ± 0.3°C using a heating lamp and warming blanket during ischemia and drug infusion. Since this study was designed specifically to analyze the influence of duration of ischemia on motor performance, we eliminated the trauma that can be produced by femoral vessel cannulation. Therefore, we did not record blood pressure, pH, Po2, or Pco2 changes during surgery.
A separate group of rats were administered CNS-1102 15 min after induction of ischemia with the bolus of 0.5 mg/kg i.v. immediately followed by intravenous infusion at a constant rate of 0.345 mg/kg/h (6 μl/min) for 3 h. For delivery of CNS-1102, we catheterized the cephalic vein ∼6–7 mm proximal to the jugular vein. Untreated animals subjected to this cannulation procedure suffered no visible neurological impairment or any abnormalities when subjected to the behavioral tests.
Behavioral testing
The investigator who performed the behavioral tests was blind as to the treatment and duration of ischemia that each rat underwent. Animals were housed individually in plastic cages and maintained on a 12:12 h light/dark cycle with free access to food and water. Testing was conducted during the light cycle by the same investigator, without moving the rats from their familiar environment. Preferentially, when possible, testing was carried out in the rats' home cages. Other than those tests that required a baseline score, we did not test the animals on the day of surgery to avoid the direct effect of anesthesia as well as to eliminate the effect of stress produced by surgery itself. A total of nine behavioral tests were evaluated. Most of these tests were previously employed in studies of the behavioral effects of cortical lesions (Kolb and Whishaw, 1983; Schallen and Whishaw, 1984; Barth et al., 1990) and analyzed strength/fatigue, activity, balance, and coordination.
Rope suspension—one and two arms. Rats were suspended by their forepaw(s) from a 4-mm-diameter nylon rope, stretched horizontally 70 cm over a foam pad. For one-arm tests, the cotton end of a cotton-tipped applicator was secured to the ipsilateral (to ischemic hemisphere) forepaw with surgical tape, so that the rats were incapable of grasping with that paw. They were then suspended by their (contralateral) forepaw(s) from the rope. Time to falling from the rope was measured using a stop watch. Animals were tested twice a day (two trials per test) on the 2nd and 3rd day after surgery.
Box. Rats were placed in a 30 × 30 cm Plexiglas box with wire mesh bottom, enclosed on all sides but one. The animals were given 15 s to orient themselves while the box was still horizontal. The box was then rotated 90°, so that the open side was face down, and the duration that the rats were able to stay in the box was measured. Animals were tested twice a day (two trials per test) on the 2nd and 3rd days after surgery.
Stationary balance beam. Rats were placed on a 25-mm-diameter, cylindrical, unpolished wooden beam, suspended 40 cm above a foam pad, and time to falling off was measured with a stop watch, for a maximum of 90 s. Rats were tested twice a day (two trials per test) on the 2nd and 3rd days after surgery.
Running beam. The rats were trained to run from one end of a 2-cm-wide, 1.2-m-long rectangular wooden beam to the other using adverse stimuli (white noise). Time to run the length of the beam was measured as the first variable (Dixon et al., 1991). A second variable of contralateral (to MCA/CCA occlusion) minus ipsilateral forelimb slips (errors) was counted and calculated as a percentage of the total number of steps taken as the animal ran along the beam (percent error). Training occurred 1 day prior to surgery (two training sessions of two trials each), and baseline variables were collected the morning of surgery. Animals were tested once a day (two trials per test) on days 2, 3, 5, 7, and 10 after surgery.
Running grid. We constructed a ladder to place over the length of the running beam, consisting of two long bars of 1-cm-diameter Plexiglas and 1.4-mm-diameter wire rungs placed every 2 cm. Again, both variables, the time to run from one end to the other and number of forepaw slips, we recorded as described for the running beam. Training and testing were done exactly as described for the running beam.
Wheel. Using a rat running wheel of 12-in diameter and rung spacing of 2 cm, we closed the sides of the wheel using pasteboard, leaving a securable flap that could be opened and closed to place and retain the animals inside the wheel. The rats were secured inside the wheel, which itself was placed in the home cage, and then videotaped as they ran. One hundred steps taken with the forepaw contralateral to the infarcted hemisphere were observed, and the coincident number of slips of this paw between the rungs of the wheel were counted. The same observations were taken of the ipsilateral forepaw and hindlimbs, and values of slips (error) per steps taken were calculated for each limb. The number of errors was then obtained by subtracting the ipsilateral errors from the contralateral ones. Each animal was tested once a day on postoperative days 2, 5, and 21.
Arm flex. The rats were lifted by their tails so that their ventral surface was exposed to the observer. Cumulative duration of asymmetrical arm flexure during the initial 10 s after lifting was timed using a stop watch, and the “score” was equal to this duration. No flexure resulted in a score of 0, while continuous flexure during the initial 10 s was scored as 10. The animals were tested once a day (two trials per day) on postoperative days 1–8 and again on the 21st day.
Tape test. This test for asymmetry and recovery from asymmetry was previously described and characterized in detail by Schallert and Whishaw (1984). Briefly, Avery self-adhesive labels (1-cm-diameter circles) were placed on each forepaw of the rat in the distal-radial region of the wrist. The time required for the animal to touch and remove each label, and the order (contralateral versus ipsilateral) in which this occurred was established and used to approximate ipsilateral asymmetry. By then affixing larger labels to the wrist less preferred and correspondingly smaller labels to the other wrist, the bias could be extinguished and simultaneously quantified, dependent upon the sizes of labels required. The bigger the ratio between surface of ipsilateral versus contralateral patches (from 1:1 to 1/8:15/8) used to neutralize bias, the higher the score (from 1 to 7) that was assigned to the rat, reflecting more extensive damage. We did no pretraining, other than extensive handling of the animals for at least 7 days prior to the surgery. Each animal was tested on postoperative days 2–4, 6, 8, and 21 (one trial per day).
Composite test scoring and analysis of correlation between duration of ischemia and degree of behavioral damage
Results from those tests that demonstrated positive correlation between degree of behavioral impairment and duration of ischemia (wheel, arm flex, and tape test) were collected and presented as number of errors (slips/steps), time, and score (tape surface ratio necessary to extinguish bias). The same tests that displayed positive correlation between duration of ischemia and amount of behavioral impairment were later utilized to assess the therapeutic effect of CNS-1102. We analyzed each test separately as well as a composite score. Within each test, the two lowest values were averaged and subtracted from the average of the two highest values. This range was then divided into five equal quantities and subsequently scored from 0 to 4. Finally, we evaluated the results by determining the composite score of all three tests, assigning equal weight to each of them. Each rat was therefore scored on three separate tests and given a composite score of performance, for a maximum score of 3 × 4 = 12. Composite score values of individual rats were entered into the computer and analyzed by the curve-fitting computer program ALLFIT to generate a curve describing the correlation between behavioral impairment and duration of ischemia in terms of maximal behavioral dysfunction (BDmax) produced by prolonged ischemia, the time necessary to induce half-maximal behavioral dysfunction (BD50), as well as curve shape and steepness. The computer program (ALLFIT) employed to perform this analysis uses the logistic function y = (a − d)/[1 + (x/c)b] + d, where y is the behavioral dysfunction score, x is the duration of ischemia, a is the response when x = 0, d is the BDmax, b is a slope factor that determines the steepness of the curve, and c is the BD50. This program was developed for the simultaneous fitting of families of sigmoidal dose-response curves and was obtained from the Laboratory of Theoretical and Physical Biology at NIH. This identical curve-fitting method has been used to correlate duration of ischemia versus infarct volume and the effect of CNS-1102 in the same animal model in our laboratory (Aronowski et al., 1994).
Statistical analysis
An analysis-of-variance model with both repeated measures (days) and between group (duration of ischemia and treatment condition) measures was used to evaluate differences in arm flex, wheel, and tape tests. Following this omnibus test, two sets of group comparison were calculated to more closely characterize the nature of differences. Comparison of differences between duration of ischemia within each treatment group separately was calculated using the Newman–Keuls procedure that corrects for multiple testing. Individual comparisons of treated/untreated differences within each duration level were calculated using Student's t test. Regression slope and intercept scores were calculated to each animal based on its tape surface ratios (tape test) raw score over time. Group differences in slopes were then evaluated as a function of duration of ischemia and treatment condition. Intercept values were treated as covariates.
RESULTS
Physiological variables
As mentioned in Materials and Methods, to avoid the effects of blood vessel catheterization on motor performance, we did not monitor blood pressure changes and did not collect blood samples for gas and pH analysis during ischemia. These parameters, however, were monitored previously in animals that were subjected to an identical protocol (including CNS-1102 treatment) and were reported not to be different among the animals (Aronowski et al., 1994).
Behavioral analysis
The first set of analyses was aimed at selecting behavioral test(s) that would allow a grading of the amount of damage after focal ischemia using the duration of ischemia as an index of the amount of damage produced, assuming that greater damage is caused by prolongation of MCA/CCA occlusion (Aronowski et al., 1994). The second set of experiments was to demonstrate that our method of measuring behavioral dysfunction would detect a positive effect of a therapy known to produce reduction of infarct volume.
Initial analyses were directed toward testing changes in strength (or fatigue) of the animals after 0–120 min of ischemia. We tested animals for rope suspension using one (contralateral to ischemic hemisphere) and both forelimbs and also tested the animal's ability to remain on a vertical wire mesh floor (box test). Performance was graded according to the time that the animal could hold onto the rope or mesh floor before falling. Neither of these tests was able to discriminate between the degree of brain damage produced by varying durations of ischemia, as indicated in Table 1.
Analyses of strength/fatigue
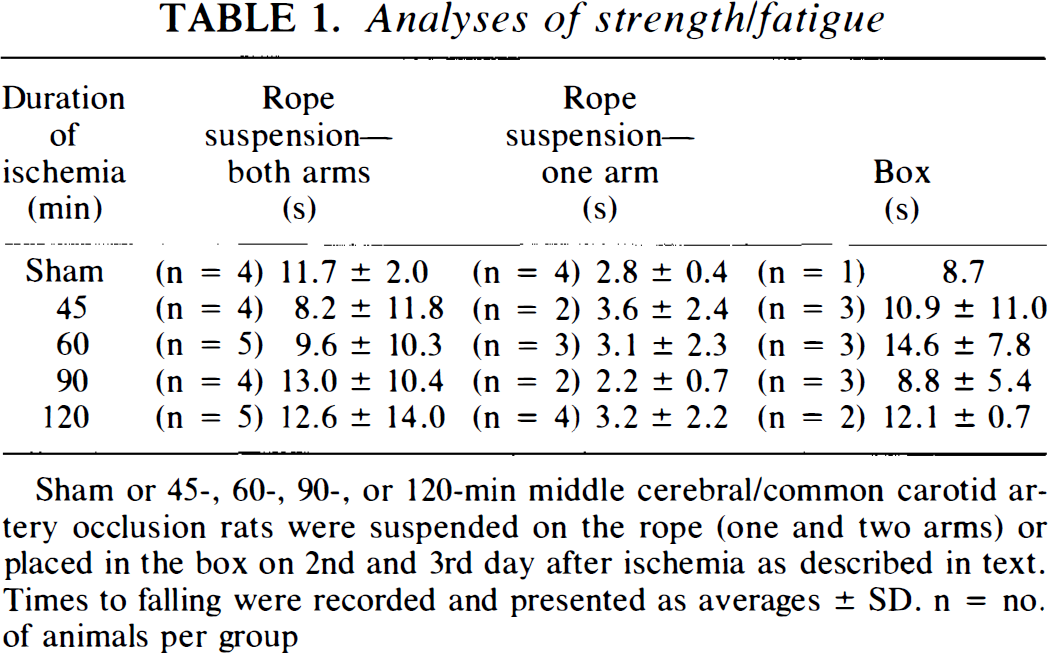
Sham or 45-, 60-, 90-, or 120-min middle cerebral/common carotid artery occlusion rats were suspended on the rope (one and two arms) or placed in the box on 2nd and 3rd day after ischemia as described in text. Times to falling were recorded and presented as averages ± SD. n = no. of animals per group
The stationary balance beam, which measures the length of time that the rat can remain on a cylindrical beam before falling off (for a maximum of 90 s), could not discriminate between the various durations of ischemia (Table 2). The running beam and running grid tests were good indicators of the amount of behavioral dysfunction produced by ischemia; however, this observation was made quite serendipitously. The original purpose of these tests was to measure the time necessary to traverse the length of the beam (or overlaid grid), but we did not find any correlation between this time and the duration of ischemic insult (Table 2). We observed, however, that injured animals slipped more times with the forepaw that was contralateral to the hemisphere of occlusion. These slips were counted as a supplementary measure and quantified as a percentage of missed (error) versus total steps taken while running on the beam/grid. Animals subjected to 45 min of ischemia had 16% (p < 0.07) and 14% more missed steps than sham-operated rats, and there was an additional increase of 23 and 18% of missed steps among animals after 60 min of ischemia for running beam and running grid, respectively.
Analyses of balance and coordination
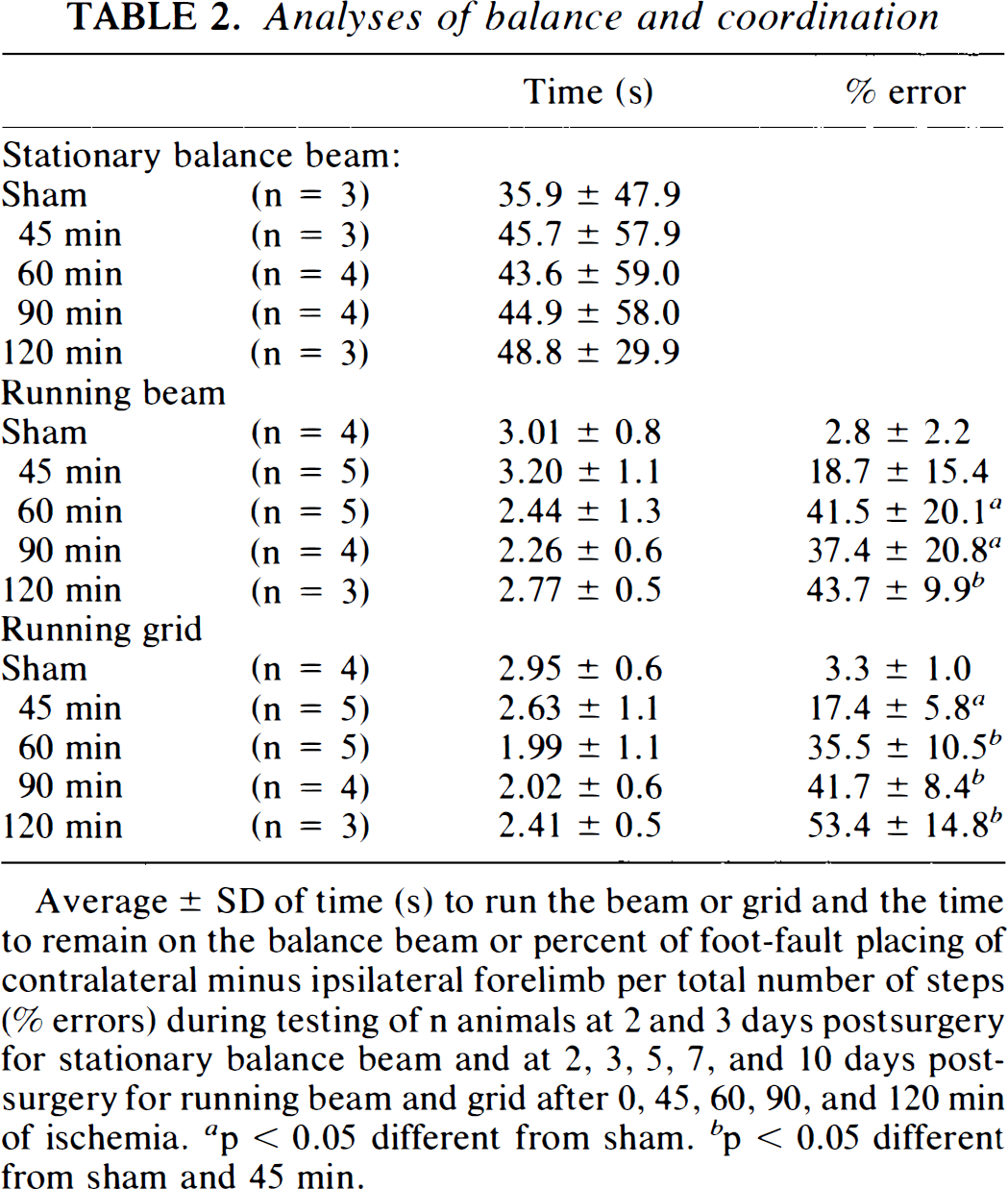
Average ± SD of time (s) to run the beam or grid and the time to remain on the balance beam or percent of foot-fault placing of contralateral minus ipsilateral forelimb per total number of steps (% errors) during testing of n animals at 2 and 3 days postsurgery for stationary balance beam and at 2, 3, 5, 7, and 10 days postsurgery for running beam and grid after 0, 45, 60, 90, and 120 min of ischemia, a p < 0.05 different from sham, b p < 0.05 different from sham and 45 min.
The wheel test represents a modification of the running grid and analyzes the frequency of foot-fault placement of the forepaws while stepping on the rungs within the wheel. Recording of the animal's movement in the wheel, however, was continuous, allowing for the assessment of many more steps per trial. Optimal scoring of this test required videotaping of the animal's performance to enable careful reviewing and quantitation of errors. No signs of ptosis were noticed in any of the analyzed animals. The results of the repeated measures analysis of variance indicated that the number of errors on the wheel task differed as a function of duration of ischemia and treatment group, but did not differ across days. Since the number of errors did not differ across days, implying that damage was permanent for up to 21 days, an average error score was calculated for each animal. Using this test, we observed that increased durations of CCA/MCA occlusion resulted in a gradual increase in contralateral foot-faults (errors) while running (Fig. 1A). There was no effect of any duration of ischemia on the errors made by the ipsilateral forepaw or the hindlimbs (data not presented). Assuming that 120 min of ischemia produces maximal dysfunction, we found that 45 min of ischemia resulted in a behavioral dysfunction that was 46% of maximum (Fig. 1A). We had also found that the same duration of ischemia resulted in half-maximal histological damage (Aronowski et al., 1994). We observed 70 and 88% of maximal dysfunction after 60 and 90 min of ischemia, respectively.
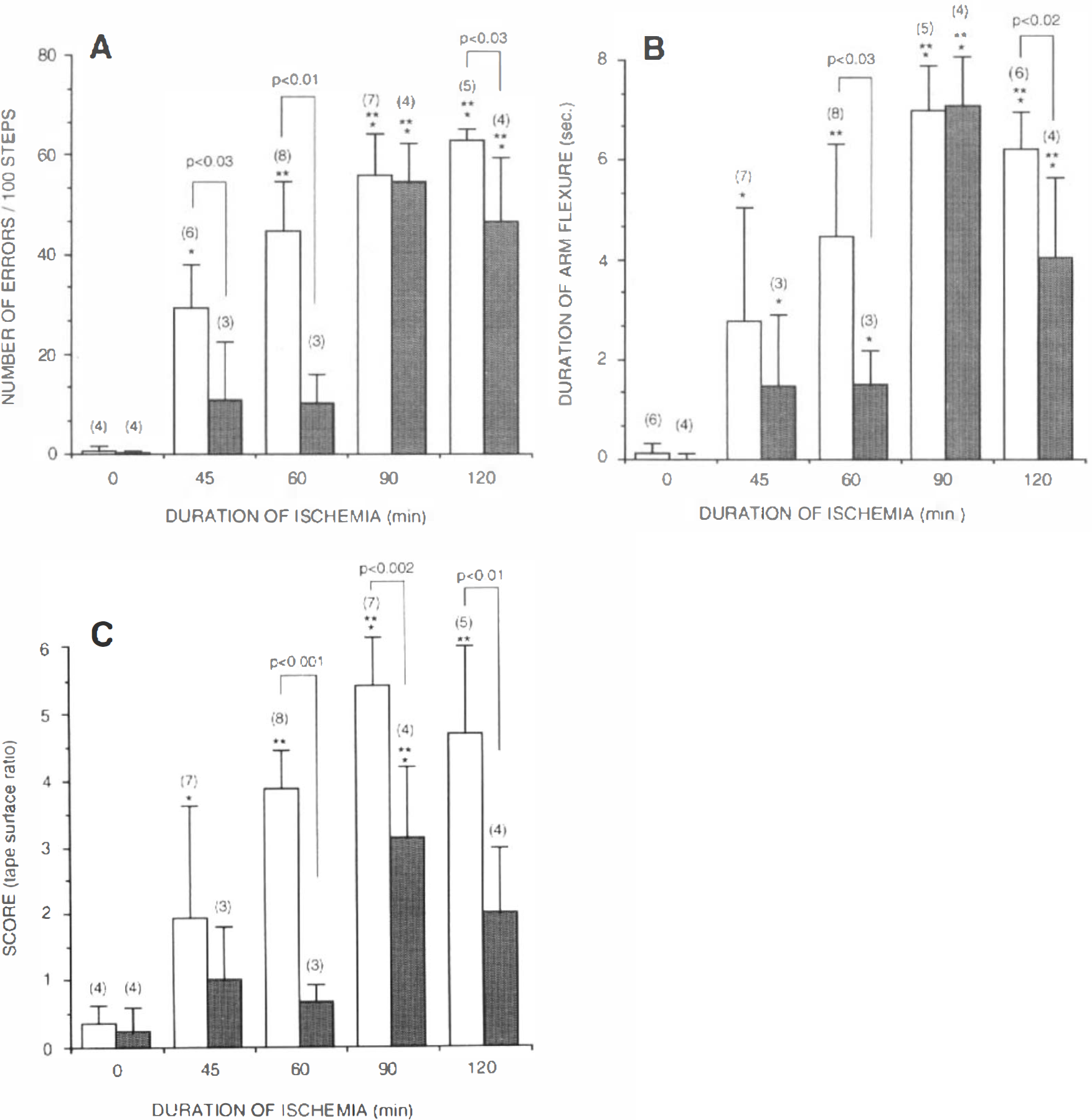
Control (open bars) and CNS-1102-treated (shaded bars) rats after 0, 45, 60, 90, and 120 min of ischemia were tested and scored for impairments analyzed with the use of either wheel foot-fault placement
CNS-1102 produced a substantial decrease in number of errors, reducing the dysfunction by 62, 77, and 27% for 45, 60, and 120 min of ischemia, respectively, as compared with untreated rats.
The amount of impairment produced by all durations of ischemia, measured by foot-fault placing in the wheel, was permanent for at least 21 days after ischemia, and CNS-1102, apart from reducing the initial impairment (Fig. 1A), did not help to improve its recovery (data not included).
Measuring the time that the animal retained its forelimb (contralateral to the infarcted hemisphere) flexure while being suspended by the tail for 10 s was a good indicator of the extent of cortical damage after CCA/MCA occlusion. Since the duration of flexure did not differ across days within each duration of ischemia, an average error score was calculated for each animal. Rats with longer durations of ischemia flexed their forelimb longer than those rats subjected to shorter durations (Fig. 1B). This analysis of change in posture graded the amount of damage produced by durations of ischemia up to 90 min and impairment produced by 45 min of ischemia resulted ∼40% maximal impairment under the assumption that damage produced by 90 min of ischemia was 100%.
Treatment with CNS-1102, similar to results with the wheel test, improved the amount of dysfunction produced by all durations of ischemia except 90 min. CNS-1102 shortened duration of flexure by 46, 66, and 35% after 45, 60, and 120 min of ischemia, respectively.
The amount of impairment produced by the various durations of ischemia, as measured by the arm flex test, was irreversible for at least 21 days, and CNS-1102, apart from reducing the initial amount of impairment, did not have a significant effect on promoting recovery (data not presented).
The tape test (sensorimotor asymmetry test) showed that all animals after CCA/MCA occlusion preferentially contacted and removed a sticky tape from the forelimb ipsilateral to the ischemic hemisphere, when labels of identical size and shape were applied to both forelimbs. This preference was neutralized by increasing the size of the tape placed on the contralateral forelimb with simultaneous decrease of the size of the tape on the ipsilateral forepaw. Animals after longer durations of ischemia required a higher ratio (score) between the size of contra- versus ipsilateral tape to extinguish the asymmetry. The tape test performed on the second day after ischemia was able to distinguish between graded ischemic durations of up to 90 min, but did not discriminate between 90 vs. 120 min of ischemia (Figs. 1C and 2).
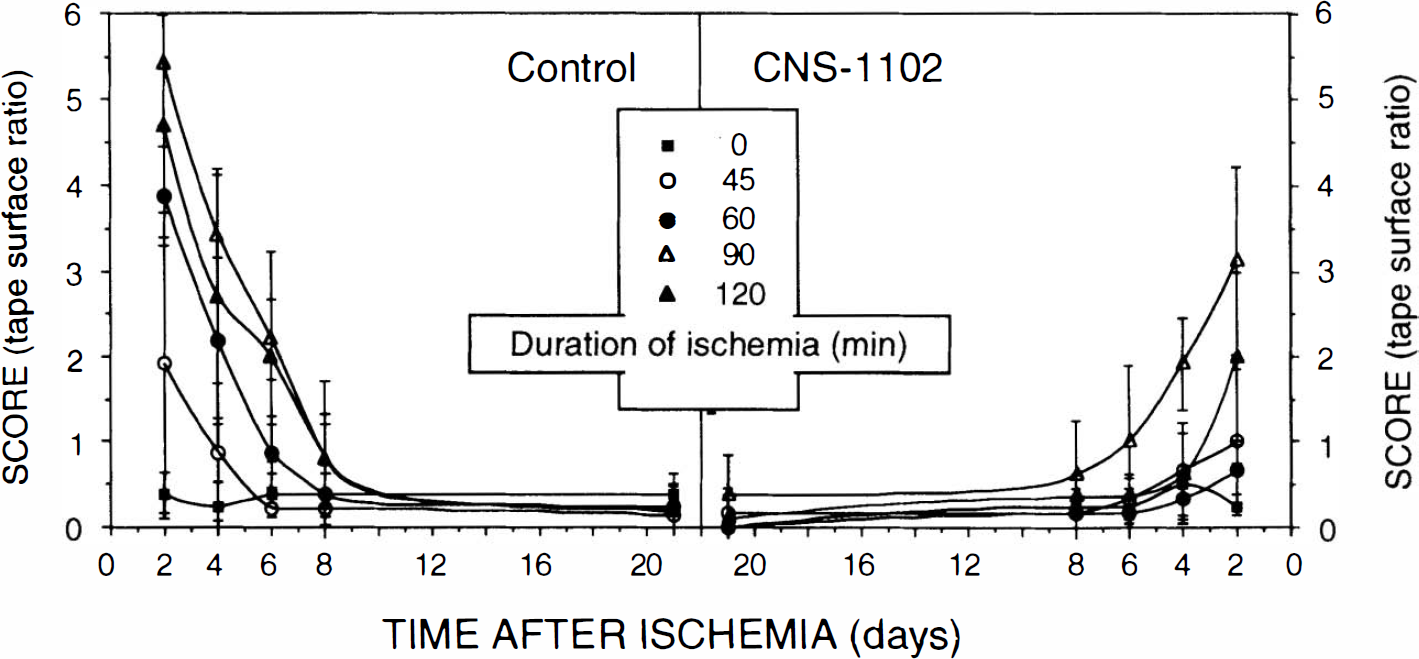
Postischemic recovery of impairment analyzed using the tape test. Presented values represent mean ± SD scores for the tape test during testing at 2, 4, 6, 8, and 21 days after 0 (sham), 45, 60, 90, and 120 min of ischemia using (n) rats for each duration of ischemia as indicated in Fig. 1C.
CNS-1102 greatly reduced the asymmetry produced by ischemia, protecting animals for up to 60 min and reducing impairment produced by 90 and 120 min of ischemia by ∼40–50% (Fig. 1C).
The dysfunction produced by ischemia and quantitated using the tape test recovered within days after ischemia (Fig. 2). Recovery rate was proportional to the degree of initial damage; animals after 45 min of ischemia recovered within the first 6 days, whereas the recovery rate from 60 min of ischemia was ∼8 days. None of the animals showed any signs of impairment 21 days after ischemia (Fig. 2).
CNS-1102, apart from reducing the initial amount of impairment, did not accelerate its recovery. The results of analysis of recovery (slopes) indicated that once the initial damage due to ischemia (intercept score) was controlled statistically, no differences between treatment or duration of ischemia were found (all F < 1.0).
Composite score analysis
The behavioral analyses of individual tests described and analyzed allowed us to select the three tests, namely, arm flex, wheel, and tape tests, that best discriminated between duration of ischemia and degree of impairment.
Since the use of multiple behavioral tests to analyze extent of injury after stroke in clinical studies might provide the best overall description of outcome (Tang et al., 1993), in addition to analyses the individual tests, we decided to combine tape, arm flex, and wheel test scores, assigning equal weight to each of them, to create a final score as described in Materials and Methods. Additionally, our recent observations have indicated that the relationship between duration of ischemia and infarct volume can be fitted and described by a sigmoidal function (Aronowski et al., 1994). Therefore, we decided to present our behavior composite analysis using the same criteria as for histological evaluation. Computer analysis of the composite scores (Fig. 3) indicates that the predicted duration of ischemia that produces half-maximal neurological dysfunction occurs after 53.6 min of ischemia in controls and that half-maximal damage will not occur before 67.8 min of ischemia in animals treated with CNS-1102. Average maximal behavioral dysfunction was also reduced by CNS-1102 treatment from 11.43 ± 0.51 to 8.08 ± 7.43 (mean ± SE) (Fig. 3). This curve-fitting method is very reproducible; the curves presented in Fig. 3 comprise values obtained during four consecutive experiments for untreated and two experiments for treated rats that individually produced very similar curves, including BD50, BDmax, and the curve slope (data not included).
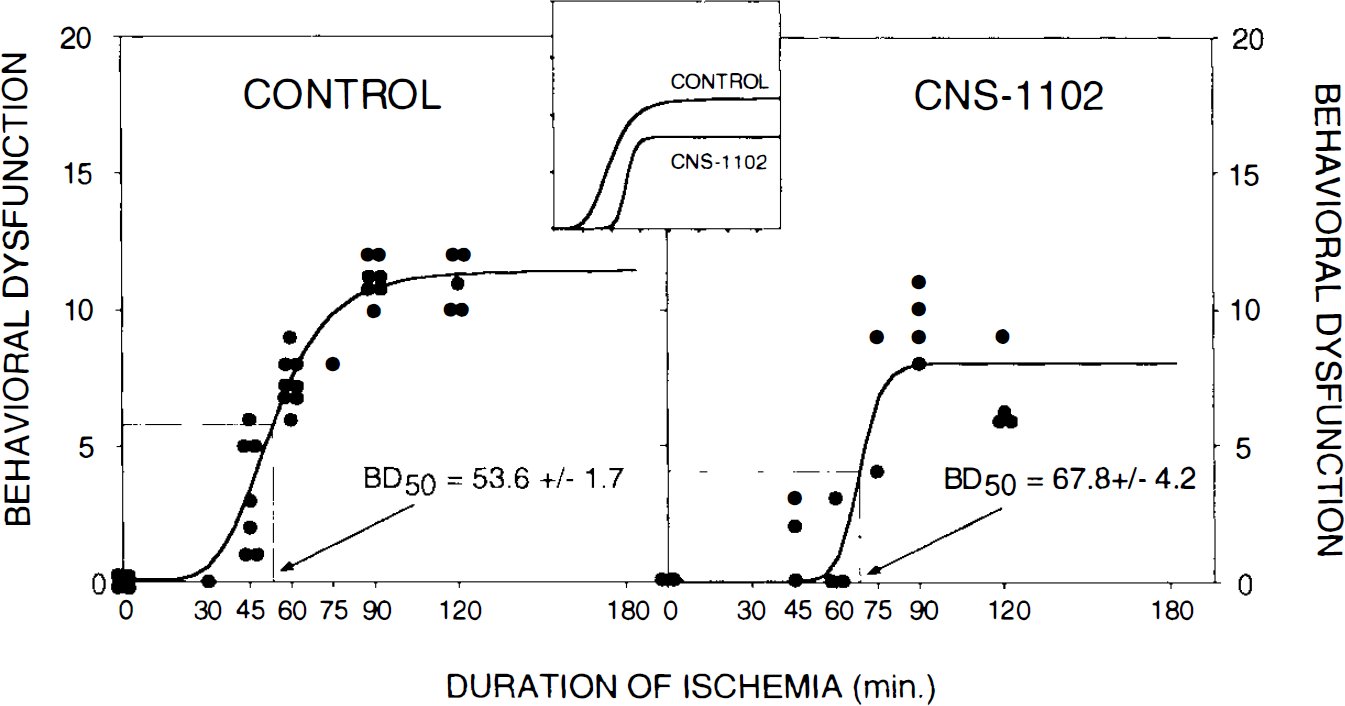
Curve representing the correlation between duration of ischemia (min) and behavioral dysfunction (higher score = greater dysfunction) obtained using the logistic function as described in the text. Each symbol indicates the behavioral dysfunction from one experimental animal. Control and CNS-1102-treated animals were analyzed. Time (mean ± SE) required to produce half-maximal dysfunction (SD50) is indicated.
DISCUSSION
The purpose of this study was to establish a convenient, reliable, and predictable method for analyzing the nature and extent of behavioral dysfunction that results from varying degrees of cortical damage produced by tandem MCA/CCA occlusion. Recently, we reported on behavioral abnormalities using the balance beam test (presently stationary balance beam) and the prehensile traction test (rope suspension—both arms). However, these tests identified behavioral impairments only after permanent MCA occlusion, and large numbers of animals were required to produce these results (DeGraba et al., 1994). Our objective in this study was to develop a test that was sufficiently sensitive to discriminate neurological deficits after shorter durations of ischemia, consequently presenting more covert behavioral problems while utilizing fewer animals. Therefore, we evaluated nine tests to find those that were most sensitive to the deficits produced by the cortical infarction resulting from short to intermediate durations of ischemia in our model.
The rope suspension (both arms and one arm) and the box primarily measure strength, and while this is an important aspect of motor performance, our data show that these tests are not sensitive enough to differentiate between rats with relatively small differences in occlusion times. Similarly, the speed component of the running beam and running grid tests showed no correlation between the behavioral variable and duration of ischemia.
The remaining tests, all displaying a significant degree of correlation, were the error (foot-fault) component of the running beam and grid tests, the wheel, the arm flex, and finally the tape test. Among these tests, data obtained from the last two provide unique information, whereas the first three can be redundant. The narrow beam walking test has been suggested to be a sensitive indicator for detecting hindlimb rather than forepaw deficit (Kolb and Whishaw, 1983; Barth et al., 1990). Using this test, we did not find any significant deficit in hindlimb placement, suggesting that the sensorimotor region of the cortex responsible for hindlimb placement was not affected by the durations of ischemia produced in this study. The running beam, grid, and wheel tests all measure the number of slips made per total number of steps taken. Since the wheel can be performed in the animal's cage and because the rats cover a greater distance on the wheel as compared with the beam and grid, thereby increasing the sensitivity and accuracy of the test, we included only the wheel test and not the running beam or grid in the behavioral battery used to calculate BD50.
The arm flex test used in our study represents a modification of the test described by Bederson et al. (1986), where instead of using the binomial scale determining the presence or absence of dysfunction, we introduced ranking of the impairment based on the time that the animal retains its contralateral forelimb flexure. We observed that there was good correlation between duration of ischemia and duration of arm flexure.
Finally, we evaluated the tape test, or somatosensory neutralization test, which was first characterized and described by Schallert and Whishaw (1984) as a sensitive measure of the extent of sensorimotor impairment. This test has also been used in studies to characterize the nature of damage to the cerebral cortex including those produced by ischemia (Markgraf et al., 1992). This test is clinically analogous to the “simultaneous extinction” exercise used in patients with frontal or parietal cortex lesions.
The deficits analyzed by the tape, arm flex, and foot-fault placing (Wheel) tests, which were capable of detecting behavior changes produced by our model of focal ischemia, appear to be at least in part under the control of proprioception and motor and sensorimotor processing and integration, which correspond to the area of cortex found in our histological analyses to be the infarcted region after tandem MCA/CCA occlusion (DeGraba et al., 1993, 1994; Aronowski et al., 1994). Distinct fields of cerebral cortex were proposed to be responsible for the presence and extent of dysfunction tested either by tape or foot-fault placing (wheel) tests (Barth et al., 1990). While the arm flex tests forepaw posture, presumably reflecting changes in motor/proprioception functions affecting muscle tone, the wheel test can quantify the accuracy of limb placement, presumably reflecting impairment in motor coordination and proprioception, although a deficit in posture precluding normal coordinated limb placement cannot be excluded. Finally, the tape test analyzes impairment of both somatic and sensorimotor functions, which, unlike the previous impairment variables, showed significant recovery over time. The recovery rate from impairment measured by the tape test was shown to vary between electrolytic lesions produced in distinct subfields of the cerebral cortex (Barth et al., 1990). This implies that the functional impairment tested by the tape test does not necessarily coexist with the two other impairments, indicating different anatomical origins of injury. Alternatively, the result may also suggest the same anatomical origin of injury, but a different mechanism or locus of recovery. It can also be postulated that impairment produced by ischemia and characterized by aberrant asymmetry (tape test) may reflect the degree of cerebral edema and that the eventual reduction of edema would result in recovery of asymmetry.
For these reasons, we considered each of the three tests to be complementary with the others in evaluating the extent of damage induced by ischemia, and this suggested a need for multiple test measurements (including recovery) to reliably measure the extent of damage.
As has been already suggested (Bederson et al., 1985; Wahl et al., 1992; Tang et al., 1993), a battery of behavioral tests, as opposed to a single test, may be optimal to characterize the nature of impairment produced by ischemia. Theoretically, if behavioral outcome is used to assess the amount of neuroprotection obtained with effective neuroprotective agents, improvement of only one of several behavioral functions should be noted, but might be considered as a sign of only partial improvement, proportional to the total number of tests not affected (or only partially affected), and used to evaluate improvement. The more tests used to evaluate improvement (or deterioration), the more valid the reading of general outcome is to be expected.
Many systems in biology can be described by a sigmoidal relationship (Zivin and Waud, 1992). Glutamate release and subsequent excitotoxicity are recognized as a major contributor to neuronal death after ischemia, and the dose–response curve of glutamate toxicity on neuronal survivability fits a sigmoidal curve (Finkbeiner and Stevens, 1988). The evolution of infarct volume (visualized by triphenyltetrazolium chloride staining) after MCA/CCA occlusion is directly correlated to the duration of occlusion. We know that brief periods of ischemia will produce no infarction, and when infarction is complete, further increases in duration of ischemia cannot cause any further damage. Thus, the data will fit some type of sigmoidal curve better than any line. Recently, we found that the correlation between duration of ischemia and infarct volume can be described by a sigmoidal curve (Aronowski et al., 1994). Since behavioral dysfunction should be proportional to the extent of histological damage, the relationship between duration of ischemia and behavioral dysfunction might also be described by a similar sigmoidal relationship. Recently, spontaneous neurological recovery after variable durations of impairment in stroke patients has been reported and also appears to fit a sigmoidal function (Minematsu et al., 1992).
We have shown that after global ischemia, neuroprotective therapy can produce more functional than histologic improvement (Grotta et al., 1988, 1990). In the present study, for logistic reasons, we did not measure histologic damage in the same animals undergoing behavioral analysis. However, in a previous study using an identical model, ischemic durations, and dose of CNS-1102, we reported the relationship of cortical infarct volume to duration of ischemia (Aronowski et al., 1994). While behavioral dysfunction in this study was proportional to the amount of histologic damage we had measured previously (Aronowski et al., 1994), we found that CNS-1102 reduced the BDmax by 30%, whereas it had no effect on the maximal infarct volume. This difference in drug effect confirms our earlier observation that in models of cerebral ischemia, functional measures can detect pharmacoprotection that is not detected by light microscopic examination. We hypothesize that our behavioral tests measure integrated sensorimotor tasks controlled by brain regions that extend beyond the infarcted regions. Therefore, they can be improved by drugs that have an ameliorating effect on receptor function, ion flux, subcellular signaling, etc., in brain regions where histologic damage cannot be measured.
In conclusion, we have developed a reliable and reproducible method for testing functional outcome after focal cortical infarct in rats that should add validity to the preclinical evaluation of potential therapies.
Footnotes
Acknowledgment:
This work was supported by National Institutes of Health NINDS grant NS23979 (J.C.G.). CNS-1102 was supplied by Cambridge Neuroscience (Cambridge, MA, U.S.A.). We thank Dr. Timothy Shallert for his help in setting up the tape test assay and Dr. Randolph Nudo for critical reading of the manuscript.
