Abstract
Tacrolimus (FK506), an immunosuppressive drug, is known to have potent neuroprotective activity and attenuate cerebral infarction in experimental models of stroke. Here we assess the neuroprotective efficacy of tacrolimus in a nonhuman primate model of stroke, photochemically induced thrombotic occlusion of the middle cerebral artery (MCA) in cynomolgus monkeys. In the first experiment, tacrolimus (0.01, 0.032, or 0.1 mg/kg) was intravenously administered immediately after MCA occlusion, and neurologic deficits and cerebral infarction volumes were assessed 24 hours after the ischemic insult. Tacrolimus dose-dependently reduced neurologic deficits and infarction volume in the cerebral cortex, with statistically significant amelioration of neurologic deficits at 0.032 and 0.1 mg/kg and significant reduction of infarction at 0.1 mg/kg. In the second experiment, the long-term efficacy of tacrolimus on neurologic deficits and cerebral infarction was assessed. Vehicle-treated monkeys exhibited persistent and severe deficits in motor and sensory function for up to 28 days. A single intravenous bolus injection of tacrolimus (0.1 or 0.2 mg/kg) produced long-lasting amelioration of neurologic deficits and significant reduction of infarction volume. In conclusion, we have provided compelling evidence that a single dose of tacrolimus not only reduces brain infarction but also ameliorates long-term neurologic deficits in a nonhuman primate model of stroke, strengthening the view that tacrolimus might be beneficial in treating stroke patients.
Keywords
Whereas a number of neuroprotective agents have reached phase 3 clinical trials for stroke, no promising results have been achieved (Clark et al., 2001; Diener et al., 2000; Lyden et al., 2002). The reasons for failure of neuroprotectants in clinical trials have been the subject of intense discussion (Gorelick, 2000; STAIR, 1999; STAIR-II, 2001). One reason for failure is the difficulty in translating the preclinical data obtained from rodents into a clinical setting. Whereas stroke models in rodents are easy to conduct and ischemic outcomes are generally uniform, allowing for robust detection of neuroprotective efficacy, human strokes exhibit considerable heterogeneity in both time-course and area of infarction. Furthermore, the obvious behavioral, anatomic, and circulatory differences between rodents and humans undermine the predictive value of these models.
Considering the failure of many clinical trials mentioned above, it is highly desirable to evaluate the neuroprotective efficacies of candidate compounds in larger animal models, which presumably mimic human stroke better and predict the clinical benefit, as recommended by STAIR-I (STAIR, 1999). Nonhuman primate stroke models offer distinct advantages to address the limitations of traditional rodent models. Nonhuman primates show striking similarity to humans in their microvascular collateral circulation and brain architecture, such as the ratio of white matter to gray matter (Gillilan, 1968). Occlusion of middle cerebral artery (MCA) in nonhuman primates produces tissue damage in the sylvian cortex, putamen, and subcortical white matter in the affected MCA territory, as commonly seen in human stroke patients (Darby et al., 1999; Watanabe et al., 1977). The damage in the white matter could be of particular importance because the ratio of the white matter to gray matter in monkeys is much closer to the ratio in humans than that in rodents. It is also desirable to establish a thrombotic MCA occlusion model in which the MCA is spontaneously recanalized. As many stroke patients show reperfusion of the MCA spontaneously or by thrombolytic therapeutic intervention (Ringelstein et al., 1992), these models would mirror human strokes more closely and allow better prediction of clinical outcomes. Furthermore, nonhuman primates exhibit more integrated behavioral changes following strokes, providing clinically relevant outcome measures generally lacking in rodent models, where assessment of infarct volume remains a common endpoint despite the unclear relationship between histopathologic changes and clinical outcomes. Thus, numerous neuroprotectants have produced robust reductions in infarct volume, only to generate disappointing outcomes in clinical trials. Assessing both behavioral and histopathologic changes in nonhuman primate would provide a vital platform for extrapolating to efficacy in humans. Such a platform also allows for the assessment of long-term behavioral changes, which more closely matches the later endpoints used in clinical trials.
Tacrolimus (FK506, Prograf) is an immunosuppressant used clinically to prevent allograft rejection and is known to inhibit calcineurin-mediated T-cell activation by forming a complex with FKBP12 (Liu et al., 1991). The drug has been well-characterized to have neuroprotective action on ischemic brain damage in rodents (Bochelen et al., 1999; Furuichi et al., 2003; Maeda et al., 2002; Sharkey and Butcher, 1994; Yagita et al., 1996). Because the neuroprotective effect of tacrolimus was first demonstrated to be mediated by inhibition of calcineurin activity (Sharkey and Butcher, 1994), the mechanism for antiischemic effects of tacrolimus have been studied, and antiinflammatory actions (Tsujikawa et al., 1998; Wakita et al., 1998) and antiapoptotic actions (Herr et al., 1999; Wang et al., 1999) have been proposed. The ischemic brain damage, including inflammatory reactions (Jean et al., 1998) and apoptotic cell death (Tsukada et al., 2001), similarly have been observed in primate stroke models. The aim of the present study was to evaluate the long-term neuroprotective efficacy of tacrolimus in a robust nonhuman primate stroke model, that is, MCA occlusion in cynomolgus monkeys. Using this model, we assessed neurologic deficits and infarction volumes following a single bolus dose of tacrolimus, both acutely (24 hours after the ischemic insult) and over a longer time course (28 days).
MATERIALS AND METHODS
Animals
Experiments were carried out using 3- to 5-year-old male cynomolgus monkeys weighing 3.1 to 5.5 kg purchased from Shin Nippon Biomedical Laboratories (Kagoshima, Japan). Animals were housed individually in cages 60 cm wide by 70 cm high by 70 cm deep (in accordance with the NIH requirement), at 23 ± 2°C, 55 ± 5% humidity, under 12-hour light/dark cycle (light on at 7:00 a.m.) for at least 1 week before experimental procedures. In the short-term study, 24 monkeys were used. There was no demise during the experimental period. In the long-term study, two monkeys died beyond 1 week after ischemia; one death was caused by severe brain edema, and the other monkey died possibly of infection. Thus, 24 monkeys were used for the long-term study, and 22 of them survived to 28 days. These studies were approved by the Ethics Committee for Laboratory Animal Experiments at Fujisawa Pharmaceutical Co., Ltd. The experimental protocol for the present study is schematically represented in Fig. 1.
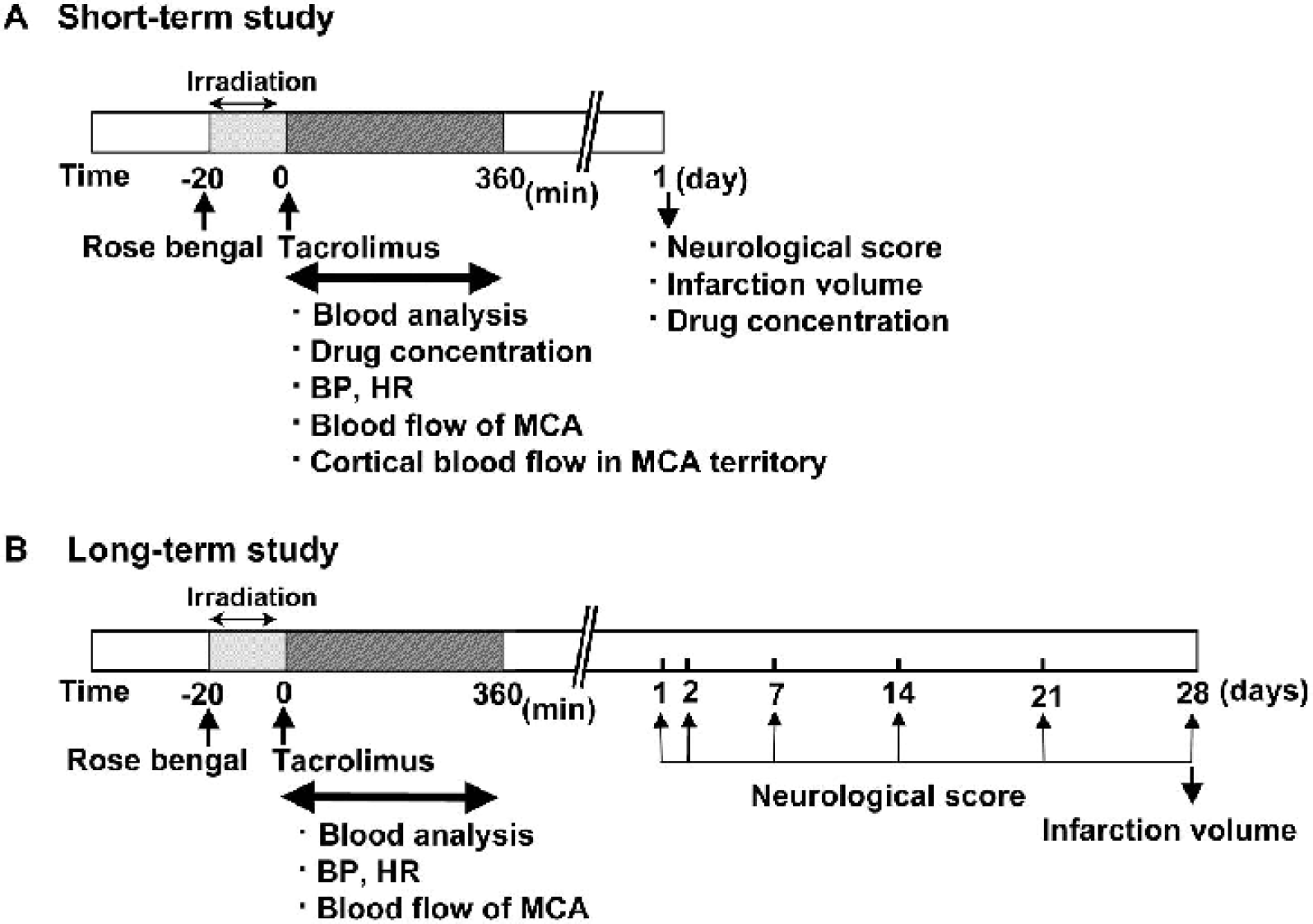
Experimental protocols. A short-term study
Surgical procedure
Surgery was carried out according to the method reported by Kaku et al. (1998). Cynomolgus monkeys were lightly anesthetized by intramuscular injection of ketamine hydrochloride (10 mg/kg), tracheostomized, and artificially ventilated immediately after intramuscular injection of atropine (0.05 mg/kg) and pancuronium bromide (0.05 mg/kg). Anesthesia was maintained with 0.6 to 1% isoflurane in a N2O:O2 gas mixture (N2O:O2 = 7:3), and immobilization was accomplished with 0.05 mg/kg of pancuronium bromide given intramuscularly every 2 hours for up to 6 hours after ischemia. The right femoral vein was cannulated with a polyethylene tube (PE50) for administration of rose bengal or test drug. The right femoral artery was also cannulated for measurement of blood pressure and heart rate, as well as blood sampling for blood gas analysis and determination of drug concentration. The right MCA was occluded by a transorbital approach. Under an operating microscope, the temporalis muscle was sectioned, and subtemporal craniotomy was performed without removing the zygomatic arch. The orbital content was dissected and excised. A window approximately 10 mm in diameter was opened just anterior to the foramen of the mandibular nerve at the skull base. The main trunk of the right MCA was visible through the window underneath the dura mater. After opening the dura mater, an optic fiber for green light irradiation was placed on the proximal site of the main MCA trunk, and the probe for a pulsed Doppler flowmeter (Model PDV-20, HHP-20, Crystal Biotech America, Northborough, MA, U.S.A.) for blood flow measurement was placed next to the optic fiber. Photoillumination with green light (wavelength, 540 nm; intensity, 4,000,000 lux) was achieved by using a xenon lamp (L-4887, Hamamatsu Photonics, Hamamatsu, Japan) with a heat-absorbing filter and a green filter. Irradiation was directed by the optic fiber mounted on the MCA trunk, and rose bengal (20 mg/kg) was infused intravenously for 6 minutes. Photoillumination was carried out for 20 minutes. After wound closure, buprenorphine hydrochloride (4 μg/kg) and cefazolin sodium hydrate (25 mg/kg) were intramuscularly administered to avoid pain and infection. Thereafter, anesthesia was stopped. After confirming good spontaneous breathing, each animal was returned to the home cage. Although most animals could feed themselves within 24 hours after surgery, some animals that could not sit upright and feed themselves were orally treated with eutrophic liquid, and their body temperature was maintained under a heating lamp. In the long-term study, analgesic and antibiotic were given daily for the first week. Rectal temperature was maintained at 37.5 ± 0.5 °C with a heating pad (TR-100, PS-100, Fine Science Tools, Canada) during surgery and regional cerebral blood flow (rCBF) measurement afterward.
Measurement of cerebral blood flow
Complete MCA photothrombotic occlusion was confirmed by the decrease of blood flow monitored for 6 hours after MCA occlusion by a pulsed Doppler flowmeter placed on the proximal MCA trunk. The time from rose-bengal injection to complete occlusion of the MCA and the time to first recanalization were measured. In the short-term study, rCBF at the surface of the cerebral cortex was also monitored for 6 hours after MCA occlusion. To perform continuous monitoring of cortical rCBF, the scalp and temporalis muscle were cut, and a small window was made 5 mm anterior and 35 mm lateral to the bregma. CBF was monitored by a laser-Doppler flowmeter (Omegaflow, FLO-N1, Neuroscience, Osaka, Japan), with the probe placed over the dura mater at the site. Change in rCBF is expressed as percentage of the baseline values.
Measurement of physiologic parameters
Arterial blood pressure, heart rate (AP-601G, AT-601G, Nihon Kohden, Tokyo, Japan), po2, pco2, pH, hematocrit, hemoglobin, and blood glucose (pH/blood gas analyzer i-STAT,i-STAT Corporation, Princeton, NJ, U.S.A.) were measured through a femoral arterial line at baseline and at 90, 180, 270, and 360 minutes after thrombotic occlusion for each group in the first acute experiment.
Assessment of neurologic deficits
Animals were subjected to a neurologic examination at 24 hours after MCA occlusion for the short-term study (Fig. 1A) and at day 1, day 2, and every 7 days for 28 days after MCA occlusion for the long-term study (Fig. 1B). Neurologic assessment was carried out in a blind fashion. Drugs were prepared and administered by the investigator who carried out surgery, and the scoring of neurologic deficits was carried out by another investigator who was a specialist for large animal care and was blinded to the treatment. The scoring method reported by Kito et al. (2001) was adopted as described in Table 1, where neurologic deficits were divided into the following categories: consciousness, motor system, muscle coordination, and sensory system. During the long-term study, animals were examined daily for obvious signs of pain or discomfort. Food intake and general health were also noted.
Neurologic scores for the monkey stroke model

“ipsi-” and “contra” indicate ipsilateral and contralateral to MCA occlusion.
Measurement of the volume of ischemic brain damage
For the short-term study, each monkey was deeply anesthetized with pentobarbital (50 mg/kg, i.p.) after neurologic examination, perfused transcardially with saline containing heparin (50 U/mL), and sacrificed. The brain was removed, and the cerebrum was coronally sectioned in 4-mm thickness using a brain slicer for cynomolgus money (Neuroscience, Osaka, Japan). The fourth coronal section was then sliced in 2-mm thickness to use the rostral slice for measurement of the drug concentration and the caudal slice for histologic assessment. The coronal sections for histologic assessment were immersed in neutrally buffered 10% formalin/PBS for at least 2 days. Each section was embedded in paraffin and cut into 3- to 4-μm thickness to make 21 consecutive slices and stained with hematoxylin-eosin. For the long-term study, the consecutive coronal slices were prepared in the same manner as for the short-term study and stained with hematoxylin-eosin. Using photographs of 22 consecutive slices, the area of ischemic brain damage and the whole area of the cerebrum were calculated using a computerized image analysis system. The area of infarction was calculated by subtracting the area of intact tissue in the ipsilateral brain region from the whole area of the contralateral brain region. The brain damage in each animal was expressed as the percentage of the sum of the area of damaged brain in comparison with the sum of the whole area of the cerebrum, and the volume of ischemic brain damage was calculated using the trapezoid method (Rosen, 1990).
Measurement of brain and blood concentration of tacrolimus
In the short-term study, arterial blood samples were collected from the cannulated femoral artery at 5, 15, 30, 90, 180, and 360 minutes and 24 hours after tacrolimus administration. Brain samples were taken at 24 hours after drug administration. Cortical and basal ganglia punches were made for measurement of drug concentration. Blood and brain samples were stored at −20°C until drug concentration was measured by enzyme immunoassay.
Drug
The injection formulation of tacrolimus prepared at Fujisawa Pharmaceutical Co. Ltd. (Osaka, Japan) was diluted with physiologic saline and administered intravenously at the doses of 0.01, 0.032, 0.1, and 0.2 mg/kg immediately after MCA occlusion. The vehicle ampoule prepared at Fujisawa Pharmaceutical Co., Ltd. was similarly diluted with physiologic saline and administered to monkeys for the vehicle-treated group. The injection volume was adjusted to 1 mL/kg throughout all experiments.
Statistical analyses
All values are mean ± SD. Statistical significance was evaluated using analysis of variance (ANOVA) followed by post hoc Dunnett's multiple comparison test for physiologic parameters, rCBF, and ischemic brain damage. Neurologic scores were analyzed with nonparametric Kruskal-Wallis test and post hoc Dunnett's multiple comparison test. P values less than 0.05 were considered significant.
RESULTS
Short-term efficacy of tacrolimus
In the first set of experiments, neurologic deficits and volumes of cerebral infarction were determined 24 hours after the ischemic insult to assess the dose-response effect for neuroprotective actions of tacrolimus. Physiologic variables, cerebral blood flow, and the concentration of tacrolimus in the blood and brain were also measured.
Cerebral blood flow
The MCA was occluded by thrombus formation, leading to a gradual decrease of cortical CBF. In most animals, cyclical reperfusion and reocclusion of the MCA were observed. The time from rose-bengal injection to complete occlusion of the MCA was 12.2 minutes for the vehicle-treated group and 13.8, 12.1, and 13.6 minutes for the groups treated with 0.01, 0.032, and 0.1 mg/kg of tacrolimus, respectively (Fig. 2A). The average time to first recanalization was 89.0 minutes for the vehicle-treated group and 66.8, 121.7, and 66.1 minutes for the groups treated with 0.01, 0.032, and 0.1 mg/kg of tacrolimus, respectively (Fig. 2B). There were no statistically significant differences in the time to occlusion and first recanalization among groups. Cortical CBF gradually decreased in most animals following MCA occlusion, although cortical CBF showed various patterns in individual animals. The average reduction of CBF at 360 minutes after MCA occlusion was 53.2% for the vehicle-treated group and 30.6%, 38.5%, and 34.8% for the 0.01, 0.032, and 0.1 mg/kg tacrolimus groups, respectively (Fig. 2C). There were no significant differences among groups.
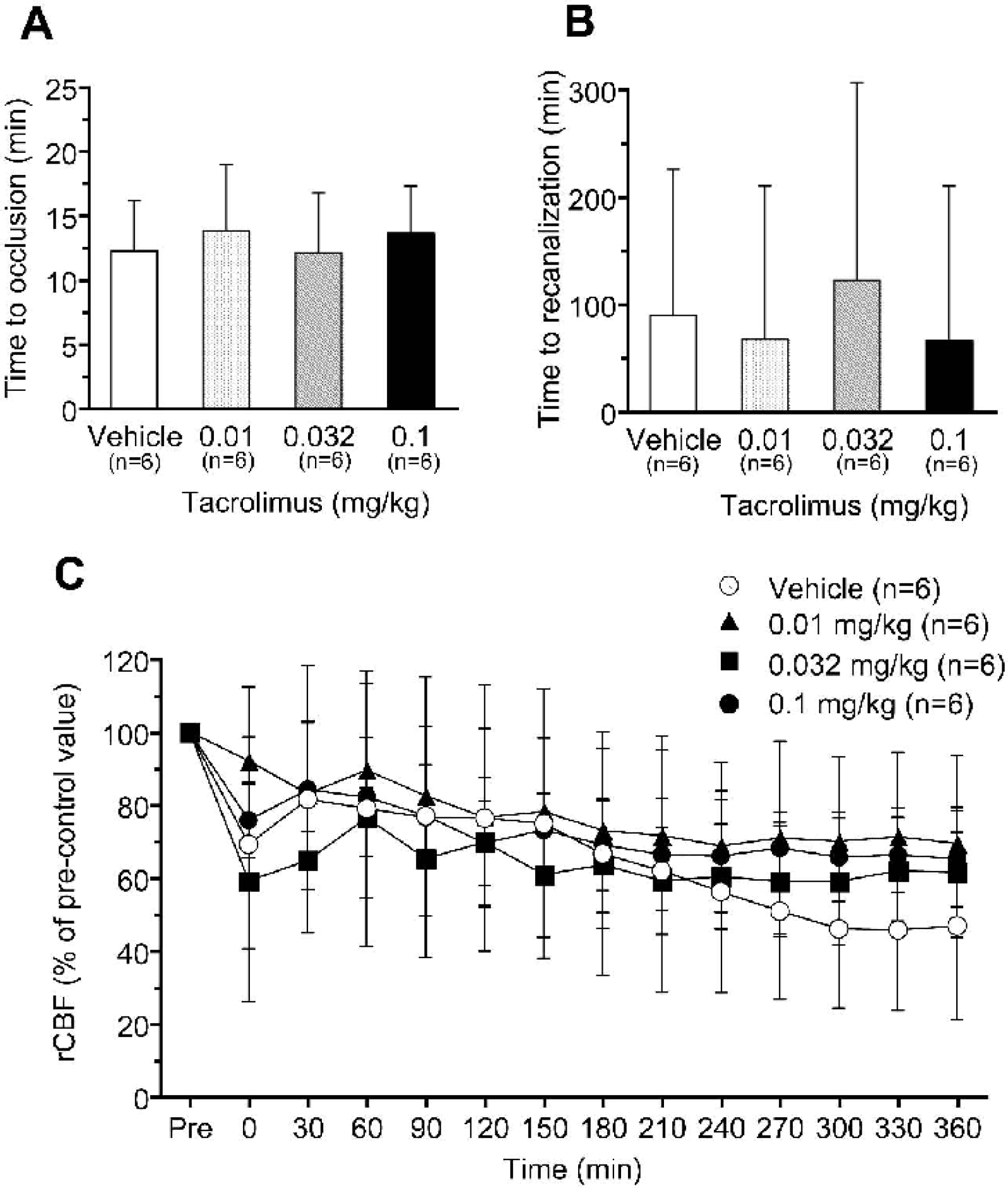
Effects of tacrolimus on the time to complete occlusion of the middle cerebral artery (MCA)
Physiologic parameters
Table 2 shows the physiologic parameters. In the vehicle-treated group, mean blood pressure (MBP) and heart rate (HR) gradually decreased and blood glucose increased after MCA occlusion. The value of pco2 at 90 minutes after MCA occlusion significantly increased but was within physiologically normal range. There were no significant differences between vehicle- and tacrolimus-treated groups except for MBP, where the tacrolimus-treated groups showed significantly higher MBP at some time points compared with the vehicle-treated control group. However, these effects showed neither time dependency nor dose dependency. Other parameters, including HR, po2, pco2, pH, hematocrit, hemoglobin, and blood glucose, were not influenced by the administration of tacrolimus.
Effects of tacrolimus on physiological parameters after thrombotic MCA occlusion in monkeys
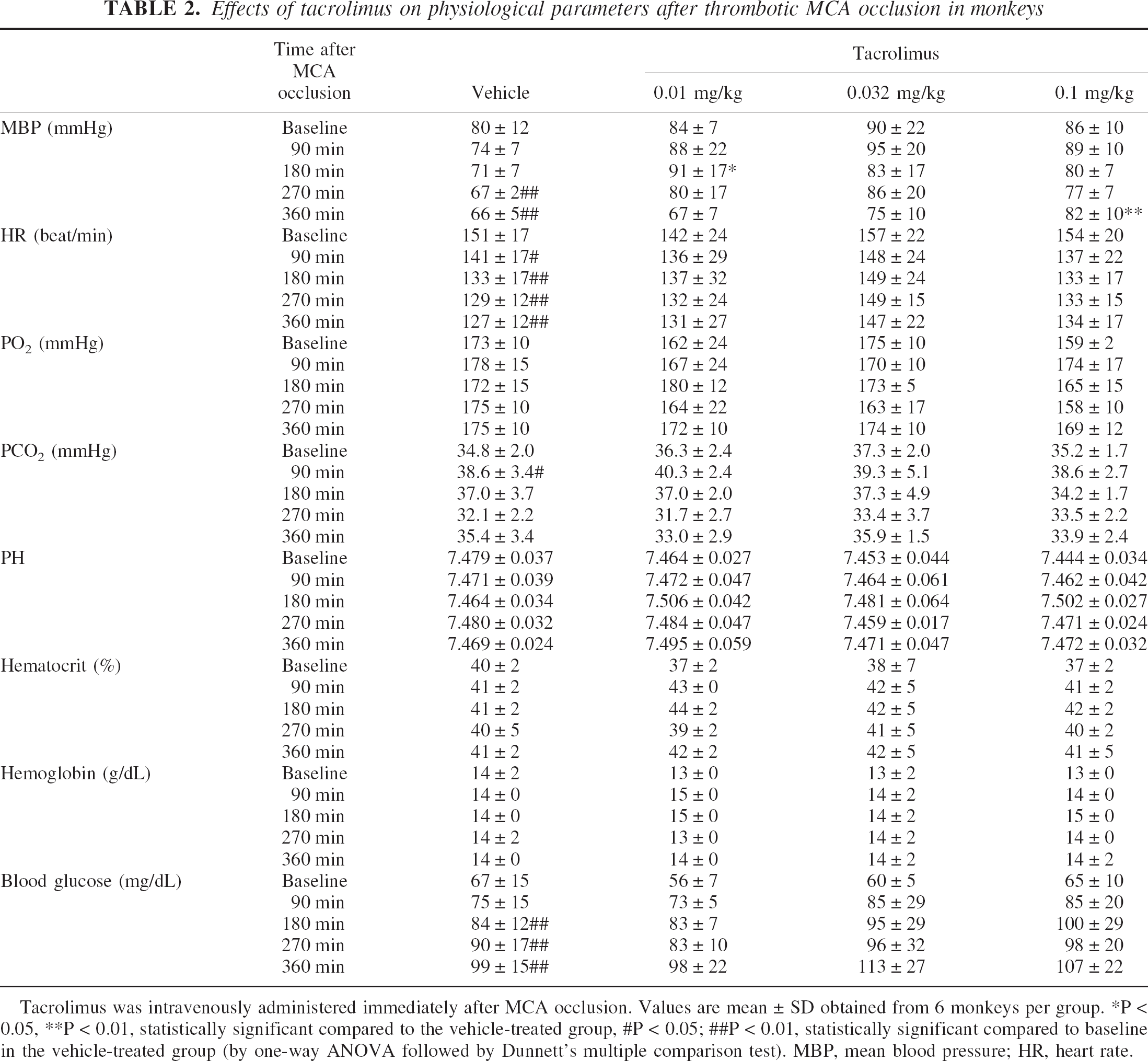
Tacrolimus was intravenously administered immediately after MCA occlusion. Values are mean ± SD obtained from 6 monkeys per group.
P < 0.05
P < 0.01, statistically significant compared to the vehicle-treated group
P < 0.05
P < 0.01, statistically significant compared to baseline in the vehicle-treated group (by one-way ANOVA followed by Dunnett's multiple comparison test). MBP, mean blood pressure; HR, heart rate.
Blood and brain concentration of tacrolimus
The blood concentrations of tacrolimus at a dose of 0.1 mg/kg declined with a half-life of 12.19 ± 1.45 hours. The brain concentrations of tacrolimus at 24 hours after administration in the ischemic and nonischemic cortices were similar. In the basal ganglia, the concentrations of tacrolimus were also comparable between ischemic and nonischemic sides (Table 3).
Blood and brain concentrations of tacrolimus in monkeys with middle cerebral artery occlusion
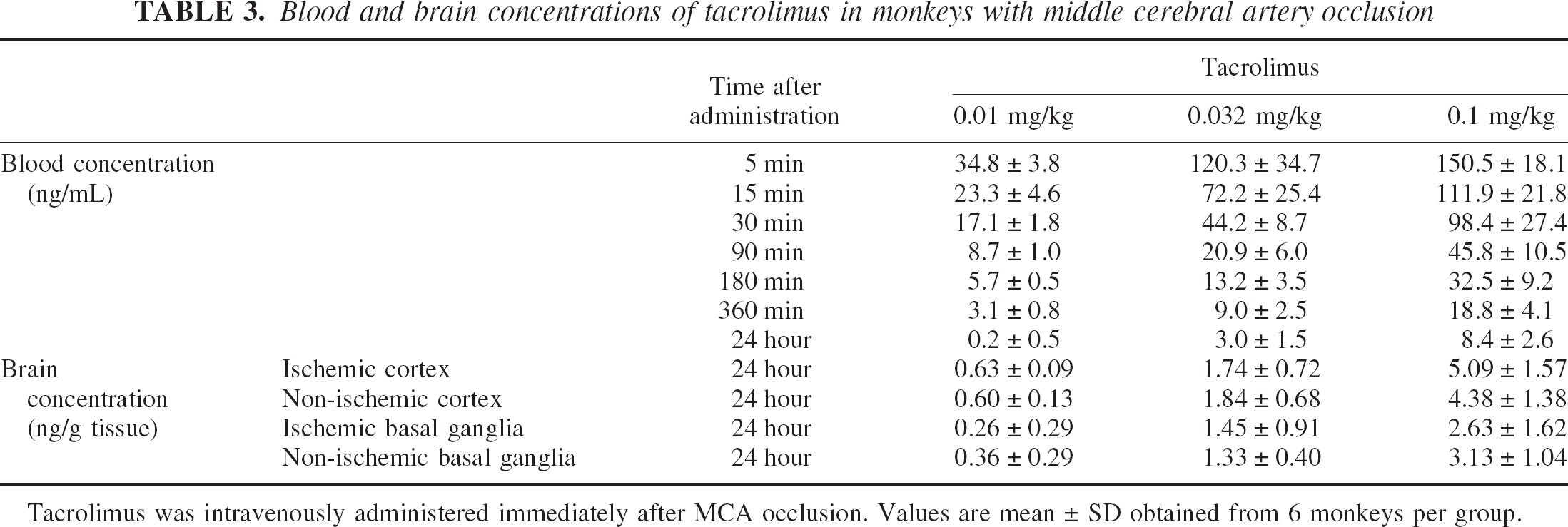
Tacrolimus was intravenously administered immediately after MCA occlusion. Values are mean ± SD obtained from 6 monkeys per group.
Neurologic score
Vehicle-treated animals displayed severe neurologic deficits at 24 hours after MCA occlusion (Fig. 3). The common neurologic deficits were drowsiness, disappearance of pain reflex, paralysis of the upper and lower limbs, decrease of muscle tone, and incoordination. Although most animals showed neurologic signs only on the contralesional side, neurologic deficits in the sensory and motor system appeared on the ipsilesional side as well in some monkeys who showed severe ischemic damage. There were no deaths within 24 hours after MCA occlusion. The neurologic total score for the vehicle-treated group was 59 ± 19.6 points. Tacrolimus dose-dependently ameliorated the neurologic deficits in all subcategories of neurologic parameters. The effects of tacrolimus on the total neurologic score at 0.032 and 0.1 mg/kg were statistically significant.
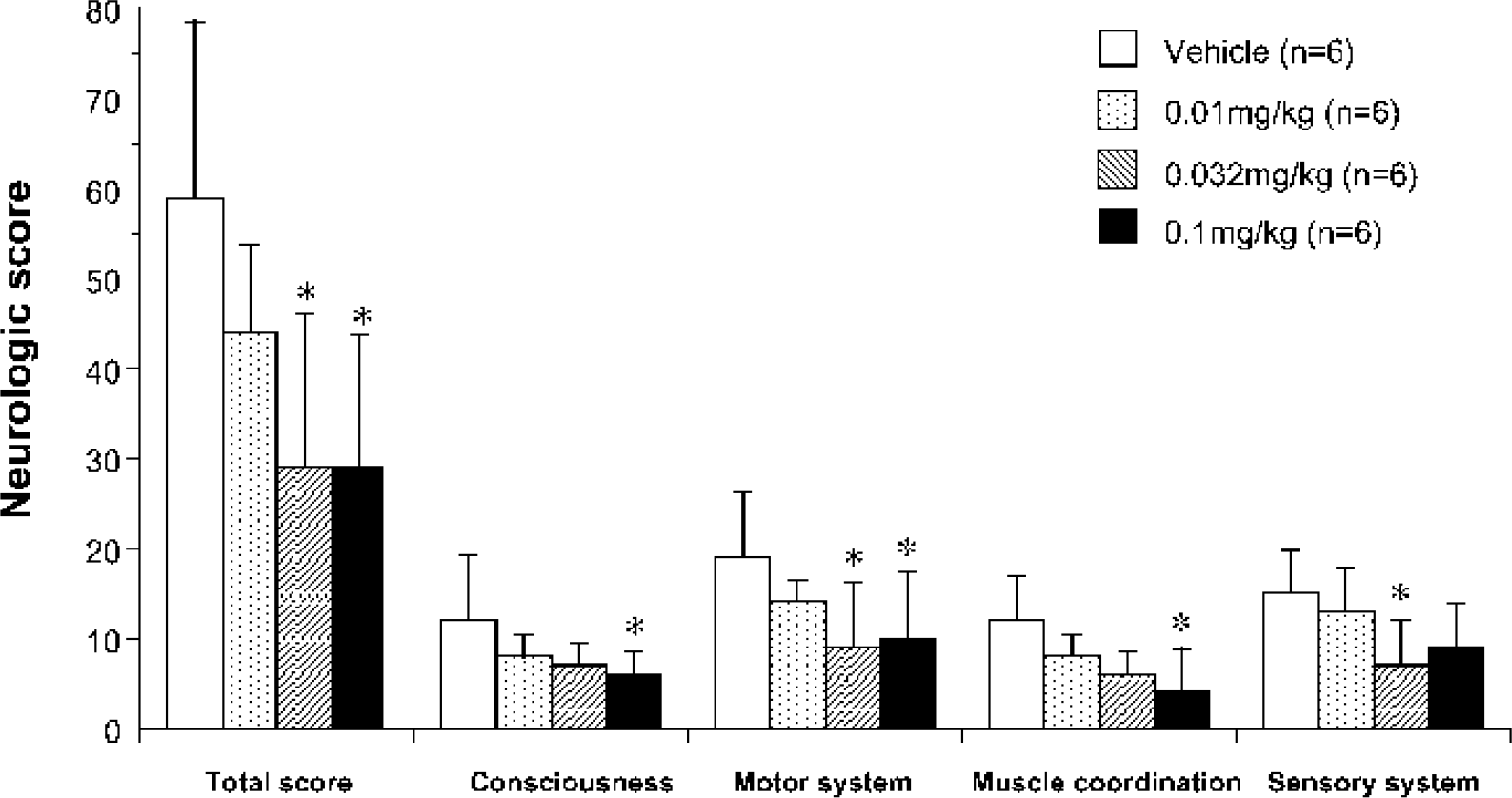
Effects of tacrolimus on neurologic deficits induced by middle cerebral artery (MCA) occlusion in monkeys. Tacrolimus was administered intravenously immediately after MCA occlusion. Neurologic deficits were scored at 24 hours after occlusion. Values are mean ± SD. ∗ P < 0.05; statistically significant compared with vehicle-treated group (by Kruskal-Wallis followed by post hoc Dunnett's multiple comparison test).
Size of ischemic brain damage
Twenty-four hours after MCA occlusion, infarction was detected primarily in the cerebral cortex, including the precentral gyrus, superior temporal gyrus, insular cortex, and medial temporal gyrus. Basal ganglia and white matter, including the internal capsule, were also damaged extensively (Fig. 4A). Tacrolimus reduced the cortical infarction around the Sylvian fissure posteriorly (Fig. 4B and Fig. 4C). The infarct volume in the total area, cortex, basal ganglia, and white matter were 6256.7 ± 2186.5, 3618.1 ± 1788.5, 1494.8 ± 238.2, and 1143.9 ± 451.1 mm3, respectively. Tacrolimus reduced the size of cortical infarction by 58%, 43%, and 85% at doses of 0.01, 0.032, and 0.1 mg/kg, respectively, and a significant effect was obtained at 0.1 mg/kg. Although the neuroprotective action of tacrolimus was primarily observed in the cortex, tacrolimus also tended to reduce infarction in the basal ganglia by 10%, 15%, and 26% at doses of 0.01, 0.032, and 0.1 mg/kg, respectively (Fig. 5).
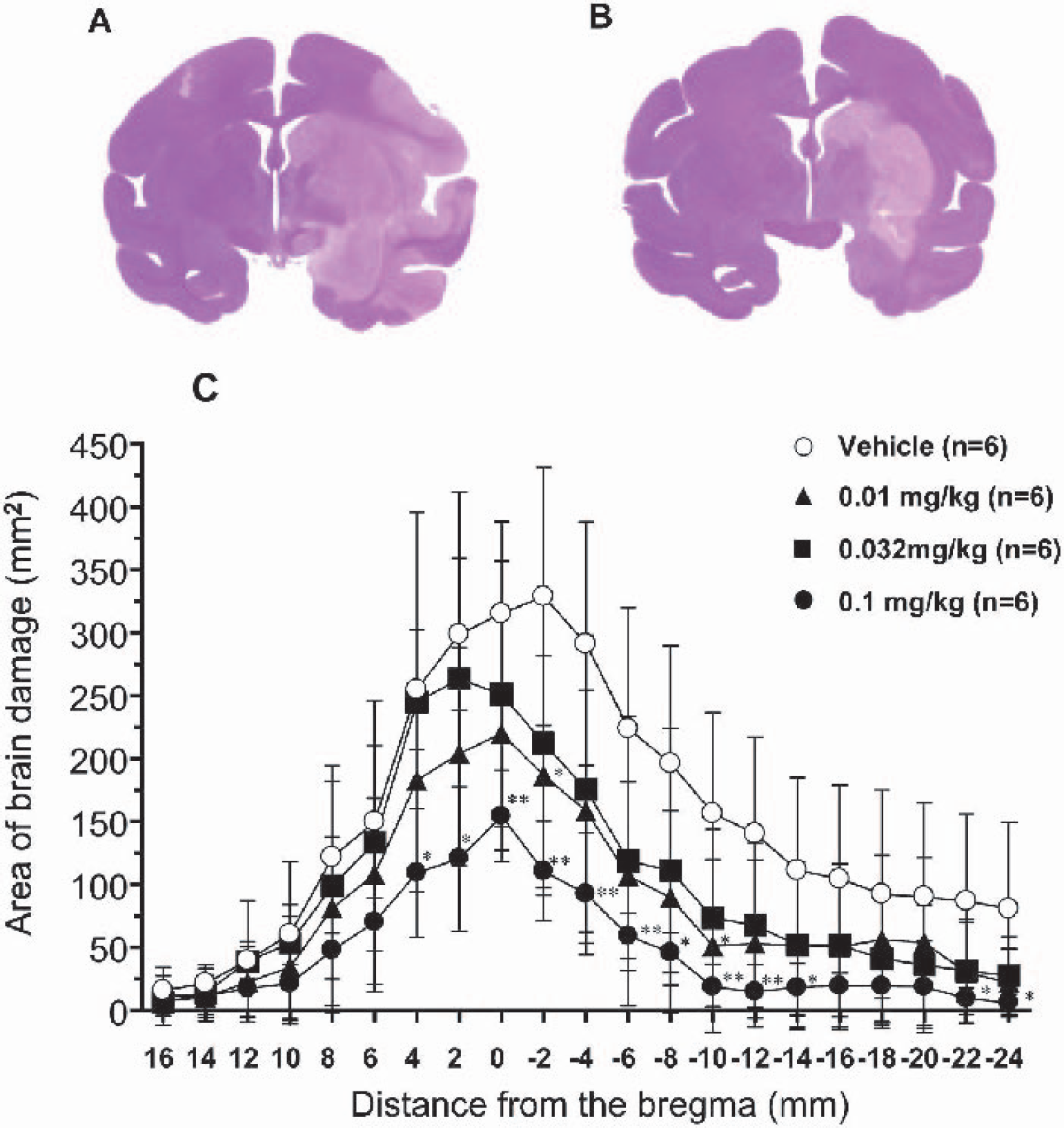
Neuroprotective effects of tacrolimus on brain damage induced by middle cerebral artery occlusion in monkeys. Tacrolimus was administered intravenously immediately after occlusion. Representative coronal sections of vehicle-treated
Neuroprotective effects of tacrolimus on brain damage induced by middle cerebral artery (MCA) occlusion in monkeys 24 hours after ischemic insult. Tacrolimus was administered intravenously immediately after MCA occlusion. Values are mean ± SD. ∗ P < 0.05, ∗∗ P < 0.01; statistically significant compared with the vehicle-treated group (one-way ANOVA followed by Dunnett's multiple comparison test).
Long-term efficacy of tacrolimus
Based upon the results of the short-term experiment, the study was extended to determination of the long-term efficacy of tacrolimus, and neurologic deficits were monitored for 28 days after focal ischemia in subsequent experiment. Brain damage was also measured at 28 days after MCA occlusion. Tacrolimus at 0.1 mg/kg, the dosage which was the most effective in the first study, was used in the second study, but 0.2 mg/kg was also added to verify the long-term neuroprotective action of tacrolimus.
Cerebral blood flow
Whereas the cortical rCBF was not monitored in the long-term study to minimize the insult to animals, blood flow at the MCA trunk was continuously monitored for 6 hours after MCA occlusion. As in the short-term study, tacrolimus (0.1 and 0.2 mg/kg) hardly altered the patency of MCA. The time to complete occlusion of the MCA was 9.9 minutes for the vehicle-treated group and 12.5 and 11.6 minutes for the groups treated with 0.1 and 0.2 mg/kg of tacrolimus, respectively, and the average time to first recanalization was 133.4 minutes for the vehicle-treated group and 122.2 and 125.8 minutes for the groups treated with 0.1 and 0.2 mg/kg of tacrolimus, respectively.
Physiologic parameters
As in the first study, occlusion of the MCA in vehicle-treated animals slightly decreased MBP and HR but increased blood glucose level, but it did not alter any of other physiologic parameters (data not shown). Tacrolimus at 0.2 mg/kg slightly but significantly elevated blood glucose compared with the vehicle-treated group at 270 minutes after MCA occlusion (data not shown). Otherwise, none of parameters, including MBP, HR, po2, pco2, pH, hematocrit, and hemoglobin, were significantly influenced by the administration of tacrolimus.
Neurologic score
Severe neurologic deficits were observed in vehicle-treated animals at 24 hours after MCA occlusion, which worsened at 48 hours after MCA occlusion (Fig. 6). The disturbance of consciousness and sensory dysfunction tended to show slight recovery 7 to 28 days after MCA occlusion. In contrast, motor system dysfunction did not show spontaneous recovery. Tacrolimus at 0.1 and 0.2 mg/kg administered as a single bolus injection immediately after MCA occlusion dose-dependently ameliorated the neurologic deficits in every category. Amelioration of neurologic deficits by tacrolimus lasted for 28 days, and improvement for muscle coordination was more pronounced.
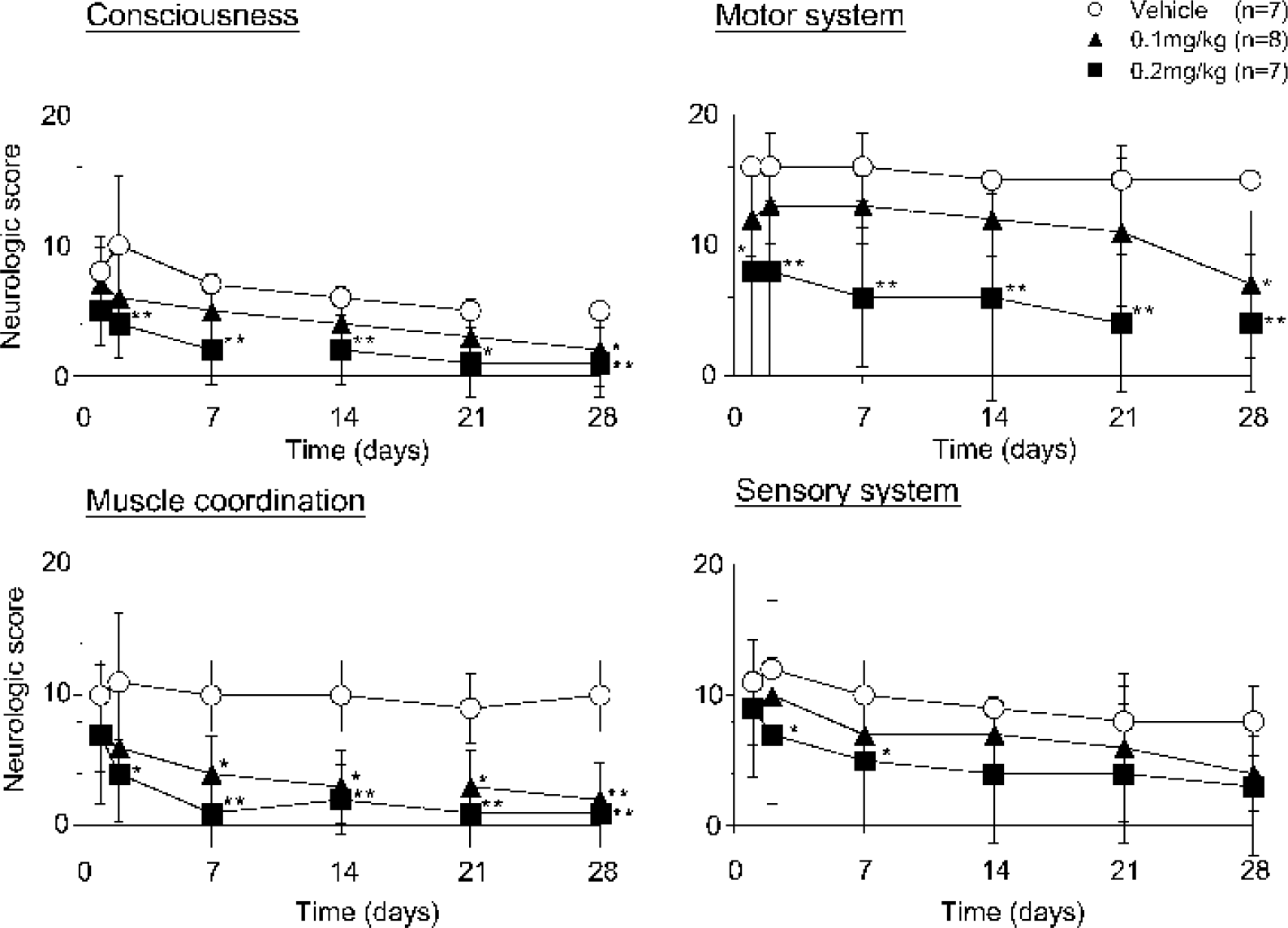
Long-term effects of tacrolimus on neurologic deficits induced by middle cerebral artery occlusion in monkeys. Tacrolimus was administered intravenously immediately after occlusion. Neurologic deficits were scored at 1 day, 2 days, and then every 7 days for 28 days after occlusion. Values are mean ± SD. ∗ P < 0.05, ∗∗ P < 0.01; statistically significant compared with the vehicle-treated group (by Kruskal-Wallis test followed by post hoc Dunnett's multiple comparison test).
Size of ischemic brain damage
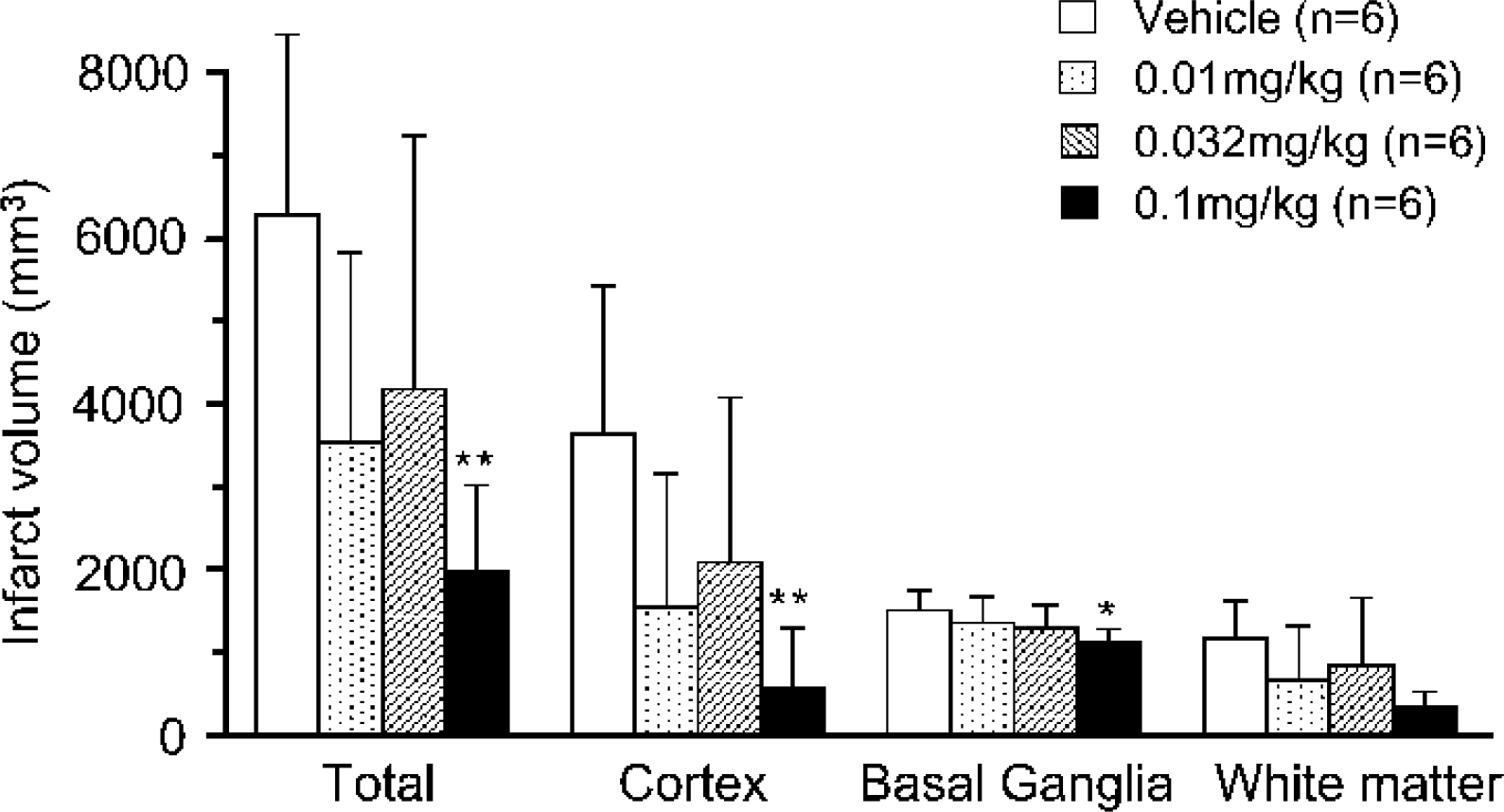
Atrophy was observed in the cerebral cortex, basal ganglia, and white matter at 28 days after occlusion of MCA. The infarct volume was 5352.4 ± 1257.7, 2398.7 ± 714.9, 1341.7 ± 287.4, and 1612.0 ± 512.9 mm3 in the whole cerebrum, cortex, basal ganglia, and white matter, respectively. Tacrolimus at 0.1 and 0.2 mg/kg significantly reduced the infarct volume by 50% and 67%, respectively. In addition to the reduction of the cortical infarct, tacrolimus at 0.2 mg/kg significantly reduced infarction in the basal ganglia and white matter at 28 days after ischemia (Fig. 7).
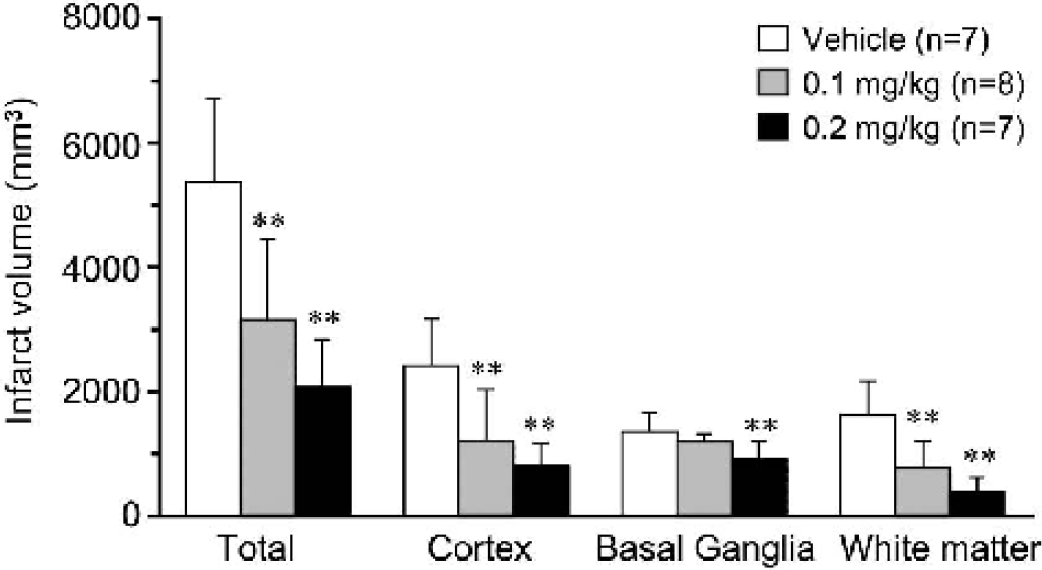
Neuroprotective effects of tacrolimus on brain damage at 28 days after middle cerebral artery (MCA) occlusion in monkeys. Tacrolimus was administered intravenously immediately after MCA occlusion. Infarct volumes in different brain areas were measured at 28 days after MCA occlusion. Values are mean ± SD. ∗∗ P < 0.01; statistically significant compared with the vehicle-treated group (by one-way ANOVA followed by Dunnett's multiple comparison test).
DISCUSSION
Previous studies have shown that tacrolimus has potent neuroprotective effects in a variety of rodent models of stroke, focal ischemia in rats (Bochelen et al., 1999; Butcher et al., 1997; Furuichi et al., 2003; Sharkey and Butcher, 1994), thrombotic focal ischemia in rats (Maeda et al., 2002; Takamatsu et al., 1998), and transient global ischemia in gerbils (Furuichi et al., 2003; Yagita et al., 1996). However, the predictive value of these models is limited given the numerous neuroprotectants that have failed in clinical trials. Therefore, evaluating neuroprotective efficacy in larger animal models should permit a more robust prediction of clinical benefit. Although a recent report showed attenuation of early ischemic neuronal death by tacrolimus at 8 hours after MCA occlusion by MCA clipping in monkeys (Takamatsu et al., 2001), the study did not address dose-dependency of the effect on infarction and neurologic deficits at more extended time points. In the current study, we show that a single administration of tacrolimus dose-dependently and permanently reduced both ischemic brain damage and neurologic deficits in a nonhuman primate stroke model.
In nonhuman primates, the microvascular collateral circulation and the ratio of white matter to gray matter resemble those in humans. Thus primate stroke models should have better predictive value for clinical benefits in stroke patients. In cynomolgus monkeys, collateral circulation is sufficient to prevent formation of reproducible infarction after clipping of the proximal part of the MCA trunk (data not shown). However, we have successfully established a reproducible thrombotic occlusion model, where vascular endothelial injury is induced by a photochemical reaction between rose bengal and green light (Kawai et al., 1995), putatively causing blockage of collateral vessels by thrombus formation. The cyclical reperfusion and reocclusion of the MCA were observed in the majority of animals as previously observed with squirrel monkeys (Kaku et al., 1998). In control animals, the MCA was occluded approximately 12 minutes after rose bengal injection, but the MCA was recanalized at approximately 89 minutes after the occlusion. The cortical CBF measured by laser-Doppler flowmetry did not reduce rapidly after MCA occlusion but gradually decreased in all animals, and the reduction of cortical blood flow at 360 minutes after MCA occlusion was only 53.2%. In spite of the small reduction of cortical blood flow, the inner layer of the cerebral cortex was infarcted extensively in most animals. Therefore, we suspect that either microthrombi that are generated locally at the peripheral sites of the cortex or emboli dislodged from the MCA following repeated recanalization of MCA associated with cyclical flow reduction participated in the formation of stable infarction in the cortical area in the present model. Because we monitored blood flow of a limited cortical surface by the laser-Doppler probe, accurate cortical circulation could not be determined. Further studies using positron emission tomography or other imaging procedures will be required to measure cortical circulation accurately in this stroke model and to better understand the mechanism of the progression of cerebral infarction.
Tacrolimus administered immediately after MCA occlusion reduced ischemic brain damage without affecting the patency of MCA or cortical blood flow. This result is consistent with a lack of the effect of tacrolimus on rCBF or recanalization by t-PA in a rodent thrombotic MCA occlusion model (Maeda et al., 2002). Therefore, tacrolimus works through a neuroprotective mechanism rather than through a direct effect on cerebral circulation or hemodynamics. In fact, the neuroprotective effect of tacrolimus has been suggested to involve the antiapoptotic action (Wang et al., 1999), antiinflammatory effect (Tsujikawa et al., 1998), and the attenuation of free-radical production (Siesjo et al., 1999) in previous in vivo studies. In rodent MCA occlusion models, we have observed that tacrolimus attenuates both apoptotic and necrotic components of neuronal cell death and also inhibits the activation of inflammatory cells driven by activated microglia and infiltrated leukocytes (Furuichi et al., unpublished observation, 2003). There are reports suggesting that apoptotic neuronal death and reperfusion-driven inflammatory reactions are involved in ischemic brain injury in primates (Haring et al., 1996; Tsukada et al., 2001). It would be reasonable to assume that similar mechanisms are involved in the neuroprotective action of tacrolimus on cerebral infarction in the present monkey model, although further studies are required to elucidate the mode of neuroprotective actions of the drug.
Tacrolimus showed a similar pharmacokinetic profile in ischemic and normal monkeys, as well as similar concentrations in ischemic and normal brain tissue, suggesting that tacrolimus readily penetrates the ischemic brain area, in accordance with a previous study in rat focal ischemia (Butcher et al., 1997). Furthermore, the concentrations of tacrolimus in the blood and brain after administration of an effective dose at 0.1 mg/kg were comparable with those in rats given with effective doses of tacrolimus in focal ischemia models (Butcher et al., 1997; Furuichi et al., unpublished observations, 2002). Of interest, however, is the fact that the neuroprotective efficacy of tacrolimus in the present model ranks highly compared with other animal models (Furuichi et al., 2003; Maeda et al., 2002), with an 85% reduction of cortical infarction size at 24 hours and over a 50% reduction after 28 days. In addition to reducing infarct size, tacrolimus remarkably improved the motor system deficits and muscle coordination (Fig. 6 and Fig. 7), perhaps by ameliorating ischemic damage in the basal ganglia and white matter, in addition to the reduction of cortical infarction.
Another important finding was that the efficacy of tacrolimus in ameliorating neurologic deficits was long-lasting. Although the infarct volume has been used as an endpoint in animal models, endpoints for clinical trials are not infarct volumes but instead are neurologic and functional outcomes at 28 days or 90 days after stroke onset (STAIR-II, 2001). Thus, sophisticated behavioral assessments over an extended time period would allow better extrapolation to efficacy in humans. Furthermore, although tacrolimus has been shown to ameliorate skilled motor deficits for up to 2 weeks in a rodent stroke model (Sharkey and Butcher, 1994), functional deficits after ischemia in rodents could be different from those in humans. For example, impairment of consciousness, paralysis, or sensory deficits may develop and persist in stroke patients. Behavioral assessments relevant to these conditions are difficult in rodents. In our model, however, almost all animals in the vehicle-treated control group showed drowsiness, disturbance of muscle coordination and tone, paralysis of the upper and lower limbs, and disappearance of pain reflex. With the exception of consciousness and sensory function, which showed slight improvement, these neurologic deficits did not exhibit spontaneous recovery in vehicle-treated control animals. Thus our model more closely reflects the pattern and time course of deficits seen in stroke patients, and, importantly, provides sophisticated and extended behavioral assessments that confirm the neuroprotective potential of tacrolimus.
One reason for failure of many neuroprotectants in clinical trials is difficulty in translating the preclinical data obtained from animal studies into a clinical setting. In the case of tirilazad, it was proven to be effective in permanent focal ischemia in rodents but ineffective in transient focal ischemia in cats (Park and Hall, 1994; Takeshima et al., 1994). In a nonhuman primate stroke model, tirilazad was demonstrated to prevent edema 6 hours after focal ischemia (Boisvert and Hall, 1996) and reduced infarction 2 weeks after focal ischemia (Mori et al., 1995). Thus tirilazad was shown to reduce the edema formation in an appropriate ischemic condition and consequently reduce the infarction. In the case of clomethiazole, neuroprotective efficacy was demonstrated in transient global, permanent focal, and transient focal ischemia in rodents (Cross et al., 1995; Sydserff et al., 1995). Furthermore, it was shown to protect against hemineglect and to improve functional outcome in focal ischemia in the marmoset, a new world primate (Marshall et al., 1999, 2000). However, permanent MCA occlusion by elecrocoagulation in marmosets produced large infarction in the cortex presumably because of the poor collateral circulation, in contrast to that in cynomolgus monkeys and humans. Subsequent clinical trials demonstrated that both of these drugs did not work in stroke patients (Lyden et al., 2002; RANTTAS Investigators, 1996). These facts suggest that preclinical assessment should be performed in the setting close to the clinical situation as much as possible and the results should be carefully interpreted. On the other hand, it would also be important to make an appropriate clinical design based upon the information of the preclinical study. Attention should be drawn to construct a good preclinical/clinical design interfacing for the success of clinical trial.
In conclusion, we demonstrated that ischemic brain damage and long-lasting functional disability were dramatically ameliorated by a single administration of tacrolimus in a nonhuman primate model of stroke. The present results encourage a clinical trial with this drug in patients with acute stroke.
Footnotes
Acknowledgment
The authors thank Mr. Yuji Awaga for his care of animals and behavioral observations, Ms. Eriko Fujita for performing the drug concentration measure-ments, and Dr. Raymond D. Price for his critical review of the manuscript.
