Abstract
This study was conducted to determine the protective efficacy and mechanisms of thrombopoietin (TPO) intervention in experimental focal stroke. Male rats underwent 2 hours of left middle cerebral artery occlusion (MCAO) followed by 22 hours of reperfusion. Vehicle or TPO (0.03 to 1.00 μg/kg) was administered intravenously immediately after reperfusion. Brain infarct and swelling, neurologic deficits, matrix metalloproteinase-9 (MMP-9), tissue inhibitor of metalloproteinase-1 (TIMP-1), TPO and c-Mpl (TPO receptor) mRNA, MMP-9 enzyme activity and protein expression, and the integrity of the blood—brain barrier (BBB) were subsequently measured. MCAO reperfusion produced a large infarct and swelling after stroke. Thrombopoietin significantly reduced these in a dose-dependent manner. The most effective TPO dose, 0.1 μg/kg, when administrated immediately or 2 hours after reperfusion, significantly reduced infarct and swelling and ameliorated neurologic deficits after stroke. Stroke-induced increases in cortical MMP-9 mRNA, enzyme activity and protein expression, TIMP-1 mRNA, and Evans blue extravasation were reduced by TPO intervention. Thrombopoietin did not alter cortical TPO or c-Mpl mRNA expression, blood pressure, heart rate, blood hematocrit, or platelets. This is the first demonstration of TPO's efficacy in reducing ischemic brain injury and improving functional outcome, partly by inhibiting the stroke-induced increase in MMP-9 and the early, negative effects on the BBB.
Keywords
Introduction
Recombinant tissue plasminogen activator (tPA), administered intravenously within 3 hours of symptom onset, is the only US Food and Drug Administration-approved treatment for ischemic stroke. Even though it has been recommended for use up to 4.5 hours after stroke, few patients (<5%) are eligible for this treatment (Del Zoppo et al, 2009). Therefore, it is imperative that additional treatment interventions be developed and made available for ischemic stroke. Hematopoietic growth factors that have been approved for other indications also have generated significant interest as potential new interventions for stroke.
Hematopoietic growth factors are circulating proteins that regulate the bone marrow production of red cells, white cells, and platelets. However, these cytokines can exhibit extended actions well beyond their hematopoietic properties. For example, erythropoietin (EPO) provides neuroprotection and promotes neurogenesis and angiogenesis after stroke (Belayev et al, 2005; Bernaudin et al, 1999; Pacary et al, 2006; Siren et al, 2001; Wang et al, 2004). Although EPO has shown promise in preliminary trials (Ehrenreich et al, 2002), there are significant concerns regarding EPO use due to increased risk of thromboembolic events and increased clinical mortality when used in conjunction with tPA (Bohlius et al, 2006; Ehrenreich et al, 2009). Thrombopoietin (TPO), the primary hematopoietic growth factor responsible for platelet production, shows significant homology with EPO (23%) at the amino-acid level and with neurotrophins (e.g., the highest homology with BDNF, 36%) in the N-terminal region (Li and Dai, 1995). The biological actions of TPO are initiated by specific binding to its receptors (i.e., c-Mpl) that are expressed on the surface of target cells. Signal transduction from activation of the c-Mpl receptor following TPO binding has been demonstrated in many cell types. Thrombopoietin signals biological functions such as cell proliferation, survival, and maturation through several intracellular pathways (e.g., janus-activated kinase-signal transducer and activator of transcription (JAK-STAT), Ras/mitogen activated protein kinase (MAPK) and phosphoinostitide 3-kinase/protein kinase B (PI3K/Akt)) (Alexander, 1999). Thrombopoietin can enhance angiogenic responses (Amano et al, 2005; Eguchi et al, 2008) and protects against myocardial and hind limb ischemia (Baker et al, 2008; Eguchi et al, 2008). Thrombopoietin's cardioprotective effects are independent of its effects on platelet production (Baker et al, 2008), demonstrating that it possesses additional pleiotropic actions beyond platelet production.
Additional, end-organ protective actions of TPO can be very important therapeutically and require systematic investigation. No study has evaluated TPO's cerebral protective effects. Therefore, the purpose of the present study is to investigate the protective efficacy and mechanisms of TPO in a rodent model of focal stroke. Here, we report that poststroke TPO administration provides dose-dependent brain protection from focal ischemic stroke. We also show that forebrain protective TPO administration inhibits stroke-induced increases in matrix metalloproteinase-9 (MMP-9) expression and the associated negative consequences of increased MMP-9 on the integrity of the blood—brain barrier (BBB) after stroke.
Materials and methods
Transient Middle Cerebral Artery Occlusion Model and Thrombopoietin Treatment
All animal protocols used in this proposal have been approved by the Animal Care and Use Committee of the State University of New York at Downstate Medical Center. Male Sprague-Dawley rats weighing 300 to 350 g from Charles River Laboratories (Wilmington, MA, USA) were subjected to middle cerebral artery occlusion (MCAO) (Jasmin et al, 2007). Briefly, under isoflurane anesthesia, a 3-to-0 monofilament suture (Ethicon, Somerville, NJ, USA) with heat-blunted tip and coated with poly-
Brain Injury Measurement
For 2,3,5-triphenyltetrazolium chloride staining (Rosenbaum et al, 1998), rats were killed and forebrains were removed and cut into 2 mm-thick coronal sections using a brain matrix (Kent Scientific, Torrington, CT, USA). The brain sections were then immersed in 1% 2,3,5-triphenyltetrazolium chloride (Sigma) for 20 minutes. After scanning, the image was analyzed and calculated using an image analysis system (IMAGE J 1.37v, NIH, Bethesda, MD, USA) by an observer masked to the study treatments. Total infarct was calculated over all sections as a percent of the normal right hemisphere (i.e., (right hemisphere−left noninfarct area)/right hemisphere × 100%; thus correcting for brain swelling). Percent brain swelling was calculated independently over all sections as a percent increase over the normal right hemisphere (i.e., (left hemisphere−right hemisphere)/right hemisphere × 100%). For substructure analyses, cortical infarct (%) was calculated as (right cortical area−left noninfarct cortical area)/right hemisphere × 100%. Striatal infarct (%) was calculated as (right striatal area−left noninfarct striatal area)/right hemisphere × 100%. Cortical swelling (%) was calculated as (left cortical area−right cortical area)/right hemisphere × 100%. Striatal swelling (%) was calculated as (left striatal area−right striatal area)/right hemisphere × 100%.
Sensory-Motor Neurologic Score
The modified Neurological Severity Score (mNSS) evaluation and the foot-fault tests were performed 1 day after MCAO. The mNSS was used to grade neurologic deficits including motor, visual and tactile responses, proprioception, performance on a balance beam, reflex responses, and abnormal movements (scale: 0 to 18; normal score, 0; maximal deficit score, 18); thus, the composite score reflects combined motor, sensory, and reflex functions (Chen et al, 2001).
Foot-Fault Test
Rats were tested 1 day after MCAO for placement dysfunction of limbs while walking on elevated metal grids with randomly missing support bars. The horizontal grids were 85.5 × 26.5 × 20 cm3 with a glass enclosure for observation. With each weight-bearing step, the forelimb can fall or slip between the metal wire support bars, which is recorded as a foot fault. The total number of forelimb steps and the total number of foot faults were recorded. The percentage of forelimb foot faults to total steps that occurred within 2 minutes was calculated (Ding et al, 2004).
Real-Time PCR
Briefly, cortical total RNA was isolated using an RNeasy Mini kit (Qiagen, Valencia, CA, USA), and treated with DNase I (Qiagen) as directed by the manufacturer. Complementary DNA (cDNA) was synthesized by reverse transcription of 2 μg of total RNA using the SuperScript III First-Strand Synthesis kit (Invitrogen, Carlsbad, CA, USA) according to the manufacturer's instructions. Quantitative real-time PCR was performed on StepOnePlus Real-Time PCR System (Applied Biosystems Inc, Foster City, CA, USA). Real-time PCR was performed in a 20-μL reaction mixture containing 10 μL of SYBR Green PCR Master Mix (Applied Biosystems Inc), 2 μL of cDNA, and primer set at 0.3 μmol/L each. The primers were designed using Primer3 software (http://frodo.wi.mit.edu/cgi-bin/primer3/primer3_www.cgi) as follows: 18S rRNA, ACC GCG GTT CTA TTT TGT TG (sense) and CCC TCT TAA TCA TGG CCT CA (antisense); MMP-9, CAC TGT AAC TGG GGG CAA CT (sense) and AGA GTA CTG CTT GCC CAG GA (antisense); tissue inhibitor of metalloproteinase-1 (TIMP-1) TCC CCA GAA ATC ATC GAG AC (sense) and TCA GAT TAT GCC AGG GAA CC (antisense); TPO, CTG AAG CCC TTC TTC ACC TG (sense) and CCT CTC CCC TGA ATC TTT CC (antisense); and c-Mpl, CTG CCA CTT CAA GTC ACG AA (sense) and TAG CTG TGA ATG GCA CCT TG (antisense). The amplification consisted of one cycle at 95°C for 10 minutes, followed by 40 cycles at 95°C for 15 seconds and at 60°C for 60 seconds. Serial dilutions of sample cDNA were used to obtain a calibration curve. The individual target mRNA was quantified by determination of the cycle threshold (Ct) and by use of calibration curves. At the end of amplification, the specificity of the PCR was confirmed by melting temperature. The relative amount of target mRNA was normalized to 18S rRNA that was measured from the same sample, and the data were expressed as this ratio (Zhou et al, 2005).
Western Blots
Cortical homogenates from TPO-treated MCAO, PBS-treated MCAO, or sham rats were prepared in TBS buffer with 0.1% Triton X-100 and protease inhibitors. The total protein concentrations were determined using the Bradford assay (Sigma). The MMP-9-positive controls (EMD Chemicals Inc., Gibbstown, NJ, USA; recombinant human pro-MMP-9 proenzyme, 92 kDa and recombinant human MMP-9 active enzyme, 83 kDa) were also used on the blots for comparison. The MMP-9-positive controls and equal amounts of protein from supernatants (20 μg) were electrophoresed on 10% NuPage Novex Bis-Tris Midi Gel (Invitrogen) and then transferred to polyvinylidene difluoride membranes (Invitrogen). After blocking (1 × blocking buffer; Sigma) for 1 hour at room temperature, the membranes were probed with the MMP-9 primary antibody (anti-MMP-9 rabbit polyclonal antibody, 1:2000; Millipore, Billerica, MA, USA) overnight at 4°C. After washing, the membranes were incubated with horseradish peroxidase-conjugated anti-rabbit IgG (Invitrogen) for 1 hour at room temperature. The resulting MMP-9 signals were visualized by the SuperSignal chemiluminescent substrates (Pierce, Rockford, IL, USA) and detected by exposure to Kodak XAR film. The intensity of each band (i.e., corresponding to active- or pro-MMP-9) was quantified by densitometry using Scion Image software. β-Actin monoclonal antibody (Sigma) was also probed similarly and used as a sample loading control for normalization, and the data were expressed as this ratio (Guo et al, 2008; Zhou et al, 2006).
Gelatin Zymography for Matrix Metalloproteinase-9
Gelatinases in brain homogenates can degrade a gelatin matrix, leaving a clear band on the matrix after staining. This method was used to measure enzyme activity as a complement to Western blot protein expression data collected as described above. Briefly, MMP-9-positive controls (see above) and equal amounts of protein from homogenized cortical sample supernatants (20 μg) obtained from TPO-treated MCAO, PBS-treated MCAO, or nontreated sham rats were loaded on Novex zymogram gels (Invitrogen); the gels were then electrophoresed, developed, and stained as directed by the manufacturer (Guo et al, 2008). After separation by electrophoresis, the gels were renatured and incubated with development buffer for 37°C for overnight. The gels were then stained using a Colloidal Blue Staining Kit (Invitrogen). Areas of protease activity appeared as clear bands against a dark background, and the gels were scanned and quantified by densitometry using an image analysis system (IMAGE J 1.37v).
Evans Blue Extravasations as a Measure of Blood—Brain Barrier Integrity
Evans blue (2% in isotonic saline, 4 mL/kg) was injected intravenously immediately after reperfusion (Kondo et al, 1997). After homogenization of cortical samples in distilled water (1:5), the homogenate was precipitated in trichloroacetic acid solution (final concentration 50%) and centrifuged at 10,000
Cardiovascular Measurements
The left femoral artery was cannulated for invasive measurement of arterial blood pressure and heart rate using a Biopac MP 100 acquisition system (Biopac Systems, Santa Barbara, CA, USA). Animals were injected with PBS or TPO, 0.1 μg/kg, intravenously and monitored for 60 minutes.
Blood Measurements
Additional animals were injected with PBS or TPO, 0.1 μg/kg, intravenously and compared with noninjected normal animals of the same age and weight. After 1, 3, and 7 days, blood was collected with a heparinized syringe directly from the heart. Platelet, hematocrit, and hemoglobin content were measured by the Clinical Laboratory at SUNY Downstate Medical Center.
Statistical Analysis
All data were expressed as means±s.e.m. For comparison between multiple groups, statistical analyses were performed using one-way or two-way analysis of variances followed by
Results
Dose Response
2,3,5-Triphenyltetrazolium chloride-stained sections were used to measure percent ischemic hemispheric infarct size (corrected for brain swelling) and percent ischemic hemispheric swelling. The MCAO reperfusion followed by PBS administration produced a large infarct (45.1%±3.7%) and increased hemispheric swelling (23.4%±3.3%). Thrombopoietin administration in the dose range of 0.03 to 1.00 μg/kg exhibited a similar ‘U’-shaped dose—response curve for both total infarct size and hemispheric swelling (
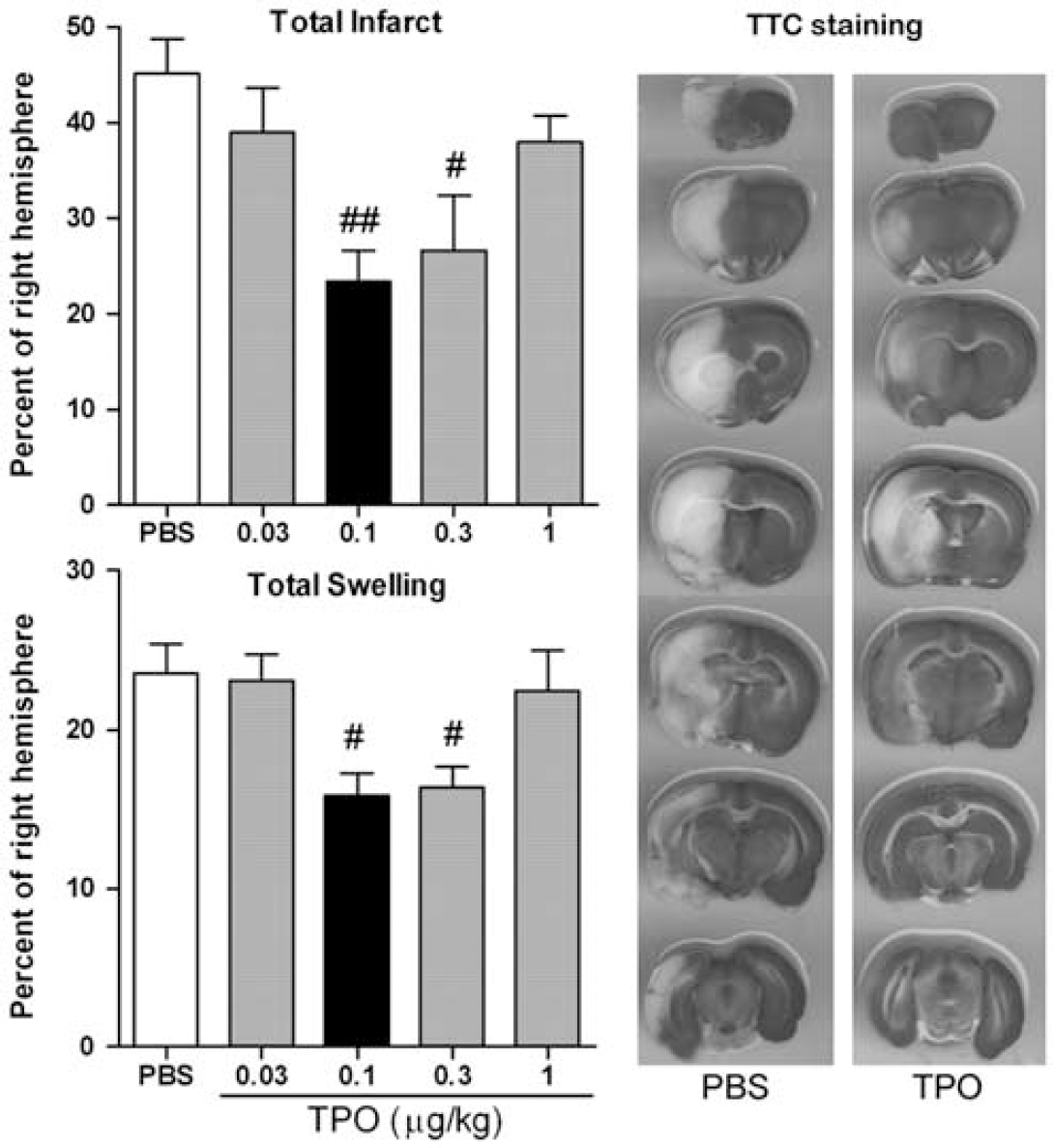
Effect of thrombopoietin (TPO) on total infarct size and hemispheric swelling after experimental stroke. Male Sprague-Dawley rats (300 to 350 g) underwent 2 hours of left middle cerebral artery occlusion (MCAO) (3-to-0 nylon suture coated with poly-
Cortical Infarct, Swelling, and Neurologic Deficits
Analyses of specific brain regions (i.e., cortex and striatum in the sections) indicated that TPO at this optimum protective dose significantly (
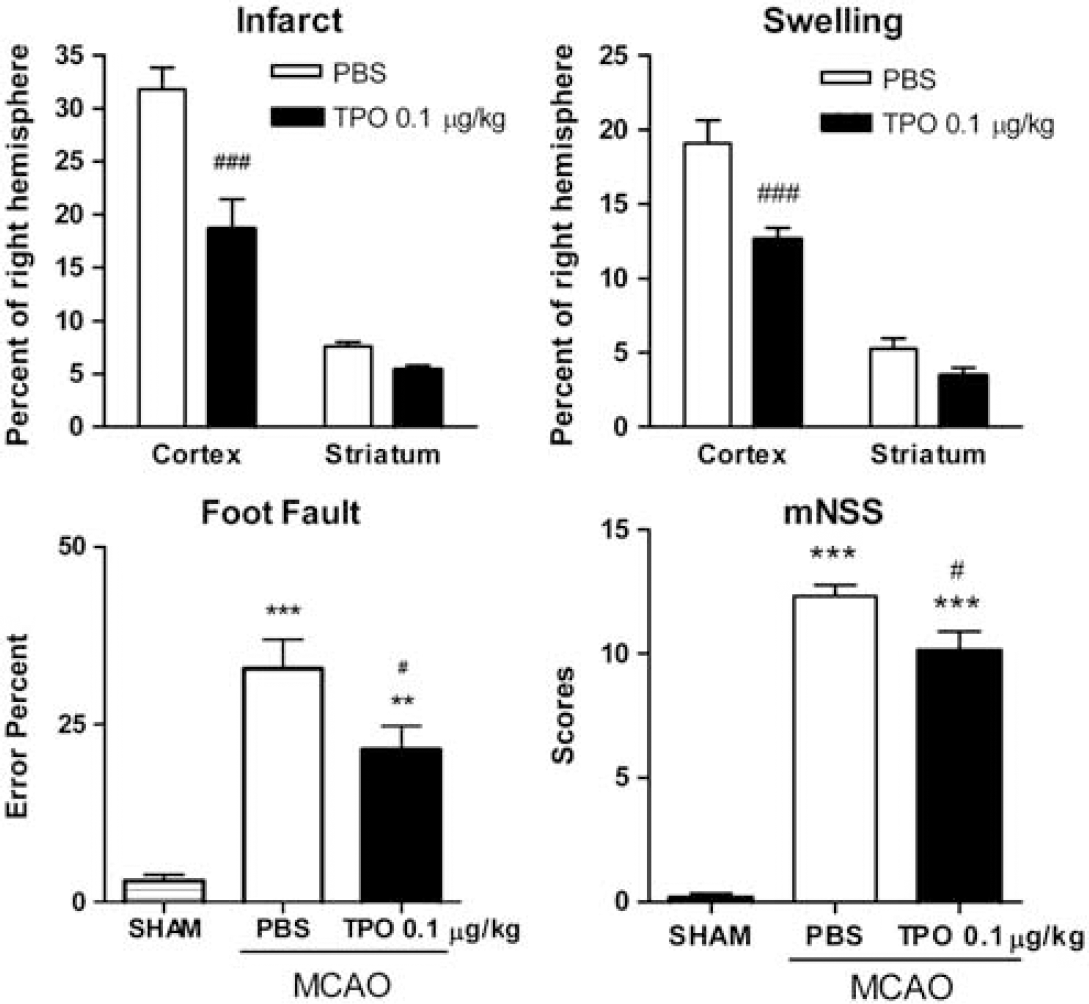
Effect of thrombopoietin (TPO; 0.1 μg/kg, on reperfusion) on infarct size and swelling in ischemic cortex and striatum and foot fault and modified neurologic severity score (mNNS) neurobehavioral tests after experimental stroke (phosphate-buffered saline (PBS) and TPO injected immediately on reperfusion). 2,3,5-Triphenyltetrazolium chloride (TTC) staining was used to evaluate each subregion focal infarct size and ischemic swelling after 2 hours of middle cerebral artery occlusion (MCAO) followed by 22 hours reperfusion. Cortical infarct (%) was calculated as ((right cortical area−left noninfarct cortical area)/right hemisphere) × 100%. Striatal infarct (%) was calculated as ((right striatal area−left noninfarct striatal area)/right hemisphere) × 100%. Cortical swelling (%) was calculated as ((left cortical area−right cortical area)/right hemisphere) × 100%. Striatal swelling (%) was calculated as ((left striatal area−right striatal area)/right hemisphere) × 100%. Values are expressed as means±s.e.m. of groups of 8 to 9 rats. ###
To measure sensory-motor impairments due to ischemic brain injury, foot fault and the mNSS were measured 22 hours after MCAO reperfusion by an investigator masked to the experimental groups and treatments. Thrombopoietin at the optimum brain protective dose of 0.1 μg/kg produced significantly (
‘Time-Window’-Type Study
After selecting the optimal dose of TPO (i.e., 0.1 μg/kg), a postreperfusion time-window study was performed. We found that TPO injected 2 hours after reperfusion (i.e., 4 hours after ischemia) still decreased total percent infarct by 29.3% (
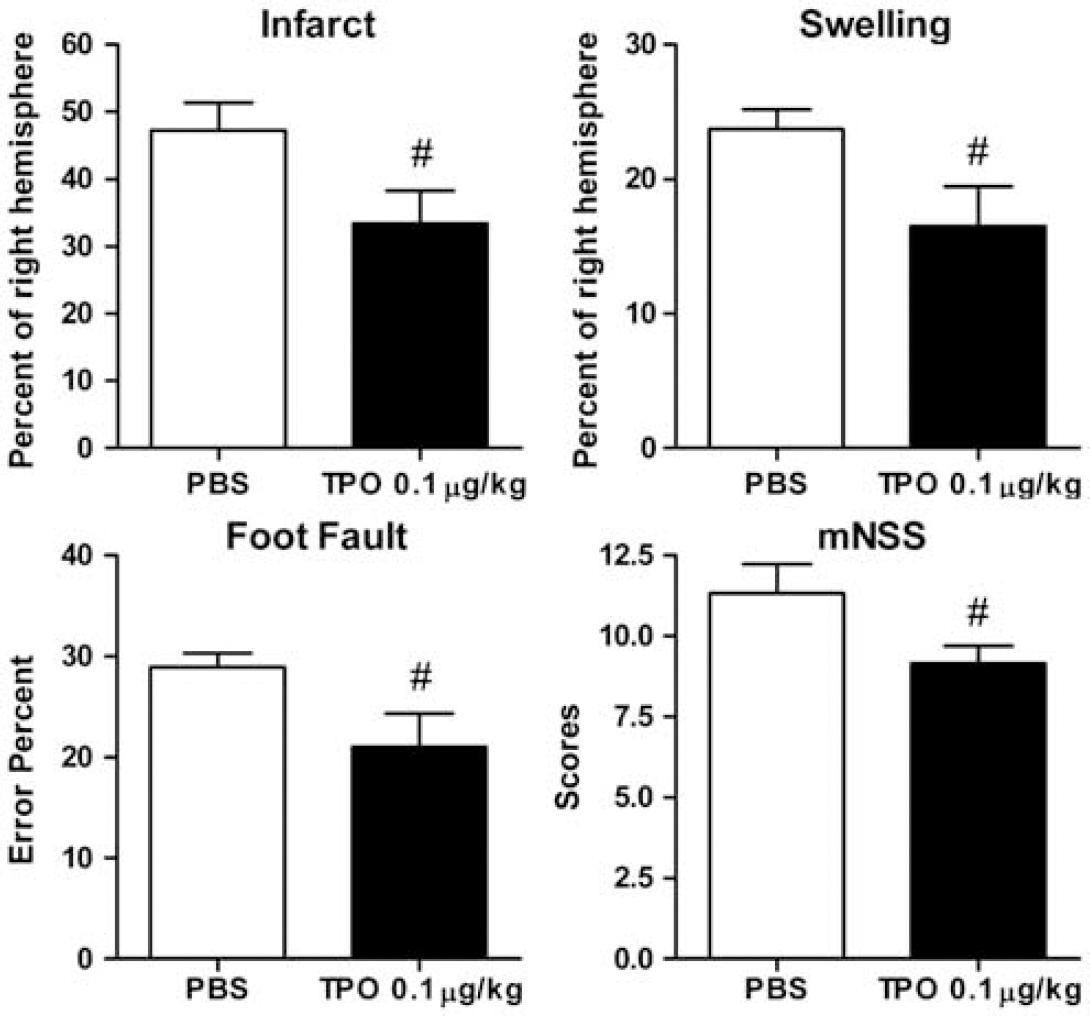
Effect of optimum thrombopoietin (TPO; 0.1 μg/kg, 2 hours after reperfusion) on ischemic infarct size, ischemic hemispheric swelling, and on neurobehavioral tests (foot fault and mNNS) after experimental stroke. In this study, TPO or phosphate-buffered saline (PBS) was administered 2 hours after reperfusion (i.e., 4 hours after stroke onset) to provide addition ‘time-window’-type information on poststroke TPO intervention. 2,3,5-Triphenyltetrazolium chloride (TTC) staining was used to evaluate focal infarct size and ischemic hemispheric swelling 1 day after ischemia. Values are expressed as means±s.e.m. of groups of 8 to 10 rats. #
Real-Time PCR
Preliminary data obtained using a rat cytokine array (RayBiotech, Norcross, GA, USA) showed that increased cortical TIMP-1 expression (i.e., highly expressed after stroke) was inhibited by TPO, 0.1 μg/kg (see Supplementary Figure 1). To confirm the effect of TPO on the MMP system, we further evaluated MMP-9 and TIMP-1 transcription by real-time PCR. A significant increase in cortical MMP-9 and TIMP-1 mRNA was induced after experimental stroke. Thrombopoietin at the optimum protective dose of 0.1 μg/kg on reperfusion after stoke significantly inhibited stroke-induced expression of MMP-9 and TIMP-1 mRNA (Figure 4). In addition, we found that TPO and TPO receptor (c-Mpl) mRNA was not changed after stroke or by TPO treatment (Figure 4).
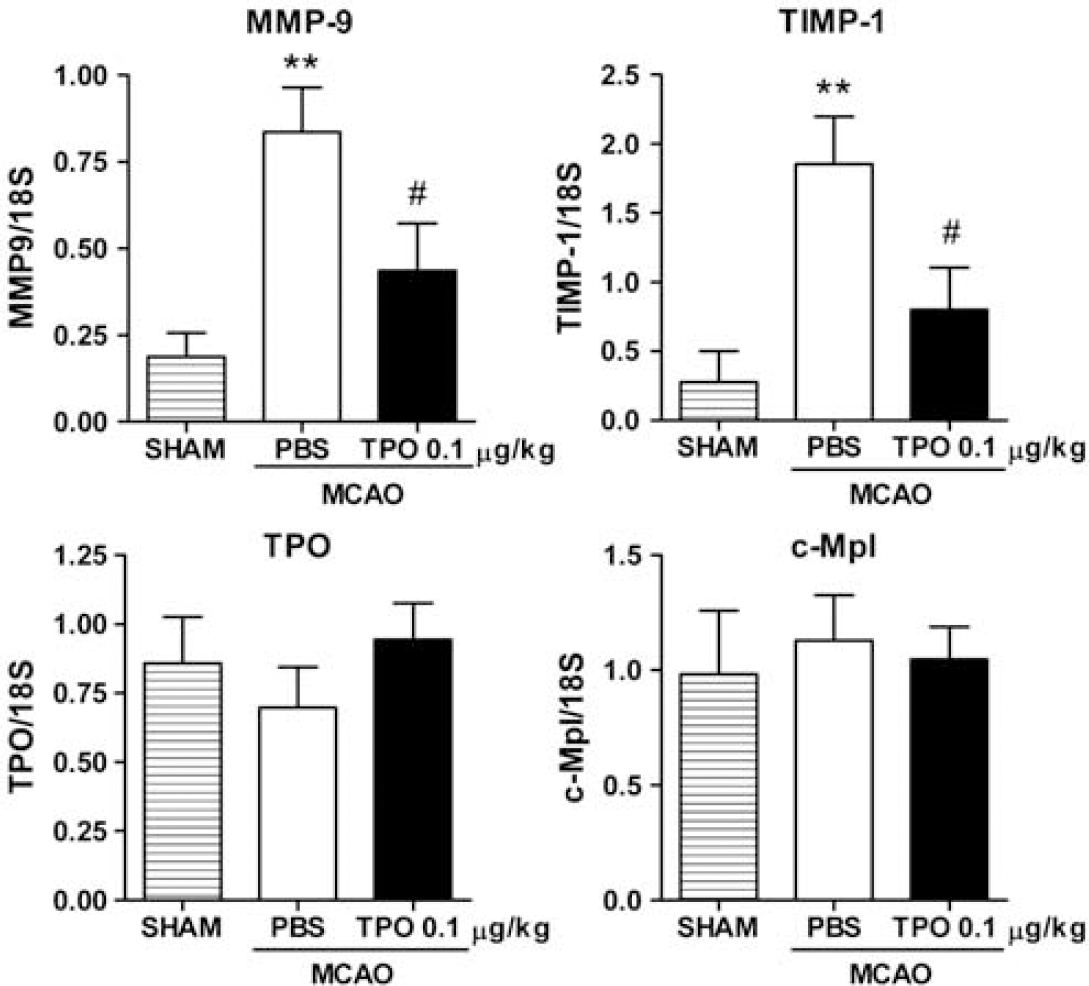
Effects of thrombopoietin (TPO; 0.1 μg/kg, on reperfusion) on cortical matrix metalloproteinase-9 (MMP-9), tissue inhibitor of metalloproteinase-1 (TIMP-1), TPO, and c-Mpl mRNA expression 1 day after stroke. Data correspond to relative levels after normalization to 18S rRNA. Values are means±s.e.m. of groups of 7 to 10 rats. ∗∗
Matrix Metalloproteinase-9 Protein Expression and Gelatinolytic Activity
The MMP-9 protein expression was determined by Western blot. Two distinct bands were identified on the gels. One was at 92 kDa (reflecting pro-MMP-9) and the other one was at 83 kDa (reflecting active-MMP-9). There was no difference in pro-MMP-9 between the sham surgery, PBS-MCAO, or TPO-MCAO groups. Active MMP-9 in the PBS-MCAO group was significantly increased by 878.8%±293.5% (i.e., compared with the sham surgery group). This stroke-induced increase in active-MMP-9 was significantly reduced in the TPO-MCAO group (i.e., it was decreased to only 259.7%±82.40% of the sham surgery group; Figure 5).
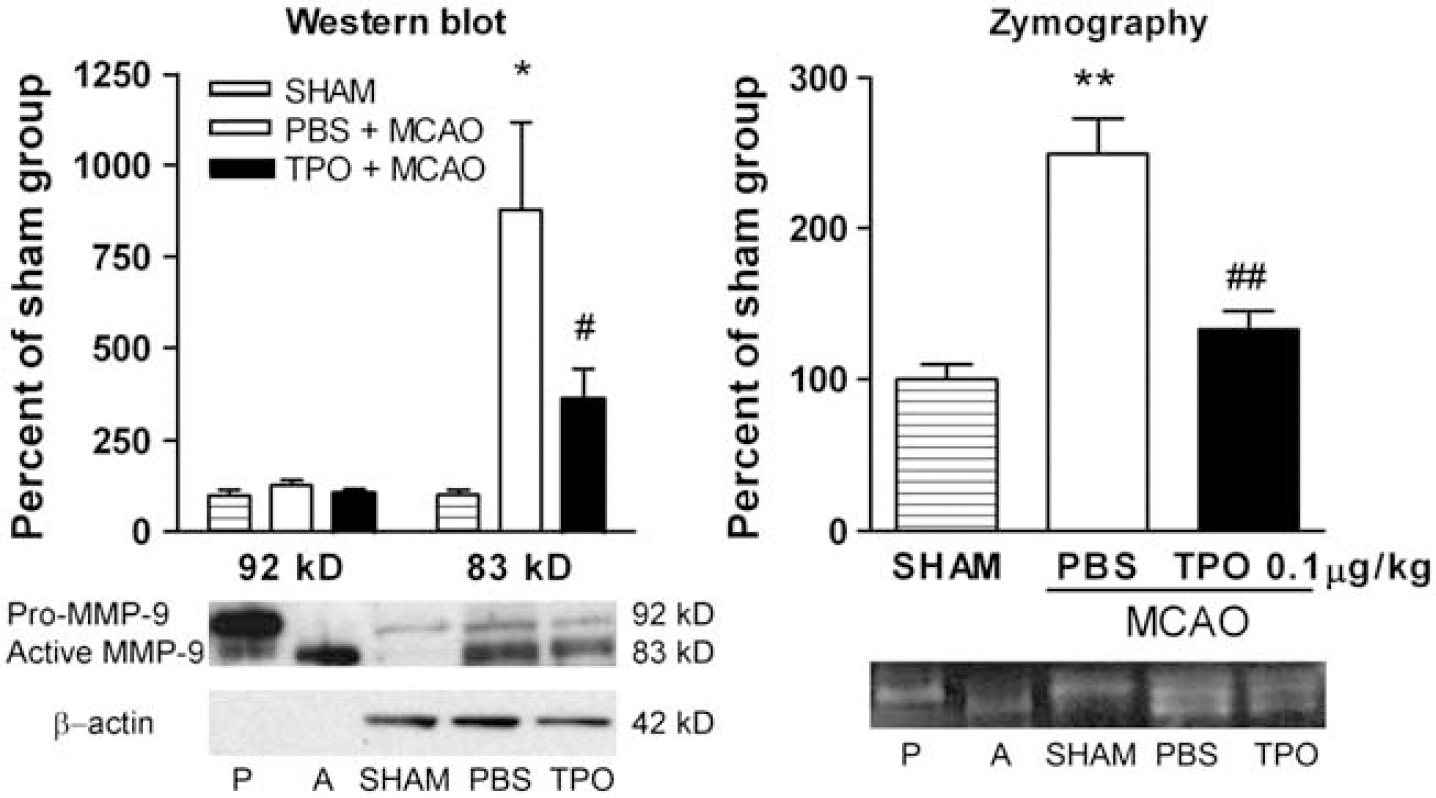
Thrombopoietin (TPO; 0.1 μg/kg, on reperfusion) decreased stroke-induced increases in cortical matrix metalloproteinase-9 (MMP-9) protein (Western blot) and metalloproteinase activity (zymography) 1 day after stroke. In the Western blots, data corresponded to relative levels of the 92 kDa (pro-MMP-9) and 83 kDa (active-MMP-9) bands normalized to β-actin. In zymograms, both active- and pro-MMP-9 were measured together as total MMP-9 activity because the discrimination of the active- versus pro-MMP-9 bands could not consistently be achieved. Both data from Western blots and zymograms were normalized to the sham surgery group. P denotes pro-MMP-9 control and A denotes active-MMP-9 control. Values are means±s.e.m. (
The MMP-9 enzymatic activity was determined by gelatin zymography. The signals for pro- and active-MMP-9 bands were observed but did not separate as clearly as in Western blots. As individual bands could not be accurately measured, total MMP-9 signals (i.e., both pro- and active-MMP-9) were compared for the three groups. Corresponding with MMP-9 protein levels, cortical MMP-9 enzyme activity in the PBS-MCAO group exhibited a significant increase of 249.5%±57.1% compared with the sham surgery group. Similar to the Western blot data, the optimum TPO dose of 0.1 μg/kg administered on reperfusion after stroke significantly decreased cortical MMP-9 activity after stroke to only 133.2%±11.9% of the sham surgery group (Figure 5).
Blood—Brain Barrier Disruption
As shown in Figure 6, Evans blue leakage was clearly seen in cortical and subcortical areas after stroke. We dissected out the cortex from both hemispheres and quantified the extent of BBB leakage by measuring cortical Evans blue content. We found that MCAO reperfusion induced significant increases in Evans blue content in the cortex of the ischemic hemisphere compared with the cortex in the nonischemic hemisphere (1219.0%±193.6%). Thrombopoietin treatment produced significantly reduced Evans blue leakage in the cortex when compared with PBS-control group (619.2%±242.6%; Figure 6). Considering the dramatic effects of TPO in reducing infarct size, Evans blue content was normalized to the PBS and TPO infarct volumes to correct for smaller lesion sizes in animals treated with TPO. Thrombopoietin and PBS cortical infarct volumes were calculated from the previous 2,3,5-triphenyltetrazolium chloride-staining measurements. After normalization, Evans blue content in the ipsilateral cortex was still significantly higher in the PBS-treated group (1.55±0.20 ng/mm3) compared with the TPO-treated group (0.89±0.32 ng/mm3) (
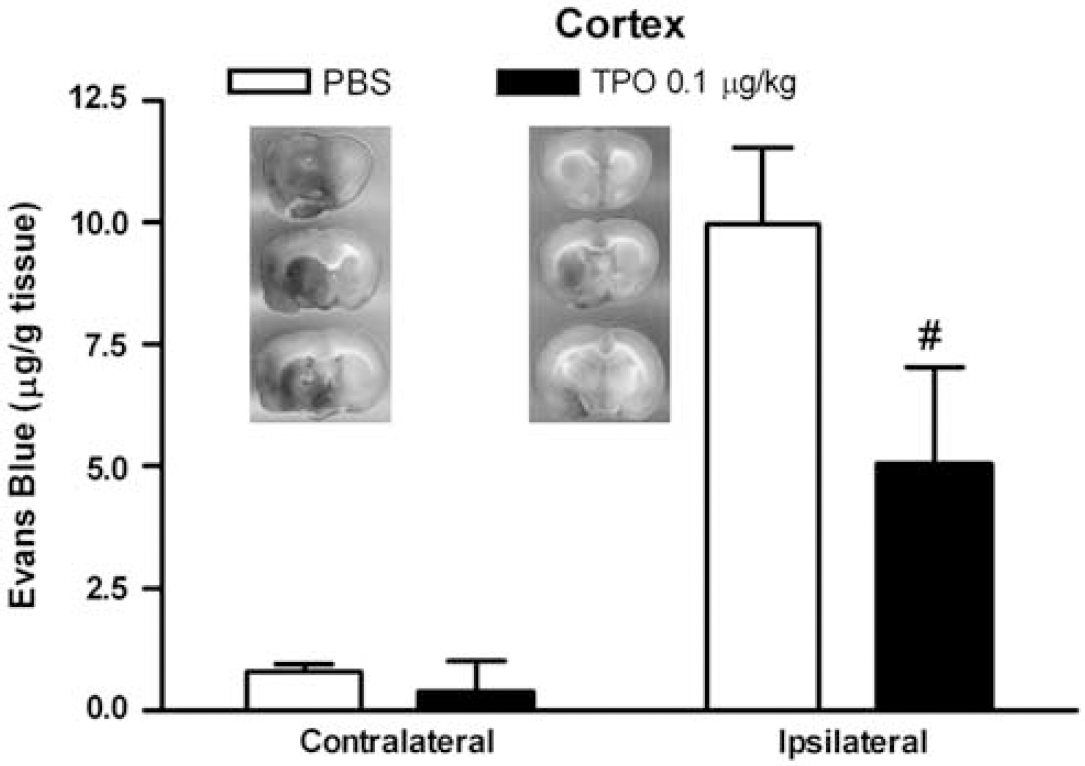
Thrombopoietin (TPO; 0.1 μg/kg, on reperfusion) attenuated stroke-induced cortical blood—brain barrier (BBB) disruption. Integrity of BBB was measured by Evans blue extravasation. Images in the figure represented typical slides of Evans blue extravasation after experimental stroke in brain sections from stroke rats receiving phosphate-buffered saline (PBS) (left) compared with TPO (right) treatments. Values are means±s.e.m. of groups of eight rats. #
Mean Arterial Pressure, Heart Rate, and Body Temperature
The mean arterial pressure, heart rate, and body temperature were quantified during baseline, 30, and 60 minutes after PBS or TPO administration under isoflurane anesthesia. At the baseline, mean arterial pressure was 97.7±3.1 mm Hg, heart rate was 340.3±12.2 beats/min, and body temperature was 37.2°C±0.1°C. After treatment with PBS or TPO, there were no significant differences between the treatment groups after 30 or 60 minutes (Table 1A,
The effects of the optimum poststroke single dose of TPO (0.1 μg/kg, intravenously) on physiological and blood measures
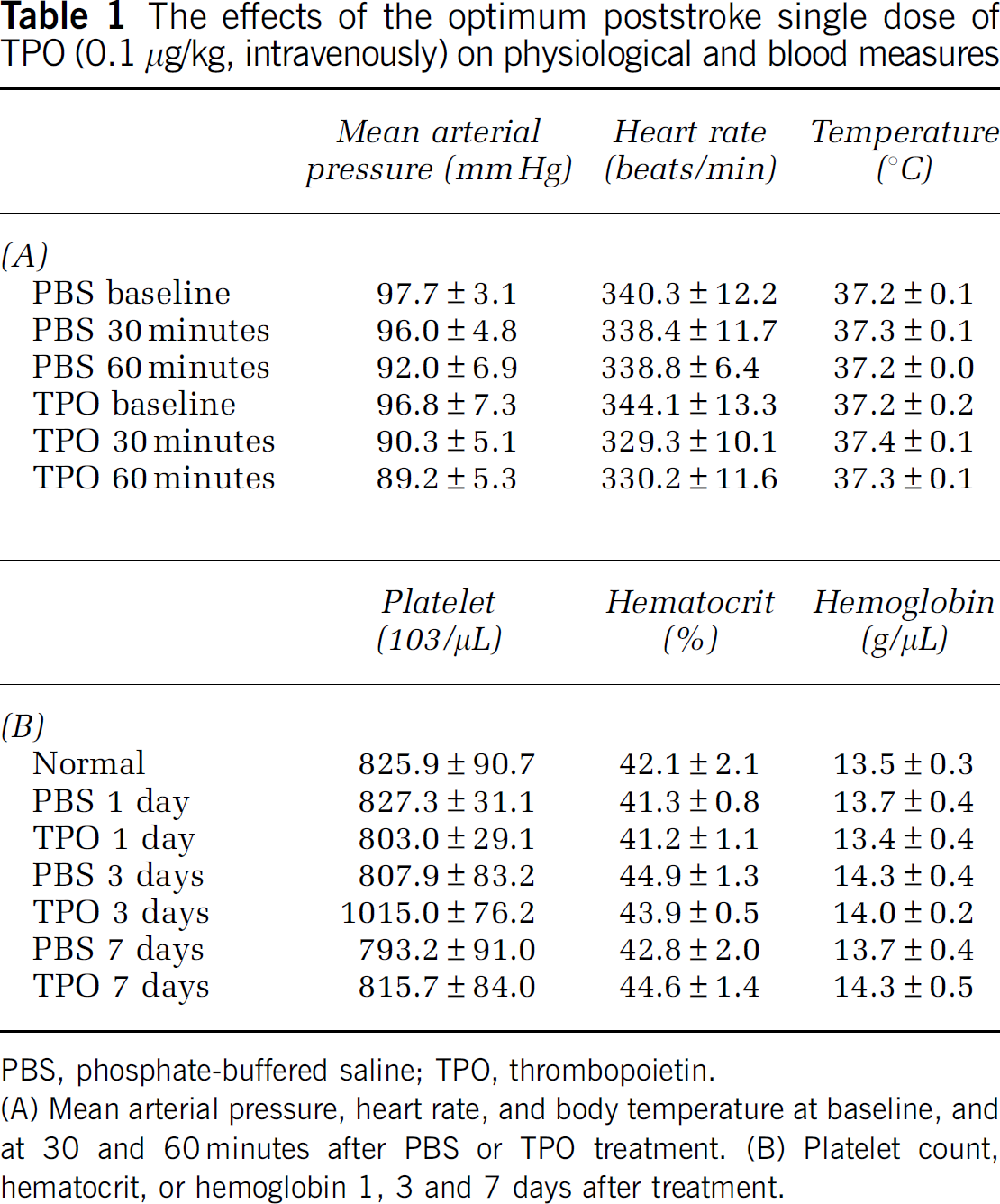
PBS, phosphate-buffered saline; TPO, thrombopoietin.
(A) Mean arterial pressure, heart rate, and body temperature at baseline, and at 30 and 60 minutes after PBS or TPO treatment. (B) Platelet count, hematocrit, or hemoglobin 1, 3 and 7 days after treatment.
Platelet, Hematocrit, and Hemoglobin Levels
To determine whether a single dose of TPO that confers cerebrovascular protection
Discussion
Thrombopoietin Provides Protection in Focal Stroke
The present study is the first to show TPO protection of the brain from ischemic injury and subsequent improvement of neurologic function. In the dose—response study, it was demonstrated that TPO administered poststroke at reperfusion provides protection from focal stroke with maximum protection at the optimal dose of 0.1 μg/kg. It is important to note that the degree of protection provided by TPO is on the order of that provided by the ‘gold standards’ of hypothermia or ischemic preconditioning. As the ultimate goal of ischemia research is to improve functional outcome after stroke, we were interested in determining whether TPO treatment would improve neurologic functional deficits produced by stroke. Using foot-fault and mNSS tests, we demonstrated that TPO significantly improved neurologic function. Moreover, we found that the protection of TPO against stroke extends to 2 hours postreperfusion (i.e., 4 hours after stroke onset), adding significant information regarding the clinical relevance of TPO. These results suggest potential new therapeutic opportunities for TPO in stroke.
Thrombopoietin Inhibits Matrix Metalloproteinase-9 and Blood—Brain Barrier Disruption
Thrombopoietin only reduced infarct size in the cortex (considered the penumbral ischemic area). Therefore, we focused on ‘protective mechanism’-type studies only in the cortex. To explore the mechanisms of TPO protection against stroke, a cytokine array was performed on cortical samples from optimum protective TPO dose versus PBS-treated stroked rats. Pathway information from the array suggested that MMPs have an important role in TPO's brain protective actions. The MMP system mRNAs, including TIMP-1, have been shown previously to be upregulated after stroke (Romanic et al, 1998; Wang et al, 1998). This initiated a series of studies. We systematically investigated the transcript, protein expression, and enzymatic activity of MMP-9 in sham surgery and stroked (receiving PBS or TPO) rats. We found, as reported previously (Romanic et al, 1998; Rosenberg et al, 1996), that MMP-9 mRNA, protein expression, and enzymatic activity was significantly upregulated after stroke. Importantly, activation of MMP-9 by stroke was confirmed by direct measurement of active-MMP-9 and pro-MMP-9 bands on Western blot and zymogram. However, TPO significantly inhibited the stroke-induced increase in MMP-9 mRNA expression, active-MMP-9 protein expression, and MMP-9 enzymatic activity.
Our data also show that increased damage of the BBB induced by ischemic stroke was significantly reduced by TPO treatment. Increased Evans blue content in the ipsilateral cortex after stroke was significantly inhibited in the TPO-MCAO group even after normalization. We did observe reduced Evans blue dye in the striatum due to TPO (
After stroke, MMPs become dysregulated and subsequently mediate acute neurovascular disruption and parenchymal injury. An early and major pathological effect of MMP activity in cerebral ischemia is the disruption of the BBB through degradation of the basal lamina that surrounds the cerebral blood vessels (Barr et al, 2010; Romanic et al, 1998; Rosenberg et al, 1996). The MMP-9 knockout mice display reduced infarct size and less BBB damage after focal ischemia (Asahi et al, 2000). Studies have reported that BB-94 (an MMP inhibitor) and an MMP-9 neutralizing monoclonal antibody provide protection in rodent models of stroke (Asahi et al, 2000; Romanic et al, 1998). Also, BB-94 improves BBB integrity after focal cerebral ischemia and ameliorates mortality after delayed tPA thrombolysis (Pfefferkorn and Rosenberg, 2003). Data from gene knockout models and pharmacologic experiments suggest that MMPs may be attractive therapeutic targets for stroke. Here, the effect of TPO on BBB disruption after stroke appears to be mediated by its downregulation of stroke-induced increases in MMP-9. Emerging data suggest that some aspects of MMP activity during the delayed neuroinflammatory response may contribute to remodeling and later recovery after stroke (Zhao et al, 2006). Thus, the challenge might be to define spatial and temporal controls that modify the MMPs and neuroinflammatory responses after stroke to optimize inhibition during these acute stages of injury without interfering with beneficial endogenous mechanisms of later neurovascular remodeling (Barone and Parsons, 2000). Further studies of prolonged TPO brain protection (i.e., well beyond 1 day after stroke) on MMPs and neurologic deficits are now required.
Our future studies will focus on long-term TPO protection and also investigate the potential opportunity for TPO to facilitate recovery of function after stroke (e.g., evaluating long term, poststroke outcome following acute early and later, and perhaps using repeated TPO interventions, respectively). This line of work will help us understand if the beneficial effects of TPO are maintained (i.e., long-term reduced infarction and improved neurologic deficits) and if behavioral deficits and cellular processes (e.g., synaptogenesis, angiogenesis, and neurogenesis) can be improved once brain injury has already occurred. Moreover, TPO protective and regenerative mechanisms at the TPO signaling level should be investigated.
Endogenous Thrombopoietin Signaling
Understanding TPO via receptor (on-target) and nonreceptor (off-target) effects is crucial for interpretation of mechanisms involved in TPO brain protection. Here, we have begun investigation of TPO and TPOR mRNA changes in brain injury/protection. Thrombopoietin or c-Mpl (TPO receptor, TPOR) mRNA expression was found in rat cortical homogenates, but neither MCAO nor TPO administration affected the mRNA levels of the ligand or receptor (Figure 4). Our additional preliminary work examining c-Mpl protein expression (Western blot) and cellular localization (immunohistochemistry), respectively, indicated that the only available rat anti-c-Mpl goat polyclonal antibody (Santa Cruz Biotech, Santa Cruz, CA, USA) did not function (i.e., did not label its target) when used in brain sections (data not shown). We plan to evaluate additional antibodies that may work better in the forebrain when they become commercially available. Alternatively, we will investigate the cellular and substructure localizations of c-Mpl using
Thrombopoietin Effects on Physiological and Blood Measures
The optimum, single dose of TPO used in our
Thrombopoietin provides both cardiac and brain protection independent of its platelet-stimulating activity, thus demonstrating that TPO possesses significant pleiotropic actions. Therefore, compared with EPO, TPO might be a more beneficial stroke intervention with fewer side effects. For example, considering the effects of stroke-induced MMP-9 and the well-known role of MMP-9 in increasing hemorrhagic conversion after delayed tPA administration (Ning et al, 2006), it will be important to evaluate TPO's ability to improve outcome after delayed tPA administration in embolic stroke in the future. This opportunity might lengthen tPA's window of safe use, thus allowing thrombolysis therapy to reach more patients in addition to providing brain protection as demonstrated in the present studies. Clearly, the present initial studies demonstrating TPO protection and potential mechanisms related to the reduced MMP-9 and neuroinflammation/BBB disruption can guide future, more systematic investigations of TPO's therapeutic opportunities and mechanisms to support its potential use as a new therapeutic intervention for treating stroke.
Footnotes
Acknowledgements
The authors thank our technician, Aleksandr Melamud, and students Jonathan Bloom, Hwai Yin Ooi, Daniel Lax, and Jacob Rube for their support in this study.
The authors declare no conflict of interest.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
