Abstract
The changes in cerebral blood flow that accompany spreading depression are well-described, as are parallel changes in cellular activity, with a wave of hyperemia followed by a prolonged oligemic phase. In this study, a cat model of the CBF changes associated with spreading depression and in vitro pharmacology were used to determine if there is a role for the powerful peptide vasoconstrictor endothelin in this response. For the pharmacological studies, the middle cerebral artery was harvested from cats postmortem. For the physiological studies, cats were anesthetized with halothane induction and α-chloralose (60 mg/kg, intraperitoneal loading; 20 mg/kg i.v. 2-h maintenance). CBF was monitored continuously in the parietal cortex using laser Doppler flowmetry (CBFLDF) after exposure of the dura mater. The in vitro work demonstrated that endothelin-1 (ET-1) mediates a strong and potent contraction of cerebral vessels. Both the selective ETA receptor antagonist FR139317 and the combined ETA and ETB receptor antagonist Bosentan caused a rightward shift of the concentration-response curve without attenuation of the maximum effect. The calculated pA2 values were 6.28 and 6.90, respectively. The slope did not differ from unity, suggesting that the ET-1-mediated contraction is evoked by a single population of ETA receptors, which were effectively antagonized by these compounds. Spreading depression was induced with a needle stick injury to the cortex. Local administration of the endothelin antagonists FR139317 (10 μM) and Bosentan (10 μM) did not affect resting blood flow in the cortex. Induction of spreading depression following local administration of FR139317 and Bosentan resulted in responses no different from those in control cortex. These data demonstrate that endothelin does not play a significant role in the vasoconstrictor portion of the CBF change seen in spreading depression, nor does it affect resting flow. Since it is widely held that spreading depression, or a very similar mechanism, underlies the aura phase of migraine, it may be suggested from these studies that endothelin antagonists are unlikely to be useful in migraine.
Headache in its many manifestations and migraine in particular make up a common and often debilitating clinical problem whose pathophysiology has been the subject of considerable investigation (Goadsby, 1994). Some 15% of patients with migraine describe a prodrome of neurological symptoms that are virtually characteristic of the condition (Bes et al., 1988). This aura phase of migraine has been the subject of considerable interest and has been proposed to be closely related to the spreading depression (SD) of Leao (1944). This association has resulted in considerable interest in the mediation of SD and possible avenues by which the phenomenon can be modulated.
The aura phase of migraine is characterized by various neurological symptoms that have particular features. Patients report phenomena such as photopsia (shimmering edges of moving light), scotomata (areas of loss of vision), limb paraesthesia, vertigo, and diplopia (double vision). The symptoms generally move across the visual field or limb in an orderly slow march. Lashley (1941), in his now classic paper, described the progress of his own aura by plotting its movement and shape as it traversed his visual field. There is considerable circumstantial evidence linking SD with the aura phase of migraine. Numerous studies have confirmed that the aura phase of migraine is associated with a reduction in CBF (Skinhoj and Paulson, 1969; O'Brien, 1971; Simard and Paulson, 1973; Skinhoj, 1973; Norris et al., 1975; Mathew et al., 1976; Edmeads, 1977; Hachinski et al., 1977; Sakai and Meyer, 1978; Olesen et al., 1981; Staehelin-Jensen et al., 1981; Lauritzen et al., 1983a; Lauritzen and Olesen, 1984; Skyhoj-Olsen et al., 1987). The visual aura and other focal neurological signs, when they are present, are often associated with this reduction in CBF (Olesen et al., 1981). In some cases, there is clear documentation of a dissociation between the aura and the headache (Olesen et al., 1990). The neurological changes parallel what is seen when the brain is directly stimulated (Brindley and Lewin, 1968; Penfield and Perot, 1963) and resemble what might be predicted if ocular dominance columns (Hubel and Weisel, 1968) were serially activated. The change in flow moves across the cortex as a spreading oligemia at 2–3 mm/min (Lauritzen et al., 1983a; Lauritzen and Olesen, 1984), corresponding to the rate that Lashley (1941) estimated.
Supporting the association of migraine and SD are reports that the oligemia is preceded by a phase of focal hyperemia (Olesen et al., 1981; Friberg et al., 1987). Such a change is exactly what would be expected if a phenomenon similar to cortical SD was involved. Following the passage of the oligemia, the cerebrovascular response to hypercapnia is blunted (Simard and Paulson, 1973; Sakai and Meyer, 1979; Harer and von Kummer, 1991), while autoregulation remains intact (Lauritzen et al., 1983b). Again, this pattern is repeated in SD. It has also been shown in the experimental animal that initiation of SD results in subcortical reductions in blood flow even in anatomically distant areas in the brain stem (Mraovitch et al., 1992). Furthermore, it has been shown that the vasodilator phase of SD, at least in the cat, involves generation of nitric oxide since the neurophysiological correlates of SD can be dissociated from the hyperemic phase by nitric oxide synthesis inhibitors (Goadsby et al., 1992). A similar blunting of the vasodilator phase has been seen in the rat (Duckrow, 1993), although a recent contradictory report (Zhang et al., 1994) suggests there is more to understand in relationship to the vasodilator phase.
Endothelin-1 (ET-1) is the most powerful vasoconstrictor substance currently described. It is a low-molecular-weight polypeptide secreted by cultured endothelial cells from cerebral vessels. The advent of antagonists of the action of ET-1 (Sogabe et al., 1993; Clozel et al., 1994) provides tools with which to determine what role this peptide may have in vasoconstrictor responses in the cerebral circulation. In this study, we characterized the ET-1 receptor mediating contraction in the cerebral vessels and thus determined the optimum pharmacological variables with which to evaluate whether an ET-1 antagonist can alter the oligemic phase of SD. Since it has been shown that ET-1 levels are increased in the plasma of people with migraines (Farkkila et al., 1992; Gallai et al., 1994), the fundamental question arises as to whether ET-1 mediates the vasoconstrictor phase of SD.
METHODS
In vitro pharmacology
Young adult cats (mean weight, 4.4 kg) were anaesthetized with α-chloralose and exsanguinated. The brain was rapidly removed and immersed in a 37°C buffer solution continuously aerated with 5% CO2 in O2. The middle cerebral artery (MCA) was dissected free from tissue under an operating microscope and cut into smaller circular segments (1 mm long). For examination, each segment was mounted on two L-shaped metal prongs (0.1 mm in diameter), one of which was connected to a force-displacement transducer (FT03C) attached to a MacLab unit for continuous recording of the isometric tension. The position of the other holder could be changed by means of a movable unit, allowing fine adjustments of the vascular tension by varying the distance between the metal prongs (Adner et al., 1993).
The specimens were immersed in temperature-controlled (37°C) tissue baths containing a buffer solution of the following composition (mM): NaCl, 119; NaHCO3, 15; KCl, 4.6; MgCl2, 1.2; NaH2PO4, 1.2; CaCl2, 1.5; glucose, 11. The solution was continuously aerated with carbogen gas, giving a pH of 7.4. A tension of 2 mN was applied to the arterial segments, and they were allowed to stabilize at this level of tension for 1 h. The contractile capacity of each vessel segment was examined by exposure to a potassium rich (60 mM) buffer solution that had the same composition as the standard buffer solution except that some of the NaCl was exchanged for an equimolar concentration of KCl. This resulted in a contraction of 3.34 ± 0.53 mN (n = 19). The effect of endothelin was examined by cumulative application of ET-1 (0.1 pM to 1 μM).
Drugs. ET-1 (Auspep, Parkville, Australia), FR139317 (R)2-[(R)-2[(S)-2[[l-(hexahydro-1H-azepinyl)]carbonyl] amino-4-methylpentanoyl]amino-3-[3-(1-methyl-1H-indolyl)]-propionyl]]-amino-3-(2-pydridyl) proprionic acid; Fujisawa Pharmaceutical, Osaka, Japan), and Bosentan (4-tert-butyl-N-[6-(2-hydroxy-ethoxy)-5-(2-methoxy-phenoxy)-2,2′-bipyrimidin-4-yl]-bezenesulf-onamide; Ro 47–0203; Roche, Switzerland) were dissolved in distilled water containing 1% bovine serum albumin. All other compounds were obtained from Sigma. For the experiments, the drugs were further diluted in buffer solution or saline.
Calculations for in vitro work. The data from the concentration-response curves were characterized by Emax values (maximum contractile effect as a percentage of the effect of 60 mM K+–buffer solution) and pD2 values (negative logarithm of the molar concentration that produced half-maximum contraction). The pA2 value (negative logarithm of the molar concentration of the antagonist that reduces the effect of the agonist to half the dose) analysis was done as described by Tallarida and colleagues (1979). To compare the difference between treatment and controls, Wilcoxon signed-rank test was used on in vitro data with p < 0.05 considered to be significant.
Cerebral blood flow
Cats weighing 2.7 ± 0.3 kg (mean ± SD) were anesthetised with α-choloralose (60 mg/kg, i.p., with supplements of 20 mg/kg i.v.) after induction with 4% halothane. They were intubated and ventilated (Harvard Pump, MA, U.S.A.) and paralyzed with gallamine triethiodide (6 mg/kg, i.v.). End-expiratory CO2 and fraction of inspired O2 were continuously monitored (Datex, Finland). Polyethylene catheters were placed into the femoral artery for monitoring blood pressure and into the vein for fluid and drug administration.
Cerebral blood flow with laser Doppler flowmetry. Cerebral cortical perfusion was measured continuously using laser Doppler flowmetry (LDF). The principles of operation of LDF have been thoroughly described (Stern et al., 1977), and its use in this laboratory has been described in detail (Goadsby, 1991). Briefly, infrared light from a laser diode with a wavelength of 780 nm is directed to the surface of the brain via a fiber optic cable. Analysis of the Doppler shift caused by that portion of light scattered by the red blood cells gives the velocity of flow for those cells, while the proportion of light backscattered is directly related to the volume of moving red cells in the brain sampled. A flow index can thus be derived from these signals. In these experiments, the BPM 403A (TSI, MN, U.S.A.) instrument was used with a pencil probe (P-433, TSI) mounted in a stereotaxic manipulator (David Kopf Instruments, CA, U.S.A.) and the signals for flow, velocity, and volume were continuously monitored. Biparietal craniotomies were carried out with a low-speed dental drill that was cooled with saline to prevent underlying thermal injury, and the dura was left intact under the probe although it was cut adjacent for drug administration. Cerebrovascular reactivity to a brief period of hypercapnia (6–8%) was tested before collecting data and during the experiment as part of the protocol.
To determine accurately the changes in cerebrovascular dynamics measured with the laser Doppler, the physiological variables were monitored on-line by a microcomputer. The blood pressure, heart rate, end-expiratory CO2, inspiratory O2, and laser Doppler volume, flow, and velocity signals were passed to a signal-conditioning device and then to an analog-to-digital convertor (LabMaster DMA, Scientific Solutions, OH, U.S.A.) in an 80386/80387-based microcomputer. Signals were continuously monitored and displayed on the computer screen using locally written software in C language. All data were stored on disk for later analysis and plotting.
Surgery and stimulation. The animals were placed in a stereotaxic frame (Kopf, Tujunja, CA, U.S.A.), primarily to facilitate the measurement of CBF with LDF (CBFLDF). The parietal cortex and overlying dura mater were exposed bilaterally. The bone was carefully drilled away using a low-speed dental drill and saline irrigation. The area was exposed over some 15 mm to allow access for the stimulating needle. Spreading depression was induced with a 23-gauge needle lowered in a stereotaxic holder to a distance of 5 mm into the brain and then removed.
Experimental design and statistics. We (Goadsby et al., 1992; Kaube and Goadsby, 1994) and others (Duckrow and Beard, 1992) have seen that in some circumstances the oligemic phase of SD depends to a great extent on the resting CBF and particularly on whether SD has been previously induced. Whether a SD has been induced can be determined by testing for the CBFLDF hypercapnic response, which is known to be blocked after passage SD (Lauritzen et al, 1983b). When SD has been induced, the oligemic phase is generally not seen on subsequent inductions. In this study, the animals were compared left to right so that one of either side had FR139317 (10 μM) or Bosentan (10 μM), and the other side had vehicle administered; SD was then elicited on each side. The blockade was tested first by locally injecting a glass micropipette of ET-1 and reapplying the peptide after local application of the antagonist. Local application of ET-1 (10−8 M) produced a vasoconstriction, with a 37 ± 7% reduction in CBFLDF.
The CBFLDF data were transformed to percentages of change from the control period (Kaiser, 1989) to allow for animal-to-animal variation and expressed as mean ± SD. Data were compared using a Mann-Whitney U test (Siegel, 1956) and assessed at the p < 0.05 level for significance.
RESULTS
In vitro pharmacology
ET-1 produced strong and potent concentration-dependent contractions of cat MCA (Fig. 1). The Emax was 115 ± 39% relative to potassium contraction and potency, and pD2 was 8.42 ± 0.07. FR139317 (1 and 10 μM) had no contractile effect by itself but caused a shift to the right of the ET-1 concentration-response curve (pD2 was 7.87 ± 0.38 and 6.96 ± 0.21, respectively) without changing the maximum contractile response (Fig. 1; Table 1). Schild plot analysis resulted in a pA2 value of 6.28 with a slope of 1.12 (not different from unity). Similarly, Bosentan had no contractile effect alone but caused a shift to the right of the ET-1 dose-response curve at all concentrations (0.1, 1, and 10 μM) studied. The Schild plot analysis of Bosentan demonstrated a slope of 0.90 and a pA2 of 6.90 (Fig. 2), showing a greater potency than seen with FR139317.
In vitro pharmacological data from FR139317 and Bosentan in cat MCA
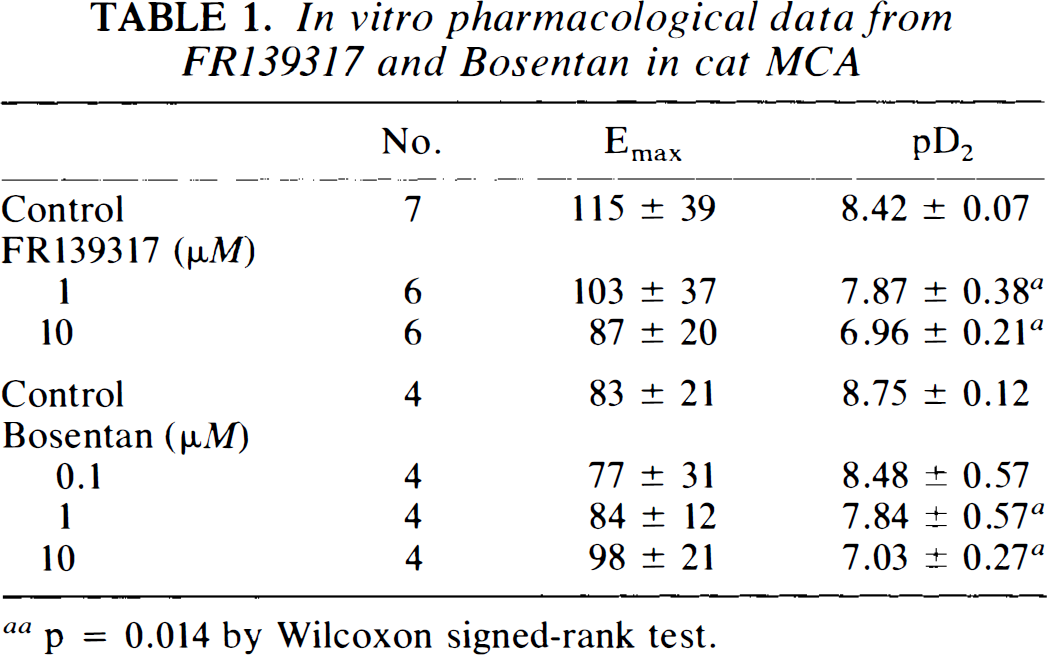
p = 0.014 by Wilcoxon signed-rank test.
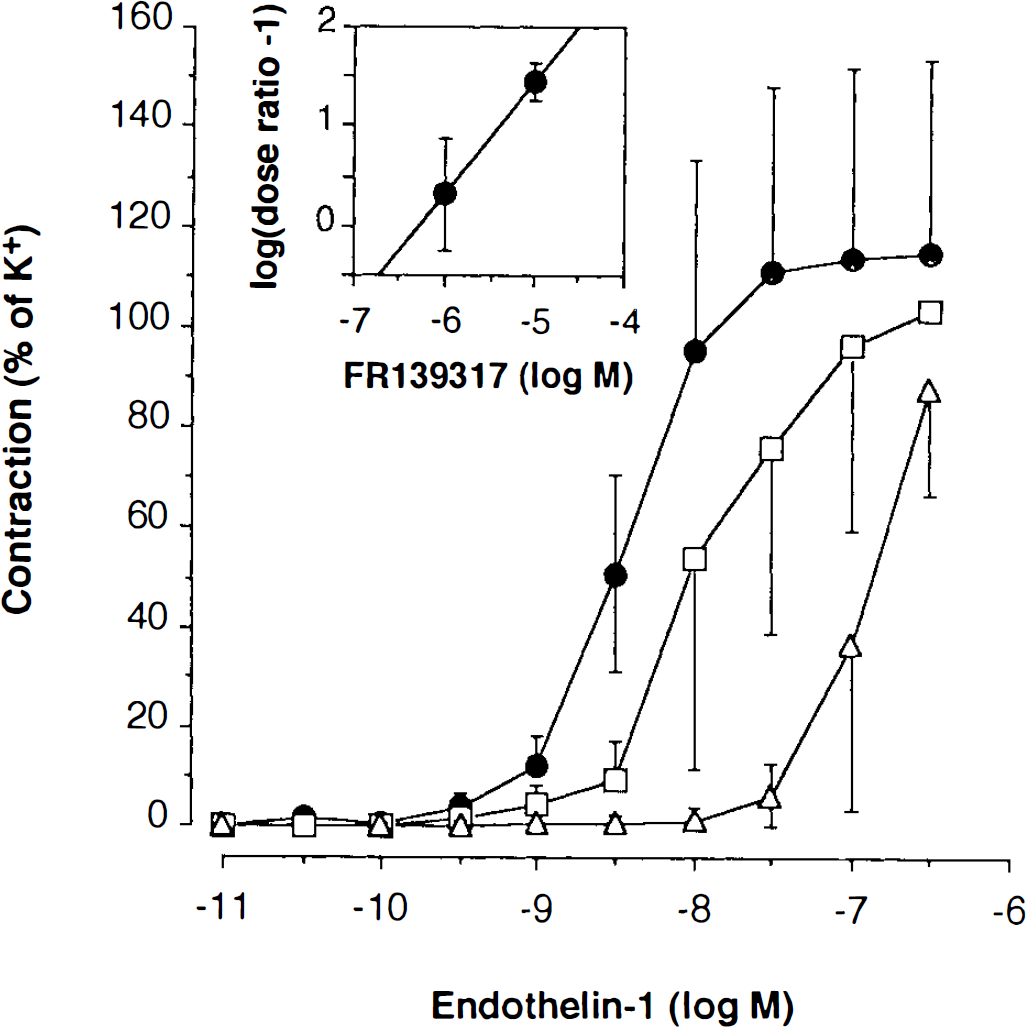
Dose-response curves are shown for ET-1 (control, circles) and in the presence of FR139317, 1 μM (squares) and 10 μM (triangles). The data are expressed as percentages of constriction (mean ± SD) compared with K+ and demonstrate potent inhibition of the vasoconstrictor response of endothelin. The inset shows a Schild plot analysis yielding a pA2 value of 6.28 and a slope of 1.12.
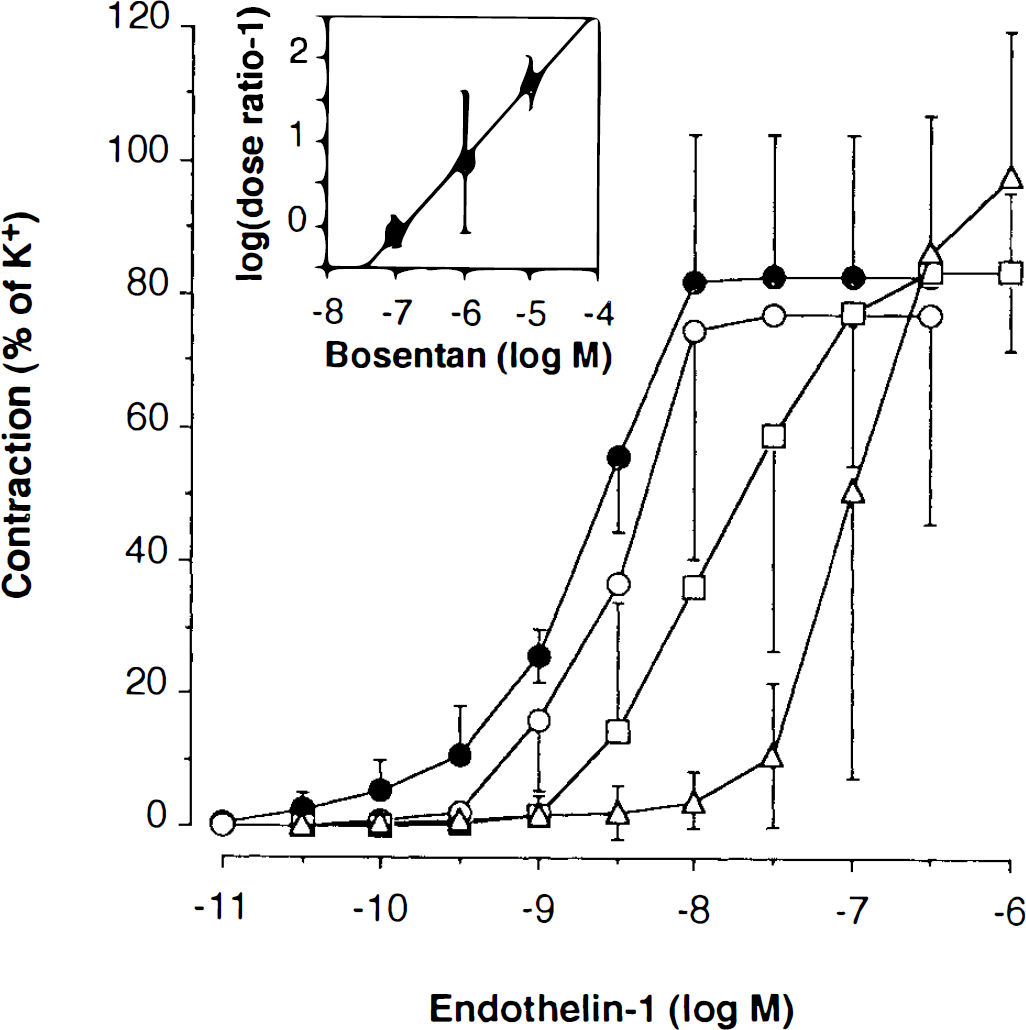
Dose-response curves are shown for endothelin-1 (control, black circles) and in the presence of Bosentan, 0.1 μM (white circles), 1 μM (squares), and 10 μM (triangles). The data are expressed as percentages of constriction (mean ± SD) compared with K+ and demonstrate potent inhibition of endothelin-1-induced vasoconstriction by Bosentan. The inset shows a Schild plot analysis yielding a pA2 of 6.90 and slope of 0.90.
Spreading depression
All nine animals included in the analysis had blood pressure, heart rate, and respiratory blood gas data within normal limits for the anesthetized cat.
CBFLDF. Local administration of FR139317 (10 μM) did not alter CBFLDF (8 ± 10%; not significant), but it did block the vasoconstrictor response to ET-1 (10−8 M; p < 0.05; n = 5). On the control side, needle stick induction of SD resulted in an initial vasodilator phase, with a 263 ± 61% increase in CBFLDF followed by a 21 ± 13% reduction in flow. Following administration of FR139317 (10 μM), induction of SD resulted in a 276 ± 65% increase in CBFLDF and a 20 ± 12% reduction in the oligemic phase (Fig. 3). Neither response was affected by the presence of FR139317 (n = 5; Fig. 4). In a further series of experiments, Bosentan (10 μM) similarly failed to attenuate the hyperemic or oligemic phases of SD (n = 4; Fig. 5).
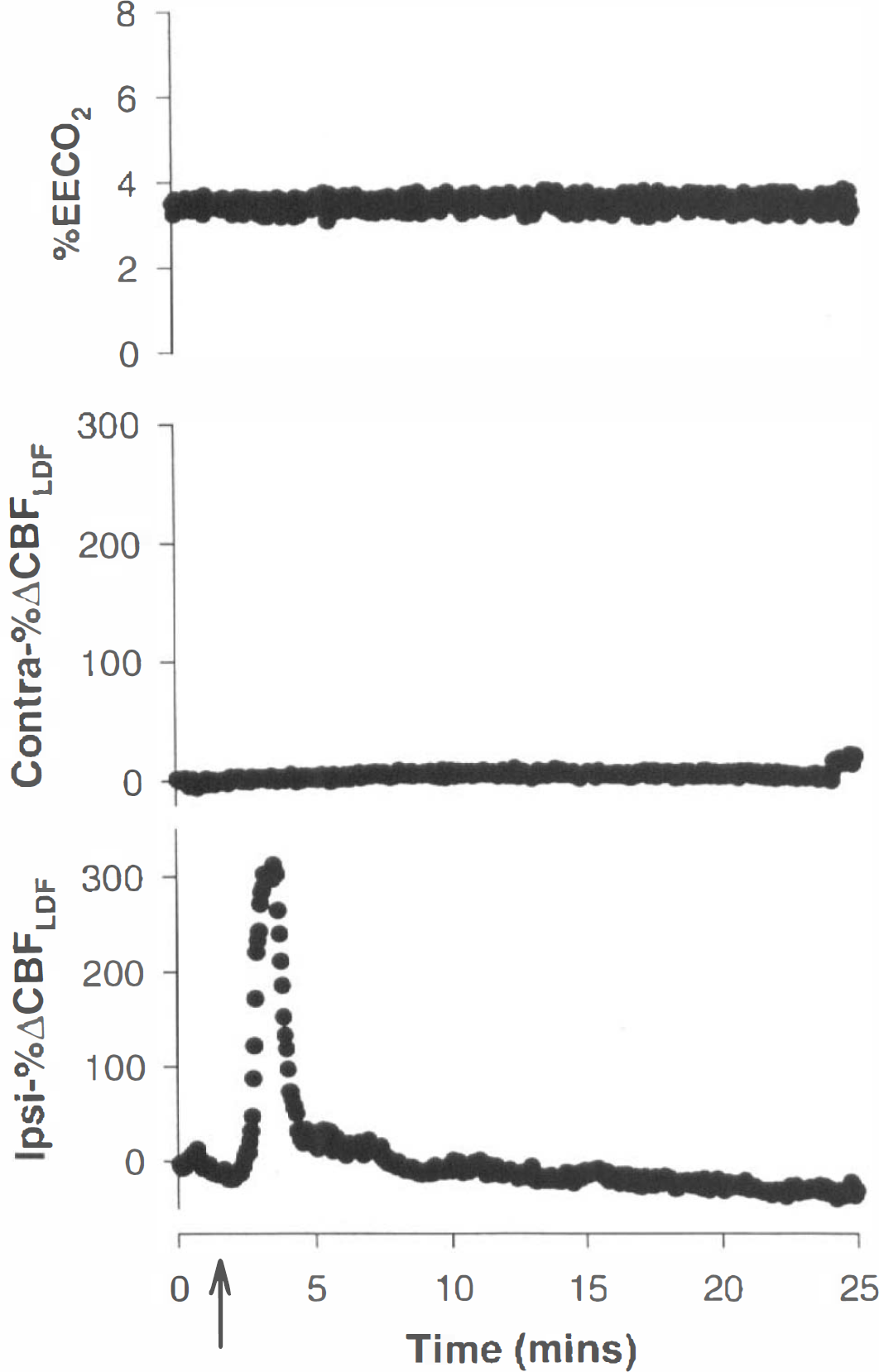
CBFLDF response to needle stick injury of the cortex following administration of FR139317 is shown. The data for CBFLDF are shown ipsilateral to the administration of FR139317 and the induction of SD (lpsi-%ΔCBFLDF) and for the contralateral untreated cortex (Contra-%ΔCBFLDF). The ipsilateral plot demonstrates the marked vasodilatation associated with passage of a wave of SD under the Doppler probe and the subsequent oligemic phase. The time at which the needle stick was induced is shown (arrow), with the × axis being time in minutes. The top trace is the recording of end-expiratory CO2 (%EECO2) during the SD.
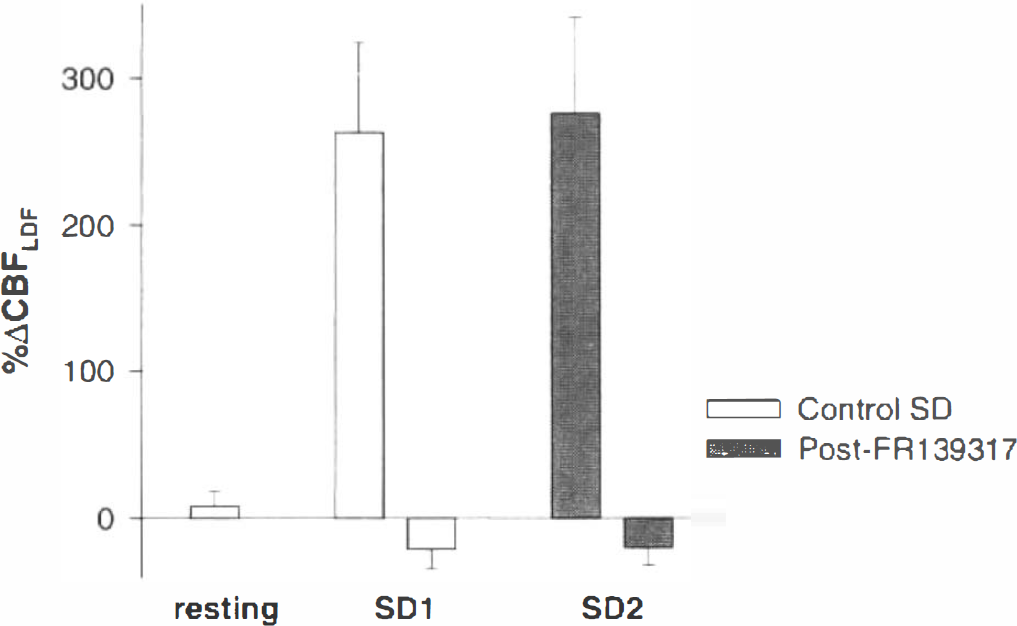
This histogram summarizes the lack of effect of FR139317 (10 μM). The compound had no effect on resting CBFLDF nor did it affect the oligemic phase of spreading depression. SD1 represents the mean (± SD) magnitude of the SD-induced vasodilation (positive histogram) and vasoconstriction (negative histogram) on the control side, while SD2 represents the vasodilator and vasoconstrictor responses after treatment with FR139317. The results in the two groups were not different (p > 0.05).
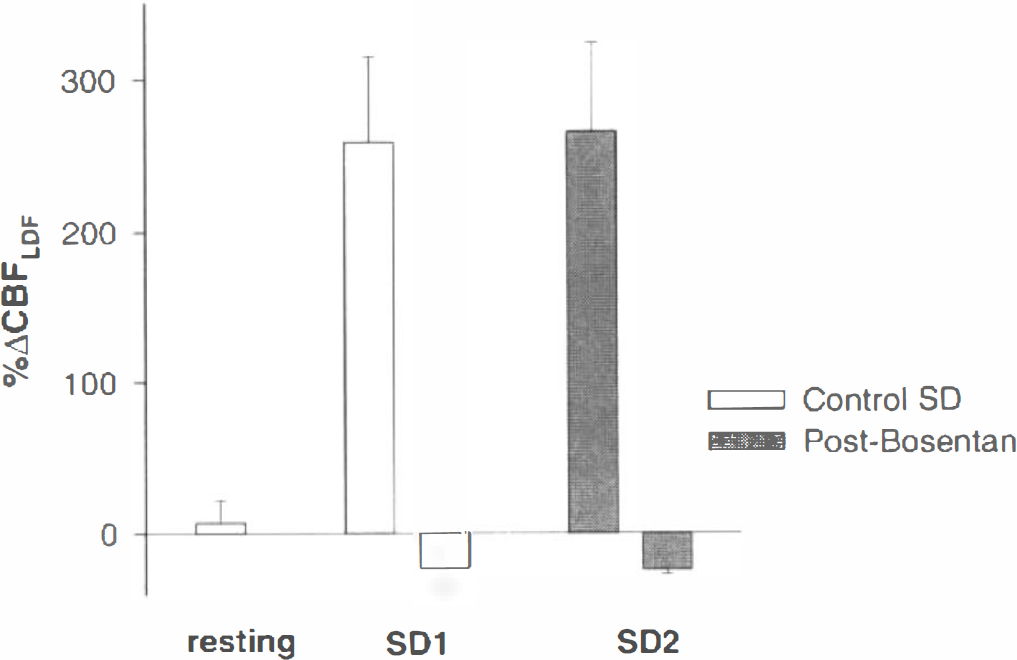
This histogram summarizes the lack of effect of Bosentan (10 μM). The compound had no effect on resting CBFLDF nor did it affect the oligemic phase of spreading depression. SD1 represents the mean (± SD) magnitude of the SD-induced vasodilation (positive histogram) and vasoconstriction (negative histogram) on the control side, while SD2 represents the vasodilator and vasoconstrictor responses after treatment with Bosentan. The results in the two groups were not different (p > 0.05).
DISCUSSION
These data have demonstrated that ET-1 is a strong and potent constrictor of cat cerebral vessels. This vasoconstriction was competitively blocked by the nonpeptide compounds FR139317 and Bosentan. Furthermore, blockade of endothelin receptors with two potent endothelin antagonists did not alter any aspect of the vascular manifestations of SD. The data demonstrate that neither the vasodilator nor oligemic phases of SD were affected by endothelin blockade, even in the presence of doses of antagonists that block the effects of exogenous ET-1. These results suggest that endothelin might not be expected to have a role in pathophysiological settings in which SD is thought to be activated.
The data presented here for FR139317 and Bosentan are consistent with findings in other vascular beds that this compound is an effective endothelin ETA receptor antagonist. Although Bosentan has some activity as an antagonist at the ETB receptor, the data, taken together with those in the literature, suggest the ETA receptor as the main site for the cerebral circulation. This is in contrast to the lack of effect of FR139317 in models in which the endothelin ETB receptor is activated (McMurdo et al., 1993; Sudjarwo et al., 1993). The pA2 in the MCA is most like that of the guinea pig pulmonary artery (pA2 = 6.65) (Cardell et al., 1993) and differs from findings in other vascular beds, such as the guinea pig isolated iliac artery (pA2 = 5.82) (Schoeffter et al., 1993). At the concentrations of FR139317 and Bosentan used in the physiological study, it would be expected that any role for the ETA receptor in SD would have been revealed. Indeed, the positive control in this study (demonstrating in vivo that the vasoconstrictor response to exogenous ET-1 could be eliminated) confirms that biologically relevant concentrations of the antagonists were used. Moreover, FR139317 has been shown in another pathophysiological setting, experimental subarachnoid hemorrhage, to reduce endothelin-mediated vasoconstriction (Nirei et al., 1993). A role for endothelin in the mediation of vasospasm has thus been postulated. The primary objective of this study was to determine if there is a role for the endothelin ETA receptor in the oligemic phase of SD. This receptor site was chosen because of the clear demonstration in pharmacological studies that the ETA receptor is the dominant receptor in the cerebral circulation (Adner et al., 1993) and in view of the suggestion of a possible role for endothelin during migraine (Edmeads, 1991; Farkkila et al., 1992; Gallai et al., 1994). The cerebral circulatory changes associated with SD have been well-characterized. The first phase is a marked hyperemic phase, which is followed by a more prolonged oligemic phase (Lauritzen et al., 1982; Lacombe et al., 1992). In addition, cortical hypercapnic responses are blocked (Lauritzen, 1984), while autoregulation may (Florence et al., 1994) or may not (Lauritzen, 1984) be impaired. The oligemic phase is thought to be the pathophysiological substrate for the neurologically negative symptoms of migraine, such as scotoma and hemiparesis. This interpretation may, however, need to be widened. Recent positron emission tomography data carefully documented a patient with a migraine who had no aura yet had what would be regarded as a classical wave of spreading oligemia (Woods et al., 1994). These new data, albeit from a single case, are compelling enough to require further experimental study of this phenomenon.
Can a SD occur without major neurological symptoms? The design of our study was important to the interpretation of the results. We used the cortex in a paired fashion, with one side receiving antagonist and the other vehicle. This approach was necessary since in our model induction of SD usually blocks or inhibits subsequent oligemia as measured with LDF, even though further hyperemias are relatively easy to induce. Care not to induce SD during the preparatory surgery was crucial since this would result in false-positive blockade of the oligemic phase. We (Goadsby, 1992) and others (Duckrow and Beard, 1992) have addressed this issue previously to examine the role, if any, of free radical generation in the oligemic response (Hall and Smith, 1991).
In summary, these data demonstrate that ET-1 mediates a strong and potent constriction of cat cerebral vessels. The selective ETA receptor antagonist FR139317 and the ETA and ETB antagonist Bosentan competitively antagonized the ET-1-evoked contraction, suggesting that it is mediated through a single population of ETA receptors. These data suggest, since doses of FR139317 and Bosentan capable of blocking endothelin-mediated effects do not block the oligemic phase of SD, that neither ETA nor ETB receptors mediate this phenomenon. If SD or a closely homologous human process is the pathophysiological substrate for the neurological disturbance in migraine with aura, then such compounds are unlikely to alter the course of migraine headache. The ultimate test of this view will be clinical studies in patients with aura. These data predict that such studies will demonstrate no effect of endothelin antagonists in migraine.
Footnotes
Acknowledgment:
The authors thank Karen L. Hoskin for her excellent technical assistance in the physiological studies. These experiments were supported by grants from the Wellcome Trust, the Migraine Trust, the Swedish Medical Research Council (#5958), and Allenburys. P.J.G. is a Wellcome Senior Research Fellow.
