Abstract
In this work, we introduce 6-[18F]fluoro-L-m-tyrosine (6-FMT) and compare its in-vivo kinetic and biochemical behaviors in monkeys and rodents with those of 4-FMT and 6-[18F]fluoro-L-3,4-dihydroxyphenylalanine (DOPA) (FDOPA). These radiofluorinated m-tyrosine presynaptic dopaminergic probes, resistant to peripheral 3-O-methylation, offer a nonpharmacological alternative to the use of catechol-O-methyltransferase inhibitors. Like FDOPA, 4-FMT and 6-FMT are analogs that essentially follow the L-DOPA pathway of central metabolism. After i.v. administration in nonhuman primates and rodents, these new radiofluorinated m-tyrosine analogs accumulate selectively in striatal structures and allow for the detection of additional innervation sites (e.g., brain stem) rich in aromatic amino acid decarboxylase. Biochemical analyses in rodents and monkeys revealed the specificity of their central and peripheral metabolism. Molecular and enzymatic mechanisms involved in their retention in central brain structures are consistent with involvement of dopaminergic neurons. The high signal-to-noise ratios observed make these radiofluorinated m-tyrosine analogs outstanding candidates for probing the integrity of central dopaminergic mechanisms in humans.
Positron emission tomographic (PET) scanning with 6-[18F]fluoro-L-3,4-dihydroxyphenylalanine (FDOPA) provides a means to visualize the dopaminergic system in central brain structures (Garnett et al., 1983a; Calne et al., 1985; Nahmias et al., 1985; Chiueh et al., 1986; Firnau et al., 1986; Leenders et al., 1986a; Martin et al., 1986; Guttman et al., 1989). Investigations in rodents (Cumming et al., 1987; Melega et al., 1990a, b ), nonhuman primates, (Garnett et al., 1983b; Firnau et al., 1987; Doudet et al., 1989; Melega et al., 1991a, b ; Hoffman et al., 1992), and humans (Nahmias et al., 1985; Hoffman et al., 1992; Snow et al., 1993) have unquestionably demonstrated that accumulation of 18F activity in the basal ganglia is related to the integrity of dopaminergic neurons since that activity could be attributed to the aromatic amino acid decarboxylase (AAAD, EC 4.1.1.28) mediated formation of 6-[18F]fluorodopamine (FDA) and its metabolites. Moreover, in double-labeling experiments in rats, it had been shown that the cerebral metabolism of FDOPA and that of [3H]-L-DOPA were correlated (Melega et al., 1990b). These results demonstrated that FDOPA was centrally decarboxylated by AAAD to FDA, which was mainly stored in a slow turnover rate functional pool. Very strong correlations between FDOPA kinetics and severe nigrostriatal degeneration were also demonstrated in both monkeys (Pate et al., 1993) and humans (Snow et al., 1993). In 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP)-treated hemiparkinsonian monkeys, FDOPA kinetics correlated with biochemical data indicating that striatal concentrations of endogenous dopamine, 3,4-dihydroxyphenylacetic acid (DOPAC) and homovanillic acid (HVA) were significantly reduced on the side of the nigrostriatal lesion (Melega et al., 1991b).
Nevertheless, the peripheral metabolism of FDOPA can complicate interpretation of the in vivo data. Tracer kinetic modeling approaches to quantitate FDOPA transport, decarboxylation rates, and metabolite clearance should take into account the transformation of FDOPA into 3-O-methyl-6-[18F]fluoro-L-DOPA (3-OMFD) in peripheral tissues, as well as its brain transport and tissue distribution (Huang et al., 1991; Gjedde et al., 1991). Catechol-O-methyltransferase (COMT, EC 2.1.1.6), the enzyme responsible for this transformation, can be inhibited in vivo (Guttman et al., 1993), reducing the formation of 3-OMFD and its effects on the kinetic data. In this work, we report a substantially different approach to address this problem: the development of fluorinated amino acid analogs that are substrates of AAAD, but inert towards COMT. Herein, we present a comparative biochemical and kinetic evaluation of two such radiofluorinated analogs, 6-[18F]fluoro-L-m-tyrosine (6-FMT) and 4-FMT (Melega et al., 1989), with FDOPA. These new analogs also provide further insight into peripheral and neuronal dopamine biochemistry, allowing for a more accurate representation and understanding of the kinetic data obtain in vivo with FDOPA (Huang et al., 1991; Gjedde et al., 1991).
MATERIALS AND METHODS
Chemicals
Monobasic sodium phosphate, 1-octanesulfonic acid (OSA) sodium salt and arylsulfatase (Type VI, prepared from Aerobacter aerogenes) were purchased from Sigma Chemicals (St. Louis, MO, U.S.A.); perchloric acid (70% aqueous solution), and HPLC-grade methanol were purchased from Fisher Scientific (Los Angeles, CA, U.S.A.); and sodium metabisulfite and ethylene diamine tetraacetic acid (EDTA) disodium salt from EM Science (Gibbstown, NJ, U.S.A.). L-α-hydrazino-α-methyl-β-(3,4-dihydroxyphenyl)propionic acid (carbidopa) was a gift from Merck, Sharp, and Dohme.
Synthesis methods
FDOPA, 4-FMT, and 6-FMT (specific activity: 1–5 Ci/mmol) were prepared by using the radiofluorodestannylation procedures previously described (Namavari et al., 1992, 1993; Satyamurthy et al., 1994). The enantiomeric purities of the L-form of these radiolabeled amino acids were >99%.
Tomographic imaging in primates
Tomographic studies were performed in adult primates [Cercopithecus aethiops (vervet), 6–8 kg, n = 9]. On the evening prior to the PET study, the monkey was maintained on a low-protein diet and kept fasted after midnight. This requirement controls competing large neutral amino acids, the presence of which would reduce the brain transport of the radiofluorinated L-DOPA analogs at the large amino acid carrier transport site at the blood–brain barrier (BBB) (Leenders et al., 1986b). The morning of the study, all necessary equipment was sterilized and the animal examined and its condition noted. If the animal was found to be in a normal state, initial anesthetization was carried out with 4–6 mg/kg ketamine i.m. The animal was also given 5.4 μg/kg atropine sulfate and was then transported to the PET Laboratory. A peripheral i.v. line was placed and the animal given 15 mg/kg of pentobarbital (nembutal) or maintained anesthetically by halothane inhalant techniques (1–2%). Animals were never rescanned within a period of ≤2 weeks. Tomographic data were obtained with a high resolution animal PET scanner (CTI, Knoxville, TN, U.S.A.) that collects 15 planes of data simultaneously in an axial span of 5 cm (Cutler et al., 1992). Interleaved scanning (axially) increased the number of planes to 30, improving axial sampling and leading to a uniform resolution of ∼5 mm. The animal was then placed supine in the tomograph bed with the head in the gantry on a custom cradle designed to support and prevent head movement. One hour prior to scanning, 5 mg/kg of carbidopa i.m. was administered to minimize the spectrum of labeled metabolites formed systemically and to increase bioavailability of the radiolabeled amino acid probe (Melega et al., 1990a). The animal was then positioned in the tomograph and a transmission scan obtained using a Ge-68 ring source for attenuation correction. An intra-arterial catheter was placed in the femoral artery opposite to the lower extremity using a venous catheter. One hour after carbidopa treatment, FDOPA, 4-FMT, or 6-FMT (∼1 mCi/kg) was administered i.v.
A dynamic scanning protocol consisting of regular scans (10 × 90 s and 3 × 300 s) over the first 30 min and 18 5-min interleaved scans over the next 90 min was performed. Interleaved scanning was accomplished by shifting the scanner bed position back and forth by 0.1687 cm (i.e., half the regular plane distance) between adjacent time frames. Images were reconstructed with a reconstruction filter (Hann filter with cut off at the Nyquist frequency), which gives an in-plane resolution comparable to the axial resolution (∼5 mm full width at half maximum). Arterial blood samples were obtained at 12-s intervals for 2 min after injection. Thereafter, samples were obtained every minute from 2–5 min and then at 7, 10, 30, 60, 90, and 120 min for determinations of time activity-curves and plasma concentrations of peripheral metabolites of the radiofluorinated amino acid. Emission scans were corrected for attenuation using the measured transmission scan. During the study, pulse rate and blood oxygenation were monitored with an oxymeter. Body temperature was monitored and maintained constant with a heating blanket.
Determination of radiolabeled metabolites in plasma
Blood samples from tomographic studies were obtained and processed as previously reported (Melega et al., 1991a).
Central and peripheral metabolism in rodents
Male Sprague–Dawley rats (weighing 275–325 g) were anesthetized with halothane and provided with an intra-jugular cannulae. Animals were allowed to recover and were housed individually for 1–2 days. On the day of the experiment (injection time between 9:30 and 11:00 a.m.), animals were pretreated with carbidopa (5 mg/kg s.c.) 60 min before injection of the radiolabeled amino acid (1.4 mCi/kg i.v.) via the cannulae, in a volume of 0.4–0.8 ml. For experiments in absence of carbidopa, a similar protocol was used. Blood samples were withdrawn for peripheral metabolite analysis and were centrifuged (5,000 × g, 5 min) in an Eppendorf microfuge at 4°C, and the plasma removed. To the plasma was added an equal volume of a cold solution containing 0.8 M perchloric acid, 1.0% Na2S2O5, and 0.1% EDTA; the resulting suspension was centrifuged and then filtered (0.2 μm) before HPLC analysis. After blood samples were taken, animals were immediately killed by decapitation. Striata and cerebellum were dissected rapidly (within 3 min) on an ice-chilled aluminum plate, blotted, immersed in liquid nitrogen, weighed, and counted for total 18F activity. Tissues were homogenized in 0.5 ml of a solution containing 0.4 M perchloric acid, 0.5% Na2S2O5, and 0.05% EDTA, centrifuged, and filtered (0.2 μm) before HPLC analysis. Extraction of 18F radioactivity from plasma was essentially quantitative (>95%); from striatum and cerebellum, it was >85% (Melega et al., 1990b). Values reported are uncorrected for extraction efficiency and corrected for 18F decay.
Analytical methods
The HPLC system consists of a Beckman 210 solvent delivery system with the following mobile phase for plasma analysis: 80% 0.1 M NaH2PC>4, 1.7 mM (2.3 mM for brain samples) octanesulfonic acid sodium salt, 0.1 mM EDTA, pH 3.1 (pH 3.3 for brain samples), and 20% MeOH at a flow rate of 1.0 ml/min. A guard cell (+0.45) (ESA, Chelmsford, MA, U.S.A.) preceded a Rheodyne 7125 injector (Cotati, CA, U.S.A.) fitted with a 100 μl loop. A guard column (30 × 4.6 mm, 5 μm; Brownlee, Foster City, CA, U.S.A.) was used in conjunction with a C18 reverse phase column (250 × 4.6 mm, 5 μm; Beckman, Fullerton, CA, U.S.A.). The analytical cell (model 5011, ESA) was set at an applied potential of +0.02 V (detector 1) and +0.4 V (detector 2) and coupled to a coulochem controller (model 5100A, ESA) to monitor endogenous catecholamine and carbidopa levels. Fractions (1 ml/min) were collected and counted for radioactivity (decay corrected) in an Nal well counter. For FDOPA, radioactivity peaks were identified by comparison of their retention times with those of authentic metabolite standards (Luxen et al., 1987).
Data analysis
Separate regions of interest (ROIs) (∼0.9 cm2 on a cross-sectional plane of largest striatal area) for the left and right striata were defined according to the apparent boundary of the structure on the PET image (summed from 30 to 120 min). ROIs (∼2.0 cm2) for the cerebellum were similarly defined, but were based on summed PET images 1–10 min postradiotracer injection. These ROIs were then applied to the serial PET images at various scan times to give striatal and cerebellar time activities. Curves from the left and right sides were averaged to give one striatal and one cerebellar curve for each study.
Arterial plasma time-activity curves (TAC) of radiolabeled amino acids (6-FMT, 4-FMT, FDOPA) were obtained from the total plasma radioactivity time-activity curves and measured fractions of the original compound, according to the modeling approach previously described (Huang et al., 1991). Tissue TAC and plasma TAC, determined above, were then used to examine the transport and uptake characteristics of the radiolabeled probe in tissue. The three-compartmental model previously developed for FDOPA kinetics (Huang et al., 1991) was used, with K, representing the forward BBB transport constant of the original labeled tracer; k2, the reversed BBB transport rate constant; k3, the decarboxylation rate constant; and k4, the clearance rate constant of decarboxylated species (and/or their metabolites) from tissue to plasma. For FDOPA, the peripheral metabolite 3-OMFD crosses the BBB and a single reversible compartment was included to describe the kinetics of 3-OMFD (Huang et al., 1991). For 6-FMT and 4-FMT, no peripheral 3-O-methylation occurs; therefore, no plasma labeled metabolites were assumed to cross the BBB in a significant manner. The model fitting procedure was performed using BLD (Carson et al., 1981), a software package for kinetic data modeling and simulation. The uptake constant (K3) of the labeled tracer from plasma to tissue was calculated from the estimated values of K1, k2, and k3 as K1k3/(k2 + k3). The value of K3 was also estimated by Patlak analysis (Patlak et al., 1983) using tissue TACs from 30 to 60 min and the entire plasma TAC of the labeled tracer (excluding all labeled metabolites). Analysis of variance (ANOVA) was used to determine whether the results among these three tracers were different. A p-value of <0.05 was considered statistically significant in the analysis.
Tomographic three-dimensional (3D) imaging process
Stereo paired 3D images of brain FDOPA PET images were prepared using the AVS5.1 software system (AVS Inc., Waltham, MA, U.S.A.) on a Sun SPARC 10/41 workstation. Brain FDOPA PET images were first segmented by thresholding the image pixel values into three levels, resulting in images consisting of only three different regions, namely, (a) one of high specific tracer uptake, (b) one of low tracer uptake, and (c) one outside of the animal's head. An edge detection technique was used to determine the surfaces of the cranium and the structures of high specific tracer uptake. Lighting and shading from the surfaces were generated by ray tracing (486 × 486 with interpolation). Moderate transparency was applied to the cranial surface (in blue), while the surfaces of specific uptake structures were opaque (yellow). Stereo-pairs were produced by 3D lateral movement of the object in generating the lighting and shading (i.e., the ray tracing step).
RESULTS
Brain and peripheral kinetics (monkeys)
Figure 1 shows typical images 90–120 min after systemic injection of the radiolabeled amino acids (6-FMT, 4-FMT, and FDOPA) in nonhuman primates [C. aethiops (vervet), n = 9]. Accumulation of radioactivity was consistent with localization of central dopamine-rich structures (Hefti et al., 1981). In all cases, specific (striatum) to nonspecific (cerebellum) ratios increased with time (Table 1). However, these ratios are dependent on the radiolabeled amino acid being higher for 6-FMT (and 4-FMT) than for FDOPA. These new radiofluorinated m-tyrosine analogs also accumulated in innervation sites rich in aromatic amino acid decarboxylase (e.g., brain stem), as shown in Fig. 2 with 6-FMT. Figure 3 shows representative arterial plasma total radioactivity and 6-FDOPA (t1/2 = 23 min), 4-FMT (t1/2 = 32 min), and 6-FMT (t1/2 = 57 min) clearance curves in monkeys. Table 2 shows the metabolite composition at selected time points (90 and 120 min postinjection) obtained from the monkey arterial plasma activity curves. The plasma FDOPA metabolite profile comprised mainly FDOPA and 3-OMFD, consistent with previous observations in monkeys (Melega et al., 1990a, 1991a). Other metabolites (e.g., FDA sulfoconjugate, 7.1 ± 2.3%) derived from incomplete inhibition of peripheral AAAD with carbidopa were also observed. Both radiofluorinated L-m-tyrosines, 4-FMT and 6-FMT, showed different arterial plasma metabolite profiles. As expected, neither 4-FMT (Melega et al., 1989) nor 6-FMT are substrates for COMT and, hence, no COMT-moderated products were present in plasma. However, under the conditions of the experiments, both 4-FMT and 6-FMT showed products of AAAD-mediated peripheral decarboxylation even with carbidopa administrations, paralleling results obtained with FDOPA. Interestingly, the peripherally formed 6-[18F]fluoro-m-tyramine (FMA) appears moderately resistant to monoamine oxidase (MAO) oxidation, but susceptible to O-sulfation mediated by phenolsulfotransferase (EC 2.8.2.1) (Roth, 1986). Similarly, 4-FMA, formed by AAAD-mediated decarboxylation of 4-FMT, is also subsequently conjugated by phenolsulfotransferase in peripheral tissues. In contrast to 6-FMA, however, 4-FMA suffers extensive MAO oxidation to form 4-fluoro-3-hydroxyphenylacetic acid (FPAC) as its final metabolite, as estimated by HPLC. Similar results were also observed peripherally in rodents (Fig. 4).
Striatum/cerebellum ratios in monkeys a
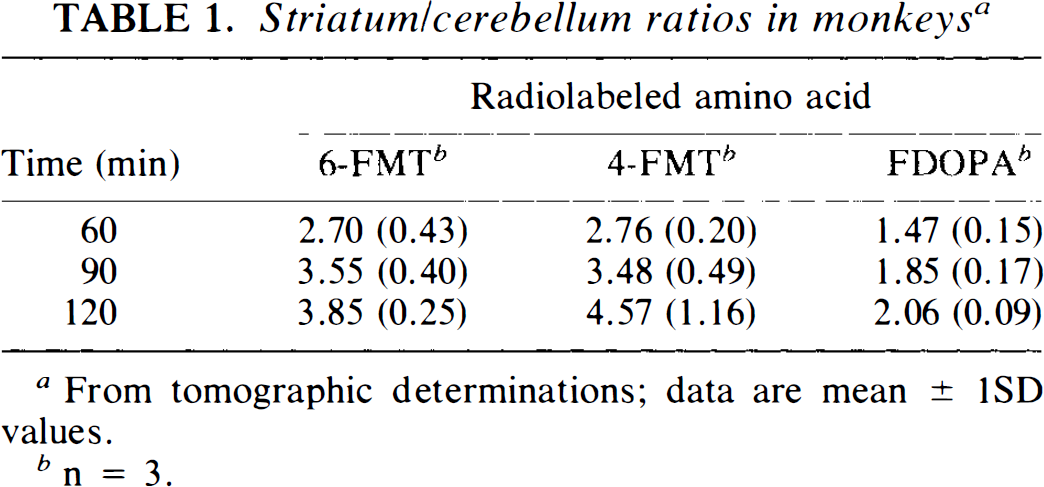
From tomographic determinations; data are mean ± 1SD values.
n = 3.
Arterial plasma metabolites in nonhuman primates a
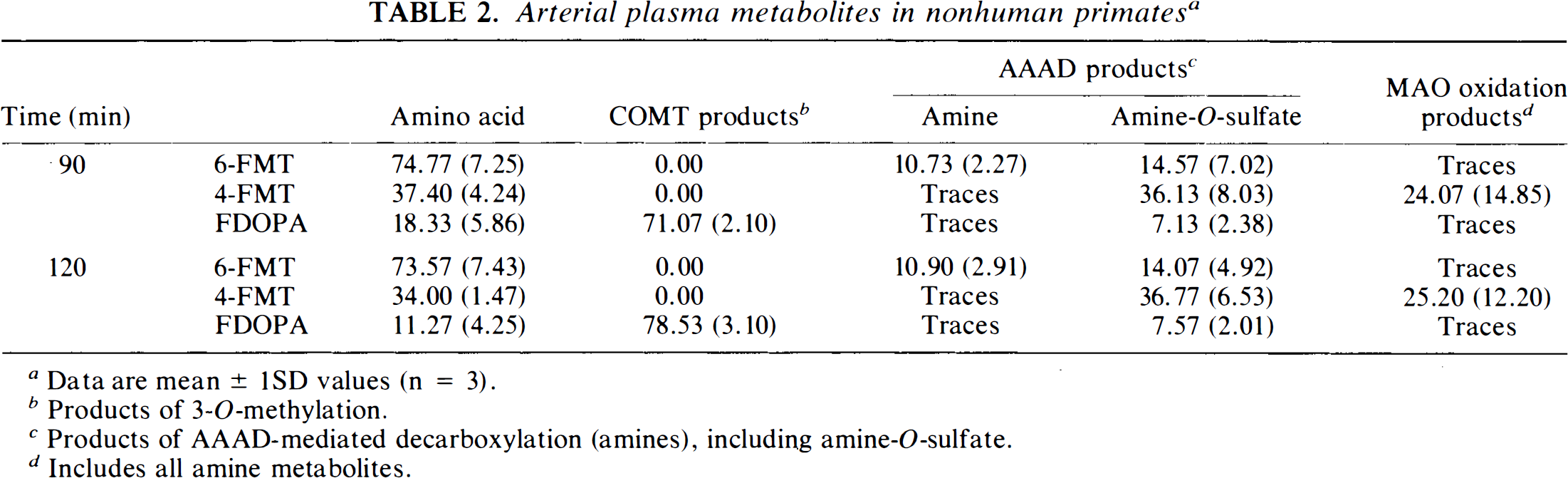
Data are mean ± 1SD values (n = 3).
Products of 3-O-methylation.
Products of AAAD-mediated decarboxylation (amines), including amine-O-sulfate.
Includes all amine metabolites.
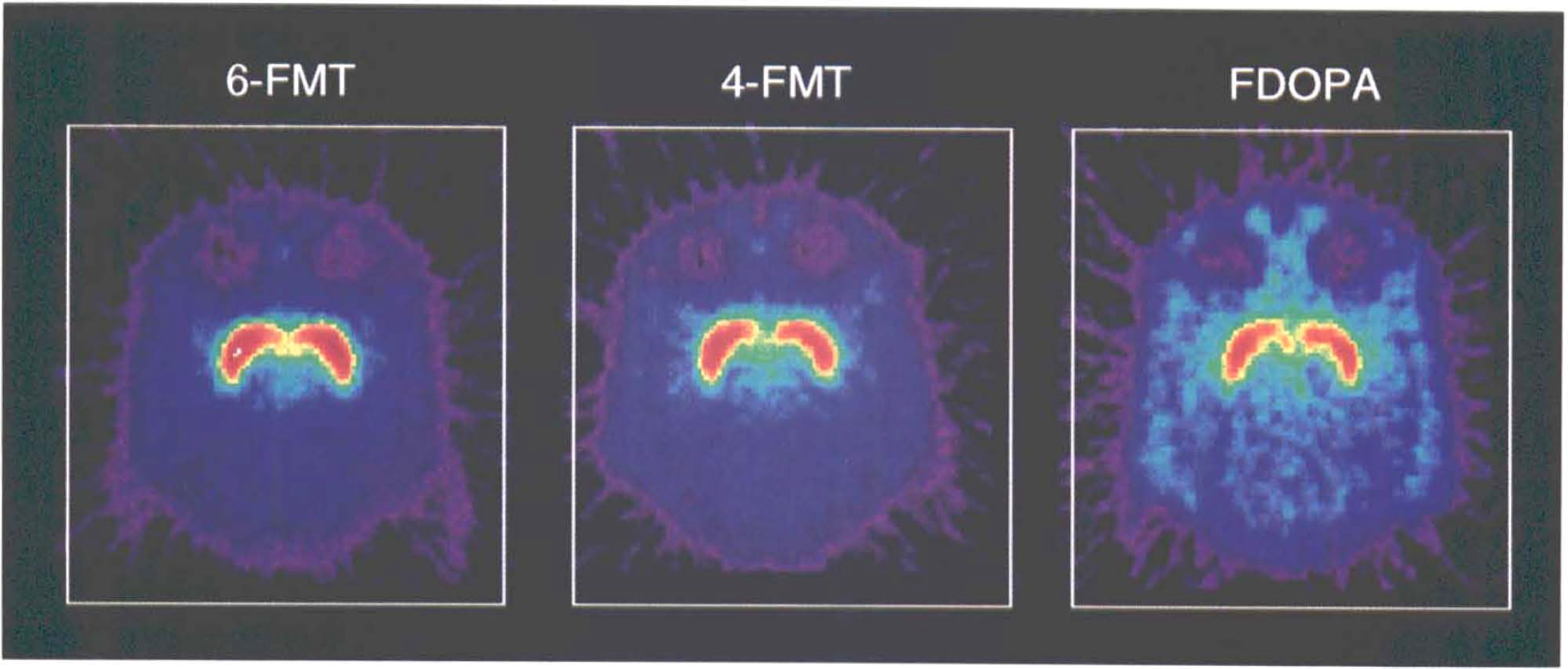
Representative tomographic sum images obtained 90–120 min postinjection of the radiofluorinated amino acids in vervet monkeys (see Material and Methods). Note the higher signal-to-noise (specific: non specific) ratios observed with the radiofluorinated L-m-tyrosines than with FDOPA.
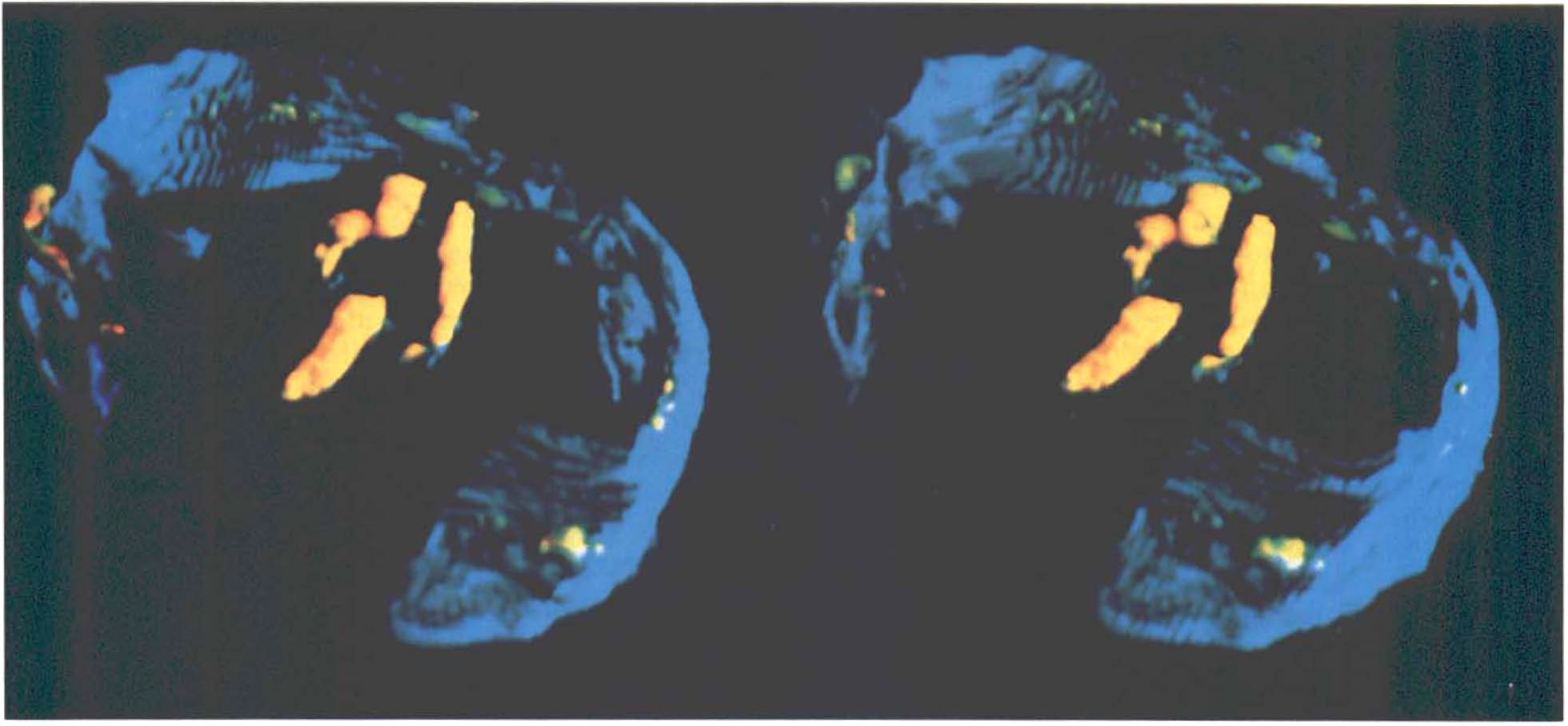
Three-dimensional stereopaired PET images (posterior view) of a monkey brain, integrated from 90 to 120 min after injection of 6-FMT. Note distribution of activity (yellow) in the basal ganglia and brain stem. Cranial outline is shown in blue; a portion of cranial outline has been erased for clarity.
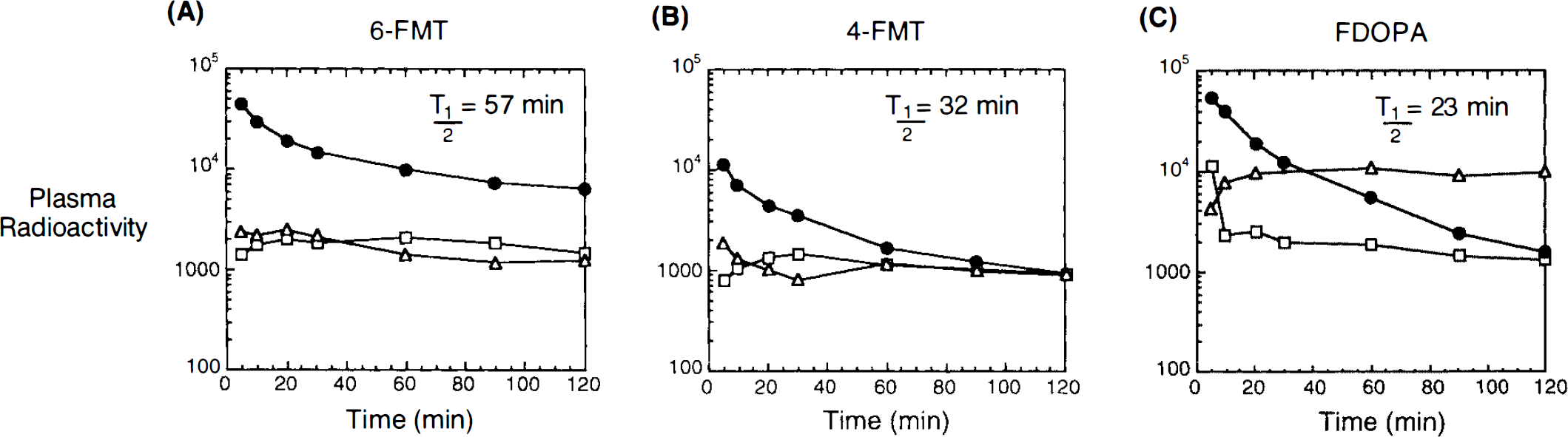
Representative arterial plasma time-activity curves and metabolite(s) profile after i.v. injection of 6-FMT (
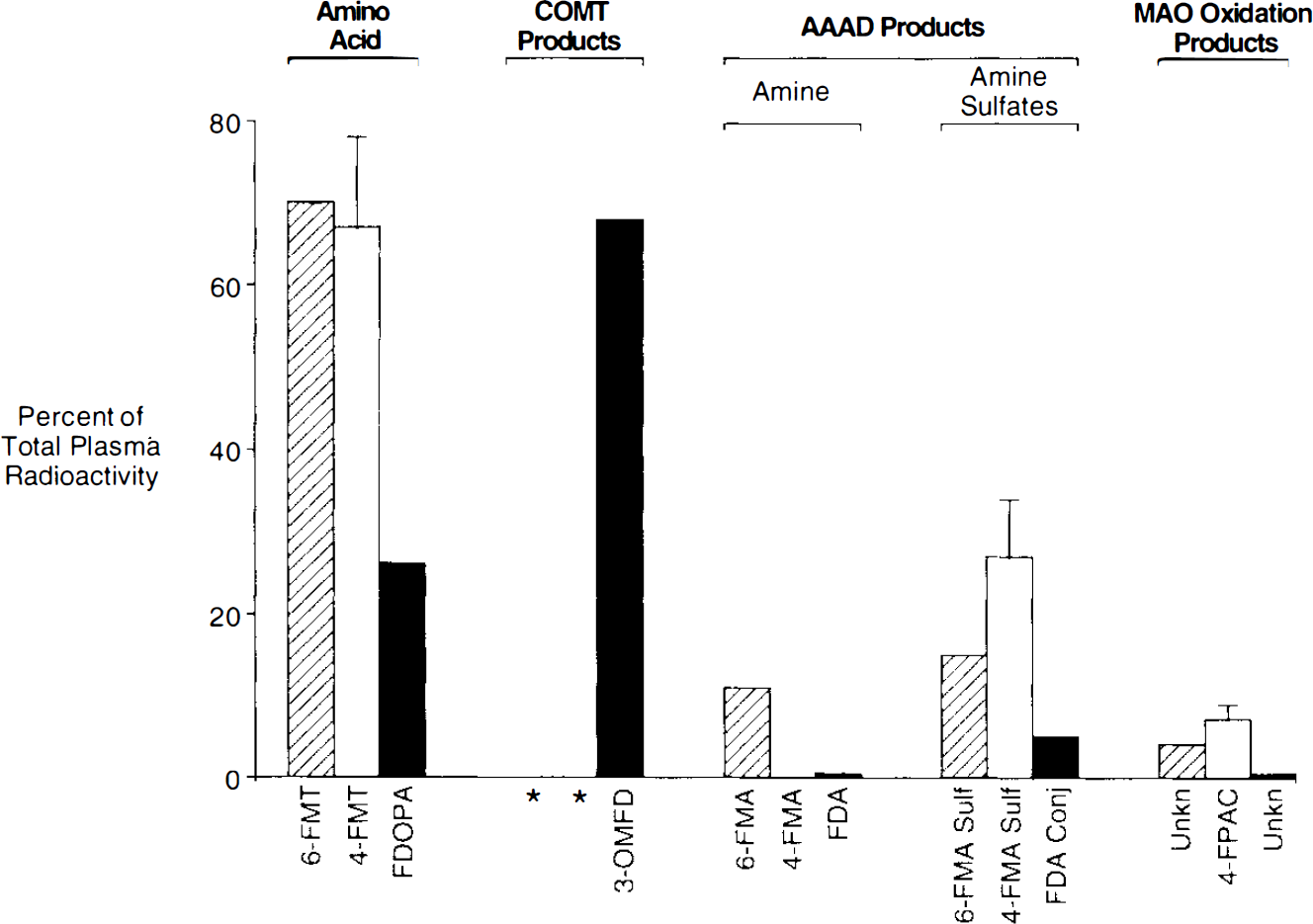
Arterial plasma metabolite profile in rats (carbidopa, 5 mg/kg), 30 min after injection of 6-FMT (shaded), 4-FMT (white), or FDOPA (black). Note the absence of COMT products with the radiofluorinated L-m-tyrosine analogs (*); 3-OMFD is the most prominent metabolite in arterial plasma at the same time point. However, an increase in AAAD products and, consequently, MAO oxidation products, is observed with the radiofluorinated L-m-tyrosine derivatives (see text for discussion). 4-FMT: mean ± 1SD; FDOPA and 6-FMT: mean of two determinations. Conversion rate constants in plasma (Huang et al., 1991) are as follows: 6-FMT: 0.0082 ± 0.0070 min−1; 4-FMT: 0.0195 ± 0.0019; and FDOPA: 0.0115 ± 0.0013.
Figure 5 shows tissue TACs in striatum and cerebellum of monkeys for the three radiolabeled amino acids and their model-fitted curves. For the same injected dose (1 mCi/kg), striatal uptakes were in the order of 6-FMT > 4-FMT > FDOPA. The continuing rise of striatal radioactivity 30 min after 6-FMT injection was consistent with the relatively high 6-FMT time-dependent plasma concentration (Fig. 3). The slower plasma clearance for 6-FMT compared with those of 4-FMT and FDOPA was also reflected in the cerebellar activity. In the case of FDOPA, the relatively high cerebellar level at late times was mostly due to the presence of 3-OMFD (Melega et a., 1990b), which crossed the BBB from the plasma. It should be noted that the apparent decrease of the striatal 4-FMT accumulation after 30 min (Fig. 5) was related to the fast decrease in cerebral 4-FMT concentrations that followed its rapid rate of plasma clearance and was not due to a larger clearance rate of labeled metabolites produced in brain tissue (see model-fitting results below). For FDOPA, the striatal activity and plasma FDOPA concentrations also fell rapidly; this was offset by an increase in plasma 3-OMFD, which was transported from plasma across the BBB. Therefore, the fall of the striatal FDOPA curve was not as apparent as that of 4-FMT.
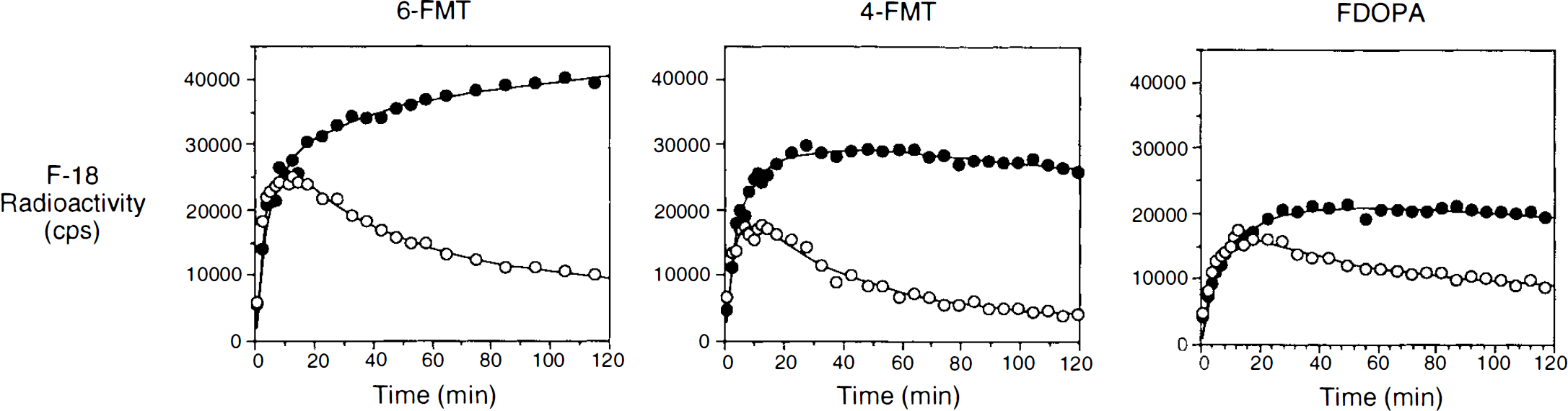
Representative tissue time activity curves in striatum (closed circles) and cerebellum (open circles) after i.v. injection of 6-FMT, 4-FMT, and FDOPA in presence of carbidopa. Solid curves through the data points are the model-fitted curves by the compartmental model originally developed for FDOPA (see text for details). Curves have been normalized for the injected dose in mCi/kg. Units of the y-axis were converted from the PET image values to equivalent counts/s/ml in a well counter (detection efficiency for positron emission is 0.42).
Model fitting of the striatal TAC of 6-FMT and 4-FMT showed that the three-compartmental model developed originally for FDOPA (without the component for the peripheral metabolite 3-OMFD) can adequately describe the striatal kinetics of these tracers (solid curves in Fig. 5). Estimated values of the model parameters for 6-FMT and 4-FMT are summarized in Table 3 along with those for FDOPA. The k4 value for FDOPA was fixed to zero in the model fitting. Allowing it to be variable gave slow convergence and large correlation among different estimated parameters (a sign of over-parameterization). This was not the case for 6-FMT and 4-FMT. Only the values of distribution volume and K3 were found to be significantly different (at p < 0.05 level) among the three tracers. The uptake constants (K3) of both 6-FMT and 4-FMT were each larger than the corresponding value for FDOPA, but only the decarboxylation rate constant (k3) of 6-FMT was significantly larger than that of FDOPA. The uptake constants, as calculated from the model fitting results, were found to be comparable to the values separately estimated by Patlak analysis, which assumes unidirectional uptake of tracer in tissue (Patlak et al., 1983). This consistency further indicates that the clearance rates (k4) of the three tracers from the striatum were relatively small during the first 2 h after their injection. Comparable values of K1 and k2 among the three tracers indicated that their BBB transport rates were not significantly different.
Estimated transport rate constants according to a compartmental model developed originally for FDOPA
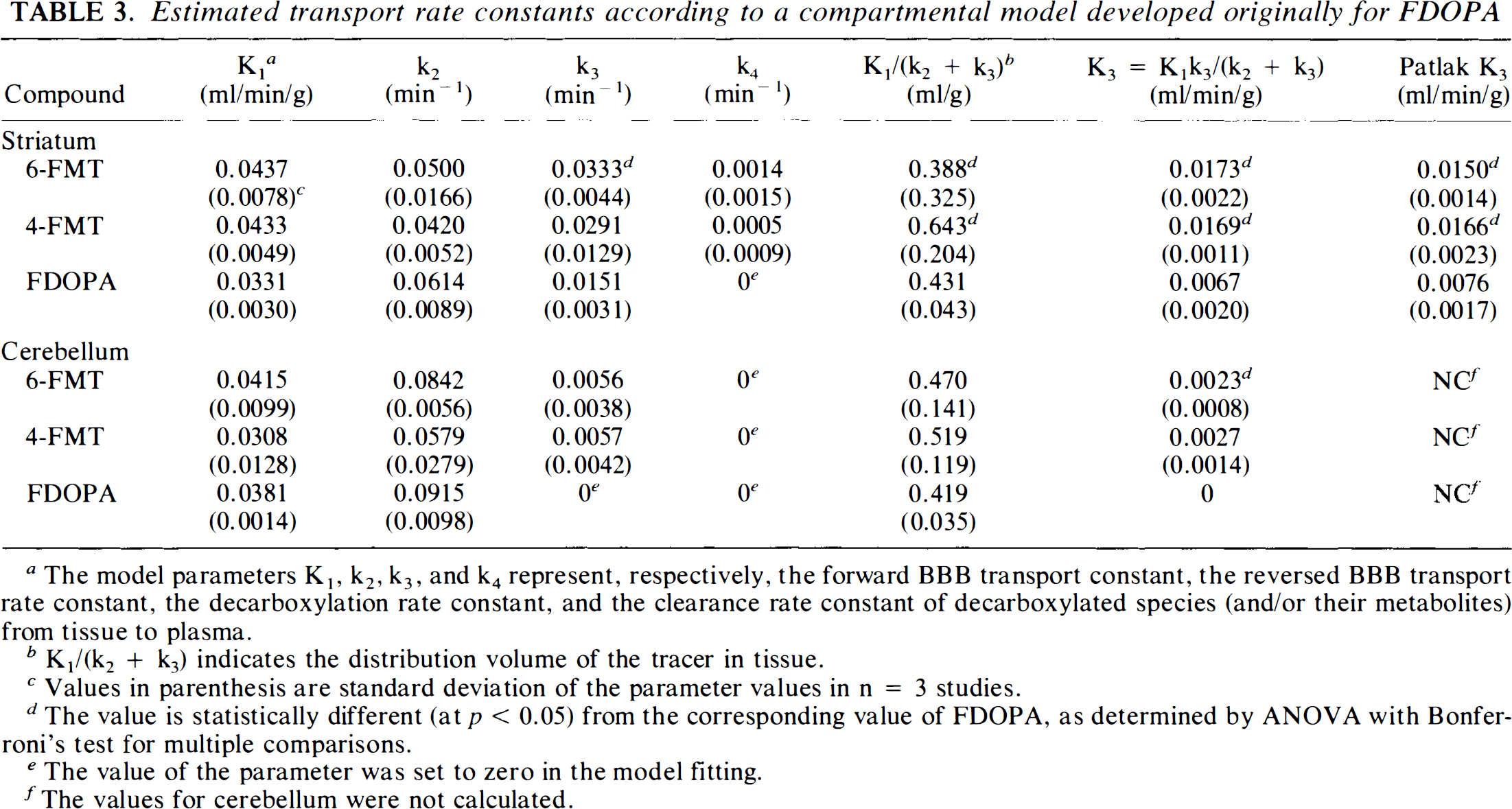
The model parameters K1, k2, k3, and k4 represent, respectively, the forward BBB transport constant, the reversed BBB transport rate constant, the decarboxylation rate constant, and the clearance rate constant of decarboxylated species (and/or their metabolites) from tissue to plasma.
K1/(k2 + k3) indicates the distribution volume of the tracer in tissue.
Values in parenthesis are standard deviation of the parameter values in n = 3 studies.
The value is statistically different (at p < 0.05) from the corresponding value of FDOPA, as determined by ANOVA with Bonferroni's test for multiple comparisons.
The value of the parameter was set to zero in the model fitting.
The values for cerebellum were not calculated.
The cerebellar tissue kinetics can also be fitted well by the same compartmental model, except that the clearance rate constant (k4) was relatively small and could not be determined reliably with the available data. The values summarized in Table 3 were obtained by setting the k4 value to zero. None of the estimated values were statistically different among the three tracers except for K3. The K3 value of 6-FMT in cerebellum is nonzero and was significantly larger than that of FDOPA. However, the modeling analysis of the present data could not determine whether this was due to decarboxylation of the 6-FMT in cerebellum or due to a nonzero BBB permeability of plasma 6-FMA.
Tissue and peripheral metabolism (rodents)
A comparative distribution of radioactivity profiles in rodent brain striatum 30 min after radiolabeled amino acid administration is shown in Fig. 6. For FDOPA, FDA (33 ± 4%) and 3-OMFD (51 ± 4%) were the predominant metabolites; small amounts of FDOPA (1 ± 0.3%) and FDA metabolites (FHVA, FDOPAC and conjugates, 4 ± 0.5%) were also observed. Consistent with the observations in peripheral tissues in both monkeys (Table 2) and rodents (Fig. 4), 6-FMT (28 ± 5%) and 6-FMA (53 ± 6%) and its amine conjugate (4 ± 3%) were observed in rodent striatum. Similarly, 4-FMT results in the same tissue paralleled those observed peripherally in rodents (Fig. 4) and monkeys (Table 3). Interestingly, the product of 4-FMA oxidation was the most significant radiolabeled product observed in striatal tissue (Melega et al., 1989). Distribution of total activity profiles was also determined in rodent striatum, cerebellum, and arterial plasma 30 min after i.v. administration of 6-FMT in the presence of carbidopa (Fig. 7). The presence of 6-FMA in cerebellar tissue is consistent with low, but significant, levels of AAAD in rat cerebellum (Rahman et al., 1981a). The paralleled FMT metabolite) profiles between cerebellum (Fig. 7B) and plasma (Fig. 7C), however, may not discount 6-FMA BBB permeability. Accordingly, in absence of carbidopa, when significant peripheral decarboxylation of 6-FMT exists, an increase in 6-FMA in cerebellum was observed (Fig. 8C) when compared with the same experiment in the presence of carbidopa (Fig. 8B). Similarly, total 6-FMA activity in striatum was decreased (Fig. 8A).
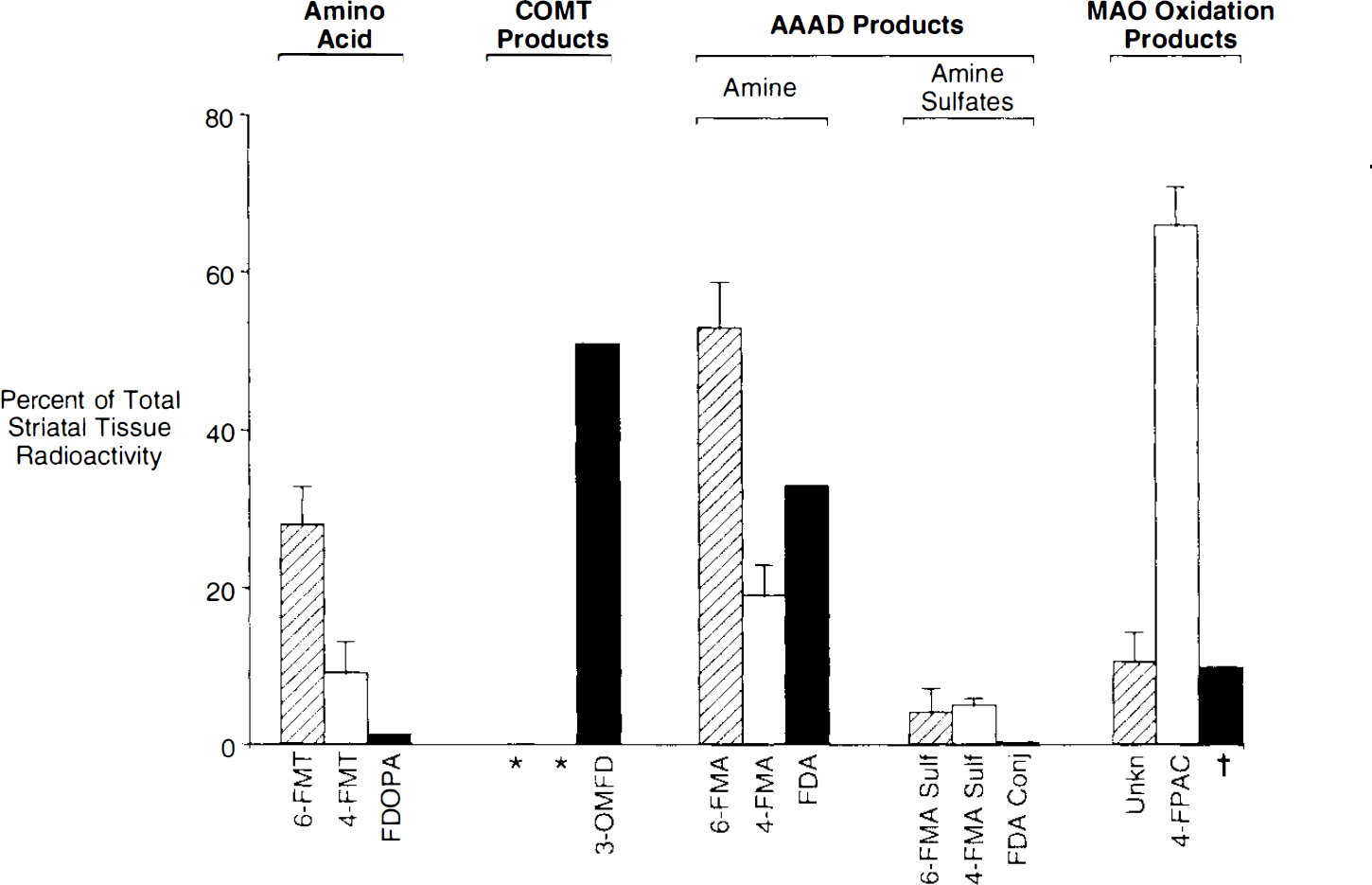
Striatal brain tissue metabolite profile in rats (carbidopa 5 mg/kg), 30 min after i.v. administration of 6-FMT (shaded), 4-FMT (white), or FDOPA (black). After administration of FDOPA, 3-OMFD was the most abundant metabolite found in rat striatum, contributing significantly to tissue-background activity. FDOPA metabolites (MAO oxidation products)† are a combination of FDOPAC and FHVA. No COMT products are observed for 6-FMT or 4-FMT. 6-FMA, resulting from the AAAD-dependent decarboxylation, was the major metabolite found from 6-FMT. Products of MAO oxidation, after AAAD-dependent decarboxylation, were prevalently found from 4-FMT (see text for discussion). 6-FMT and 4-FMT: mean ± 1SD (n = 3); and FDOPA, mean of two determinations.
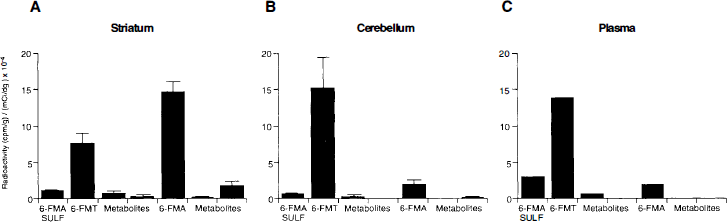
Absolute concentrations of metabolites in striatum (
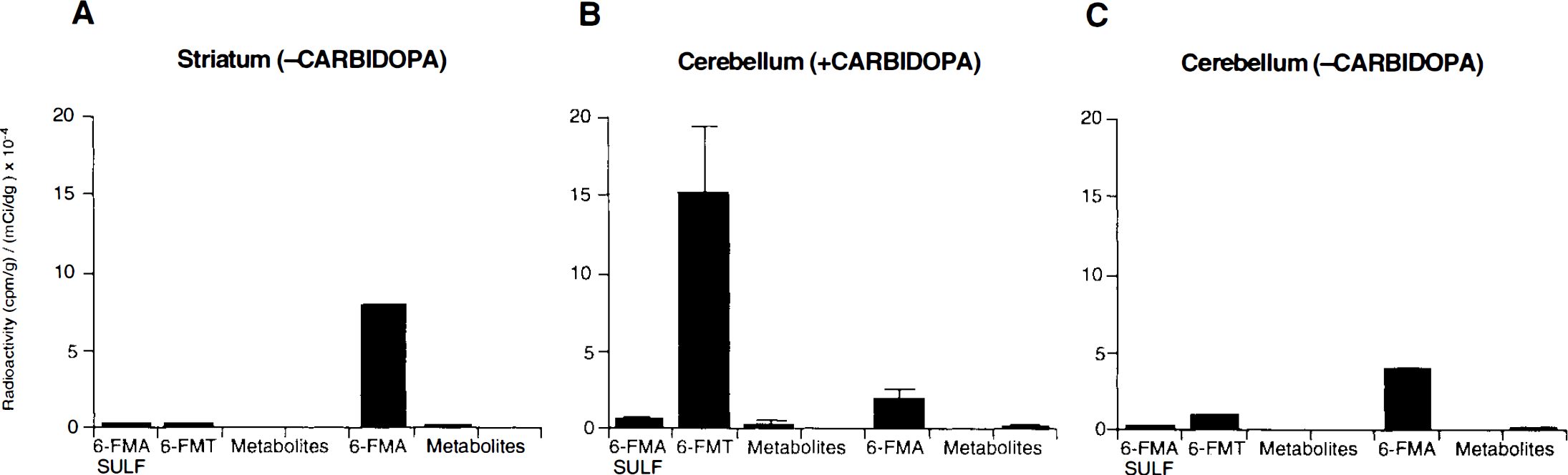
Effects of carbidopa on the absolute concentrations of metabolites after systemic (i.v.) administration of 6-FMT in rats. (–Carbidopa) indicates no administration of carbidopa; (+ carbidopa), 5 mg/kg.
DISCUSSION
Brain kinetics: kinetic rate constants in striatum (monkeys)
It has been shown that FDOPA presents excellent specificity for AAAD, and, indeed, probes central dopaminergic mechanisms in vivo (Barrio, 1991). Specific accumulation of 18F activity in the basal ganglia is attributed to the AAAD-mediated formation of FDA and its metabolites (Cumming et al., 1987; Melega et al., 1990b). However, interpretation of FDOPA kinetic data is far from clear, and important issues still remain unanswered about the in vivo use of FDOPA with PET. Beyond the biochemical uncertainties associated with FDOPA determinations, among others, the quantitative relationship between FDOPA kinetics and endogenous dopamine synthesis (Barrio, 1991), the high nonspecific tissue background, and the use of ROIs still present problems for quantitative interpretation of the data (Yu et al., 1993). The issue of FDOPA sensitivity is particularly critical with dopaminergic cell degeneration in idiopathic Parkinson's disease or MPTP exposure (Barrio et al., 1990).
The introduction of fluorinated L-m-tyrosine analogs is designed to develop new in vivo approaches and new tools to characterize biochemical processes occurring at neuronal terminals. The use of fluorinated m-tyrosine analogs is supported by a strong biochemical and pharmacological rationale. For example, we find that (a) m-tyrosine is an excellent substrate for AAAD (Awapara et al., 1962; Srinivasan and Awapara, 1978; Borri-Voltattorni et al., 1983); the Km and Vmax values derived from Lineweaver-Burk plots indicate that L-m-tyrosine behaves as a better substrate than does L-DOPA (Borri-Voltattorni et al., 1983); (b) BBB transport of 4-FMT is significant (Melega et al., 1989); (c) newly obtained in vitro data (Barrio et al., 1995) on the neuronal vesicular transport of radiofluorinated m-tyramines indicate that, similar to dopamine, radiofluorinated m-tyramines are substrates for the energy-dependent vesicular transporter and, therefore, stored in vesicles after its AAAD-mediated formation; (d) in in vitro evaluation of affinity at dopamine D-1 and D-2 receptor subtypes, 4-FMA had a similar affinity (∼twofold less affinity) for D-1 and D-2 binding sites than dopamine (Claudi et al., 1992). Other fluorinated phenylethylamines also show significant affinities for postsynaptic receptors, implicating that the catechol moiety is not required (Cardellini et al., 1988); (e) structural requirement analysis of substrates for the striatal dopamine reuptake carrier has shown that the 3-hydroxy group of dopamine is critical for transport (Meiergerd and Schenk, 1994); indeed, m-tyramine inhibited the transport of dopamine as effectively as a catechol-containing derivative (Km = 2.6 ± 0.4 μM versus 1.1 ± 0.1 μM for dopamine); and (f) both 4-FMA and 6-FMA are substrates for MAO A oxidation (Barrio et al., 1995).
The data presented herein confer validity to the assertion that, like FDOPA, both 4-FMT and 6-FMT have, a priori, all the given conditions to be appropriate in vivo probes for dopaminergic mechanisms. Further support comes from evidence suggesting that phenylethylamine and dopamine and their metabolites are co-localized in the nigrostriatal system, more specifically, in tyrosine hydroxylase-containing neurons (Juorio et al., 1991).
Indeed, this work demonstrates that both radiofluorinated m-tyrosine analogs of L-DOPA accumulate selectively in striatal structures (Fig. 1) and have comparable or higher uptake constants than those of FDOPA (Table 3); in this regard, they compare favorably with FDOPA because of their increased signal-to-noise ratios (Table 1). Striatal tissue is better delineated with both 4-FMT and 6-FMT than with FDOPA, a particularly important feature when central dopaminergic structures had suffered degeneration (e.g., Parkinson's disease) or in determinations with small animals (e.g., squirrel monkeys) (data not shown). Moreover, 4-FMT and 6-FMT allow for a more sensitive detection of additional innervations sites (e.g., brain stem), with high AAAD concentrations in all animal species investigated (Kuntzman et al., 1961; McCaman et al., 1965; MacKay et al., 1978; Rahman et al., 1981b) (Fig. 2). Lastly, a lower background level allows the use of properly validated methods (Müller-Gärtner et al., 1992) to correct for partial volume effects (Hoffman et al., 1979) to give more accurate and reliable measurements.
The substantially increased sensitivity with the radiofluorinated L-m-tyrosines (Table 3) appears related to at least two factors. The first is that striatal uptake (normalized by amount injected and body weight) follows the order 6-FMT 3= 4-FMT ≥ FDOPA (p < 0.05). The increased uptake constant (K3) (Hoffman et al., 1992) from plasma to tissue, with 6-FMT > FDOPA (p < 0.05) and 4-FMT > FDOPA (p < 0.05) is a reflection of increased rates of AAAD-mediated decarboxylations. The second is the lack of peripheral 3-O-methylation for 4-FMT and 6-FMT, which reduces the significant tissue background from this source (e.g., 3-OMFD) (Table 3). As a result of the two factors stated above, the striatal-to-cerebellum ratios follow the order 6-FMT ≅ 4-FMT > FDOPA (with 6-FMT > FDOPA, and 4-FMT > FDOPA) (p < 0.05).
Brain biochemistry: striatal metabolites (rodents)
The three-compartmental model configuration (Huang et al., 1991) describes the tissue and plasma kinetics for all radiofluorinated analogs as measured with PET in nonhuman primates. From the kinetic data, this modeling approach provides information on BBB transport, AAAD-mediated decarboxylation rates, as well as the rate of release of tissue metabolites. However, even though similar delineation of central brain structures and tissue kinetics can be described for FDOPA, 4-FMT and 6-FMT (Fig. 1 and Table 3), the molecular species involved in tissue trapping differ. Biochemical analyses of rat striatal tissue revealed that after 6-FMT i.v. administration (Fig. 6), the main metabolite found in striatum at 30 min was the product of AAAD-mediated decarboxylation, namely 6-FMA (54 ± 4%). The remainder was unmodified 6-FMT (28 ± 5%) and 6-FMA (18 ± 4%) oxidation metabolites. This is similar to the metabolite composition found after FDOPA administration, except for the absence of the peripherally generated 3-OMFD, which is competitively transported from plasma to the brain.
It has been shown earlier that conversion of FDOPA (and L-DOPA) to FDA (and dopamine) and metabolites occurs in corpus striatum mainly in dopaminergic neurons (Hefti et al., 1991; Melega et al., 1990b). Also, since noncatechol-containing phenylethylamines have distribution and metabolism similar to dopamine (Juorio et al., 1991), it can be inferred from these data that 6-FMT nigrostriatal metabolism occurs predominantly in dopaminergic neurons. In addition, 6-FMA, by analogy with FDA (and dopamine) is not extensively metabolized to further products of central oxidation of monoamines, and, therefore, also appears to be mainly stored in a slow turnover rate functional pool (Barrio et al., 1990; Melega et al., 1990b). This is to be expected from newly obtained in vitro data on the neuronal vesicular transport and MAO A oxidation rates of 6-FMA, 4-FMA, and FDA (Barrio et al., 1995).
By virtue of the cerebral distribution of radioactivity accumulation, 4-FMT metabolism is also expected to occur in tyrosine hydroxylase-containing neurons. However, metabolite distribution differed from that encountered with FDOPA or 6-FMT. At 30 min after 4-FMT administration in rats, most of the striatal activity (66 ± 5%) was attributed to 4-FPAC, the formation of which is mediated by central MAO oxidation of 4-FMA, the product of 4-FMT decarboxylation. The remaining activity was 4-FMT (9 ± 4%), 4-FMA (19 ± 3%), and its sulfoconjugate (5 ± 1%). The presence of the MAO oxidation product, 4-FPAC, as the predominant striatal metabolite (Melega et al., 1989), suggests that after 4-FMT central decarboxylation, 4-FMA MAO central oxidation competes favorably with its vesicular transport. It is also apparent from the kinetic data in nonhuman primates, that the central release of monoamine metabolites from the brain is slow (Melega et al., 1989), complicating interpretation of k4 (Huang et al., 1991). Ideally, k4 should represent the rate of release of metabolites formed in striatal tissue. Whereas k4 ≠ 0, metabolite release appears limited by diffusion out of brain tissue.
Cerebellar and peripheral metabolites (rodents)
Chemical analysis of the radioactivity accumulated in striatal tissue with all radiofluorinated probes supports the premise that its time-dependent accumulation can be attributed to central dopaminergic biochemistry. We have also examined in rodents the origin of the nonspecific (cerebellum) activity with 6-FMT. From these data (Figs. 7 and 8) and the peripheral metabolism of 6-FDOPA, 4-FMT, and 6-FMT in monkeys (Table 2) and rodents (Fig. 4), several conclusions can be extracted. Under conditions (carbidopa, 5 mg/kg) that cause almost complete inhibition of peripheral AAAD in both nonhuman primates and rodents, as judged by L-DOPA and FDOPA arterial plasma metabolite compositions (Melega et al., 1990a), both 4-FMT and 6-FMT produced peripherally measurable amounts of decarboxylation products (and metabolites). This is consistent with the observation that o-and m-tyrosines are better substrates for AAAD than are the catechol counterparts (e.g., L-DOPA or FDOPA) (Borri-Voltattorni et al., 1983), and, therefore, even low amounts of active AAAD would produce some decarboxylation products. Also, monkey species have consistently showed high peripheral levels of AAAD (Rahman et al., 1981b), and complete inhibition of peripheral AAAD, even at plasma concentrations of carbidopa well above those known to cause complete inhibition in humans (Hoffman et al., 1992), is rarely achieved. However, caution should be exercised with the interpretation of the conversion rate constants of 4-FMT and 6-FMT in plasma (Huang et al., 1991) (Fig., 4), because these results may reflect partial decarboxylation as well as differences in whole body volumes of distribution and/or excretion kinetics of the radiolabeled probes.
The presence of the radiofluorinated m-tyramines in the circulation, however, raises the possibility of their transport across the BBB. Whereas catecholamines (e.g., dopamine) are known to be poorly transported from plasma to brain tissue (Hardebo and Owman, 1980), little reliable information exists on BBB permeability of m-tyramines. Tyramines, organic cations at physiological pH, may penetrate organic barriers by diffusion (Iseki et al., 1993), analogous to the transport of ammonia. Low, but measurable, amounts of 6-FMA were found in cerebellar tissue, where local metabolism of 6-FMT may seem improbable (Nakazato et al., 1992). It must be recognized, however, that AAAD immunoreactive cells have been observed in cerebellum of hamsters, rats, and mice (Baker et al., 1991), for which a priori cerebellar AAAD-mediated decarboxylation of highly sensitive substrates (e.g., 4-FMT and 6-FMT) cannot be discounted. In the absence of carbidopa, however, cerebellar 6-FMA concentrations are higher (Fig. 8C), suggestive of the blood-borne origin of cerebellar 6-FMA.
Since the relative rates of transport across the BBB should be dependent on relative plasma amine concentrations, it is clear that if any such penetration of plasma metabolites is to occur in the brain, it will be much lower in humans than in rodents or nonhuman primates. Variations in interspecies kinetics in the peripheral metabolism of radiopharmaceuticals has been recognized earlier (Barrio et al., 1989). These variations are related to body size, blood flow, and enzyme concentrations and distributions (Boxenbaum, 1982). In addition to all the physiological differences between species, AAAD activity in human serum is ∼1 pmol/min/ml, whereas it is 44.7 pmol/min/ml in monkey and 60.0 pmol/min/ml in rat (Rahman et al., 1981b). Therefore, a more efficient carbidopa inhibition may be expected in humans, considerably decreasing (or even eliminating) any background tissue activity due to plasma-borne amines (e.g., 4-FMA or 6-FM A). Comparative results with FDOPA in humans, monkeys, and rodents support this hypothesis (Melega et al., 1990a).
In part due to the complexities of its metabolism, which reduces its sensitivity, FDOPA has endured intense scrutiny. Earnest efforts have been long underway to simplify quantitation of data and reduce undesirable peripheral metabolism (Guttman et al., 1993). Two radiofluorinated L-m-tyrosines investigated in this work show significant promise. In particular, 6-FMT appears to follow the dopaminergic pathway with decarboxylation, amine vesicle transport, and metabolism in a manner similar to that found for FDOPA, but without its drawbacks. The promising initial results shown recently in humans with 6-FMT (Nahmias et al., 1995) seem to support this notion. Clearly, preliminary work with these radiofluorinated m-tyrosines (Melega et al., 1989) and the data presented in this work indicate that additional work with MPTP-lesioned animals is warranted. Clinical work with Parkinson's disease patients should follow.
Footnotes
Acknowledgment:
We wish to thank the staff of the Biomedical Cyclotron and Animal Tomograph Units for their assistance with this work. We also gratefully acknowledge the support of the Department of Energy Grant, DE-FC0387-ER60615, NIH Grant RO1-NS-33356, and donations from the Jennifer Jones Simon and Ahmanson Foundations for this research.
