Abstract
Near infrared spectroscopy (NIRS) was used to measure the changes in concentration of cerebral oxy-and deoxygenated haemoglobin ([HbO2] and [Hb]) in six healthy adult volunteers spontaneously breathing against increased expiratory pressures (IEPs) between 0 and 20 cm H20. During expiration, an increase in [HbO2] was recorded, accompanied by a smaller decrease in [Hb], producing a small increase in total cerebral haemoglobin concentration ([Hbsum]). The mean ± SD change in [Hbsum] at the maximum IEP of 20 cm H2O was 1.2 ± 0.7 μmol L−1 (equivalent to 1.4%). Changes in [Hbsum] correlated with IEP level (
Keywords
An important application of the technique of near infrared spectroscopy (NIRS) has been in the assessment of cerebral haemodynamics in adults (Elwell et al., 1994; Kirkpatrick et al., 1994). Since current NIRS instruments can measure quantified changes in oxy- and deoxyhaemoglobin concentration ([HbO2] and [Hb]) at a rate of 2 Hz, rapid changes in cerebral haemodynamics can now be investigated. Alternative monitoring methods such as positron emission tomography or Xe computed tomography can provide high spatial resolution but give little information about changes in [HbO2] and [Hb] over short time periods (<10 s). Transcranial Doppler provides rapid acquisition rates but is limited to measurement of blood flow velocity in selected vessels. Laser-Doppler flowmetry is rapid and has previously been used to study cerebellar cortical blood flow changes due to Valsalva manoeuvres. Correlation with changes in MABP has been shown (Wendling et al., 1994), but since surface tissues must be removed, the technique is inherently invasive.
The ventilatory-linked changes in MABP and cardiac output have been described (Weatherall et al., 1987), as has the cerebral protection mechanism of autoregulation (Kety, 1960; Lassen, 1964). In a previous NIRS study, respiratory-linked changes in [HbO2] and [Hb] were noted in subjects breathing a hypoxic gas mixture (Elwell et al., 1994). The purpose of this study was to use NIRS to investigate whether, in a group of normal adults, the cardiovascular changes known to occur with ventilation against an increased expiratory pressure (IEP) are reflected in the cerebral circulation and to determine if the changes are dependent upon the magnitude of the IEP.
THEORY OF NIRS
The theory of NIRS is described in detail elsewhere (Cope and Delpy, 1988; Cope, 1991). The technique uses a modified Beer–Lambert law to convert the measured changes in optical attenuation to quantified changes in the concentrations of Hb and HbO2 in micromoles per litre, with knowledge of the tissue differential pathlength factor (Delpy et al., 1988). Since the absolute concentration of chromophore is unknown, all measurements are expressed as absolute concentration changes from an arbitrary zero baseline at the start of the measurement period. In this article the term [Hbdiff] will be used to represent the difference between the [HbO2] and [Hb] concentrations and the term [Hbsum] will be used to represent their sum.
METHODS
The subjects for this study comprised six healthy adults (age range 25–44 years, median 27 years) with no known respiratory or cardiovascular disorders. This study was approved by the University College London Faculty of Clinical Science Committee on the Ethics of Clinical Investigation, and informed consent was obtained from the subjects before each investigation.
The optodes from an NIRO 500 spectrometer (Hamamatsu Photonics, Japan) were secured high on one side of the forehead 4–5 cm apart and shielded from ambient light. A sampling interval of 0.5 s was used, and the changes in [HbO2] and [Hb] were calculated with a previously established algorithm (Wray et al., 1988; Essenpreis et al., 1993) assuming an adult differential pathlength factor of 6.26 (Duncan et al., 1995).
MABP and heart rate (HR) were monitored continuously and noninvasively (Finapress 2300; Ohmeda) via a cuff placed on a finger that was kept stationary at the level of the heart. Transcutaneous carbon dioxide tension (TcPCO2) was monitored continuously using a transcutaneous blood gas electrode (Novametrix 850) placed on the upper arm. MABP, HR, and TcPCO2 data were continuously recorded and sampled synchronously with the NIRS data.
Procedure
All measurements were made with the subject in a supine position. Subjects breathed from a circuit in which the inspiratory arm was left open to room air while the expiratory arm was placed under water in a measuring cylinder to provide the required IEP (varying from 0 to 20 cm H2O in 5-cm increments). The subjects initially expired against atmospheric pressure, then against each progressively higher level of IEP for ∼2 min. Respiratory rate was not controlled.
Data analysis
All data, which were sampled at 0.5-s intervals, were smoothed by applying a rolling average over three points. After rejection of obvious movement artefact, three representative sections of data during expiration were selected from each level of IEP in all six subjects. Within these sections the positions of the maxima and minima of [HbO2] were defined and the magnitude and direction of the changes in [Hb], [HbO2], [Hbsum], and [Hbdiff] between those points were calculated. A time offset between 0.5 and 1.5 s was present between the MABP and NIRS data. This was accounted for in the calculation by defining the maxima and minima of the MABP signal independently from that of the NIRS data.
The magnitude and direction of the changes in MABP, HR, and TcPCO2 were then calculated (measured peak to peak). [HbO2], [Hb], [Hbsum], [Hbdiff], MABP, HR, and TcPCO2 were averaged over the three data sections for each level of IEP in all six subjects. [Hbsum] was then correlated with the level of IEP and MABP.
RESULTS
The NIRS, MABP, and HR data collected from one subject breathing against an IEP of 20 cm H2O are shown in Fig. 1 (left) with the data for a single breath (between the two vertical lines) shown on an expanded scale in Fig. 1 (right). The start of the traces coincides with the start of an expiration. On each expiration, MABP increases and HR decreases. This is accompanied by an increase in [HbO2] and a smaller decrease in [Hb], producing a small increase in [Hbsum]. These changes are reversed on inspiration. The mean change in TcPCO2 (0.2–0.34 kPa) was not related to the level of IEP.
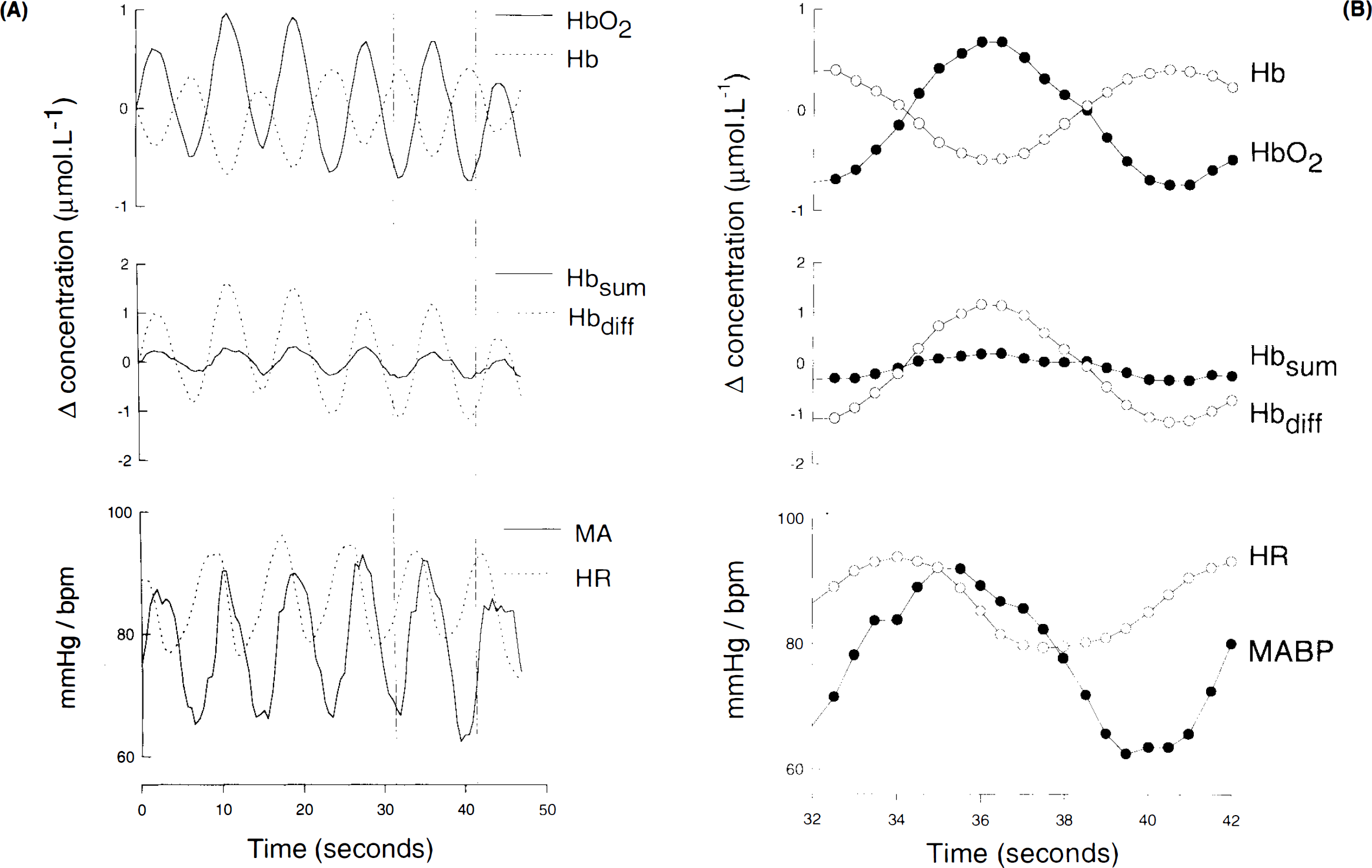
Near infrared spectroscopy, MABP, and heart rate (HR) data collected from one subject breathing against an increased expiratory pressure of 20 cm H2O over several breaths
Table 1 shows the magnitude and direction of the mean changes recorded in the six subjects during expiration. The regression of [Hbsum] (
Mean changes in NIRS, MABP, and HR data recorded during expiration at each level of IEP for all subjects (mean ± SD)
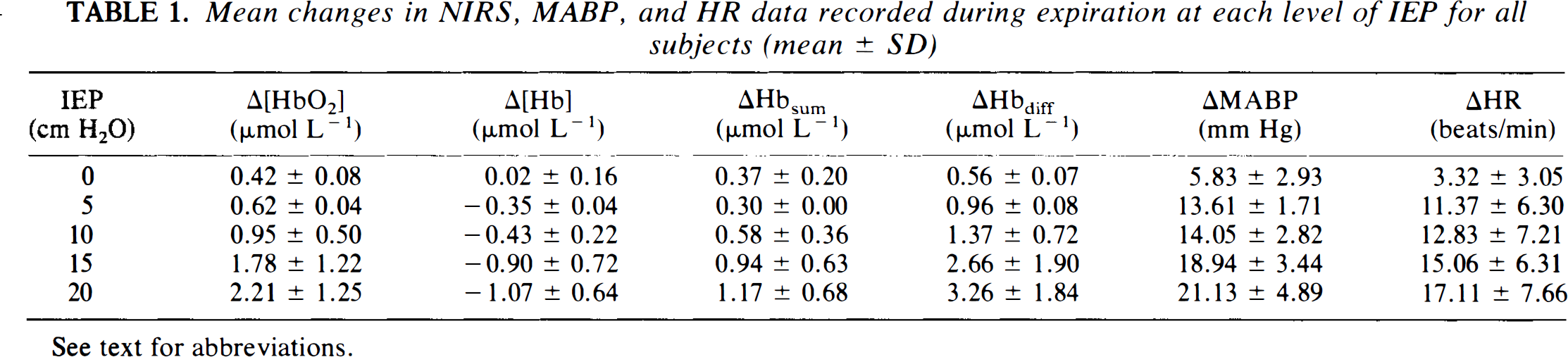
See text for abbreviations.
Assuming a cerebral haemoglobin volume (CHV) of 84 μmol L−1 (Sakai et al., 1985), the changes in [Hbsum] can be expressed as a percentage change in total CHV. The mean percentage change in CHV varied from 0.4% at 0 IEP to 1.4% at 20 cm H2O IEP (Table 2).
Percentage changes in CHV and MABP for each level of IEP (mean ± SD)
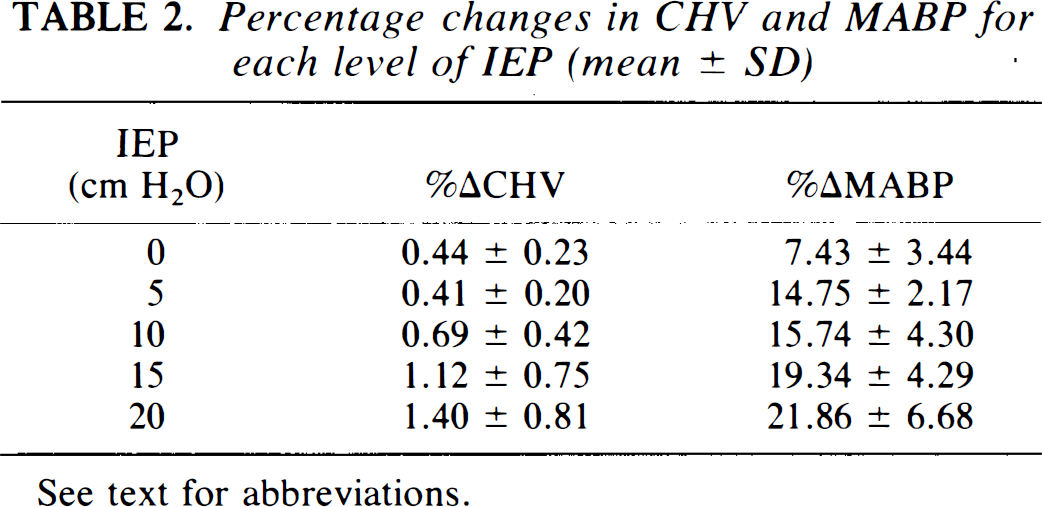
See text for abbreviations.
DISCUSSION
This article presents measurements of quantified changes in cerebral Hb and HbO2 during inspiration and expiration. In these normal, spontaneously breathing adults, CHV increased by up to 1.4% during expiration, the magnitude of the change in CHV being strongly correlated with the level of IEP.
When interpreting the results, it is necessary to first consider the theoretical problems associated with measuring small changes in [HbO2] and [Hb] in the adult head using NIRS. Since the changes in [HbO2] and [Hb] may come from any compartment in the illuminated tissue, the possible contribution of blood found in the scalp and skull must be considered. Recent NIRS studies in the field of functional imaging have demonstrated localised changes in cerebral haemoglobin (Hoshi and Tamura, 1993; Villringer et al., 1993). In a study performed during carotid endarterectomy, significant changes in [HbO2] and [Hb] were observed during clamping of the internal carotid artery but not during clamping of the external vessel (Kirkpatrick et al., 1994). These studies provide indirect evidence that current NIRS systems are capable of detecting changes in adult cerebral haemodynamics and that the contribution of surface tissues to the signals is small.
The effect of an overestimate of true cerebral optical pathlength on adult NIRS data has been considered elsewhere (Elwell et al., 1994). However, the conclusions drawn from the present study are predominantly concerned with the direction and relative magnitude of the changes in [Hb] and [HbO2] parameters rather than the absolute magnitude of their changes.
Although NIRS cannot separate the relative contribution of the arterial, capillary, and venous compartment to the global changes in the Hb signals, it is possible, from the data presented here, to make some deductions concerning (a) the changes in cerebral blood volume and CBF that occur during ventilation and (b) which compartments contribute to the change in the [Hbsum] signal.
The cardiovascular changes associated with expiration must first be considered. The term “pulsus paradoxus” is used to describe large changes in MABP and cardiac output seen in certain clinical situations during inspiration and expiration. These effects are accentuated in subjects breathing against an IEP, e.g., in asthmatic subjects or in patients undergoing positive pressure ventilation. On expiration, the rise in intrathoracic pressure produces an increase in central venous pressure and hence intracranial pressure, while the blood in the pulmonary circulation is squeezed into the left heart and the stroke volume increases. The HR does not entirely compensate and so cardiac output rises. Thus, during expiration, an increase in MABP and HR is expected that is reversed on inspiration. This effect is enhanced when an expiratory resistance is applied. These combined venous and arterial effects would be expected to increase cerebral blood volume by (a) distending the arterial part of the cerebral circulation and (b) impeding venous return and promoting venous distension. If the observed increase in [Hbsum] were due solely to distension of the vessels, then [HbO2] and [Hb] would change in the same direction. They do not, and hence, assuming that oxygen extraction is constant for this brief period, the results may be interpreted as an alteration in CBF.
A transient increase in CBF during expiration would also explain the decrease in [Hb]. A rise in CBF increases the delivery of arterial [HbO2] and at the same time effectively washes deoxygenated blood out of the venous compartment. Although [HbO2] and [Hb] can be expected to rise due to the increase in mean arterial and central venous pressure, the input pulse of increased MABP appears to be associated with a significant increase in CBF that exceeds any volume increase, producing a net decrease in [Hb]. This explanation is supported by correlation of the level of IEP with both [Hbsum] and MABP changes.
The time period over which these transient changes in CBF occurred depended upon the breathing rate of the subject; however, the mean time between the minimum and maximum MABP was 4 s. There are few data documenting the time response of global (i.e., nonmicrocirculatory) homeostatic control mechanisms in the cerebral circulation over such short time periods. Florence and Seylaz (1992) describe a study measuring the time course of CBF during rapid hypovolaemic hypotension. In the figures of this article, oscillations in CBF and MABP during baseline conditions can be seen that appear to be compatible with the respiratory-linked changes we have observed, although they are not commented upon in the text.
The changes in Hbsum observed are very small— on average, 1–2% of total blood volume; however, they are clearly reproducible and as such indicate the sensitivity of the NIRS technique for monitoring subtle changes in cerebral haemodynamics.
CONCLUSION
The increased temporal resolution now available with NIRS systems allows the near continuous monitoring of the cerebral haemodynamic effects of respiratory and cardiac changes during one breath. The data presented indicate that homeostatic mechanisms do not maintain CBF constant over short time periods (<10 s). The magnitude of the changes observed correlates strongly with the level of IEP.
Footnotes
Acknowledgment:
This work has been supported by grants from the United Kingdom Medical Research Council, the Wolfson Foundation, and Hamamatsu Photonics KK.
