Abstract
Regional cerebral muscarinic cholinergic receptor binding was quantified in normal young and elderly subjects employing the muscarinic antagonist radioligand [11C]tropanyl benzilate (TRB). Binding was determined by kinetic analyses of positron emission tomographic (PET) determinations of cerebral activity in conjunction with radial arterial blood sampling following intravenous radiotracer injection. A significant, but minor (8%), loss of frontal cortical receptors relative to whole brain average receptor density was found with advancing age. Parametric estimates of binding suggest small reductions in cerebral cortex binding as well as increases in brain stem and cerebellar binding underlying the observed pattern difference. However, these latter changes did not achieve statistical significance. We conclude that cerebral muscarinic receptor availability, as depicted by antagonist binding, does not undergo a major decline during normal aging of the adult human brain. The cerebral cortical cholinergic dysfunction in elderly subjects, suggested by prior clinical evidence, is not attributable to major loss of total muscarinic cholinoceptive capacity.
Central nervous system cholinergic function is believed to play an important role in memory and cognition (Longo, 1966; Drachman, 1977; Bartus et al., 1982; Sunderland et al., 1987; Molchan et al., 1992). Impaired presynaptic cholinergic activity is associated with cognitive impairment in Alzheimer's disease (Coyle et al., 1983; Whitehouse et al., 1988a) Parkinson's disease (Dubois et al., 1987; Whitehouse et al., 1988a), Down's syndrome (Yates et al., 1980), and progressive supranuclear palsy (Whitehouse et al., 1988b). These findings have led to rational therapeutic trials of direct muscarinic agonist and indirect cholinergic agonist treatments of Alzheimer's disease (Agnoli et al., 1983; Eagger et al., 1991) and progressive supranuclear palsy (Foster et al., 1989). The potential for success of these therapeutic approaches is, however, dependent on intact muscarinic cholinergic receptors.
Postmortem assays have revealed conflicting results regarding the integrity of muscarinic cholinergic receptors in these diseases as well as in normal aging (Whitehouse and Au, 1986; Gottfries, 1990). Studies of normal human brain have indicated either decreased (White et al., 1977; Rinne, 1987; Perry, 1980) or unchanged (Davies and Verth, 1977; Allen et al., 1983) muscarinic receptor binding. These discrepancies may be attributed to sources of variation, e.g., preterminal illness and nutrition, drug effects (Whitehouse and Au, 1986; Freund and Ballinger, 1989), postmortem delay to tissue procurement, and tissue storage prior to assay (Whitehouse et al., 1984; Cortés et al., 1987; Zilles et al., 1988).
Clarification of expressed levels of muscarinic cholinergic receptors in the course of normal aging is essential to the evaluation of the role of cholinergic neurotransmission and its alteration in age-associated conditions, including Alzheimer's disease, and may ultimately assist in the development of new therapies for cognitive disorders (Whitehouse and Au, 1986). In the present study, we quantitatively mapped the distribution of cerebral muscarinic receptors in vivo in neurologically normal young adult and elderly subjects with use of positron emission tomography (PET) and the antagonist radioligand [11C]tropanyl benzilate (TRB). Previously, we evaluated the tracer kinetic behavior of TRB in relation to estimation of muscarinic cholinergic receptors, our results indicating the feasibility of detecting normal or disease-related changes in human brain (Koeppe et al., 1994).
METHODS
Subjects
All studies were given prior approval by the University of Michigan Institutional Review Board and by the Subcommittee on Human Use of Radionuclides. Additionally, the use of [11C]TRB was under the auspices of an investigational new drug application, approved by the U.S. Food and Drug Administration. Written informed consent was obtained from all subjects prior to initiation of experimental procedures.
Six young (aged 24 ± 3 years, range, 20–28 years; four men and two women) and eight elderly (aged 67 ± 8 years, range, 55–76 years; five men and three women) normal subjects were studied. All subjects were free of neurologic, psychiatric, or serious medical illnesses on medical history, general physical, and neurologic examinations. Normal neuropsychometric testing results in the elderly group excluded the possibility of subclinical cognitive impairment. Evaluations performed included the Wechsler Adult Intelligence Scale-Revised (Wechsler, 1981), the Wide Range Achievement Test-Revised (Jastak, 1965), the Mini Mental State Examination (Folstein et al., 1975), the Boston Naming Test (Kaplan et al., 1978), Verbal Fluency (Lezak, 1983), the Wechsler Memory Scale-Russell Modification (Russell, 1975), the Rey-Osterreith Complex Figure Test (Kimura, 1984), and the Self-Rating(Berent et al., 1982). All subjects were free of any centrally-acting medications for a minimum period of 1 month prior to study. All were nonsmokers and all denied history of recreational drug use or excessive consumption of alcohol or caffeine.
Collection of brain and arterial tracer curves
[11CTRB was synthesized as previously described (Mulholland et al., 1992) from the desmethyl precursor and high specific activity [11CCH3I.
Scans were performed with a Siemens 931/08-12 tomograph (CTI Inc., Knoxville, TN, U.S.A), which simultaneously images 15 slices each separated by 6.75 mm (Spinks et al., 1988). Subjects were positioned supine on the scanner bed in a dimly-lit room with eyes and ears unoccluded. Antecubital vein and radial arterial cannulae were placed for injection of tracer and withdrawal of blood samples, respectively. Four-to-five fiduciary beads, labeled with 11C, were affixed to the forehead and retroauricular scalp regions of each subject at the beginning of each study.
Data acquisition commenced with bolus intravenous administration of 25–50 mCi of [11CTRB containing <10 μg tracer mass. Dynamic imaging over a 110 min interval was performed as follows: 2 × 30 s and 4 × 1, 2 × 2.5, 2 × 5, 7 × 10, and 1 × 20 min. Image data were reconstructed to an in-plane and axial spatial resolution of ∼8 mm full width at half maximum, using ellipse-calculated attenuation correction factors. Images were corrected for decay and acquisition time and then for subject motion during the image acquisition sequence. Motion correction was accomplished by noting the spatial positions of the fiduciary beads in the reference frame (50–60 min frame). Other frames in the dynamic imaging sequence were then realigned to this frame on the fiduciaries, resulting in realignments to within 0.5 mm (translational) and 1° (rotational), as determined from phantom studies (Koeppe, RA unpublished observations). These procedures resulted in a series of spatially-registered images depicting [11CTRB concentration within each scanned volume element.
Arterial plasma samples were obtained every 10 s for the first 2 min and then at 2.5, 3, 4, 5, 7.5, 10, 15, 20, 30, 45, 60, 90, and 110 min postinjeetion of [11CTRB. Plasma samples obtained at 1,2, and ≥3 min postinjeetion intervals were corrected for the presence of labeled metabolites using liquid chromatography, as previously described (Frey et al., 1992; Koeppe et al., 1994). 11C activities in total plasma samples and in chromatographic assays were determined in a sodium iodide well counter that was cross-calibrated to tomograph sensitivity.
Estimation of muscarinic receptor binding
Single, anatomically-configured midline regions of interest (ROIs) were drawn on the 50–60 min frame corresponding to the occipital (primary visual) cortex and pons. Bilateral ROIs were placed corresponding to the dorsal frontal (motor and premotor) and parietal (heteromodal association) cortices, cerebellum, caudate, putamen, and thalamus. In addition, whole brain activity was estimated from the area-weighted average of ROIs corresponding to the entire brain at each transaxial level. ROI templates were saved and subsequently applied to the entire temporal sequence of activity images (for kinetic analyses) and to pixel-by-pixel parametric images (see below).
Two previously-described methods were employed for determination of TRB binding to muscarinic receptors. The first was a pixel-by-pixel weighted integral estimation (Alpert et al., 1984) of TRB transport (K,) and binding [tissue distribution volume (DV)], calculated according to a two-parameter, two-compartment analysis of the cerebral and arterial tracer activity curves (Koeppe et al., 1994). This method provides images of both ligand delivery and binding; however, there is a systematic underestimation of binding, DV, in regions of highest receptor density. The second was a constrained, four-parameter, three-compartment kinetic analysis (Koeppe et al., 1994), which allows, more accurate but less precise binding estimates. The model in this instance includes parameters representing bidirectional exchange of tracer between blood and brain (K1 and k2′), receptor binding (k3′, equivalent to the kon × βmax product in in vitro assays) and dissociation (k4, equivalent to koff). Simultaneous estimation of the four-rate parameters was performed in the cerebellar ROI; however, this was not routinely feasible in regions of higher receptor binding due to difficulty in estimating k2′ and k4 in the presence of very rapid specific binding (high k3′). Therefore, it was assumed that the nonspecific and free ligand distribution volumes (specified by the K1/k2′ ratio) and the ligand dissociation rate from specific binding sites (k4) are identical across the examined gray matter ROIs. Thus, k4 was constrained to the population mean of all subjects' cerebellar values, and the K1/k2′ ratio was constrained to the value obtained from analysis of each subject's own cerebellar ROIs. Nonlinear least-squares curve fitting was then applied to the ROI activity curve sequences for estimation of K, and k3′.
Data analyses and statistics
Transport and binding data were averaged for the bi-hemispheric regions, providing a single estimate for each region per subject. Data from the pixel-by-pixel analyses were examined both in parametric values as well as expressed values relative to individual brain average, emphasizing more subtle pattern distinctions. Differences between groups were assessed with repeated measures analysis of variance (MANOVA), employing brain region as the repeated measure, at a significance threshold of p < 0.05. For instances in which significant between-group or group-by-region interaction effects were indicated, posthoc two-tailed Student's Mests were conducted. No formal correction for multiple parallel comparisons was employed; however, results of posthoc analyses were considered significant only at the p < 0.01 level. Additionally, linear regression analyses of cerebellar and frontal cerebral cortical binding estimates were conducted to investigate the possibility that between-group differences might be masked by age ranges within the study groups.
RESULTS
Two-compartment kinetic analyses
Parametric K1 and DV estimates did not differ between the young and elderly groups (Table 1) (F = 2.67, p = 0.15, and F = 2.42, p = 0.17, in analyses of region-by-group interactions in MANOVA of K1 and DV data, respectively). When data were expressed relative to whole brain averages, K1 was again comparable between the groups (F = 1.99, p = 0.21). Analysis of DV, however, indicated the possibility of an alteration in the pattern of muscarinic receptor binding (F = 5.14, p = 0.032). Subsequent regional analyses indicated a trend toward relatively increased binding in posterior fossa structures and decreased binding in frontal and occipital cortices with advancing age; only the reduction in the frontal cortex achieved statistical significance. Comparison of these data with untransformed parametric values suggested the presence of both neocortical and posterior fossa trends. However, the magnitude of the frontal cortex decline was only 12% (p = 0.105, not significant) in parametric estimates and 8% (p < 0.001) in binding expressed relative to the global mean.
Effect of age on the transport and binding of[11C]TRB in two-compartment kinetic analyses a
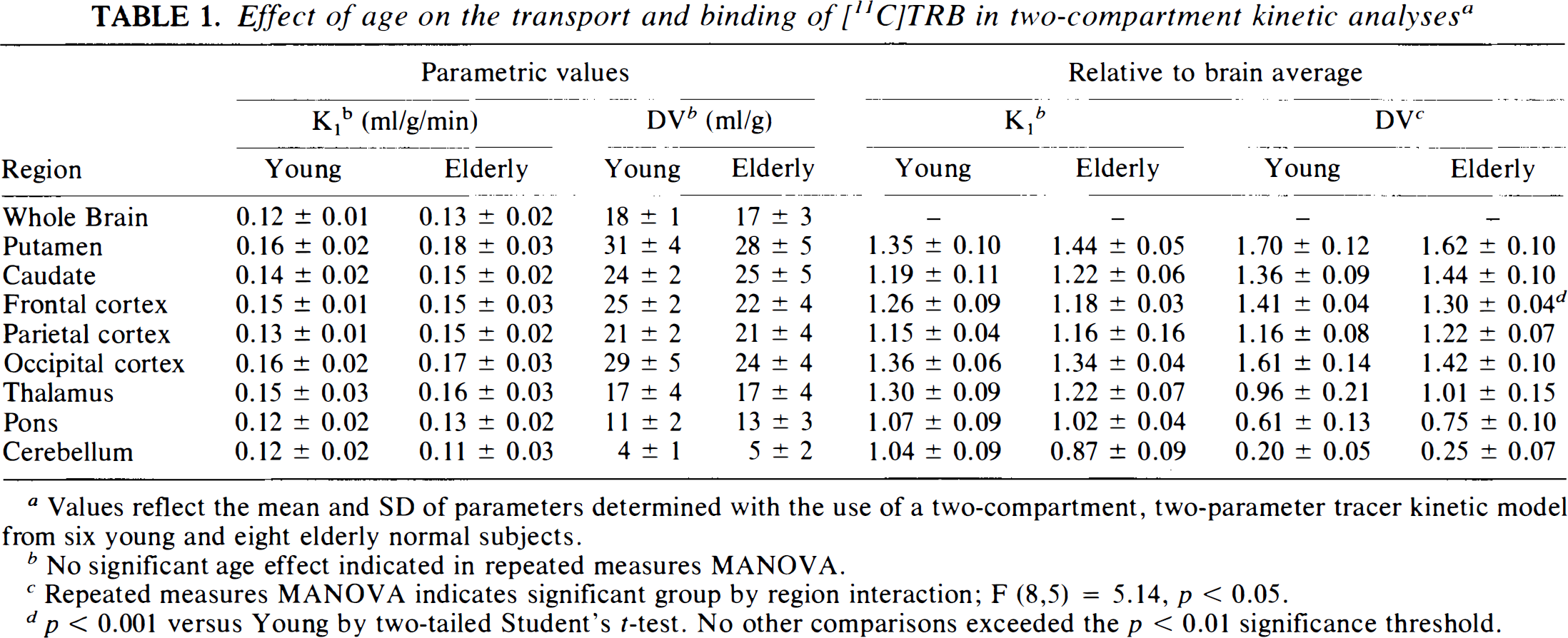
Values reflect the mean and SD of parameters determined with the use of a two-compartment, two-parameter tracer kinetic model from six young and eight elderly normal subjects.
No significant age effect indicated in repeated measures MANOVA.
Repeated measures MANOVA indicates significant group by region interaction; F (8,5) = 5.14, p < 0.05.
p < 0.001 versus Young by two-tailed Student's t-test. No other comparisons exceeded the p < 0.01 significance threshold.
Effect of age on estimates of [11C]TRB transport and binding using a constrained three-compartment tracer kinetic model a
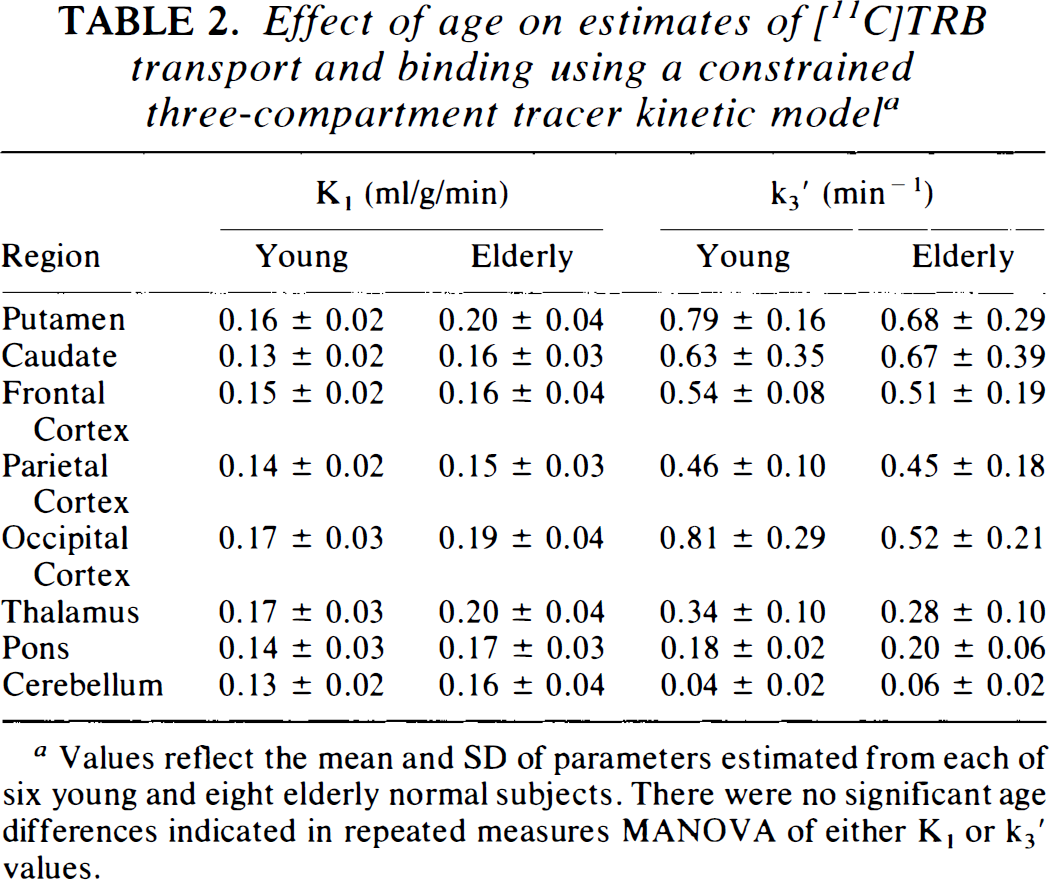
Values reflect the mean and SD of parameters estimated from each of six young and eight elderly normal subjects. There were no significant age differences indicated in repeated measures MANOVA of either K, or k3′ values.
Three-compartment kinetic analyses
Since the constrained three compartment binding estimates depend, in part, on the values assigned to the fixed parameters, potential intergroup differences in both the binding dissociation rate and in the distribution volume of nonspecific and free ligand were investigated in the cerebellum. There was no significant difference in the dissociation rate of specific TRB binding between groups (k4 estimates of 0.04 ± 0.03 min−1 and 0.03 ± 0.01 min−1 in young and elderly subjects, respectively; p = 0.29). The mean value of all subjects, 0.032 min−1, was employed in all subsequent fitting analyses. Cerebellar distribution volumes for free and nonspecific TRB (K1/k2′ ratio, estimated with k4 fixed to 0.032 min−1) were 2.5 ± 0.4 ml/g in young and 2.3 ± 1.3 ml/g in elderly subjects (p = 0.69).
Estimates of muscarinic receptor binding in receptor-rich regions from the constrained three-compartment and from the two-compartment pixel-by-pixel analyses were in good agreement; however, the more complex model estimates demonstrated greater variability (Fig. 1) (Koeppe et al., 1994). No significant differences were seen in TRB binding estimates (MANOVA revealed no significant k3′ region by group interaction; F = 2.70, p = 0.12).
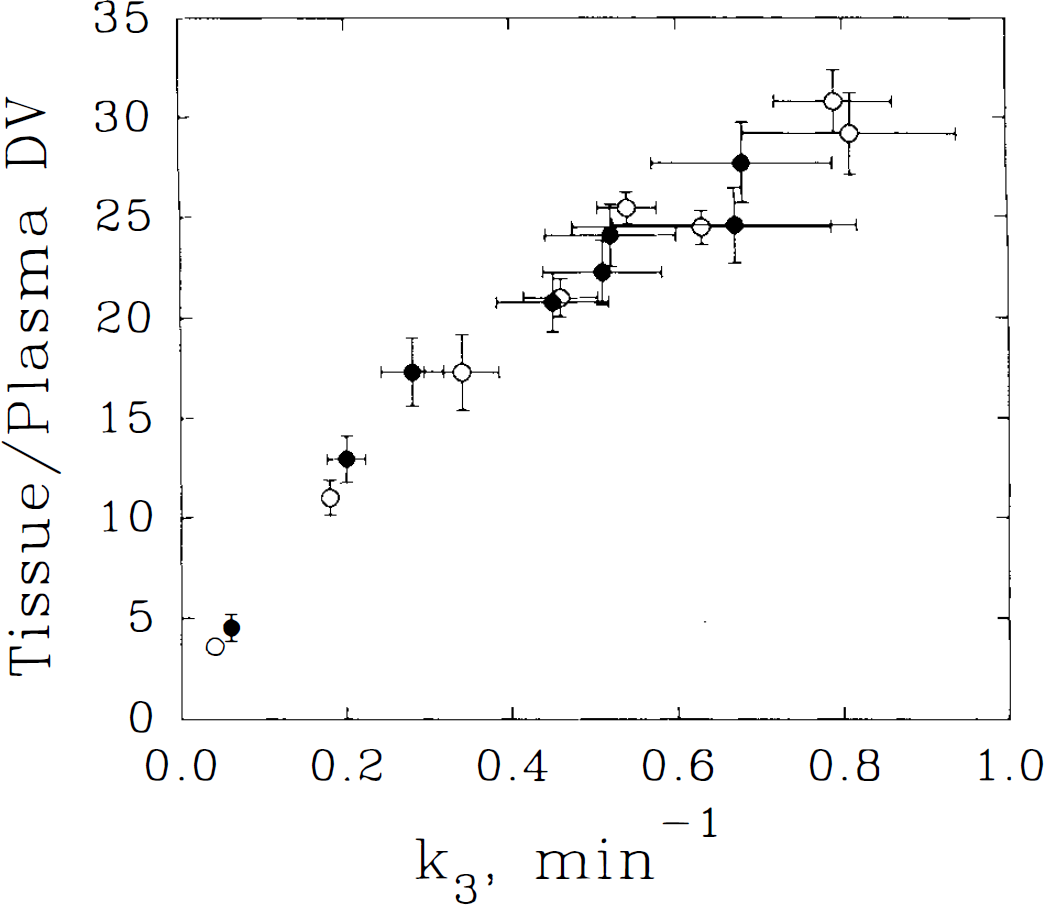
Comparison of the relationship between two different kinetic measures of muscarinic receptor density, two-model compartment distribution volume (DV) and three-compartment model k3, in young and elderly subject groups. Each point represents the mean ± SEM for a specific brain region (cerebellar cortex, pons, thalamus, parietal cortex, frontal cortex, occipital cortex, caudate, and putamen in ascending order for elderly subjects) in a group (young, ○, elderly, •). Using a quadratic regression model, the correlation between measures for these points was r = 0.99, p < 0.0001.
Linear regression analyses
The possibility that within-group variance due to age ranges might have masked detection of significant effect was investigated in the frontal cortex and cerebellum by linear regression analyses. In the frontal cortex (Fig. 2), declines of 0.19, 0.14, and 0.10%/year were indicated by DV, normalized DV, and k3′ binding estimates, respectively. Only the normalized DV data demonstrated a statistically-significant age relationship. Cerebellar binding analyses (Fig. 3) suggested increases of 0.96/(DV), 0.82/(normalized DV), and 0.84%/yr (k3′), none of which achieved statistical significance.
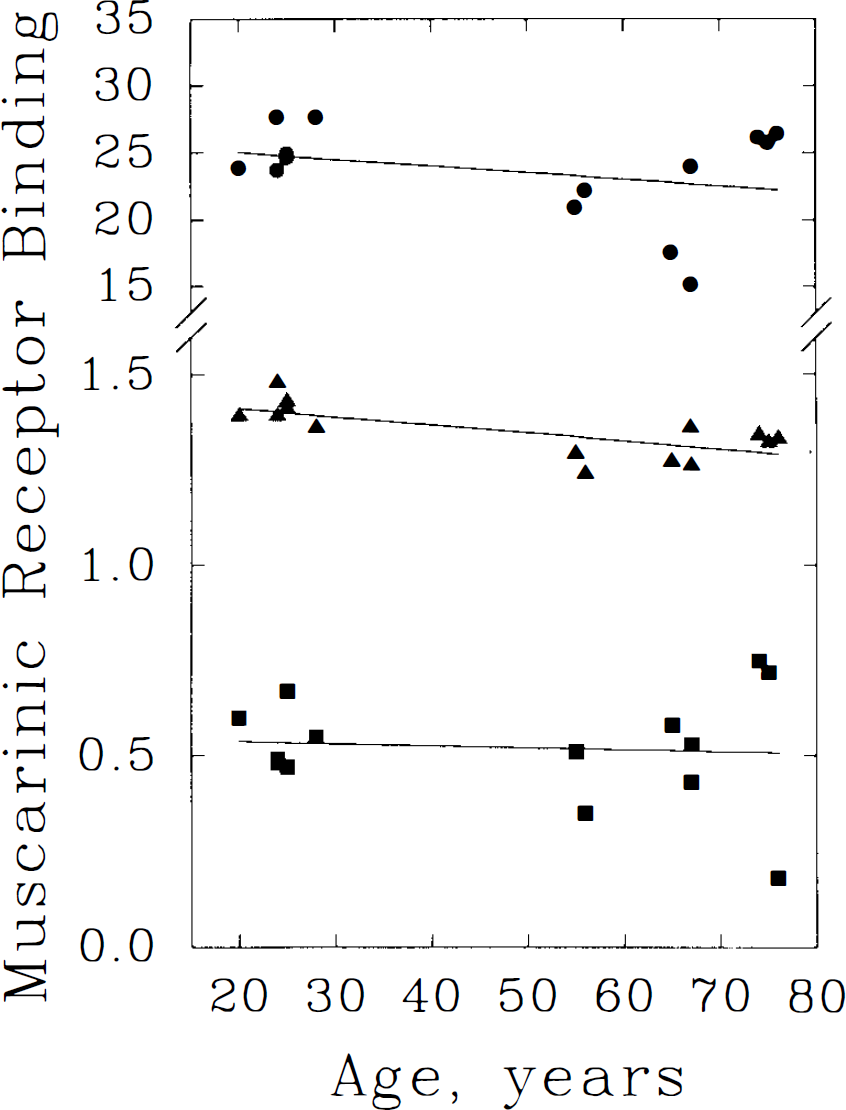
Linear regression analyses of frontal cortical [11C]TRB binding versus age. Three methods for estimation of TRB binding to muscarinic cholinergic receptors are depicted: the DV from a pixel-by-pixel analysis of a two-compartment model (•); DV expressed relative to whole brain average (DVnorm, ▴); and the rate of receptor binding (k3′, ▪) derived from regional kinetic analyses with a constrained, three-compartment kinetic model. All three methods suggest minor declines in binding with advancing age, however, statistical significance is demonstrated only for normalized DV data. Equations of the regression lines are: DV = 26.011 − 0.050 × age (r = 0.31, p = 0.29); DVnorm = 1.452 − 0.002 × age (r = 0.71, p = 0.005); and k3 = 0.548 −0. 00053 × age (r = 0.081, p = 0.78).
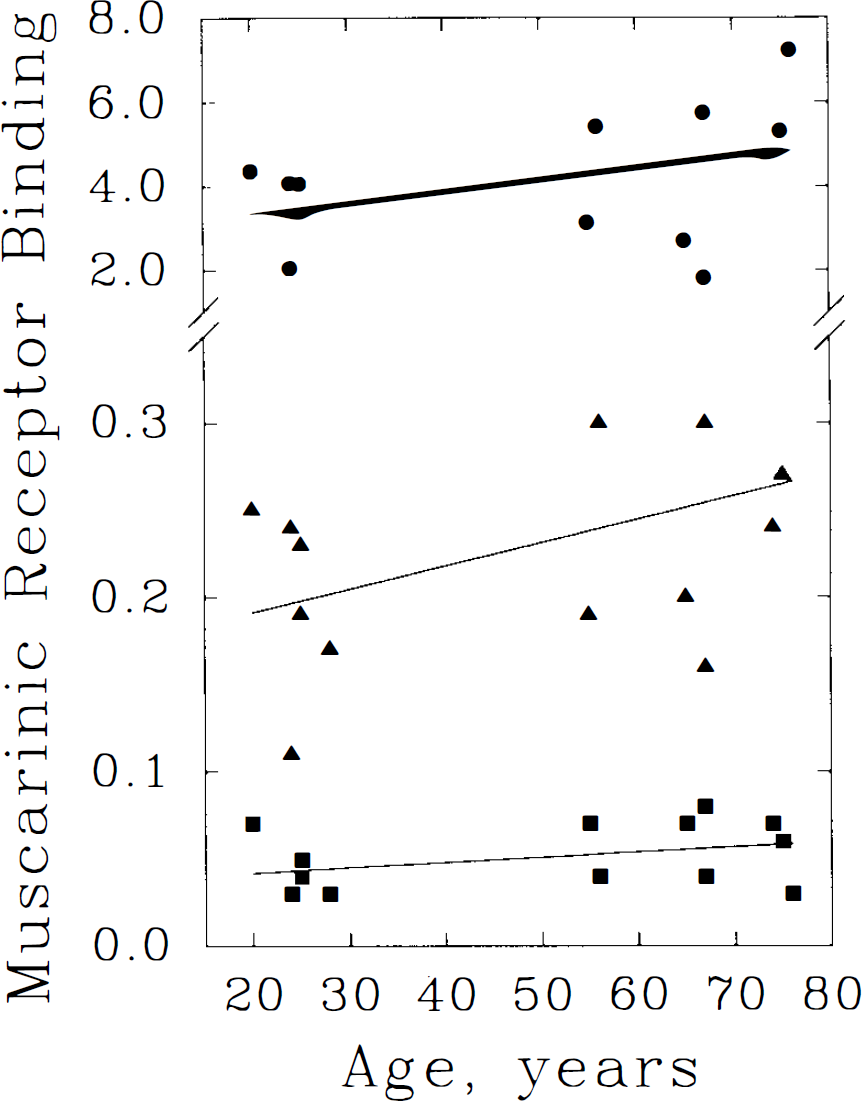
Linear regression analyses of cerebellar [11C]TRB binding versus age. The three methods for binding estimation and their symbolic depictions are given in the legend to Fig 2. Each method suggests an insignificant trend toward increased binding with advancing age. Equations of the regression lines are: DV = 2.804 + 0.027 × age (r = 0.40, p = 0.15); DVnorm = 0.164 + 0.0013 × age (r = 0.46, p = 0.10); and k3 = 0.036 + 0.00030 × age (r = 0.371, p = 0.19).
DISCUSSION
The primary finding in the present study is a lack of substantial change in the binding of the antagonist ligand [11C]TRB during normal aging in the adult human brain. Our current methods depict binding at a single, subsaturating ligand dose, permitting estimations only of the forward binding rate (proportional to Bmax × kon) or of the total tissue DV (proportional to Bmax × kd, but with an added component attributable to nonspecific binding and free ligand in tissue). There are numerous in vitro studies of aging effect on muscarinic receptors in rodent and primate brain (see below) that collectively indicate unchanged antagonist binding affinity (kd), but suggest apparently discrepant results regarding receptor density (Bmax). Thus, we interpret our findings and review the prior literature with the assumption that age-related antagonist binding changes reflect differences in receptor density. Apparent lack of agreement on the presence and magnitude of age-related changes in prior in vitro studies is resolved by considering the distinction between developmental changes that occur in advance of adulthood versus aging of the adult brain.
In vitro radioligand binding studies of mouse brain have indicated loss of receptors in the interval between the ages of 4 and 30 months (Freund, 1980; Waller and London, 1983). It has been further reported that aging effects are regional; there are ∼30% loss of striatal and 60% loss of neocortical muscarinic receptors, with unchanged or elevated receptor numbers in the cerebellum and hippocampal formation (Waller and London, 1983).
In the rat, atropine binding to synaptosomal fractions reveals an age-associated 50% decline in the cerebral cortex between ages 2 and 20 months, while cerebellar binding increases between 2 and 10 months and then declines to initial levels (James and Kanungo, 1976). Conversely, subsequent studies of Morin and Wasterlain (1980) failed to detect changes in total muscarinic receptor binding in the cerebral cortex, hippocampus, amygdala, or hypothalamus between the ages of 4 and 22 months. A significant striatal receptor decline of 30% and an insignificant trend toward receptor loss in the cerebellum (25% reduction) were observed. A subsequent study revealed modest (∼20–25%) declines in cerebral cortical, hippocampal, and striatal muscarinic receptors, with a 60% increase in the brain stem of male, but not female, brains between the ages of 3–4 and 27–30 months (Gurwitz et al., 1987). A more recent study that addressed aging effects on muscarinic receptors employed autoradiography of intact brain sections and detected minor changes in cerebral cortical (∼15% decreases) and unaltered hippocampal binding between the ages of 4 and 31 months (Biegon et al., 1988). Striatal changes were limited to an apparent 17% loss of receptors from the dorsomedial caudate putamen, while pontine and cerebellar binding were reduced by ∼30%. Thus, changes observed in several laboratories are in general agreement that minor reductions in cerebral cortical and striatal muscarinic receptor expression are observed in the aging adult rodent brain. Studies indicating larger decreases have included adolescent subjects for initial reference, suggesting that developmental, rather than senescence-related, changes predominate.
Binding results in Macaca mulatta are in agreement with rodent data. Wenk et al. (1989) found no significant decline in muscarinic Ml-type receptors in the frontopolar cerebral cortex of monkeys between the ages of 4 and 31 years. Wagster et al. (1990) reported reduction in total muscarinic binding in the temporal neocortex that is greatest between the ages of 2 and 6 years (15% decline) and least between 13 and 20 years (8% decline) of age. Subcortical regions were not assayed in these studies.
Postmortem examinations of human brain have yielded less consistent results than have the aforementioned nonhuman studies. Most investigations have been restricted to middle-aged and elderly adult subjects, and included individuals with a variety of preterminal illnesses and drug exposures. Several studies have suggested modest declines in cerebral cortical or hippocampal muscarinic receptors between the ages of 60 and 90 years (White et al., 1977; Perry, 1980; Nordberg and Winblad, 1981). Others, however, have not identified such changes (Davies and Verth, 1977; Allen et al., 1983). The series with the greatest age ranges and subject numbers (Rinne, 1987) indicates ≥50% reductions in muscarinic receptor numbers in the striatum, hippocampus, and frontal cortex between ages 4 and 93 years. Although the highest receptor densities were found in the eight subjects ≤20 years of age, age-associated declines of lesser magnitude were evident in the data from the adult subjects as well.
Recognizing the limitations inherent in human autopsy studies, two prior investigations employed in vivo neuroimaging techniques to estimate cerebral muscarinic receptor populations in human aging. The first study (Dewey et al., 1990) utilized the antagonist radioligand [1C]benztropine. Seven subjects between the ages of 19 and 82 years of age were evaluated employing arterial plasma tracer sampling with metabolite correction and dynamic PET imaging. Age-related reductions in tracer concentration were observed in all cerebral regions reported. Additional analyses of the total tissue distribution volumes of benztropine, expressed relative to the cerebellum, suggested a decline of 50% in all of the neocortical regions and in the striatum. No change was suggested in the thalamus. The second investigation of muscarinic receptor changes in aging human brain with PET (Suhara et al., 1993) made use of the more recently developed antagonist ligand [11C]N-methylpiperidyl benzilate (NMPB) (Mulholland et al., 1994) and analyzed the dynamic time-course of cerebral tracer activity, expressed in comparison with cerebellar activity as an assumed nonspecific and free tracer reference region. This analysis revealed apparent 45% decreases in binding in all neocortical regions, hippocampus, striatum, thalamus, and pons between ages 18 and 75 years.
In contrast to the prior PET studies, our present findings are in agreement with the majority of the cited in vitro and postmortem studies and do not confirm the presence of large, age-related reductions in cerebral muscarinic receptors. Rather, we observed minor, insignificant trends toward reduced frontal (12% decline) and occipital (17% decline) cortical and toward elevated cerebellar (25% increase) muscarinic binding as estimated by the untransformed DV of [1C]TRB. Directions and magnitudes of these trends were similar despite the application of two distinct analytic schemes, each with distinct relative strengths and biases (Koeppe et al., 1994). The combination of opposing cortical and posterior fossa changes may have led prior PET investigators, employing the cerebellum as a reference region, to conclude that the loss of cerebral muscarinic receptors was more substantial than that reported here. In fact, the ratios of our mean frontal to cerebellar TRB binding estimates would lead to estimated cortical reductions of 26–37% for the various analysis schemes, attributable largely to the cerebellar increase. Power analyses indicate that we have 92 and 73% probabilities of having identified 50% losses in cortical receptors in the present untransformed DV and k3 data at the p < 0.01 significance level, respectively. Thus, it is unlikely that undersampling or variability in our measures masked detection of changes as large as those previously suggested.
The present findings do not exclude changes of greater magnitude involving small subcortical brain regions or select subpopulations of muscarinic receptors. Several previous in vitro studies have suggested preferential losses of subcortical M2 muscarinic receptors, particularly those associated with basal forebrain cholinergic neurons (Biegon et al., 1988; 1989). The antagonist ligands TRB and NMPB identify cerebral muscarinic receptors as a homogeneous population, despite current knowledge that there are detectable contributions from distinct, molecularly-defined receptor subtypes designated m,-m4in most forebrain regions (Levey et al., 1991; 1994). At the present time, radioligands that adequately differentiate these subtypes are limited (Frey and Howland, 1992); those with partial selectivity do not readily enter the brain and are not amenable to in vivo applications. Thus, potential subtype-specific changes in small regions with low total receptor density cannot be directly investigated with current PET techniques.
We conclude that there is evidence for alteration in the regional pattern of muscarinic cholinergic receptor density in the human brain during normal aging. Examination of parametric binding data, however, indicates that the underlying loss of cerebral binding sites is likely to be minor and that there may be a more substantial increase in cerebellar receptors. This suggests that clinical evidence for a decline in cerebral cortical cholinergic function in aging may not be ascribed to simple loss of receptors and emphasizes the importance of presynaptic cholinergic assessments (Araujo et al., 1990; Stroessner-Johnson et al., 1992). Future studies employing concomitant measures of presynaptic cholinergic terminals (Kuhl et al., 1994) will be necessary for complete characterization of the aging cholinergic nervous system and its hypothesized role in dementing illnesses.
Footnotes
Acknowledgments:
This work was supported by grants designated P01 NS15655 and ROI NS24896 from the National Institutes of Health. The authors are indebted to the staff of both The University of Michigan Radiochemistry/Cyclotron Facility and the University Hospital PET Imaging Suite for invaluable assistance in performing these studies.
