Abstract
Nitric oxide (NO)-dependent regulation of brain blood flow has hitherto not been studied in reptiles. By observing the brain surface (telencephalon) of the freshwater turtle (Trachemys scripta) with epiillumination microscopy, we show that topical application of acetylcholine (ACh) induces an increase in CBF velocity that can be completely blocked by the NO synthase inhibitor NG-nitro-L-arginine methyl ester (
Acetylcholine (ACh) causes vasodilation of mammalian cerebral arteries, both when administered exogenously and when released from parasympathetic fibers innervating cerebral arteries and arterioles (Morita-Tsuzuki et al., 1993). The vasodilatory action of ACh is mediated by an endothelium-derived relaxing factor (EDRF), which is now generally accepted to be identical with nitric oxide (NO) or a closely related molecule (reviewed by Faraci, 1993).
Also, hypoxia causes increased brain blood flow in mammals, and the underlying mechanisms have been suggested to include NO production (Kozniewska et al., 1992; Burnstock, 1993). Moreover, NO is produced in the ischemic mammalian brain (Malinski et al., 1993; Sato et al., 1993), but the role of NO in ischemic brain damage is controversial, because it potentially may have both beneficial and detrimental effects (Choi, 1993).
Endogenous NO is synthesized from
However, very little is known about the role of NO in the regulation of brain blood flow in nonmammalian vertebrates, one exception being a recent study showing that ACh superfusion elevates brain blood flow velocity in crucian carp through a NO-dependent mechanism (Hylland and Nilsson, 1995).
With regard to the phylogeny and evolution of brain blood flow regulation, turtles are of particular interest because they represent the oldest living reptiles, the ancestors of modern turtles having deviated from other reptiles some 300 million years ago (Kardong, 1995). Nevertheless, the possible presence of NO-dependent brain blood flow regulation has not been studied in turtles. Still, few brains of ectothermic vertebrates have attracted as much attention as the brain of freshwater turtles of the genera Trachemys and Chrysemys (Lutz and Nilsson, 1994). This is because these vertebrates are among the very few that can tolerate prolonged anoxia, surviving days of anoxia at room temperature and several months at temperatures close to 0°C (Ultsch, 1989). Only the crucian carp shows a comparable degree of anoxia tolerance (Piironen and Holopainen, 1986).
It is likely that an exceptionally high demand is put on the ability of anoxia-tolerant vertebrates such as turtles to upregulate brain blood flow. The brain's high rate of energy consumption has to be met by increased anaerobic ATP production during anoxia, which is likely to require increased glucose delivery by the blood. Indeed, previous studies have shown a two- to threefold increase in CBF in turtles exposed to anoxia (Davies, 1989, 1991), and recent results suggest that adenosine is involved in the anoxia-induced elevation of turtle brain blood flow (Hylland et al., 1994). Still, it remains to be studied whether other mechanisms, such as NO-mediated vasodilation, also are involved in brain blood flow regulation in the anoxic turtle.
Thus, the aims of the present study were to find out whether ACh stimulates CBF in the freshwater turtle brain by a NO-dependent mechanism, and to examine the role of NO in blood flow regulation in anoxic turtles.
MATERIALS AND METHODS
Experiments were performed at 20 ± 1°C. Turtles (Trachemys scripta, weighing 350–500 g) were anesthetized (50 mg pentobarbital/kg i.p.), and ventilated with air via endotracheal intubation. A 5 × 5-mm area of the skull above the telencephalon was removed, and a small opening in the dura was made. The cortical surface was superfused (100 μl/min) with an unbuffered Ringer's solution (125 mMNaCl, 2.5 mM KCl, 1.3 mM CaCl2, 1.2 mM MgSO4) to avoid interference with endogenous pH changes. Nevertheless, the pH was adjusted to 7.6, which is normal for turtle CSF (Hitzig, 1982). The Ringer's solution had been equilibrated with air.
A video camera was attached to a Leitz Ortholux microscope equipped with a Leitz Ultropak epiilluminator and a water immersion objective (11 × magnification, giving 320× magnification on the video screen). This setup was used to observe and measure blood flow velocity (erythrocyte velocity) in venules on the dorsal cortical surface (where no arteries are seen in turtles), as described by Nilsson et al. (1994). At selected time points in the experiments, venule diameter was measured. Here, an objective with 22× magnification was used (giving 640× magnification on the video screen), and the diameter of the blood-filled lumen of the vessel was measured on the screen using a ruler. The mean diameter of the vessels studied was 61 ± 33 μm (range 13–117 μm). Blood pressure in the right subclavian artery was recorded on a Grass polygraph (representative traces are given in the figures).
Anoxia was achieved by N2 respiration. Drugs (from Sigma Chemical Co.) were dissolved in the Ringer's solution superfusing the brain.
Three to five venules were observed in each animal. Blood flow rates (microns per second) and vessel diameter (microns) were normalized to percentage values. The average rate or diameter observed in a vessel during the initial control period was set to 100%, whereupon an average for each animal was calculated. Changes were evaluated using one sample t test (two-tailed, null hypothesis = 100%), in which each animal (and not each vessel) was regarded as one observation.
RESULTS
When the brain surface was superfused with 10 μM of ACh during normoxia, the blood flow velocity reached 243 ± 60% (p = 0.006, Fig. 1A). This effect could be completely blocked by 10 μM of
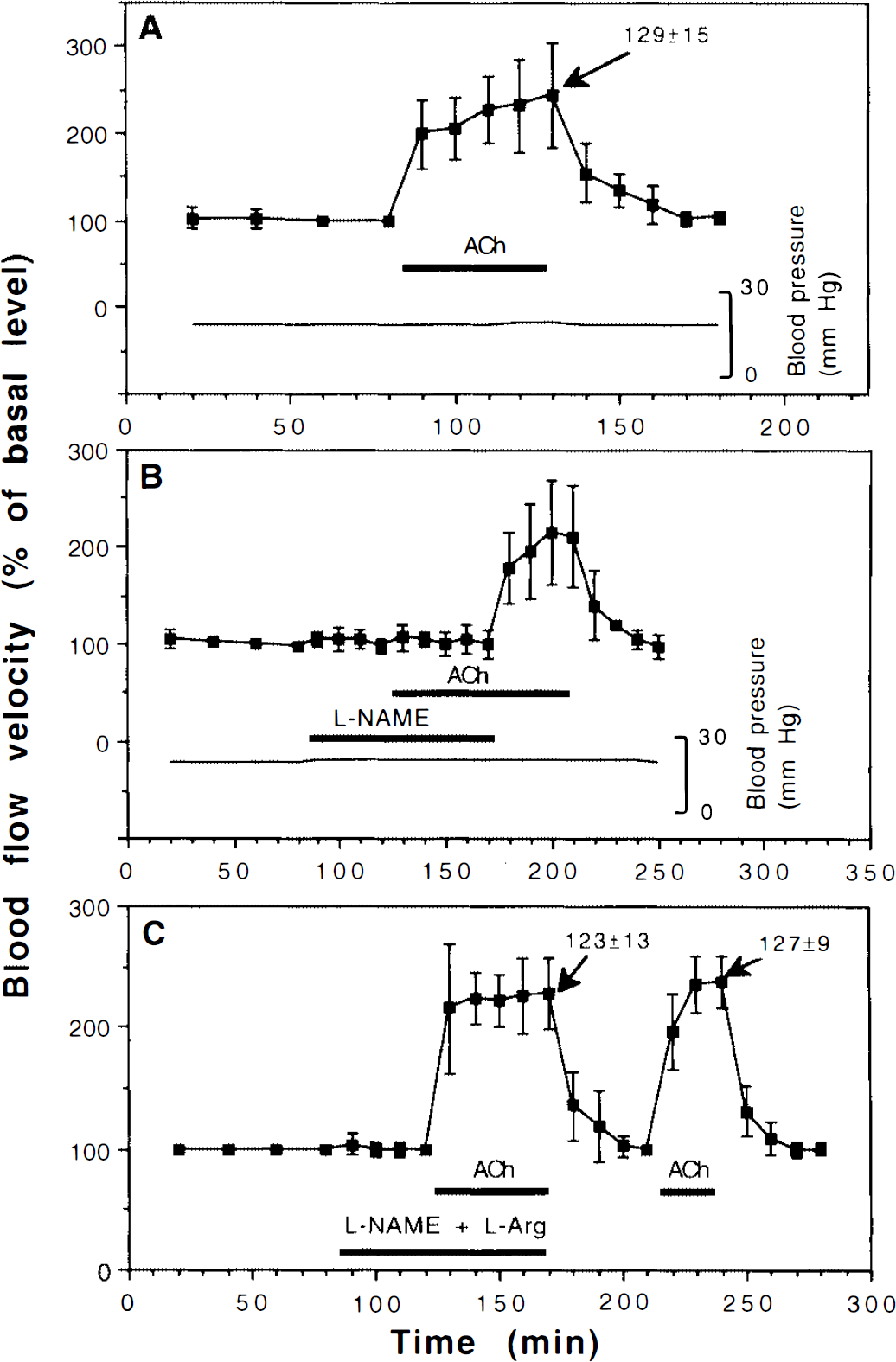
Effects of superfusing the brain with
Changes in the inner diameter (i.e., the blood-containing lumen) of the venules were recorded at the peak response to the treatments. The venule diameters (percent) are indicated by arrows in the figures (the preceding control period being set to 100%). Figure 1A shows that when the brain was superfused with 10 μM ACh during normoxia, the diameters of the venules reached 129 ± 15% (p = 0.0001). Assuming a circular lumen of the vessels, this increase in diameter indicates that the flow (volume per time) reached 404% during ACh superfusion. When the brain was simultaneously superfused with
The ability of
Superfusing the brain with 5 μM of sodium nitroprusside (SNP) also resulted in an increase in blood flow velocity, here reaching 252 ± 48% (p = 0.008, Fig. 2A). At the same time, SNP caused the venule diameter to increase to 118 ± 8% (p = 0.01, Fig. 2A), indicating that the flow reached 351% during SNP superfusion. The elevation of blood flow velocity caused by SNP could not be blocked by 10 μM of
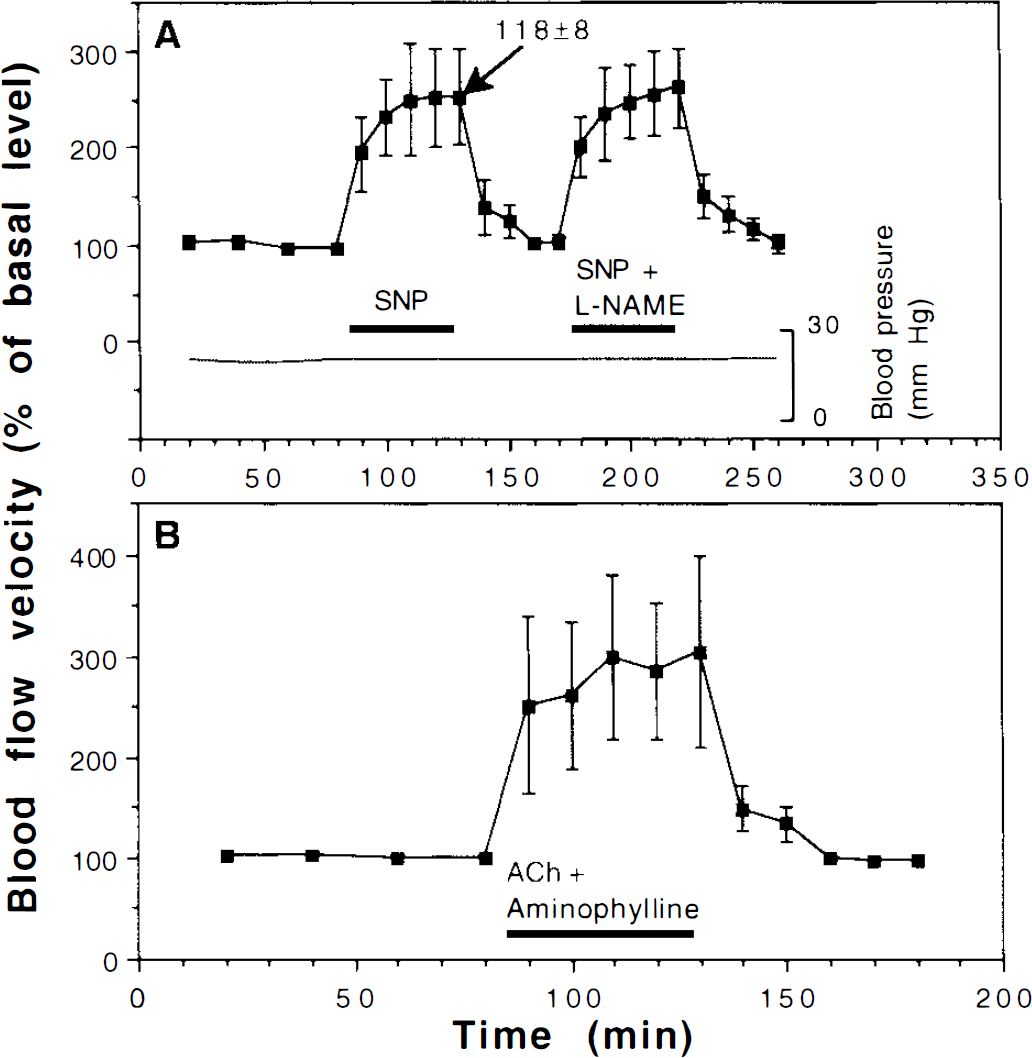
Effects of
Figure 2B shows that superfusing the brain with 2.5 mM of aminophylline (an antagonist of adenosine receptors) had no effect on the ability of ACh to elevate blood flow velocity (this time reaching 261 ± 62%), showing that the increased flow velocity seen during ACh superfusion was independent of adenosine release. Under the same conditions, aminophylline superfusion completely blocks the effects of both anoxia and adenosine on CBF velocity in the turtle (Hylland et al., 1994).
As in our previous study (Hylland et al., 1994), anoxia resulted in a temporary increase in blood flow velocity, lasting ∼1 h. Superfusing the brain with 10 μM
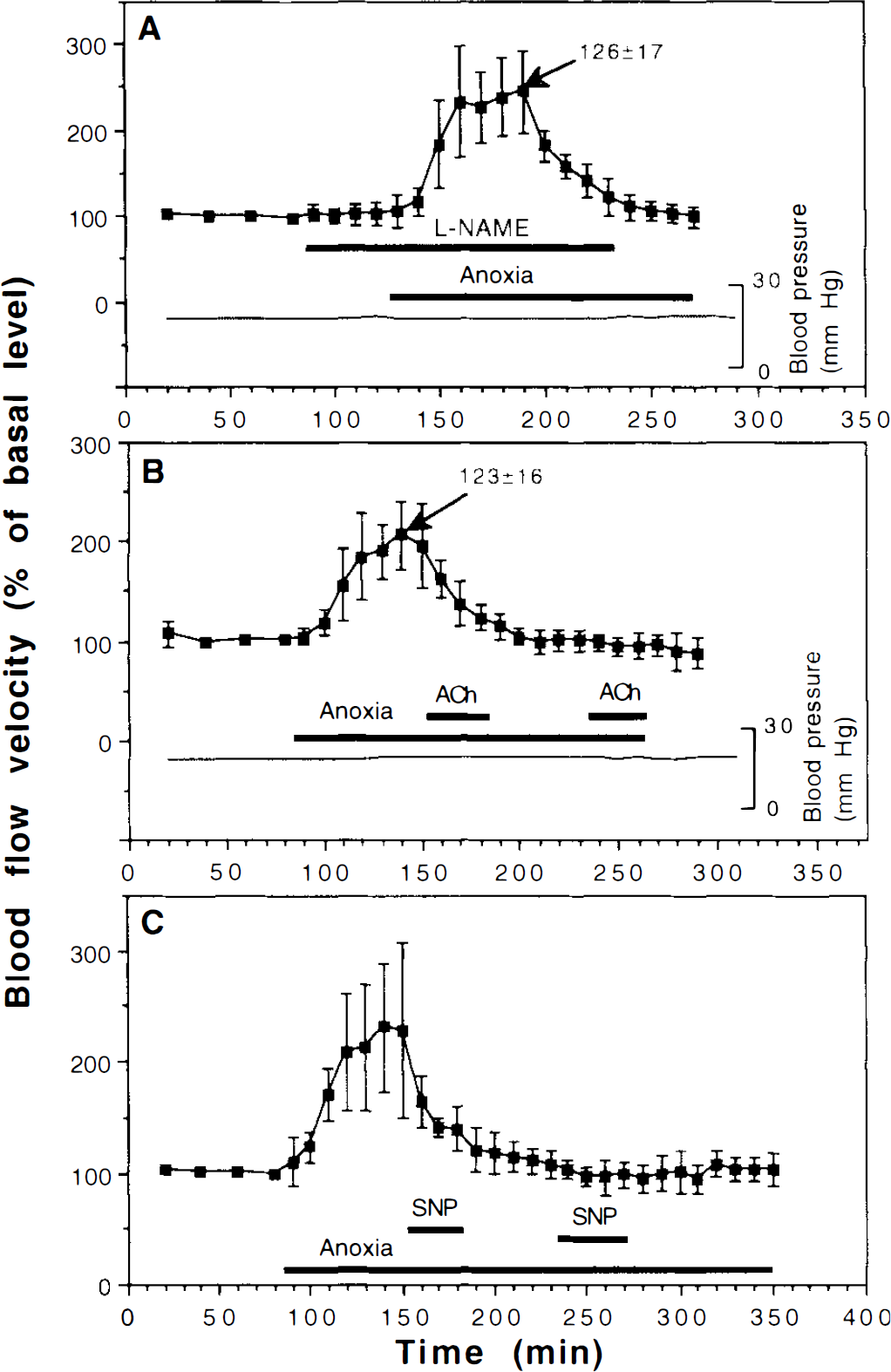
Effects of anoxia and
The experiments shown in Fig. 3B and C also revealed that superfusing the brain with 10 μM of ACh or 5 μM of SNP during anoxia induced no increase in blood flow velocity, not even if the blood flow velocity had fallen back to preanoxic values.
During anoxia the diameter of the venules increased to 123 ± 16% (p = 0.008, Fig. 3B), indicating that the flow reached 312%. Superfusing the brain with 10 μM
DISCUSSION
ACh and NO-dependent vasodilation
The present results show that the NOS inhibitor
Although the endothelium of cerebral arteries is one likely site of action for the ACh effect presently studied, NO-producing neurons or glial cells may also be involved. Because ACh is a neurotransmitter in reptile brain (Bissoli and Contestabile, 1988), it is possible that ACh superfusion produces NO release through neuronal activation and an increase in brain metabolic rate. A role for NO in the coupling between brain metabolism and blood flow has been demonstrated in mammals (Dirnagl et al. 1993). However, it deserves to be pointed out that adenosine is another known coupler of brain blood flow to metabolic rate in mammals (Morii et al., 1987), and that the present results with the adenosine receptor blocker aminophylline suggested that adenosine plays no role in the effect of ACh on blood flow velocity.
Interestingly,
Similar to our previous study on crucian carp (Hylland and Nilsson, 1995), the blocking effect of
It appears likely that the increase in blood flow velocity seen during superfusion with SNP was caused by the liberation of NO, SNP being well known to decompose to form NO in tissues (Bates et al., 1991). However, a vascular response to SNP is not in itself sufficient evidence for involvement of NO, because SNP may also decompose in tissues to form cyanide (Bates et al., 1991). Cyanide could indirectly stimulate increased blood flow by causing “chemical hypoxia.”
ACh-dependent vasodilation in tissues other than brain has been examined in some nonmammalian vertebrates. Thus, the isolated aorta of frog, cayman, and rooster dilates in response to ACh (Miller and Vanhoutte, 1986, 1992). In these studies, the role of NOS was not examined. However, more recently, Knight and Burnstock (1993) have used
From a phylogenetic point of view, birds can be considered to be reptiles, having deviated from other reptiles later than turtles did. Thus, it is interesting to note that a recent histological study on the Japanese quail (Panzica et al., 1994) has shown a wide distribution of neurons staining positively for NADPH-diaphorase (which at least in part may be identical with NOS).
Anoxia and NO-dependent vasodilation
Although the anoxia-induced increase in CBF velocity in the turtle can be fully blocked by the adenosine receptor antagonist, aminophylline (Hylland et al., 1994), blocking NO-synthase with
Administration of the NO donor SNP has been shown to cause an increase in CBF in ischemic mammalian brain (Zhang and Iadecola, 1993; Zhang et al., 1994). Superfusing the turtle brain with ACh or SNP during anoxia had no effect on blood flow velocity, even though the substances were administered after the initial temporary increase in brain blood flow had subsided, and the flow had returned to preanoxic levels. With regard to ACh, this is less surprising, because the NOS-catalyzed production of NO demands oxygen (Leone et al., 1991). However, the release of NO from SNP does not require oxygen, and it has actually been found to be blocked by high oxygen levels (Bates et al., 1991). Moreover, the lack of any effect of SNP during anoxia cannot be due to a general unresponsiveness of the vessels because, under identical experimental conditions and during the same anoxic period, adenosine superfusion more than doubles the blood flow velocity in the turtle brain (Hylland et al., 1994). Thus, it is possible that anoxia somehow makes blood vessels in the turtle brain unresponsive to NO. The mechanisms underlying this lack of responsiveness obviously deserve further experimental attention.
It should be pointed out that the open craniotomy used exposed the brain to the atmosphere, which may have increased the oxygen availability of the tissue studied. However, the profound effects that anoxia had on the brain blood flow suggest that such an effect was of minor importance.
There is a substantial increase in the NOS-catalyzed production of NO during ischemia in the mammalian brain (Malinski et al., 1993; Sato et al., 1993), and Burnstock (1993) made the suggestion that the initial vasodilation in response to hypoxia is mediated by NO. However, as mentioned in the introduction, whether the role of NO during ischemia in mammals is primarily detrimental or whether it may serve protective functions is a matter of apparent controversy. It may therefore be relevant to point out that the present results from turtles, as well as our previous study on crucian carp (Hylland and Nilsson, 1995), indicate that these well-adapted facultative anaerobes do not use NO as an endogenous vasodilator during anoxia. Actually, the present results indicate that the turtle brain vasculature becomes unresponsive to NO during anoxia.
Footnotes
Acknowledgment:
This study was financially supported by the Swedish Council for Forestry and Agricultural Research, the Swedish Natural Science Research Council, and Helge Ax:son Johnson Foundation, The Florida Atlantic University Foundation, and the Royal Swedish Academy of Sciences (The Hierta-Retzius Foundation).
