Abstract
Insulin-like growth factor I (IGF-I) is an anabolic pleiotrophic factor essential for postnatal rat brain development, especially during the first 21 days, the “critical growth period.” Cerebral hypoxic–ischemic insults occurring during the perinatal period can result in neuronal necrosis and permanent brain damage. To understand the regulation of the action of IGF-I in response to such a metabolic insult, we investigated the gene expression of IGF-I, type I IGF receptor, IGF binding protein (IGFBP) 2, and IGFBP5 during the first 72 h after hypoxia–ischemiain the immature rat. At 1 h of recovery, messenger RNA (mRNA) levels of all IGF system components were decreased throughout the hemisphere ipsilateral to the carotid artery ligation. This decrease is more pronounced at 24 h of recovery, especially in areas vulnerable to hypoxic–ischemic injury, such as the thalamus and hippocampus. At 72 h of recovery, although IGFBP2 and type 1 IGF receptor mRNA levels remain suppressed, gene expression of both IGF-I and IGFBP5 was activated in reactive astrocytes. Therefore, during the critical growth period in rats, the transcriptional levels of all IGF system components are extremely sensitive to metabolic perturbations associated with cerebral hypoxia–ischemia. The immediate decrease in IGF-I gene expression may be partially responsible for the impending neuronal death and selective vulnerability of myelinogenesis during the perinatal period.
The insulin-like growth factor (IGF) system consists of two biologically active polypeptides (IGF-I and IGF-II), two receptors (type I and type II IGF receptors), and six binding proteins (IGFBP 1–6). The developmental regulation of IGF-I gene expression supports its functional role during the “critical growth period,” which occurs perinatally in humans and during the first 3 postnatal weeks in rats (Davison and Dobbing, 1968). This critical growth period is characterized by accelerated dendritic growth, synaptic formation, and myelination of the axons, all of which are critical for brain cytoarchitecture. Any disturbance in the cellular metabolism may lead to structural and functional defects, resulting in permanent brain damage. It is during this period that IGF-I messenger RNA (mRNA) is expressed in large projection neurons, reaching maximum levels about postnatal day 12–14 (Bondy, 1991). In addition, the type I IGF receptor (Bondy et al., 1992), IGF binding protein 2 (IGFBP2), and IGFBP5 (Bondy and Lee, 1993a) are all expressed at the highest levels coordinately with IGF-I during this growth period, suggesting an autocrine/paracrine mode of action for IGF-I.
Although the functional importance of this space-and time-specific expression of IGF system genes is not well understood, cell culture studies revealed the nonspecific trophic actions of IGF-I on all cultured brain cells. For example, IGF-I acts as a mitogen to central cholinergic and dopaminergic neurons (Knusel et al., 1990), regulates the mitotic cycle in cultured rat sympathetic neuroblasts (DiCicco-Bloom and Black, 1988), and potentiates PMA-induced differentiation of SH-SY5Y neuroblastoma cells (Påhlman et al., 1991). In addition, both in vivo and in vitro evidence supports a neuroprotective role for IGF-I. For example, IGF-I protects neurons against hypoglycemia in vitro (Cheng and Mattson, 1992) and against hypoxic–ischemic injury in vivo (Gluckman et al., 1992; Guan et al., 1993). Taken together, the nondiscriminative trophic and neuroprotective effects, the ubiquitous presence of type I IGF receptor on all brain cells (Bondy, 1991), and the developmental regulation of IGF system gene expression strongly support an important functional role for IGF-I in brain development, especially during the critical growth period.
Perinatal hypoxia–ischemia remains the leading cause of severe brain damage in newborn infants (Mulligan et al., 1980; Hill and Volpe, 1981; Bjerre et al., 1983; Gray et al., 1993; Pasternak, 1993). The combination of an asphyxial insult and impaired blood flow can deprive the young brain of oxygen and other essential nutrients (Low et al., 1989). If not corrected in time, cellular oxidative phosphorylation will be severely depressed, which will lead to neuronal damage and death (Silverstein et al., 1984; Volpe et al., 1985; Yager et al., 1992; Vannucci, 1993). Although both the neuropathologic (Rice et al., 1981; Towfighi et al., 1991) and cerebral metabolic (Palmer et al., 1990; Vannucci et al., 1994) effects of hypoxia–ischemia have been described in the immature rat, the effect of hypoxia–ischemia on IGF-I gene expression has not been investigated during the critical growth period, when its normal level of expression may be critical for brain development. Previous studies, using postnatal day 21 (P21) rats, showed a hypoxia–ischemia-induced increase in IGF-I expression in reactive astrocytes at 3 days after insult (Gluckman et al., 1992). Because of the anabolic activity of IGF-I and its importance during the critical growth period (i.e., before P21), we investigated the gene expression of IGF-I and other IGF system components in immature rat brains after hypoxia–ischemia. Neonatal rats at 7 days (P7) after birth were used because the maturity of their brains is roughly comparable to that of a 32–34 weeks' gestation human fetus or newborn infant (Davison and Dobbing, 1968; Rice et al., 1981). To understand the impact of metabolic insult on IGF-I gene expression during the critical growth period, we investigated the cellular distribution of mRNAs for IGF-I, type I IGF receptor, IGFBP2, and IGFBP5 in the perinatal period during the first 72 h after hypoxia–ischemia.
MATERIALS AND METHODS
Perinatal hypoxia–ischemia model
Pregnant Wistar rats (Charles River) were housed in individual cages and fed standard laboratory chow ad libitum. Offspring were delivered vaginally, and litter size was adjusted to 10 pups/litter on the day of delivery and remained with the dam until the day of experimentation. Hypoxia-ischemia was induced in 7-day-old rat pups as previously described (Rice et al., 1981), with minor modifications. Before exposure to hypoxia (8% O2/92% N2), pups were placed in the 37°C water bath for 20 min followed by 2.5 h of hypoxic exposure. Preliminary studies demonstrated that this period of prewarming produced a more uniform degree of damage in littermates but required a reduction in the hypoxic exposure to ensure survival. Unilateral common carotid artery ligation combined with 8% oxygen produces brain damage predominantly in the territory of the middle cerebral artery of the cerebral hemisphere ipsilateral to the carotid artery occlusion (Rice et al., 1981). Damage does not usually occur in the contralateral cerebral hemisphere or in either hemisphere when the animal is exposed to arterial ligation or hypoxia alone. All rat pups were then returned to their dams for 1, 3, 5, 24, or 72 h, at which times they were killed by decapitation, and the brains were removed and frozen in isopentane at −40°C, and stored at −70°C until analyzed.
In situ hybridization
In situ hybridization histochemistry was used to examine the transcription of exogenous IGF-I gene and the expression of type I IGF receptor, IGFBP2, and IGFBP5. Rat IGF-I, type I IGF receptor, IGFBP2, and IGFBP5 chromosomal DNA (cDNA) clones were used for the synthesis of cRNA probes, and each has been described in detail previously (Zhou and Bondy, 1993). The cRNA probes were synthesized in 10 μl reaction mixture containing 100 μCi each 35S-cytidine triphosphate (CTP) and 35S-uridine triphosphate (UTP; Amersham), 10 mM NaCl, 6 mM MgCl2, 40 mM Tris (pH 7.5), 2 mM spermidine, 10 mM dithiothreitol (DTT), 500 μM each unlabeled adenosine triphosphate (ATP) and guanosine triphosphate (GTP), 25 μM each unlabeled UTP and CTP, 1 μg linearized template, 15 units of the appropriate polymerase (BRL) and 15 units RNasin (Promega). The reaction was incubated at 42°C for 60 min, after which the DNA template was removed by digestion with DNase I. Average specific activity of probes generated by this protocol was 2–3 × 108 dpm/μg.
Before hybridization, tissue sections were warmed to 25°C, fixed in 4% formaldehyde, and acetylated in 0.25% acetic anhydride/0.1 M triethanolamine hydrochloride/0.9% NaCl. Tissues were dehydrated through an ethanol gradient, delipidated in chloroform, rehydrated, and air-dried. The 35S-labeled cRNA probes were added to fresh hybridization buffer (107 cpms/ml), which is composed of 50% formamide, 0.3 M NaCl, 20 mM Tris HCl (pH 8), 5 mM EDTA, 500 μg transfer RNA (tRNA)/ml, 10% dextran sulfate, 10 mM DTT, and 0.02% each of bovine serum albumin (BSA), ficoll, and polyvinylpyrolidone. Hybridization buffer was added to sections, which were then covered with glass coverslips and placed in humidified chambers overnight (14 h) at 55°C. Slides were then washed several times in 4 × standard saline citrate (SSC) to remove coverslips and hybridization buffer, and then dehydrated and immersed in 0.3 M NaCl, 50% formamide, 20 mMTris HCl, and 1 mM EDTA at 60°C for 15 min. Next sections were treated with RNase A (20 μg/ml, Boehringer Mannheim, Indianapolis, IN, U.S.A.) for 30 min at 37°C, and then passed through graded salt solutions, followed by a 15-min wash in 0.1 × SSC at 50°C. Slides were dehydrated and air-dried and apposed to Hyperfilm-beta Max (Amersham) for 2–5 days and then dipped in Kodak NTB2 nuclear emulsion, stored with desiccant at 4°C for 6–15 days, developed, and stained with cresyl violet or hematoxylin and eosin for microscopic evaluation.
Immunocytochemistry
Immunocytochemistry was used to identify reactive astrocytes in the injured neonatal brains. A biotin-avidin-horseradish peroxidase method was used, employing a monoclonal antibody against glial fibrillary acidic protein (GFAP; Boehringer Mannheim). The immunocytochemistry was performed on fresh frozen tissue sections (16 μm thickness) adjacent to those used for in situ hybridization studies (Lee, Clemens, et al., 1992). After fixing in Zamboni fixative and blocking in 10% normal goat serum, tissue sections were incubated with a 1:5 dilution of GFAP antibody as described. Thereafter, tissue sections were treated with biotinylated horse anti-mouse serum (1:200) for 2 h followed by a 45-min incubation with steptavidin–peroxidase conjugate. The antigen–antibody complex was visualized by incubation with 3,3′-diaminobenzidin (DAB).
RESULTS
During the first 72 h of recovery from hypoxia–ischemia, there was a dramatic change in gene transcription levels for IGF-I, type I IGF receptor, IGFBP2, and IGFBP5 in neonatal rat brains (n = 3) at each time point evaluated (Fig. 1). Normally, these IGF gene components are expressed at the highest levels in the relay stations of the somatosensory and cerebellar systems (Bondy, 1991; Bondy et al., 1992; Lee et al., 1992; Bondy and Lee, 1993a). As early as 1 h after hypoxia–ischemia, a panoramic decrease in the expression of all IGF system components was seen in the whole hemisphere ipsilateral to the ligated carotid artery (Fig. 1, left column). The decrease was more pronounced at 3 and 5 h (data not shown) and most striking at 24 h of recovery (middle column). Within the ipsilateral hemisphere, this early decrease was more pronounced in the thalamus, hippocampus, and cortex. By 72 h of recovery, however, the cellular distribution of mRNAs differed among IGF-I, IGFBP2, IGFBP5, and the type I IGF receptor. Whereas IGFBP2 and type I IGF receptor mRNA levels remained decreased, there was a parallel increase in IGF-I and IGFBP5 mRNA levels in the ipsilateral hemisphere. Compared to the levels of expression observed at 24 h of recovery, the increase was more pronounced in the thalamus for IGF-I and in the hippocampus and amygdala for both IGF-I and IGFBP5 (Fig. 1, right column). However, the cellular pattern of the enhanced IGF-I and IGFBP5 gene expression was different from that in the contralateral hemisphere. Whereas their mRNA levels remained subnormal in the ventral basal thalamus, they were much higher in the rest of the thalamic region, as well as in the hippocampus and amygdala, without any nuclear pattern. By comparison, throughout the first 72 h of recovery, the cellular pattern and levels of gene expression in the contralateral hemisphere remained normal for all IGF system components examined, except for an increase in IGF-I and IGFBP5 mRNA levels in the parasagittal cortex.
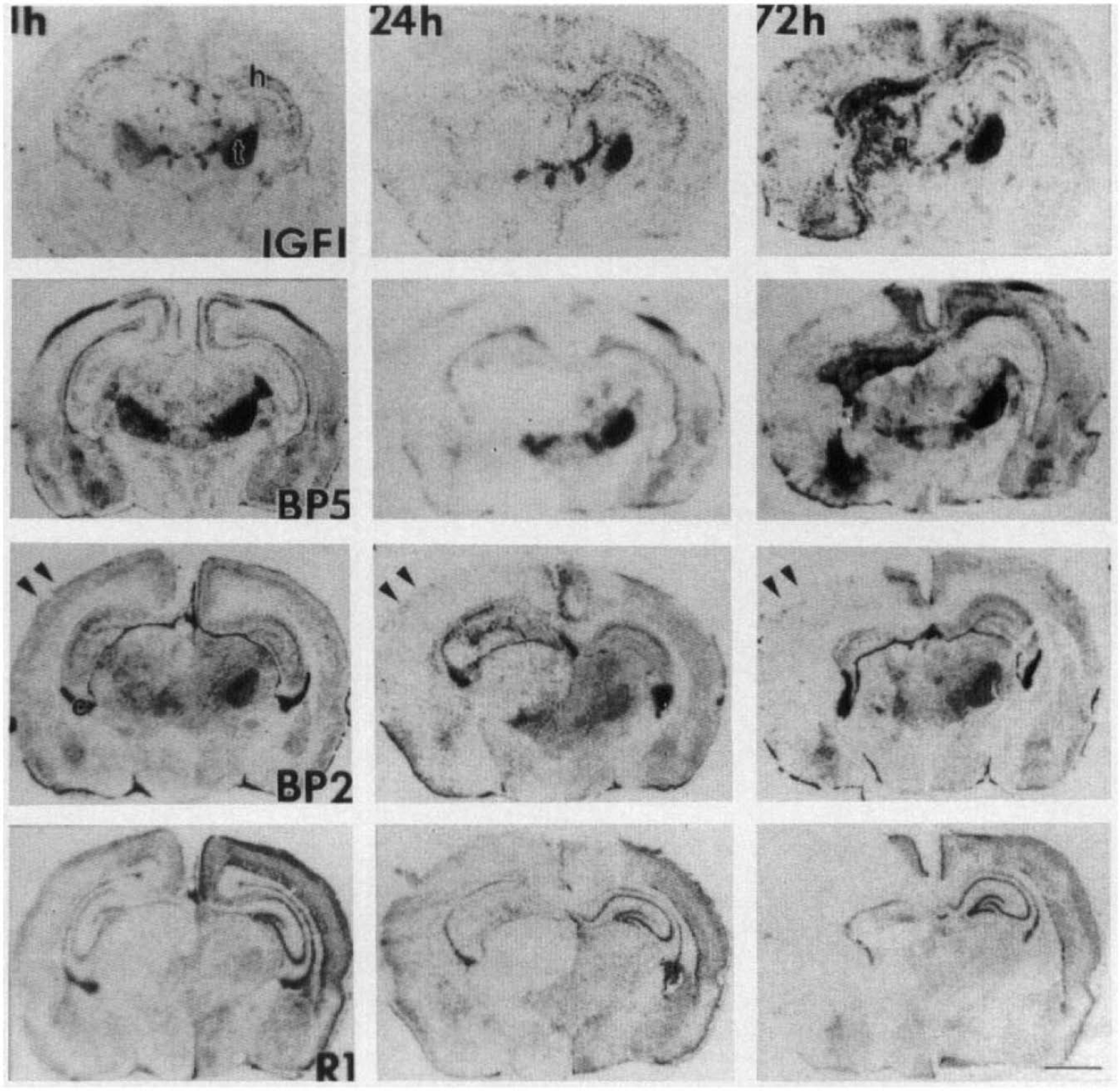
Time course and distribution of insulin-like growth factor (IGF) system gene expression during the first 72 h after hypoxia–ischemia in perinatal rat brain. Neonatal rat brains were obtained at 1, 24, and 72 h (three brains in each group) after the hypoxia–ischemia. Fresh frozen tissue sections were hybridized to IGF-I (first row), IGFBP5 (second row), IGFBP2 (third row) or type I IGF receptor (R1, fourth row) chromosomal RNA (cRNA) probes, processed, and exposed to autoradiographic film in the same experiment. The messenger RNA (mRNA) levels for all IGF system components were decreased in the ipsilateral hemisphere as early as 1 h, with the most pronounced reduction in the ventral basal thalamus and hippocampus. The decrease was more pronounced at 24 h. At 72 h of recovery, the cellular levels and distribution of IGF-I and IGFBP5 mRNA were different from those of IGFBP2 and R1. Whereas IGFBP2 and R1 mRNA levels remained suppressed, IGF-I and IGFBP5 mRNA levels were increased in the thalamus, hippocampus, and cortical regions. Bar = 2 mm.
To understand the potential action of IGF-I at the cellular level during the first 72 h after hypoxia–ischemia, we examined IGF-I and IGFBP5 mRNA cellular distribution in the ventral posterior thalamus (VPL) nuclei. There, IGF-I (Fig. 2A) and IGFBP5 (Fig. 2E) mRNA are normally colocalized in large projection neurons, as seen in the contralateral VPL, whereas IGFBP2 mRNA is in astrocytes (Lee, Javedan, et al., 1992). By 1 h of recovery, IGF-I and IGFBP5 mRNA levels decreased progressively in these large projection neurons (Fig. 2B and F). By 24 h, the neuronal expression of IGF-I and IGFBP5 was completely resolved, whereas there were apparent signs of neuronal death, as evidenced by chromolysis and pyknotic nuclei (Fig. 2C and G). At 72 h, there was an increase in IGF-I and IGFBP5 mRNA levels in the ipsilateral VPL, but this appeared to be in nonneuronal cells (Fig. 2D and H). Because most neurons are gone at this stage and a similar distribution of GFAP-positive cells was seen on adjacent sections, these small, nondescriptive cells were dispersed in the ipsilateral VPL and were most likely to be reactive astrocytes (Figs. 3B, 4A). The distribution of reactive astrocytes resembled that of the cell population expressing the IGF-I gene (Figs. 3A, 4B) as well as that for IGFBP5 (Figs. 3C, 4C) and IGFBP2 (Figs. 3D, 4E and F), but less pronounced for the latter two.
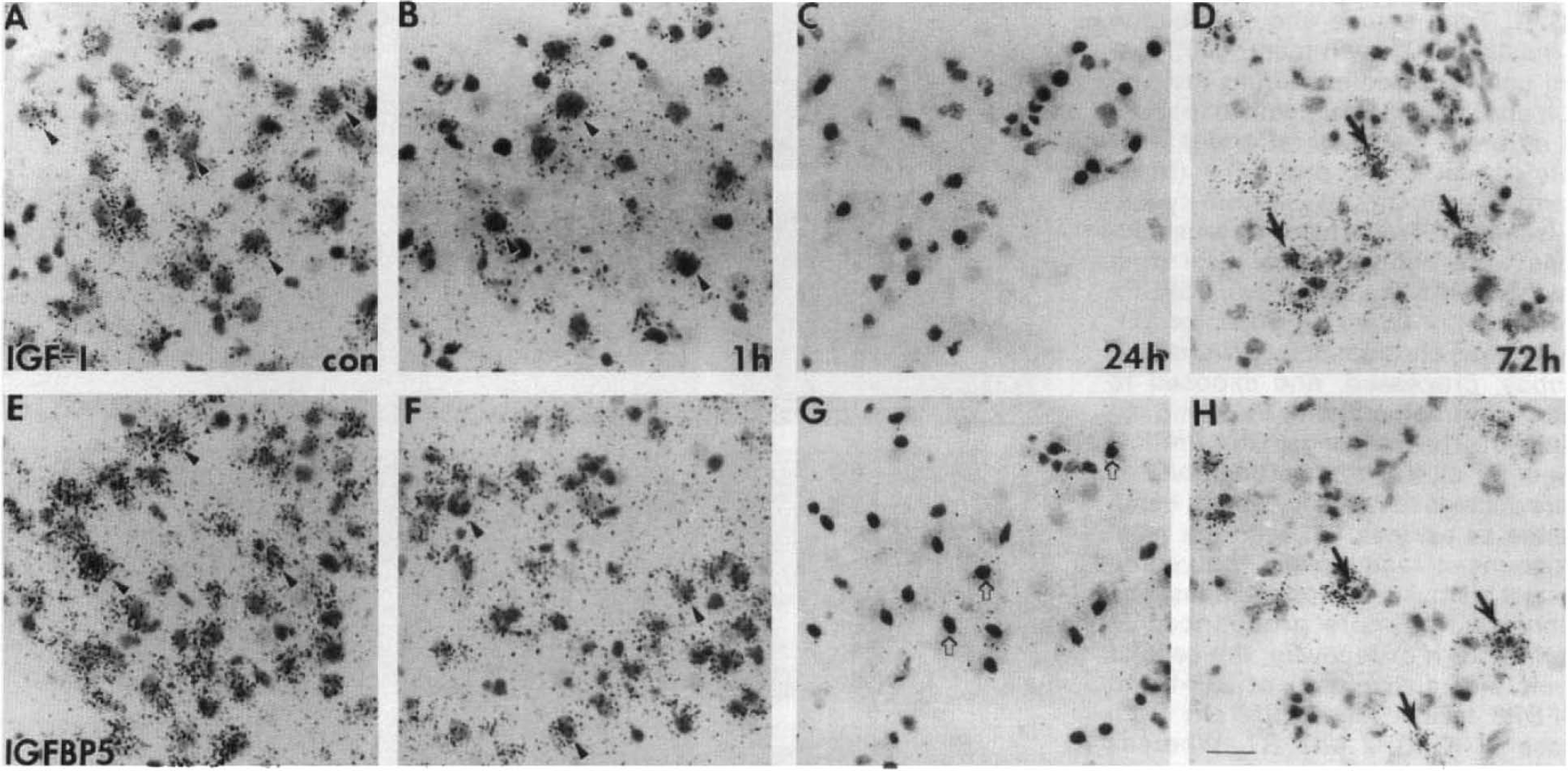
Cellular pattern of insulin-like growth factor I (IGF-I) and IGFBP5 messenger RNA (mRNA) during the first 72 h after hypoxia–ischemia-induced brain injury in neonatal rats. High magnification micrographs were taken from the ventral posterior thalamus, ipsilateral
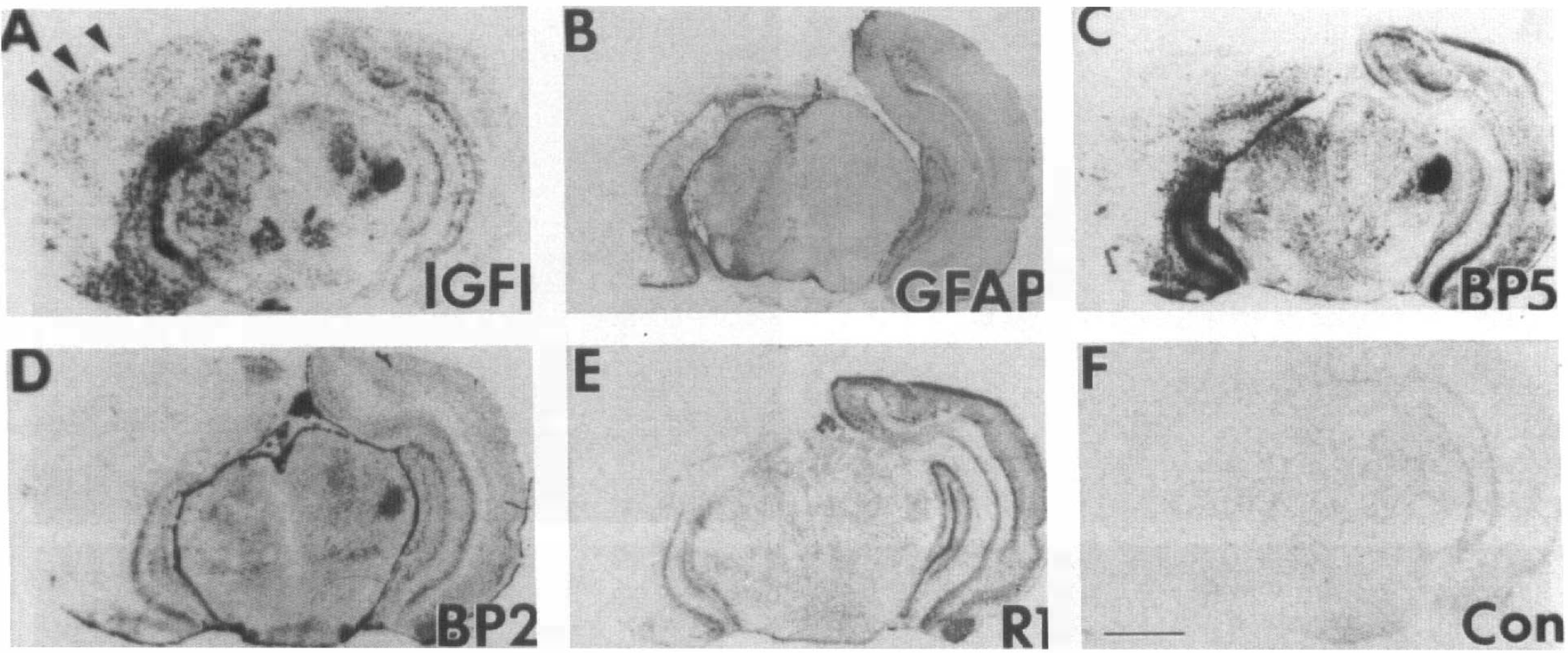
Distribution of insulin-like growth factor I (IGF-I), IGFBP2, IGFBP5, and type I IGF receptor messenger RNAs (mRNAs) and glial fibrillary acidic protein (GFAP)-positive astrocytes. The distribution of IGF-I
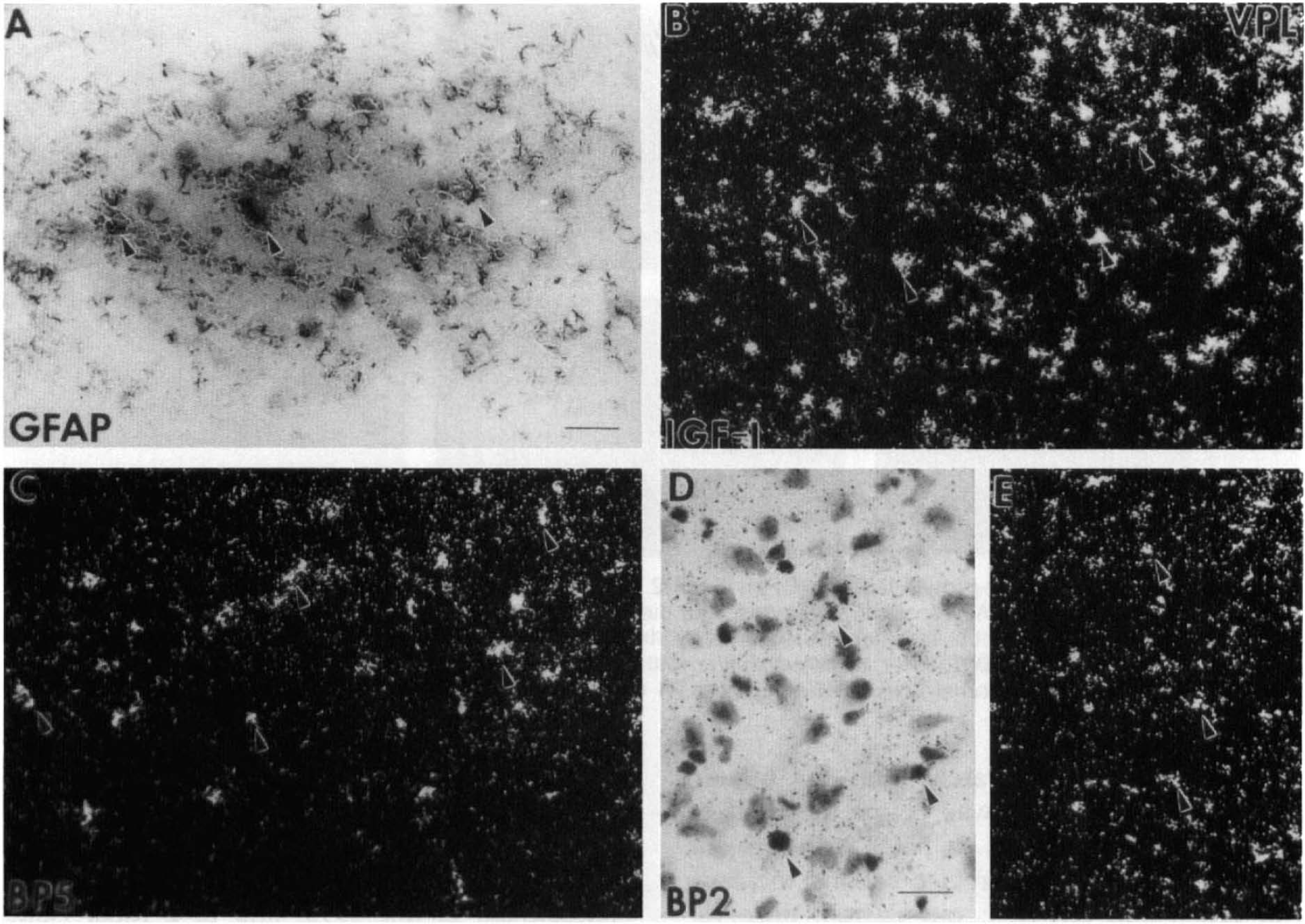
Cellular distribution of insulin-like growth factor I (IGF-I), IGFBP5, and IGFBP2 messenger RNA (mRNA) and glial fibrillary acidic protein (GFAP)-positive astrocytes in the ventral posterior lateral thalamus (VPL) at 72 h of recovery. Reactive astrocytes in the VPL are identified by GFAP-positive immunostaining (arrowheads in
A similar distribution of IGF-I and IGFBP5 mRNA and GFAP-positive reactive astrocytes was also found in the hippocampus (Fig. 5) and amygdala (Fig. 6). There IGF-I mRNA was expressed at a high level (Figs. 5A, 6A) in some of the reactive astrocytes present at 72 h after ischemia (Figs. 5D, 6D). IGFBP5 mRNA was expressed at a lower level in most of the same population of reactive astrocytes in these two areas (Figs. 5B, 6B). In contrast, IGFBP2 mRNA was below the limit of detection in both areas (Figs. 5C, 6C). Although our data cannot establish the precise cellular localization of IGF-I and IGFBP5, our current observations, also supported by previous studies, agree with the coexpression of IGF-I and IGFBP5 in reactive astrocytes. Future study using combined immunocytochemistry and in situ hybridization will be needed to determine the colocalization.
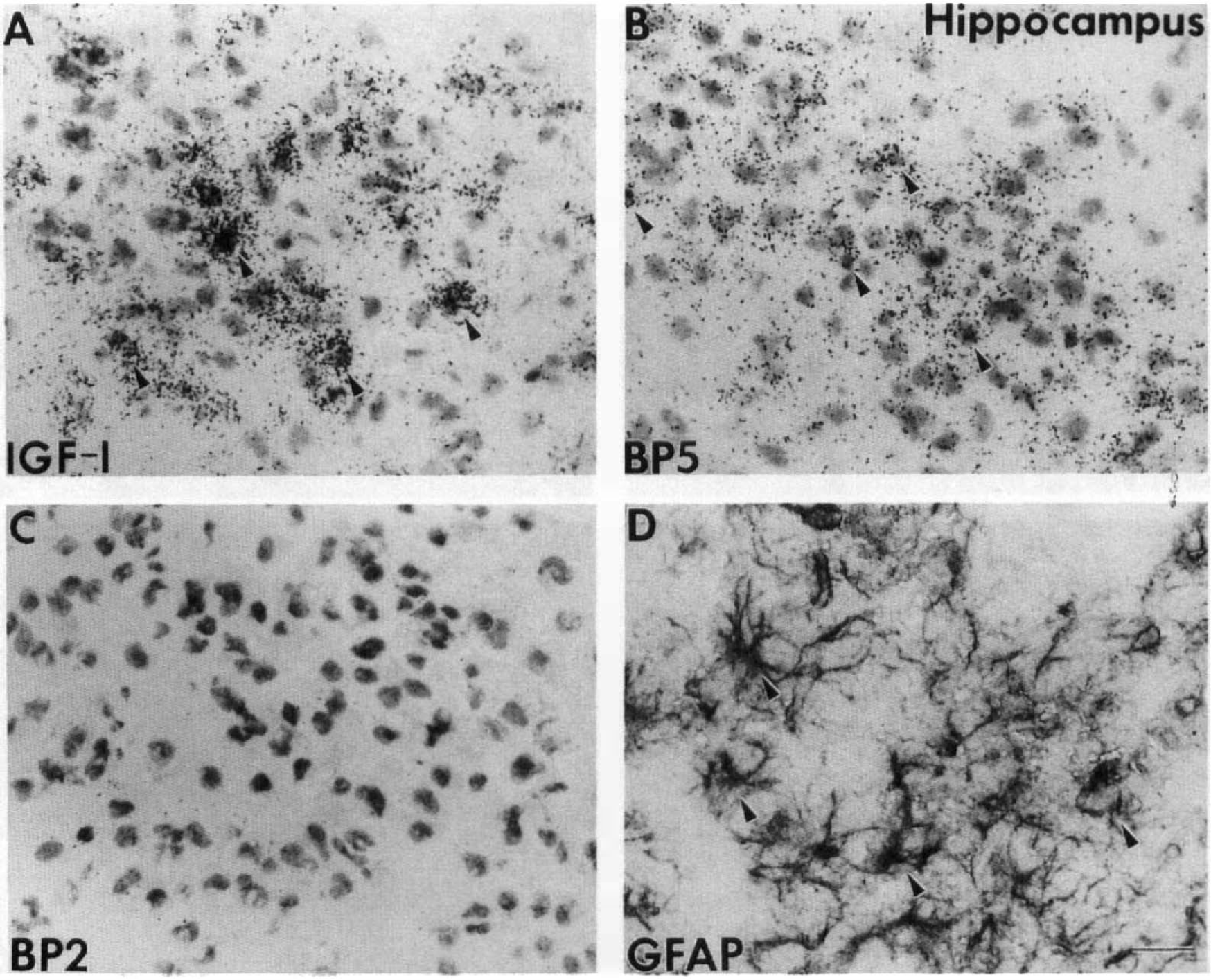
Cellular distribution of insulin-like growth factor I (IGF-I), IGFBP5, and IGFBP2 messenger RNA (mRNA) and glial fibrillary acidic protein (GFAP)-positive astrocytes in the hippocampus at 72 h of recovery. Micrographs showed granule cell layer in dentate gyrus on adjacent sections hybridized to IGF-I
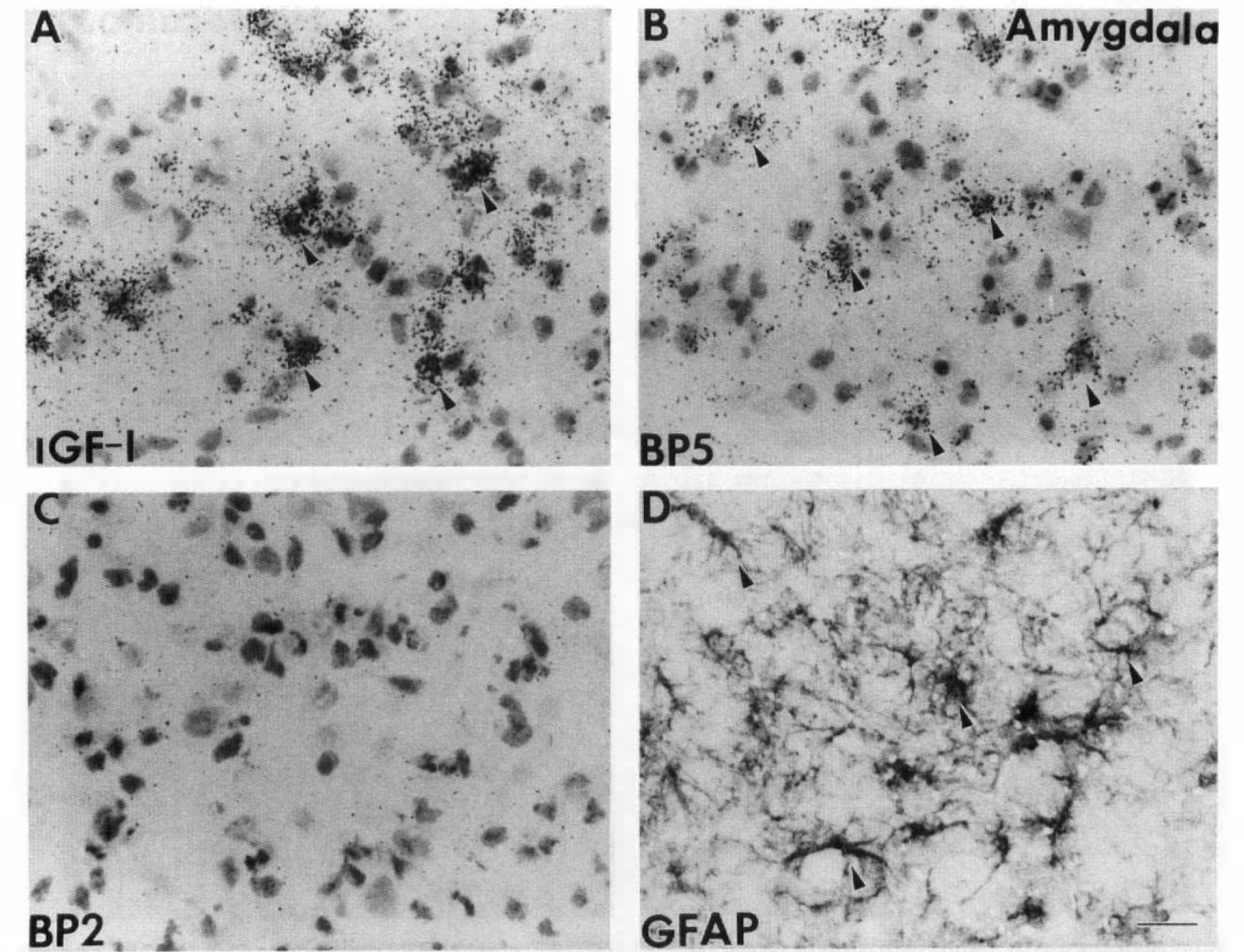
Cellular distribution of insulin-like growth factor I (IGF-I), IGFBP5, and IGFBP2 messenger RNA (mRNA) and glial fibrillary acidic protein (GFAP)-positive astrocytes in the amygdala at 72 h of recovery. Cellular distribution of IGF-I
DISCUSSION
Current evidence, including ours, supports the functional role of IGF-I during brain development, especially in the critical growth period. To understand the regulation of IGF-I gene expression during this period in response to a major disturbance in cellular metabolism, this study examined the effect of perinatal hypoxia–ischemia on the expression of IGF system genes in rat brain. Almost immediately after the hypoxia–ischemia, there was a panoramic decrease of mRNA levels of all IGF system components in the ipsilateral hemisphere, especially in those areas most vulnerable to hypoxic–ischemic injury. This early decrease in IGF system gene expression, absent in adult rats or at postnatal day 21 (Gluckman et al., 1992), appears to be unique to neonatal rats. This decrease was more pronounced at 24 h, when neuronal death is well advanced. By 72 h, IGF-I and IGFBP5 gene expression was activated in reactive astrocytes congregated at areas of injury, whereas mRNA levels of other IGF system genes remained suppressed. The astrocytic IGF-I expression was probably involved in astrogliosis associated with brain tissue repair, which has been observed after hypoxia–ischemia in older rats (Gluckman et al., 1992) and in a focal ischemia model (Lee, Clemens, et al., 1992). The coactivation of IGFBP5 gene expression in the same population of astroglia with IGF-I has not been reported in older rats (Gluckman et al., 1992). Future studies using immunocytochemistry will be needed to evaluate whether the peptide levels were also increased for IGF-I and IGFBP5 in astrocytes. Overall, we described for the first time a coordinated early decrease in neuronal IGF-I and IGFBP5 mRNA levels, as well as their late activation in reactive astrocytes.
A potential functional role of IGF-I in brain development is indicated by its specific spatial-temporal expression during the critical growth period. In rats, IGF-I mRNA levels start to increase soon after birth exclusively in large projection neurons, reach their maximum about postnatal day 12–14, and decline by the end of third postnatal week when the brains mature (Bondy et al., 1992). In addition, the type I IGF receptor (Bondy et al., 1992), IGFBP2 and IGFBP5 (Lee, Javedan, et al., 1992; Bondy and Lee, 1993a) are all expressed at the highest levels coordinately with IGF-I, suggesting an autocrine/paracrine mode of action for IGF-I during this growth period. Whereas IGFBP2 is expressed primarily in astroglia, IGFBP5 is coexpressed with IGF-I in the same group of projection neurons in the relay stations of the somatosensory and cerebellar systems (Bondy and Lee, 1993a). To date, the functional role of IGF-I during this critical growth period is not well understood, but it is likely to be involved in regulating the heightened cellular metabolism required by the developing brain (Bondy et al., 1992).
After hypoxia–ischemia, the immediate decrease in IGF system gene expression indicates that the IGF system components are subject to metabolic regulation and perturbation during the critical growth period, which is characterized by accelerated dendritic growth, synaptic formation, and myelination of the axons. All of these events require supplies of essential nutrients, as well as heightened cellular metabolism. During and after hypoxia–ischemia, cerebral oxidative metabolism is severely compromised as a direct result of decreased delivery of glucose and oxygen to the neonatal brain (Vannucci, 1993). Among all indicators of cellular metabolism, there is a persistent loss of cellular ATP, which in turn will affect those metabolic processes that require energy for their completion. Interestingly, similar hypoxic–ischemic disturbances at P21 did not result in the early decrease in mRNA levels (Gluckman et al., 1992) of any IGF system components as seen at P7. It is therefore likely that IGF system gene expression is subject to metabolic regulation during the critical growth period, to which the normal expression of IGF-I and its activity are essential. Consequently, the early decrease in IGF-I expression may be partially responsible for the impending cell death that occurs within 24 h of recovery, especially in areas of myelinogenesis (Rice et al., 1981).
IGF-I is a potent stimulator in oligodendrocyte proliferation, differentiation, and survival. Oligodendrocytes will not develop in culture unless the medium contains 10% fetal bovine serum or physiological concentrations of IGF-I (McMorris et al., 1986). Through type I IGF receptors present on oligodendrocytes (Masters et al., 1991), IGF-I stimulates their mitosis, induces immature glial precursors to differentiate into oligodendrocytes (McMorris and Dubois-Dalcz, 1988), and promotes myelin production (McMorris et al., 1986; Mozell and McMorris, 1991). More convincingly, transgenic mice that overexpress the IGF-I gene have larger brains with increased cell size, number, and myelin content (Carson et al., 1993). Conversely, brains of mice with a deletion of the IGF-I gene are hypomyelinated (Beck et al., 1995). In addition to its role in oligodendrogenesis, IGF-I also acts as a survival factor to prevent cell death in the oligodendrocyte lineage (Barres et al., 1992). Therefore, the immediate decrease in IGF-I mRNA levels may contribute to the selective vulnerability of myelinogenesis to hypoxia–ischemia in neonatal rat brain.
The late activation of IGF-I in reactive astrocytes has been reported in other models of neuronal injury. In transected sciatic nerves, immunoreactive IGF-I is increased in reactive Schwann cells, reaching a peak level in 2 weeks (Hansson et al., 1986). Interestingly, infusing IGF-I into the sciatic nerve stimulates its regeneration, whether the injury was induced by mechanical crush (Kanje et al., 1989), sectioning (Li et al., 1994), or by freezing (Silverstein et al., 1984). In the central nervous system, IGF-I gene expression is induced in reactive astrocytes during astrogliosis induced by mechanical lesion (Garcia-Estrada et al., 1992), by ischemic injury (Lee, Clemens, et al., 1992), or by hypoxia–ischemia at P21 (Gluckman et al., 1992). Normally, IGF-I is expressed exclusively in neurons throughout life. Only in the delayed phase of neuronal injury is the IGF-I gene activated in reactive astrocytes. The astrocytic IGF-I expression is most likely involved in the heightened metabolism required during astrogliosis. In fact, high levels of IGF-I mRNA were found in areas demonstrating high levels of 14C-2-deoxyglucose uptake in a focal ischemia model (Bondy and Lee, 1993), supporting an anabolic role during astrogliosis.
In neonatal brain, another unique response of IGF system genes to hypoxia–ischemia is the coordinated expression of IGF-I and IGFBP5, which was not observed at postnatal day 21 (Beilharz et al., 1993). Normally, IGF-I and IGFBP5 mRNA are coexpressed in large projection neurons in relay stations of somatosensory and cerebellar systems (Bondy and Lee, 1993a). Soon after cerebral hypoxia–ischemia in the immature rat, the neuronal expression of both IGF-I and IGFBP5 was decreased. However, by 72 h, there was a coactivation of both genes in reactive astrocytes. IGFBP5 is one of the six soluble IGFBPs that have been identified in the circulation and extracellular fluid (Clemmons, 1993). These high-affinity proteins can modulate the biological activities of IGFs in a target tissue by transporting IGFs in and out of the vascular compartment, targeting IGFs to specific cell types, and modulating the interaction between IGFs and their receptors. To date, the mechanism of action of IGFBPs is not entirely understood but may largely depend on the affinity between a specific IGFBP and IGFs relative to IGFR-I. For example, IGFBP5 has the highest affinity for IGFs of all the IGFBPs, which is 50-fold greater than that of the IGFR-I at pH 7.4 (Cohick and Clemmons, 1993). This high affinity is decreased when the IGFBP5-IGF complex binds to the extracellular matrix protein (Jones et al., 1993), where IGFs can be released to stimulate cellular growth. It is conceivable that IGF-I and IGFBP5 are made by reactive astrocytes and released in bound form, which will protect IGF-I from protease degradation. This IGF-I-IGFBP5 complex will diffuse away to other sites, such as the extracellular matrix, for the anabolic action of IGF-I.
In summary, we found significant differences between the responses of the cerebral IGF system gene expression in immature (P7) rats and that reported for older P21 rats. First, unlike those of older animals, the transcription levels of all IGF system gene components at P7 were decreased soon after metabolic disturbance in the ipsilateral hemisphere, especially in areas vulnerable to hypoxia–ischemia. This early decrease suggested that, during the critical growth period, IGF system gene expression in rat brain is subject to metabolic regulation. Second, there was a coordinated early decrease in neuronal IGF-I and IGFBP5 mRNA levels, as well as a late activation of both genes in reactive astrocytes. The specific temporal and cellular expression of IGFBP5 and IGF-I suggest a unique regulatory mechanism of the action of IGF-I in areas of injury by IGFBP5 in the developing brain.
Footnotes
Acknowledgment:
This study was supported by grants from the James Whitcomb Riley Memorial Association (W.-H. Lee) and the National Institutes of Health 1 R29 HD31521-01 (S.J.V.). We are grateful to Peter Rotwein for supplying us with the IGFBP5 clone, Mrs. Nancy Chapman for secretarial assistance, and Phil Wilson for expert photography.
