Abstract
Our previous studies on cerebral metabolic activity in genetic absence epilepsy rats from Strasbourg (GAERS) were in favor of decreased functional activity during absences and normal or increased interictal activity. To ascertain that hypothesis, in the present study we performed continuous measurements of CBF in both children with typical absence epilepsy and GAERS, using Doppler ultrasonography and laser-Doppler flowmetry, respectively. CBF fluctuations during absences were recorded in four children between 5 and 6 years of age and 16 adult GAERS. In both children and animals, CBF measured in the middle cerebral artery and cortical capillaries, respectively, significantly decreased by a median value of 20–24% under basal levels during spontaneous absences. In GAERS, CBF levels were continuously decreased during haloperidol-induced absence status epilepticus, while they were not affected by ethosuximide. Conversely, convulsive seizures induced in rats either by kainate or Picrotoxin led to a 175–664% increase in CBF levels. In conclusion, the present data show that during spontaneous absences, CBF decreases under basal levels in both cortical capillaries (GAERS) and the middle cerebral artery (children). Moreover, these fluctuations occur in vessels with normal vascular reactivity, are not mediated by changes in Po2, Pco2, or arterial blood pressure, and represent rather a response to reduced metabolic demand.
Keywords
Using positron emission tomography (PET) measurements in humans with typical childhood absence epilepsy, Engel et al. (1982, 1985) have shown a general 2.5- to 3.5-fold increase of cerebral metabolism during the scan in children with typical absence seizures. In adult patients with primary generalized absence epilepsy, local CMRglc (LCMRglc) increased by 30–50% during the ictal scan, while LCMRglc values were unchanged or decreased in young adults with secondary generalized epilepsy (Theodore et al., 1985; Ochs et al., 1987). Likewise, in the rat model of genetic absence epilepsy called GAERS (genetic absence epilepsy rats from Strasbourg) (Marescaux et al., 1992), we have shown that the rates of cerebral energy metabolism assessed by the quantitative autoradiographic [14C]2-deoxyglucose method display an overall consistent 16–50% increase over those of a selected strain of control nonepileptic rats (Nehlig et al., 1991). In both humans and GAERS, increases in LCMRglc involve all cerebral functional systems without any focal hyperactivity (Engel et al., 1982, 1985; Theodore et al., 1985; Ochs et al., 1987; Nehlig et al., 1991), although spike-and-wave discharges (SWDs) are recorded mainly in the thalamus and cortex of GAERS (Vergnes et al., 1990).
Moreover, when GAERS receive haloperidol, which induces the appearance of absence status epilepticus (Warter et al., 1988), the levels of cerebral energy metabolism decrease compared with those in untreated GAERS and become similar to those of the control nonepileptic strain (Nehlig et al., 1992, 1993). Likewise, in one patient whose PET scan was performed during absence status epilepticus, LCMRglc decreased compared with the interictal scan (Theodore et al., 1985). After the administration of ethosuximide, a specific petit mal antiepileptic drug that totally suppresses absences in GAERS (Marescaux et al., 1992), LCMRglc values remain as high as in untreated epileptic animals (Nehlig et al., 1992, 1993). Therefore, unlike convulsive seizures during which CBF and metabolism widely increase (Ingvar et al., 1984; Pinard et al., 1987; Nehlig et al., 1995), our data are in favor of a normal or decreased ictal metabolism and of an increased interictal glucose utilization in the brain with absence seizures (Nehlig et al., 1991–1993). However, because of the long duration of a 2-deoxyglucose experiment or of a [18F]fluorodeoxyglucose PET scan, i.e., 45 min, the cerebral metabolic level recorded in naive GAERS or in typical childhood absence epilepsy, respectively, is always a combination of ictal and interictal phases.
CBF is another index of cerebral functional activity that is usually coupled to cerebral metabolic demand (Sokoloff, 1981; Kuschinsky, 1987). Therefore, to confirm our hypothesis of decreased ictal and increased interictal cerebral functional activity, in the present study, we measured continuously the rates of CBF in both humans and GAERS. In children with typical absence epilepsy, CBF velocity was assessed using transcranial Doppler ultrasonography and in GAERS by means of laser-Doppler flowmetry (LDF). Control EEG recordings were performed simultaneously with CBF recordings in both children and GAERS. In children, the effect of spontaneous absence seizures on cerebral blood velocity was studied. In GAERS, in addition to spontaneous absences, CBF was also recorded after the administration of haloperidol and ethosuximide, as well as during generalized convulsive seizures.
MATERIALS AND METHODS
Clinical studies
Experimental studies
On the day before the experiment, rats underwent surgery under chloral hydrate anesthesia (300 mg/kg i.p.). First, the skin on the skull was opened and the thickness of the bone was reduced from 0.5 to 0.1 mm under light microscopy over a round surface of ∼2–3 mm in diameter on the left side of the skull. This procedure allowed visualization of superficial brain vessels by transparency. Thereafter, the right side of the skull was equipped with two single-contact electrodes over the frontoparietal cortex, as previously described (Vergnes et al., 1990), to allow continuous recording of the EEG. The electrodes consisting of stainless-steel screws connected to metal threads and the screws allowing the positioning of the laser probe device were embedded into acrylic cement over which the skin was sutured. Finally, to record arterial blood pressure and measure blood gases, the left femoral artery of the rats was catheterized with Tygon tubing (ID 0.015 mm, ED 0.030 mm; Tygon Micropore Tubing, Akron, OH, U.S.A.), and the catheter was rolled and hidden under the skin of the animal before suturing the skin opening. The animals were then allowed to recover for 24 h in their home cages with food and water
Basal levels of CBF and arterial blood pressure were recorded under simultaneous EEG control in all animals. An 80-μl blood sample was taken from the arterial catheter to measure basal values of pH, Po2, and Pco2 in a blood gas analyzer (Corning 178, Corning Medical and Scientific, Halstead, England). Thereafter, the vascular reactivity of cortical brain vessels was controlled during hypercapnia. After 45- to 60-min recording of CBF levels during spontaneous absences, the rats underwent different treatments. Five of them received an intraperitoneal injection of haloperidol (2 mg/kg), while five others were injected with ethosuximide (70 mg/kg). Arterial blood gases were monitored 5–15 min after the injection of these two drugs. At the end of the experiment and after recovery of the spontaneous occurrence of SWDs, the cerebrovascular response of GAERS during convulsive seizures was monitored in three rats after kainic acid injection (12 mg/kg i.p.) and in six others after Picrotoxin administration (3 mg/kg i.p.). The duration of the experiments was ∼2 h during which all rats remained awake and calm with occasional episodes of head motion. Rats were killed with an overdose of pentobarbital at the end of the experiments.
Statistical analysis
The percent CBF changes during and after absences and induced by ethosuximide and haloperidol were compared with basal CBF levels by means of nonparametric paired Wilcoxon statistics. The effect of haloperidol and ethosuximide on arterial blood pressure and blood gases was analyzed by Student's
RESULTS
Clinical studies
In each of the four children studied, two to nine absences lasting for a median duration of 14 s (range 9–17 s) were recorded. Cerebral blood velocity decreased by a median value of 24% (range 12–27%) during absences compared with basal levels. This cerebral blood velocity decrease occurred after a median latency duration of 5 s (range 2–8 s). At the end of the absence episode, cerebral blood velocity increased to a postictal level similar to the basal level (Fig. 1) within a median time of 11 s (range 6–14 s) after the termination of the seizure. Only in one child was the decrease in cerebral blood velocity more marked at the end of the absence episode and followed by a short period of increased flow velocity level compared with baseline.
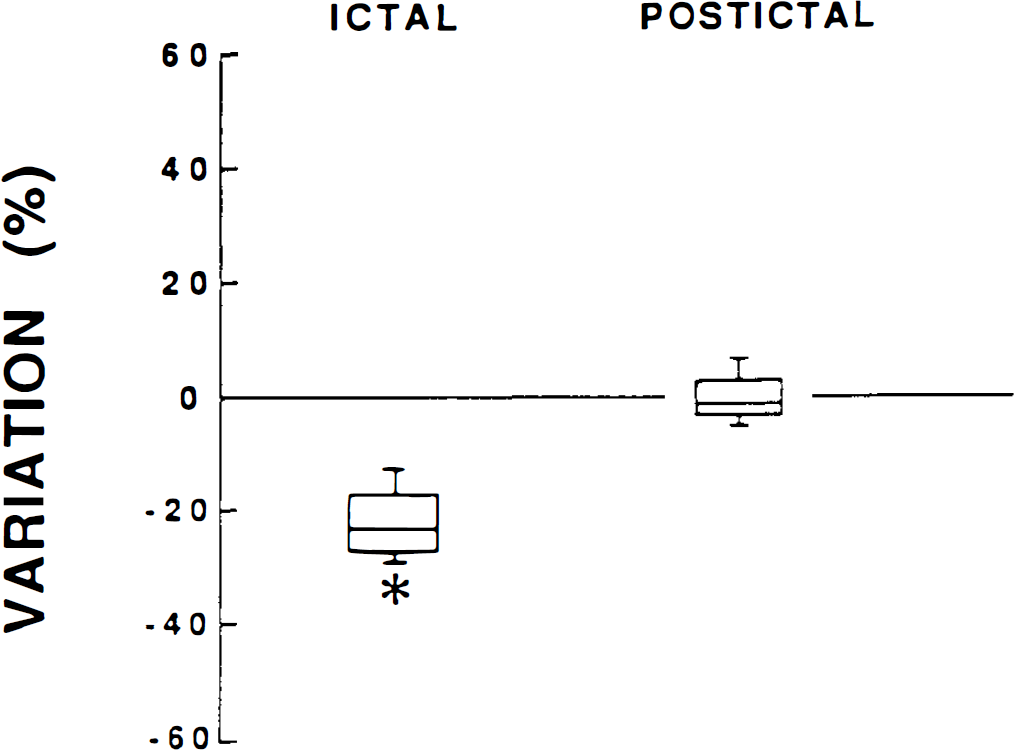
CBF changes during ictal and postictal periods in children with typical childhood absence epilepsy. Values, expressed as medians, inferior and superior quartiles, and percentiles, represent percent variation from the basal CBF level. *p < 0.05, statistically significant difference from basal CBF level.
Experimental studies in GAERS
Physiological variables in genetic absence epilepsy rats from Strasbourg (GAERS) treated with haloperidol (HAL) or ethosuximide (ESM)
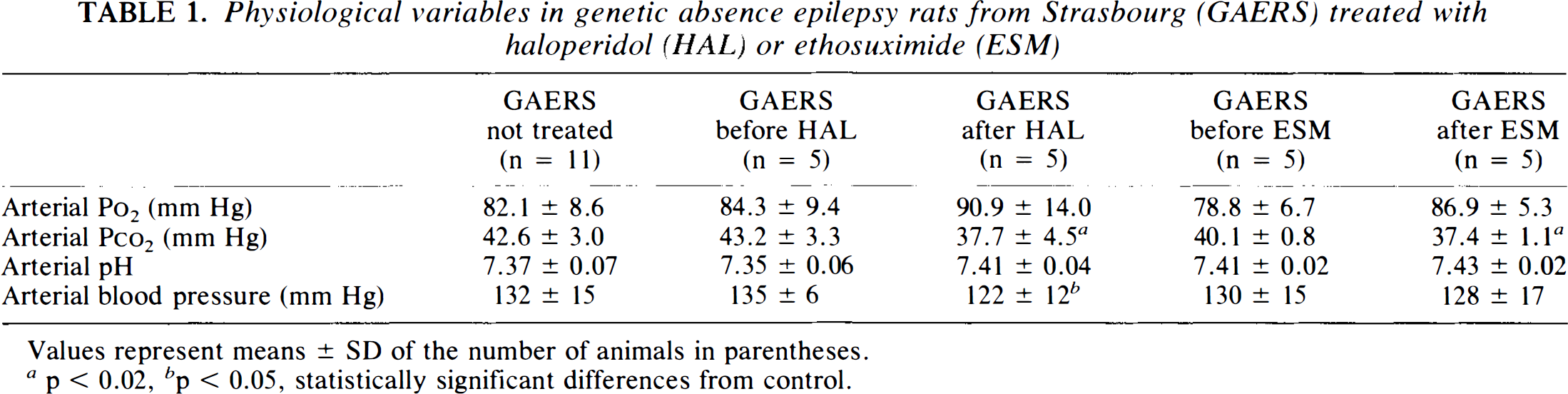
Values represent means ± SD of the number of animals in parentheses.
p < 0.02,
p < 0.05, statistically significant differences from control.
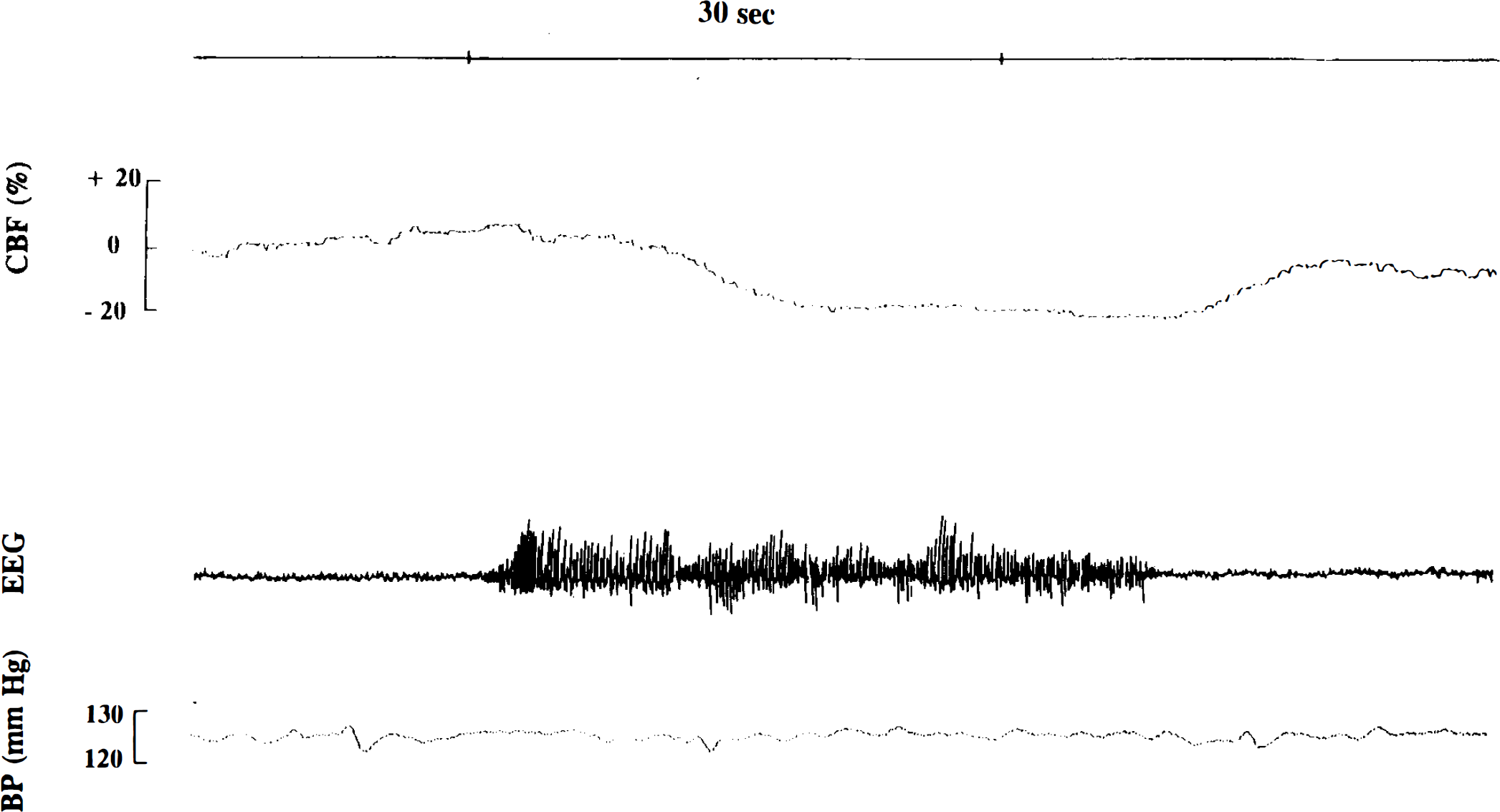
CBF variations (expressed as percent variation from the basal CBF level) during spike-and-wave discharges recorded in genetic absence epilepsy rats from Strasbourg (GAERS) at high speed. The decrease in CBF starts at 5 s after the onset of the seizure and returns to basal levels immediately after the cessation of the seizure. These CBF variations are totally independent of arterial blood pressure (BP) fluctuations.
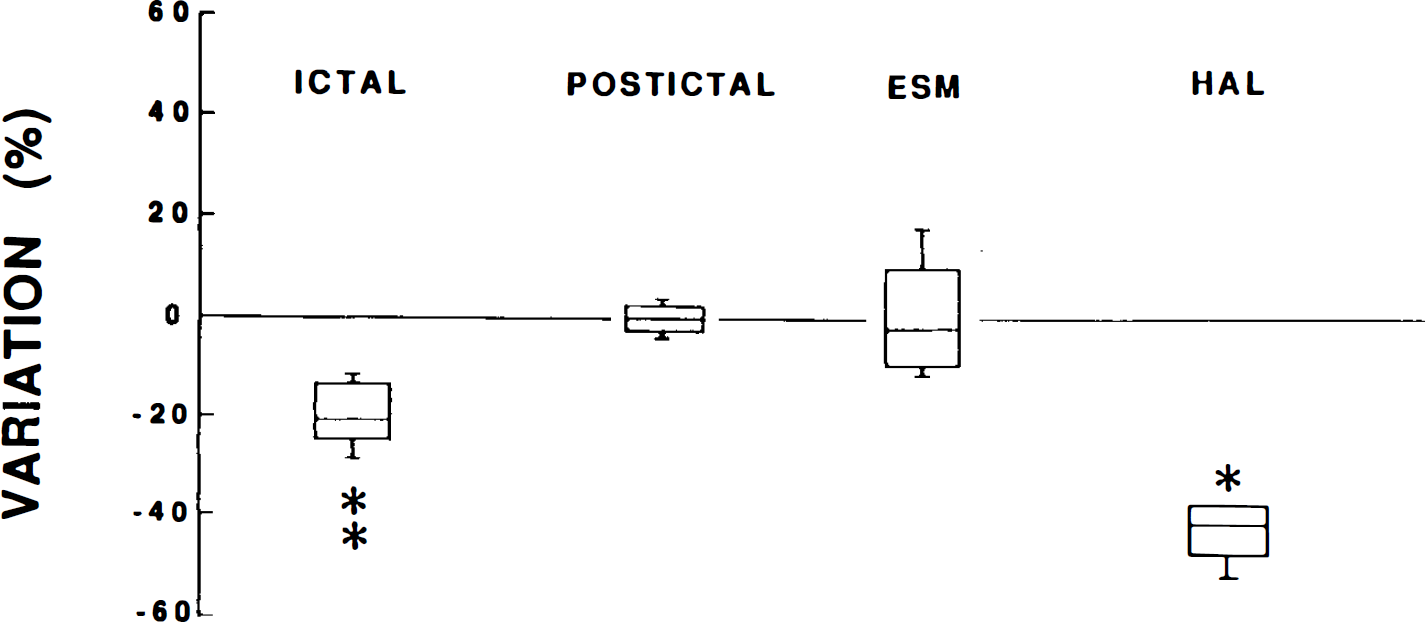
CBF changes during ictal and postictal periods in genetic absence epilepsy rats from Strasbourg (GAERS) not exposed to drugs and after the administration of ethosuximide (ESM) or haloperidol (HAL). Values, expressed as medians, inferior and superior quartiles, and percentiles, represent percent variation from the basal CBF level. *p < 0.05, **p < 0.01, statistically significant differences from basal CBF level.
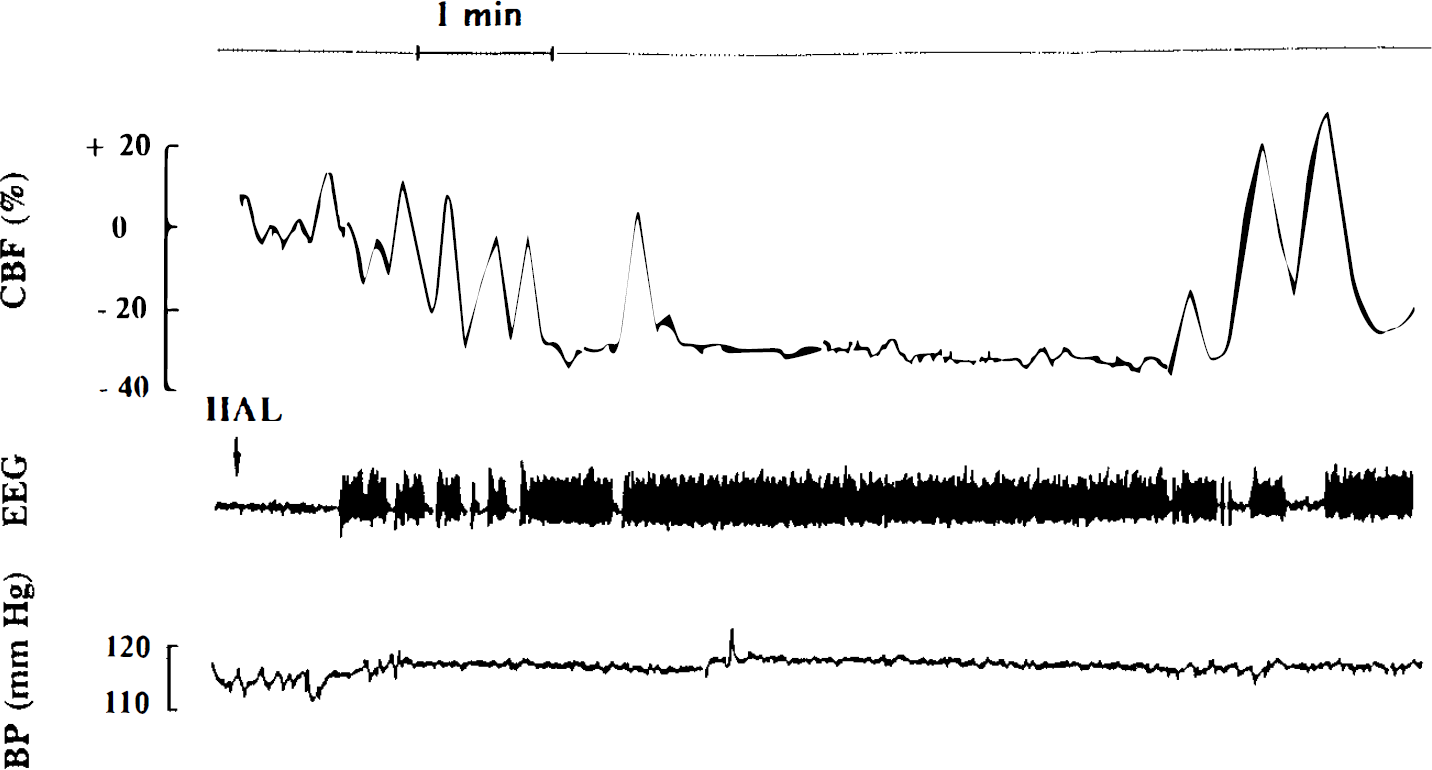
CBF variations (expressed as percent variation from the basal CBF level) after the administration of haloperidol (HAL). The drug induced the appearance of petit mal status epilepticus with continuous spike-and-wave discharges on the EEG, paralleled by a massive and constant decrease of CBF during the whole ictal period. When spike-and-wave discharges became intermittent, CBF fluctuated with high levels during interictal periods and low levels during ictal periods. These CBF changes did not affect arterial blood pressure (BP).
The administration of Picrotoxin (3 mg/kg) induced, after a latency of 11–23 min, myoclonias and generalized tonic–clonic seizures concomitant with transient increases in CBF reaching 175–664% over baseline. Changes in CBF rates induced by Picrotoxin were inversely correlated with blood pressure decreases (Fig. 5).
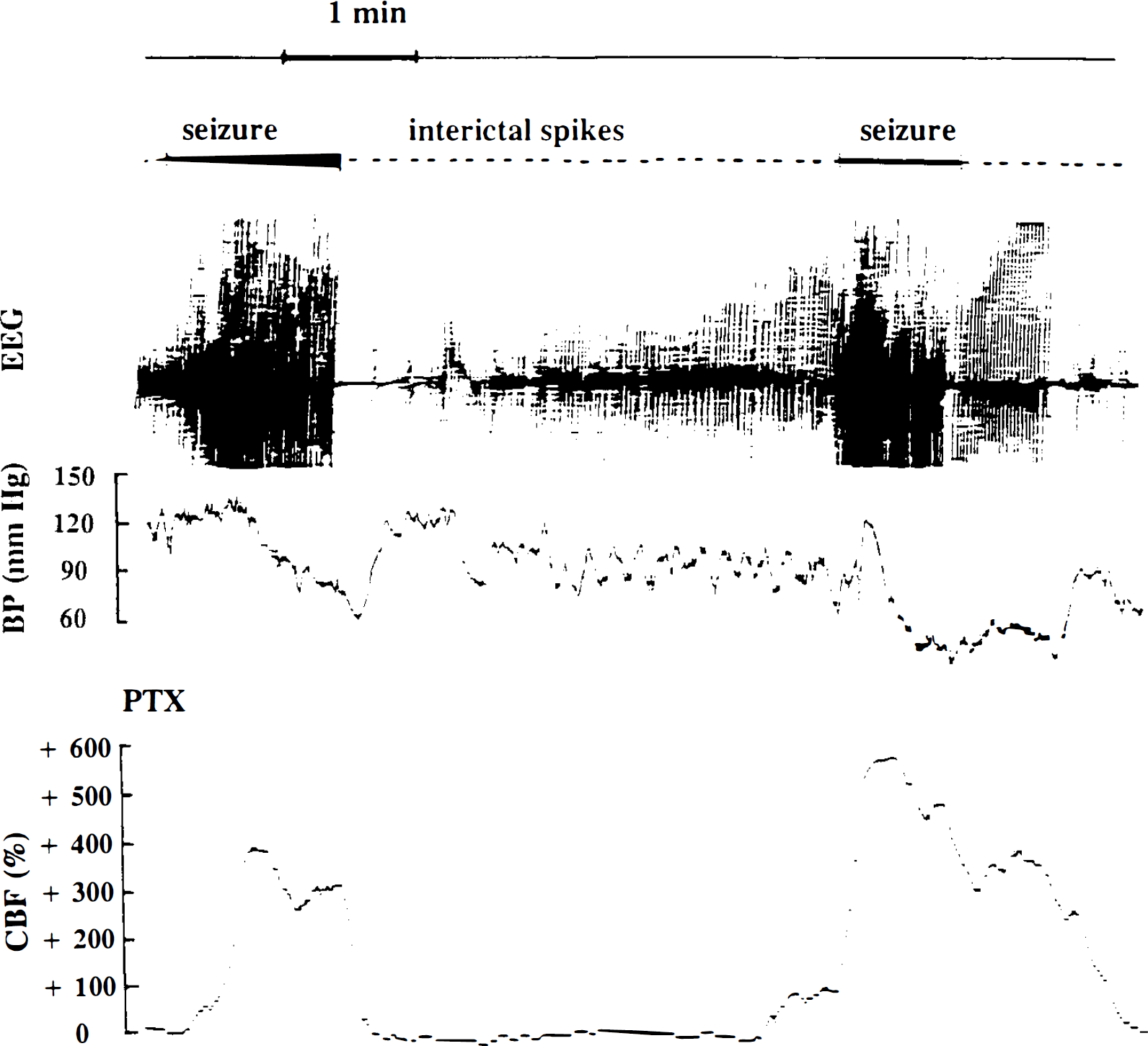
CBF changes (expressed as percent variation from the basal CBF level) at ∼15 min after Picrotoxin (PTX) administration in genetic absence epilepsy rats from Strasbourg (GAERS). During ictal episodes characterized by continuous polyspikes on the EEG, CBF dramatically increased while arterial blood pressure (BP) decreased. During isolated interictal spikes and polyspikes, CBF went back to lower levels 110–150% higher than basal levels before the administration of Picrotoxin.
DISCUSSION
The results of the present study show that during absences in both children with typical absence epilepsy and GAERS, CBF is significantly reduced compared with interictal levels.
Doppler ultrasonography records blood flow velocities that are determined by flow volume and the diameter of the vessel. A good correlation has been found between mean flow velocity in cerebral arteries and CBF (Greisen et al., 1984; Kirkham et al., 1986). Thus, the mean flow velocity in the middle cerebral artery can be taken as a semiquantitative index of CBF, provided that the diameter of the artery does not change significantly. Likewise, LDF has been applied to the measurement of CBF. This technique is based on the fact that the Doppler shift of a back-scattered laser beam from a moving red blood cell is related to its velocity. In LDF, the output signal reflects not only red blood cell velocity, as does the Doppler ultrasound method, but also red blood cell concentration in a given volume and hence blood flow (Wadhwani and Rapoport, 1988). LDF allows reliable, noninvasive, and continuous recordings of the actual time course of flow in the cerebral cortex. For quantitation of CBF by LDF, a careful calibration of LDF needs to be performed previously with other CBF methods (Wadhwani and Rapoport, 1988).
So far, only two clinical studies reported decreases in CBF in two (Sanada et al., 1988) and five (Bode, 1992) patients with typical childhood absence epilepsy. The results of the present study in both children and GAERS are in good accordance with these data. Surprisingly, CBF decreases during SWD occur in the face of largely increased (150–250%) cerebral glucose utilization in children with typical absence epilepsy (Engel et al., 1982, 1985) and more moderately increased (20–50%) LCMRglc values in some young adults with primary generalized SWD (Theodore et al., 1985; Ochs et al., 1987) and GAERS (Nehlig et al., 1991, 1992). The discrepancy between the elevated metabolic activity during typical absence epilepsy in children (Engel et al., 1982, 1985), some young adults (Theodore et al., 1985; Ochs et al., 1987), and GAERS (Nehlig et al., 1991) and the decrease of CBF rates during single absences recorded in the present study may be partly the reflection of the technical constraints of the quantitative autoradiographic [14C]2-deoxyglucose or [18F]fluorodeoxyglucose techniques. Indeed, the measurement of cerebral metabolic activity requires a time long enough to allow most of the tracer to be cleared from the blood and the cerebral tissue, i.e., usually 40–45 min. Therefore, cerebral glucose utilization measured by those methods necessarily represents a mixture of ictal and interictal periods with a predominance of interictal phases. In most situations, CBF and LCMRglc are tightly coupled so that the substrate supply remains adjusted to the cellular demand (Sokoloff, 1981; Kuschinsky, 1987). If this holds true during absences, the origin of the increase in cerebral glucose utilization recorded in children (Engel et al., 1982, 1985), in some young adults with primary generalized absence epilepsy (Theodore et al., 1985; Ochs et al., 1987), and in GAERS (Nehlig et al., 1991–1993) in the face of decreased ictal CBF remains to be elucidated.
In the present study, haloperidol, which induces the appearance of absence status epilepticus, leads to a larger decrease of CBF during SWD in GAERS exposed to the drug (40%) compared with the decrease recorded during single absences in naive animals (20%). Moreover, the treatment with haloperidol induces a generalized decrease in LCMRglc in GAERS compared with naive animals and metabolic levels become similar to those in the control strain (Nehlig et al., 1992, 1993). Likewise, in one patient with status epilepticus, LCMRglc values were decreased compared with the interictal scan (Theodore et al., 1985). All these data are in favor of an association of SWD with reduced energy consumption, hence blood flow. It is also striking that when patients suffer from secondary generalized absence epilepsy, LCMRglc is unchanged by SWD or below the normal range (Ochs et al., 1987) but never over control levels as recorded in primary generalized absence epilepsy (Engel et al., 1982, 1985; Ochs et al., 1987; Nehlig et al., 1991).
In fact, it is quite likely that the SWD-induced CBF decrease is related to a decrease in neuronal activity. Indeed, in the cat generalized penicillin model of absence seizures in which electrophysiology has been studied in detail, the bilateral synchronous SWD response occurs in conditions of diffuse mild cortical hyperexcitability that causes cortical neurons to generate an increased number of action potentials per afferent volley. This secondarily leads to the activation of the intracortical recurrent inhibitory pathway. The result is an alternation of short periods of increased cortical excitation corresponding to the EEG spike during which the cells fire bursts of action potentials with longer-lasting periods of inhibition or silence corresponding to the wave component of the SWD (Fisher and Prince, 1977; Gloor, 1978; Avoli and Kostopoulos, 1982; Inoue et al., 1993). However, the cycles of activity through which cortical neurons go do not exceed the normal range of neuronal excitatory or inhibitory states, conversely to paroxysmal depolarization shifts that occur in focal epileptogenic lesions (Gloor, 1979). Moreover, no population of cells that fire throughout the period of inhibition represented by the wave has been detected (Fisher and Prince, 1977). In fact, most cells are silent during the greater part of the wave, probably since inhibitory neurons fire only at the onset of the inhibitory postsynaptic potential that is prolonged by a persistent action of an inhibitory neurotransmitter (Krnjevic et al., 1966). Therefore, the data on the electrophysiological analysis of the SWD are coherent with the reduced functional activity, hence blood flow, during SWD, as recorded in the present study, when the period of silence in neuronal activity exceeds that of excitation.
In most situations, local rates of CBF and glucose utilization are tightly coupled so that the substrate supply remains coupled to the cellular demand (Sokoloff, 1981; Kuschinsky, 1987). Although some studies in animals have shown local mismatches between CBF and metabolism during seizures (Ingvar and Siesjö, 1983; Ingvar et al., 1984; Tanaka et al., 1990), PET studies using the 15O steady-state and bolus inhalation techniques in human status epilepticus suggest that CBF is adequate for metabolic demand (Franck et al., 1986; Duncan, 1992). Usually, the mismatch between CBF and metabolism, especially the so-called “relative hypoperfusion,” is considered potentially responsible for neuronal damage in animals. This mismatch has been reported only in situations of sudden cellular excitation related to paroxysmal events occurring during the course of convulsive seizures. Conversely, during SWD, as pointed out before, the cycles of activity through which neurons go do not exceed the normal range of neuronal excitatory or inhibitory states (Gloor, 1979). Therefore, it is highly likely that CBF rates and CMRglc remain coupled during absences. Moreover, conversely to the elevated cerebral metabolism recorded during absences in children (Engel et al., 1982, 1985) or GAERS (Nehlig et al., 1991–1993), there is no evidence in the literature that interictal CBF is elevated in children with absence epilepsy since most measurements have been performed with the nonquantitative Doppler ultrasonography technique. However, in one PET study, LCMRglc was shown to decrease in a young adult patient during status epilepticus compared with the interictal state (Theodore et al., 1985).
Some other factors potentially responsible for CBF decreases must be taken into account in the present study. First, the normal extent of vasodilation induced by hypercapnia and convulsive agents in GAERS is indicative of a normal vascular reactivity. Concerning chemical regulation of CBF, Po2 and Pco2 are not different in GAERS and Wistar rats from those in our control strain, as previously shown (Nehlig et al., 1991, 1992). In the present study, Pco2 was decreased 5–10 min after the administration of haloperidol (Table 1). This decrease could therefore at least partly explain the decrease in CBF recorded during absence status epilepticus. However, the decrease in Pco2 consequent to the administration of haloperidol is of a quite low amplitude and similar to the one induced by the injection of ethosuximide. Since the latter antiepileptic drug does not induce any significant change in CBF, it is quite unlikely that the decrease in CBF recorded after haloperidol could result from the slight change in Pco2 recorded in the present study. Arterial blood pressure is not affected by the occurrence of SWD either during a single absence (Fig. 2) or during haloperidol-induced status epilepticus (Fig. 4). The significant decrease recorded 5–10 min after the administration of the dopaminergic antagonist (Table 1) occurs before the induction of absence status epilepticus during which arterial blood pressure remains stable, as shown in Fig. 4. Moreover, the decrease in arterial blood pressure induced by haloperidol is of a low amplitude (Table 1), and since arterial blood pressure largely remains inside the cerebral autoregulation plateau, this slight decrease cannot explain the median 40% extent of CBF decrease during haloperidol-induced absence status epilepticus (Fig. 3).
Neurogenic control could also influence CBF responses during absences. Indeed, cerebral vessels are encircled by a dense network of sympathetic and parasympathetic innervation mediating cerebral vasodilation and constriction (Sercombe et al., 1979; Seylaz et al., 1988; Suzuki and Hardebo, 1993). However, the latencies in CBF decrease after the onset of absence seizures are longer than those recorded before vasodilation mediated by autonomic nerves after brainstem stimulation (Langfitt and Kassell, 1968). Therefore, neurogenic factors are not likely to be predominantly involved in the CBF decrease during SWD that represents rather a response to reduced activity, and hence to reduced metabolic demand. Indeed, during situations like absences, there is no reason why CBF would not stay coupled to metabolic demand, as in most situations (Sokoloff, 1981; Kuschinsky, 1987). To confirm that assumption in spite of variably increased metabolic activity recorded over long periods of time in children with typical absence epilepsy (Engel et al., 1982, 1985), some young adults with generalized SWDs (Theodore et al., 1985; Ochs et al., 1987), and GAERS (Nehlig et al., 1991–1993), a continuous measurement of fast changes of brain energy consumption should be performed by a short temporal resolution technique for assessment of brain metabolic activity.
An interesting point in the present study is that SWD-induced CBF decreases were recorded in both cortical capillaries in GAERS and in the middle cerebral artery in children. These data show that SWD-induced CBF changes occur in both small and big vessels located in different regions of the brain. These observations are in good accordance with previous metabolic data in children with typical absence epilepsy (Engel et al., 1982, 1985), some young adults with primary generalized SWD (Theodore et al., 1985; Ochs et al., 1987), and GAERS (Nehlig et al., 1991–1993) showing a diffuse increase with no specific anatomical substrate. Moreover, the latencies in the vascular responses are directly related to the size of the vessel, i.e., quite short in cortical capillaries and longer in the middle cerebral artery.
In conclusion, the results of the present study show that typical absences induce a decrease in CBF with a return to baseline after the end of the SWD. It is likely that this response is the reflection of decreased cerebral activity, hence decreased cerebral metabolic demand. However, this correlation remains to be demonstrated during absence seizures.
Footnotes
Acknowledgment:
This work was supported by grants from the Institut National de la Santé et de la Recherche Médicale (CAR 490019), the Centre National de la Recherche Scientifique, and the University of Paris VIL The excellent technical assistance of O. Issertial is greatly acknowledged.
