Abstract
Objective:
SN514 is a thermolysin-like enzyme under development as a debrider. Preclinical and non-clinical studies supported a first in human healthy volunteer study to predict the need for protection of periwound skin.
Approach:
Pharmacologic activity testing compared in vitro digestion of collagen, fibrin, and elastin with relevant enzymes. A Yorkshire pig model of burn injury was used to evaluate debridement over 10 days and effects on intact skin. A human 21-day cumulative irritation study using Webril patches taped to the backs of 38 healthy adult volunteers compared four enzyme concentrations (0.10%, 0.20%, 0.40%, and 0.80% w/w) with the hydrogel vehicle, saline (low irritant control), and 0.2% sodium lauryl sulfate (positive irritant control) using randomized placements and blinded evaluation.
Results:
SN514 showed excellent digestion of fibrin, elastin, and collagen in vitro. Burn wound studies in Yorkshire pigs showed efficient eschar debridement with minimal periwound erythema. Direct treatment on intact porcine skin for 5 days produced no to limited erythema. The preclinical findings of minimal irritation with SN514 were verified by a Phase 1 first-in-human 21-day cumulative skin irritation test. Irritation was observed to increase stepwise by concentration, confirming formulation accuracy. Each enzyme concentration was found to be “possibly mild in use” (Berger and Bowman method). No treatment emergent adverse events were observed during the study.
Innovation:
A fast-acting enzyme with a favorable irritation profile, prepared as a stable, ready to use hydrogel formulation, overcomes many recognized shortcomings of enzyme debriders.
Conclusion:
The overall findings support clinical dose range testing for tolerance and preliminary efficacy.
Keywords
INTRODUCTION
The promise of enzymatic debridement to be a superior approach over sharp, surgical, or mechanical methods rests with the potential to be easily and safely applied by any health care professional, acceptably rapid, selective for damaged tissue, less demanding of resources, less traumatic, and used without anesthesia or special facilities. 1 Since the 1940s, many candidate enzymes have been tested with several reaching commercial use but none have demonstrated an ideal balance of purity, potency, selectivity, stability, and safety.
Herbert B. Slade, MD

Poor quality and variability have been highlighted as perhaps the most disappointing factors in the study and use of enzymes. 2 Products that are mixtures of multiple enzymes (e.g., clostridial collagenase and stem bromelain) have a certain inherent variability, which presents challenges for potency release testing. A nonspecific substrate may be chosen such as casein, or a purified substrate for a particular enzyme, or potentially an artificial eschar material containing the relevant proteins collagen, fibrin, and elastin together. 3 Proteases typically display either narrow or broad substrate specificity. Narrow specificity can inadequately address proteins present in necrotic tissues, as has been suggested in the case of fibrinolysin where clinical benefit appeared lacking. 4 This drawback of narrow specificity can be compensated by choosing a collagenase to attack collagen, the most abundant protein in animals and a challenging protein to digest due to its structural organization. For proteases of broader specificity, activity against collagen can be improved by using high concentrations of enzyme (e.g., bromelain) or boosted with the addition of chaotropic urea, but the combination of papain with urea led to the erosion of healthy tissue at the wound base, which became a safety issue. Studies with bromelain have recently switched from using petrolatum to using a zinc oxide skin protectant on periwound skin in order to reduce unwanted damage. Further safety issues have manifested as application site pain, or allergic hypersensitization as with Ficus-fruit allergy directed against papaya fruit latex (papain) or allergy to the Bromeliaceae family (pineapple). A series of fatal anaphylactic reactions led the U.S. Food and Drug Administration (FDA) to ban papain debriders in 2008.
SN514-066b is a hydrogel formulation of the SN514 enzyme, a thermolysin-like metallo endopeptidase made by Anoxybacillus caldiproteolyticus. 5 This family of enzymes is known for exceptional thermal conformational stability, showing peak activity at 70°C (158 °F). Optimal activity is seen at pH 7–8 with a broad stable range from pH 6–9, well-matched to a wound environment. The enzyme is a single molecule rather than a blend of molecules. Unpublished toxicokinetic studies have estimated an enzyme half-life of 2–6 h following intravenous administration to minipigs. SN514 possesses collagenolytic activity to effectively degrade both intact collagen and denatured collagenous materials, while also exhibiting strong activity toward other noncollagen proteins found in eschar. Because denatured proteins, as found in eschar, have lost much of their intact three-dimensional folding, they are much more susceptible to digestion by proteases than the intact protein found in healthy skin. With good selectivity for eschar proteins, the application dose can be significantly low, improving safety.
SN514 and the hydrogel formulation SN514-066b have been extensively evaluated in relevant preclinical models. We report here key findings from studies of in vitro enzyme activity and animal burn wound debridement, followed by the results of a first in human 21-day cumulative irritation test (CIT) 6 designed to evaluate tolerability on healthy intact skin.
INNOVATION
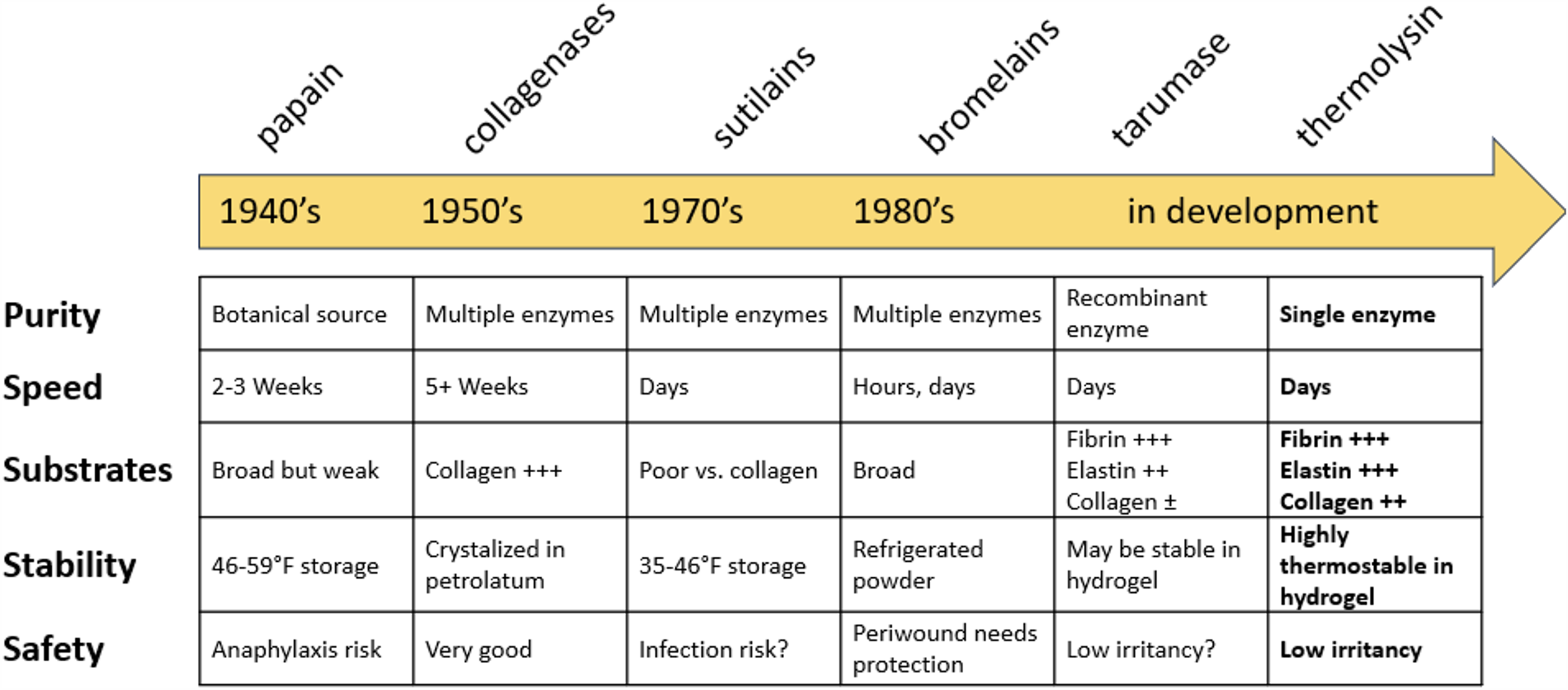
A timeline of major advances in enzymatic debridement.
Enzymatic debridement has long been considered a potentially ideal approach to making burn or chronic cutaneous wounds ready to heal without damaging healthy tissue. Experience over decades with enzymatic debriders has been disappointing, with shortcomings including variable purity, stability, quality and selectivity, excessively aggressive formulations, application site pain, Ficus-fruit latex protein hypersensitivity reactions, or slow speed of debridement.7,8 SN514 is a single, purified, thermostable protease made by Anoxybacillus caldiproteolyticus, formulated in a ready to use hydrogel stable at room temperature for years. We report here the finding of low irritancy potential when applied to intact healthy skin for 21 days.
CLINICAL PROBLEM ADDRESSED
In order for skin to heal well following burns, chronic ulceration, and various traumatic skin injuries, debridement must be performed to remove foreign debris and devitalized tissue. Where debridement cannot be permanently accomplished in a single effort, a higher frequency of repeated debridement has been found to correlate with improved rates of healing. 9 Enzymatic debriders are potentially ideal for effecting a continuous debridement, but a stable product with good purity and a high therapeutic index has been elusive.
MATERIALS AND METHODS
Electronic laboratory notebook was not used for any of these studies.
In vitro enzyme activity
SN514 in tris buffer solution (pH 7.4) was tested against type I collagen (calf skin, Elastin Products Company [EPC], Owensville MO) at 1.0 mg/mL (0.1% w/v), against fibrin at 0.01 mg/mL (0.001%), generated using fibrinogen and thrombin (Sigma, St. Louis MO), and against elastin (elastin-remazol E194S, EPC) at 0.1 mg/mL (0.01%). 10 Enzyme solutions were prepared using 50 mM tris buffer at pH 7.4 (tris[hydroxyl]aminomethane). Reactions were carried out at 37°C for 180 m, 40 m, and 60 m respectively in 96-well microplates using 200 µL each of enzyme solution and substrate solution. A microplate absorbance reader running SOFTmax PRO software was used for kinetic analyses.
Comparator enzymes were collagenase type 1 (Millipore-Sigma (Calbiochem), Burlington MA), elastase (ET947 from porcine pancreas, EPC), high-potency papain (Enzybel BSC, Waterloo Belgium or Enzyme Development Corporation, New York NY), and bovine trypsin (Millipore-Sigma) at matching concentrations. In the collagen digestion experiments, absorbance change in optical density (OD) at 495 was recorded. For the fibrinolysis and elastosis experiments, Vmax (Δ milli OD/min, the rate of an enzyme-catalyzed reaction at saturation) was calculated at 405 or 595 nm respectively to determine maximum velocity. Elastase and trypsin were not tested for collagenolysis.
In vivo porcine burn wound: study A
Animal studies were approved by the Institutional Animal Care and Use Committee at the test facility (University of North Texas Health Sciences Center in Fort Worth, TX). The protocols complied with the National Research Council’s criteria for humane care. In this study 11 on day 0, six commercially raised, specific pathogen free female Yorkshire-cross pigs (20–25 kg) each had twenty 2-cm diameter dorsal burn wounds created as 2 rows of 5 wounds per flank posterior to the axillary line and parallel to the spine, for a total of 120 burn wounds (Fig. 2a). Pigs were sedated by intramuscular (IM) injection with tiletamine/zolazepam and xylazine, received analgesia with IM buprenorphine, were intubated, and anesthesia was maintained using inhaled isoflurane. Hair was clipped then shaved, target sites were surgically scrubbed, and the 20 2-cm diameter thermal wounds were made using brass rods heated to 100°C in a sand bath (one rod per wound) then held on the skin for 45 s. Reston foam (3M, St. Paul MN) was held in place with benzoin around the wounds as protection, covered with Surgilast mesh (Medline, Northfield IL) held in place with tape wrap at ends. Postprocedural pain was managed with buprenorphine.
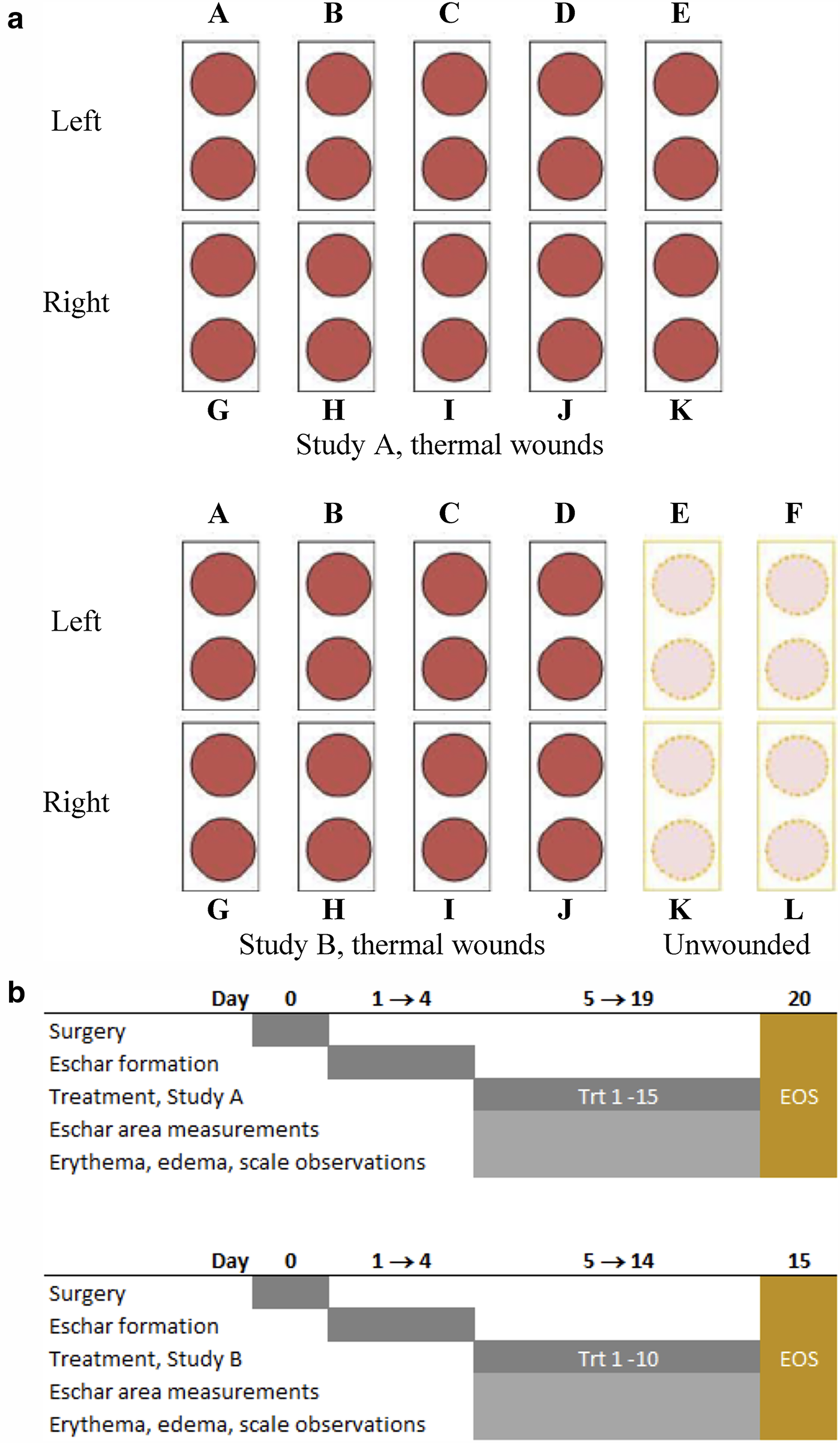
Treatments were first applied on Day 5 to allow establishment of the burn eschar prior to treatment. During the 15-day treatment period (Fig. 2b), formulations were covered with saline moistened cotton pads secured with tape, and further with body wraps and swine jackets. No losses occurred, and no data were imputed. Pigs were humanely euthanized at the end the experiment.
The wounds were photographed and measured using the SilhouetteMobile™ (ARANZ Medical, Christchurch, New Zealand) device to determine the area of the eschars. Clinical assessments were conducted daily, noting the day of completed debridement. The wounds were treated with a reagent-grade bromelain made freshly into a hydrogel formulation, a laboratory-made papain/urea ointment, and as a control, clostridial collagenase ointment (CCO) according to the randomized treatment scheme. SN514 was made into a dry film by first creating a hydrogel and then allowing a volume of the hydrogel to completely desiccate in a plastic petri dish, from which circular discs could be cut to match the size of the burn wounds. Film discs were made at 1.6 mg per 3.14 cm2 disc (low dose, 4.98% w/w of the dry film) or 4.36 mg per 3.14 cm2 disc (high dose, 9.98% w/w) using one disc per 2-cm diameter wound, resulting in doses of 0.7 mg/cm2 and 1.4 mg/cm2, respectively.
In vivo porcine burn wound: study B
A proprietary hydrogel formulation of SN514 (#066b) was evaluated in this animal study. 12 Inclusion criteria were commercially raised female Yorkshire or Yorkshire-cross pigs, 40–60 lbs. in weight and specific pathogen free. One loss occurred on day 12 due to a rectal prolapse. No data were imputed. Animals were sedated, received analgesia and anesthesia, and were surgically prepared and burn wounded with brass rods similarly to study A. Sixteen 2-cm diameter thermal wounds were made posterior to the axillary line and parallel to the spine. Any blisters were deroofed. Up to eight additional unwounded treatment locations per animal were also prepped as shown in Figure 2a. For the eschar formation period, in addition to the surrounding foam and Surgilast mesh used in study A, smaller nonstick mesh dressings (Mepitel) and dry gauze pads were applied directly in contact with the burn wounds and held in place with medical tape. Postprocedural pain was managed with buprenorphine.
Eschars were allowed to form for 5 days with treatments beginning on day 5 (treatment day 1). Each animal had one of three concentrations of SN514 (0.05%, 0.20%, or 0.80%, giving 0.08, 0.33 and 1.30 mg/cm2 of wound) and the hydrogel vehicle alone (placebo) applied randomly in duplicate on one flank (e.g., to AA, BB, CC, and DD), and with a different random scheme on the opposite flank, for a total of 24 wounds treated with each test concentration among the 6 animals. Test articles were also applied in duplicate across the midline on unwounded skin (e.g., to EE, FF, KK, LL) in random fashion. Daily applications continued on wounds through day 13 as 0.5 mL per wound, with unwounded skin treated on days 5–9 (treatment days 1–5). Dry nonstick gauze pads and film dressings (Tegaderm™, 3M) were taped in place over treated wounds, which were then further covered with body wraps and swine jackets. Daily scoring of outcome measures was conducted by a blinded evaluator at pretreatment (day 5) to day 14 (Table 1). On day 14, a tissue strip was taken under anesthesia from each wound location, and the animals were humanely euthanized.
Animal wound assessment scoring

Statistical testing was conducted using one-way ANOVA with Holm-Šídák correction for multiple comparisons.
Clinical study of cumulative irritation
Consenting subjects were adult volunteers aged 18–70 years, in good general health, willing and able to comply with study instructions and commit to all visits were included. Females were required to have a negative pregnancy test and not be lactating. Exclusion criteria included skin cancers, immune disorders or immunosuppressive treatments, skin diseases or damage that would interfere with assessment of irritation, and recent treatment for cancer or with any investigational drug. The trial was conducted in accordance with the ethical principles put forth in the 1975 Declaration of Helsinki, and in conformance with the International Conference for Harmonization Good Clinical Practice (ICH-E6), as well as the United States Code of Federal Regulations (CFR) 45 CFR Part 46, 21 CFR Part 50, 21 CFR Part 56, and 21 CFR Part 312. The informed consent, site, and protocol were overseen and approved by a central Institutional Review Board (Advarra).
SN514 enzyme was formulated into the proprietary hydrogel vehicle for each concentration, rather than making serial dilutions, then loaded into labeled 30 g aluminum tubes and crimp sealed. Each of the test articles (low irritant control [0.9% sodium chloride solution], SN514-066b hydrogel vehicle, 0.10%, 0.20%, 0.40%, and 0.80% SN514-066b, and a positive irritant control that was 0.2% sodium lauryl sulfate [SLS]) was placed into a labeled glass jar just prior to loading onto Webril patches. Qualifying subjects arriving on day 1 entered a private booth where their shirt was lifted to expose their back. The left side of the back was wiped down with 70% isopropyl alcohol and allowed to flash dry.
Test articles were applied using an occlusive square patch with rounded edges (Strukmyer Medical, Mesquite, Texas) of approximately 3.8 cm × 3.8 cm, held to the skin using a clear, nonporous polyethylene hypoallergenic adhesive tape (3M Plastic Medical Tape®). Adhered in the center of the patch was an approximately 2.0 cm × 2.0 cm square of Webril® nonwoven cotton (GWJ Company, La Quinta, CA) with an approximate thickness of 1 mm, onto which was placed a volume of 0.2 mL of the test article (giving 0.05, 0.10, 0.20, and 0.40 mg/cm2).
Patches were applied to the left side of the back in a randomized fashion. Subjects were instructed to keep the patches as dry as possible and return the following day for evaluations and fresh patch applications.
Subjects returning on days 2–21 had their patches removed by the staff and the sites wiped with distilled water. Scoring for irritation was performed 15 min (±5 min) after patch removal at every visit by a qualified blinded grader. Once a patch evoked a strong reaction (score of ≥3), the patch was discontinued and the terminating score (≥level 3) carried forward for all remaining visits.
Subjects were permitted a maximum of one missed visit. If a subject missed one visit the following day’s score would be “carried back” and recorded for the missed visit. Missing a second visit at any time would result in the subject being discontinued from the study. Subjects returning on Day 22 had their patches removed by the staff and the sites wiped with distilled water. Scoring for irritation was again performed 15 min (±5 min) after patch removal. Individual subject scores were reported by test article and visit. Frequency counts of individual subject scores by visit, and total subject irritation scores over the 21-days were also tabulated by test article. For calculating total subject irritation scores, if a subject had a score >3 at any reading, a value of 3 was entered for that reading and for all of the remaining readings of the test.
Total scores were calculated using the methodology of Berger and Bowman. 13 An “observed score” of erythema was given as 0 for no visible reaction, 0.5 for barely perceptible (faint reactivity), 1 for mild erythema (pink), 2 for moderate erythema (definite redness), and 3 for strong erythema (very intense redness). When noted, superficial effects added to the “calculated score” as Glazing +2, Peeling +2, Crusting/Scabbing +2, Hyperpigmentation +1, and Fissuring +2. Calculated scores were similarly increased when edema, dryness, papules, vesicles, erosion, bullae, spreading, weeping, induration, or pustules were noted. Once a total calculated score of ≥3 was reached, no further applications were made at that site. Scoring was overseen by a board-certified dermatologist serving as coinvestigator. The calculated scores for 0.80% and SLS control were compared using a nonpaired t-test assuming unequal variance.
RESULTS
In vitro enzyme activity
Compared with high-potency papain, SN514 showed more specific proteolytic action on wound insoluble proteins of collagen (4× higher), fibrin (3× higher), and elastin (10× higher) (Fig. 3). SN514 displayed the highest fibrinolytic activity compared with the other proteases in the test, with high potency papain, trypsin, and elastase showing equivalent activity against fibrin. SN514 was more potent than all other enzymes tested against elastin, including elastase, a protease whose specific substrate is elastin. Type I collagenase showed the highest collagenolytic activity. The results indicated a good potential for SN514 to effectively digest relevant proteins at a wound surface.
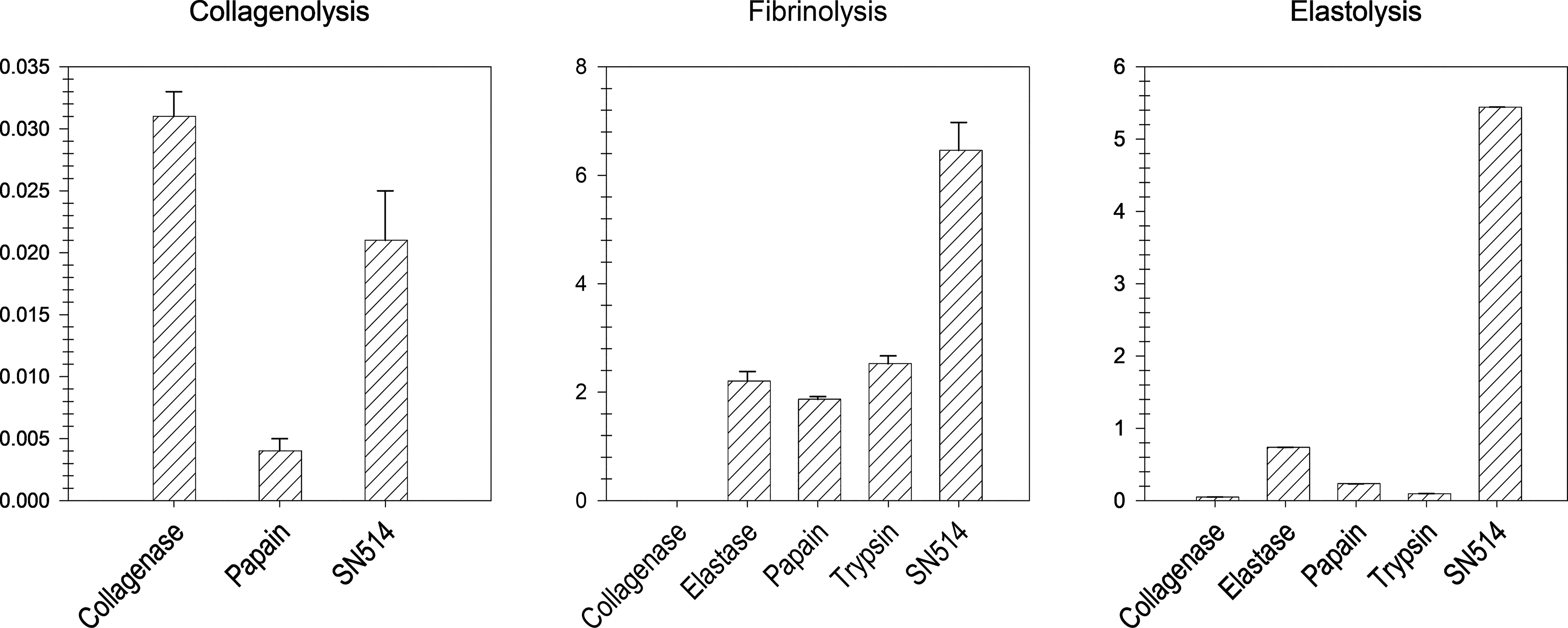
Δ milli OD495/180 min for collagenolysis, enzyme Vmax (Δ milli OD405/min) for fibrinolysis (405 nm), and Vmax (Δ milli OD595/min) for elastolysis. Elastase and trypsin were not tested for collagenolysis.
In vivo porcine burn wound: study A
Visual assessments indicated rapid debridement of the eschar volume during the first few days of the study for the SN514, bromelain, and papain/urea treatments, while the CCO-treated eschars showed little change. Based on ANOVA testing on ranks, there were significant differences found between the bromelain and CCO treatments on day 4–12, and between the SN514 low dose and CCO treatments on days 3–8. In addition, differences were found between the SN514 high dose and CCO treatments on days 3–10 and the papain/urea and CCO treatments on days 4–12. Irritation resulting from treatment application showed the bromelain treatment had the greatest amount of irritation, the SN514 and papain/urea treatments had a moderate amount of irritation, and the CCO treatment had the least amount. The findings were consistent with the previous in vitro experiments, while also showing limited periwound irritation. Note that in this study, no occlusive film dressing was applied over the treatments, raising the concern of drying such that adequate moisture may not have been maintained for optimal hydration/gelling of SN514 films.
In vivo porcine burn wound: study B
Clinical scores for debridement found an efficacy ranking for SN514 hydrogel doses of 0.8% = 0.2% > 0.05% > placebo. In addition, 0.8% SN514 was significantly superior to 0.05% SN514 on treatment days 2–6, with 0.2% SN514 superior to 0.05% SN514 on treatment days 3–6 (Fig. 4A). Erythema was evident in the burn wounds and immediate surrounding tissue in all groups including placebo and classified as moderate. Erythema increased with increased dosage and was significantly different from placebo from treatment day 3 to end of study day 14 in the two higher doses. Testing of intact skin showed no edema in most of the treated groups and up to mild edema in the high-dose group. Erythema of intact skin was evident in the 0.2%- and 0.8%-treated groups but was only mild to moderate (Fig. 4B).
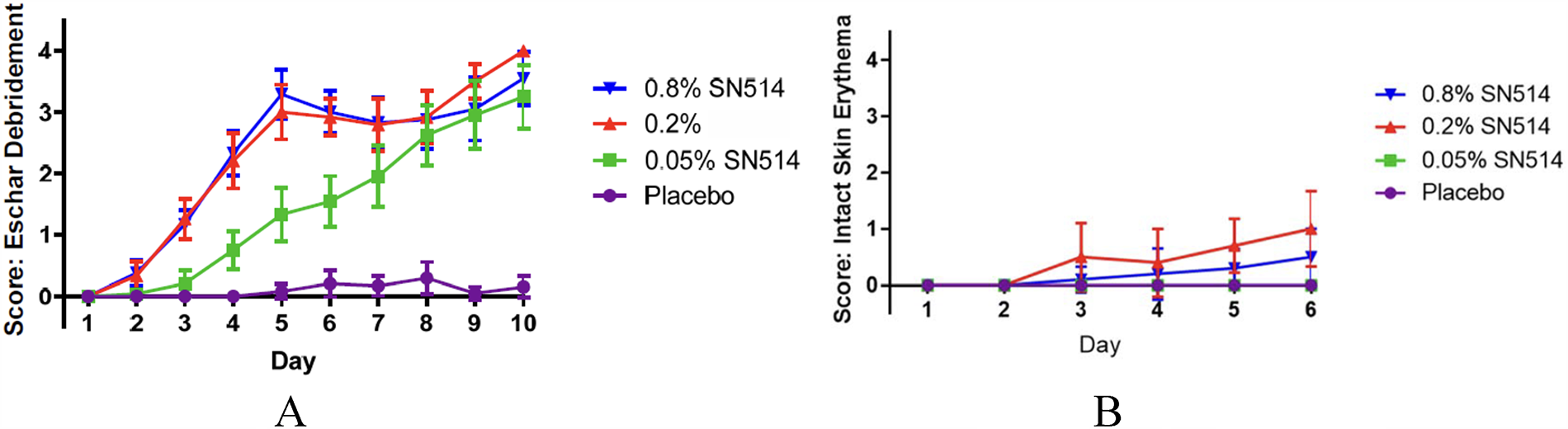
All wounds treated with 0.2% and 0.8% SN514 were fully debrided with absence of necrotic tissue within 7 days, with many of the 0.05%-treated wounds also fully debrided by day 10. Varying degrees of inflammation were noted in SN514-treated wounds as evidenced by large amounts of inflammatory cells near the wound surface. The hydrogel formulation was considered appropriate for human testing, beginning with an evaluation of irritation potential on healthy skin.
Cumulative irritation
Recruitment took place in May–June 2024 with the first subjects enrolled 03 June. Among 39 individuals screened, 38 were enrolled having a mean age of 57 years (±11.9, median 62 [range 20–69]) with 22 males (58%) and 16 females (42%). The race distribution was 32 (84%) Black, 2 (5%) Caucasian, and 4 (11%) Hispanic. Fitzpatrick skin types were predominantly dark brown (14; 37%) or very dark brown (16; 42%) with others being fair (2; 5%), white to olive (3; 8%), or moderate brown (3; 8%). Thirty-six completed all scheduled visits. One qualified subject failed to arrive for the first induction visit and was dropped as a screen failure. One enrolled subject discontinued after 3 days, and a second after 10 days, both due to tape reactions. Application of 0.8% was stopped in one subject after 3 days due to a misapplication at the test site.
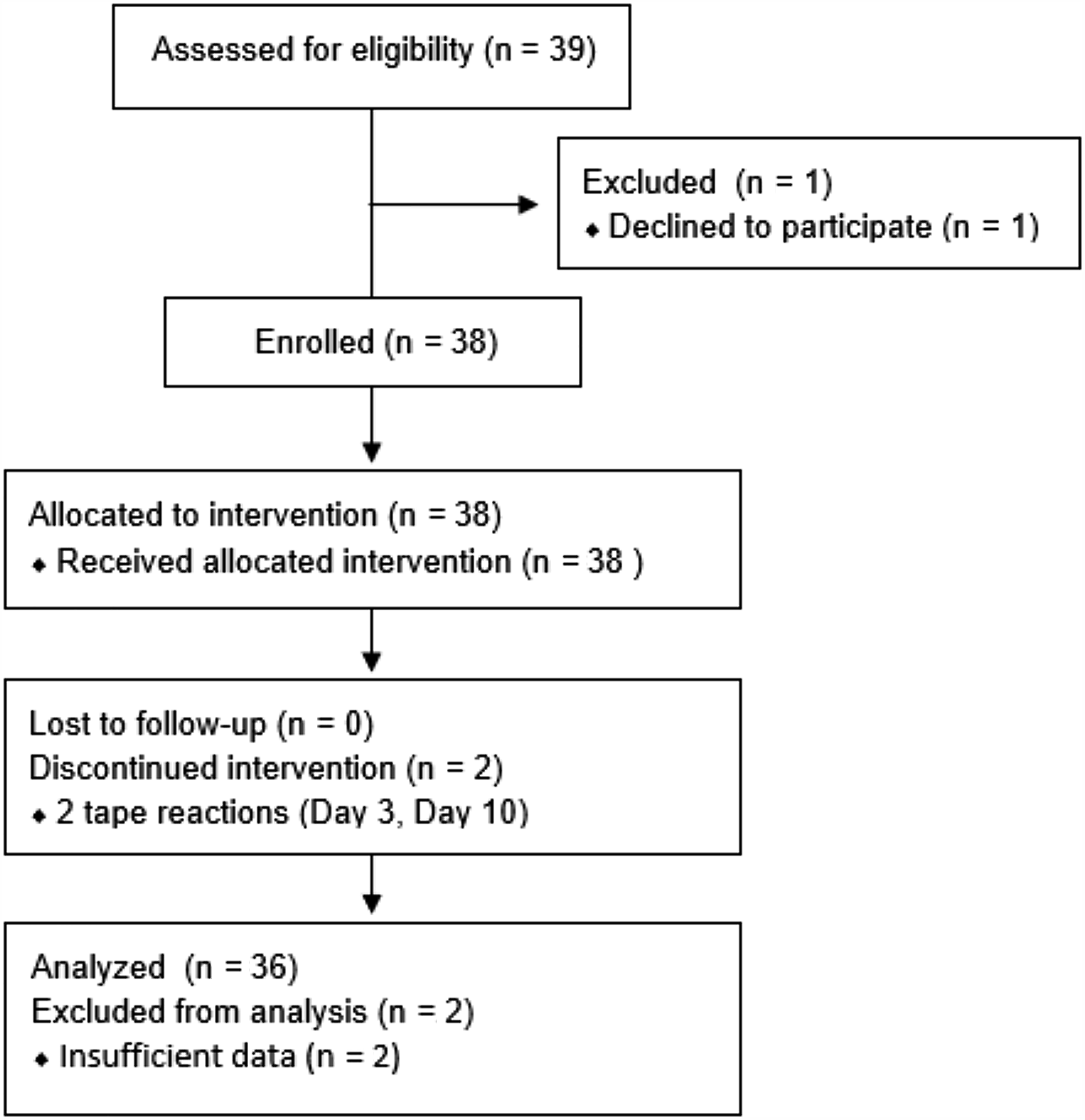
Per FDA guidance, a total of 30 subjects are required to complete a 21-day cumulative irritation study.
No test article-related adverse events were reported other than the anticipated irritation of skin. There were no reports of pain at the application sites. All test sites returned to normal appearance in approximately the same time ≤22 days following cessation of application.
Cumulative irritation scores showed the vehicle to be barely different from saline and probably mild in use, while each concentration of SN514 ranked as possibly mild in use (Table 2). The cumulative scores after 21 days for the highest concentration (0.80%) were significantly lower than those for the positive control (p = 0.0031, unpaired t test with unequal variance). The cumulative scores and the mean observed daily scores showed a dose response relative to the tested concentrations.
Cumulative irritation scoring

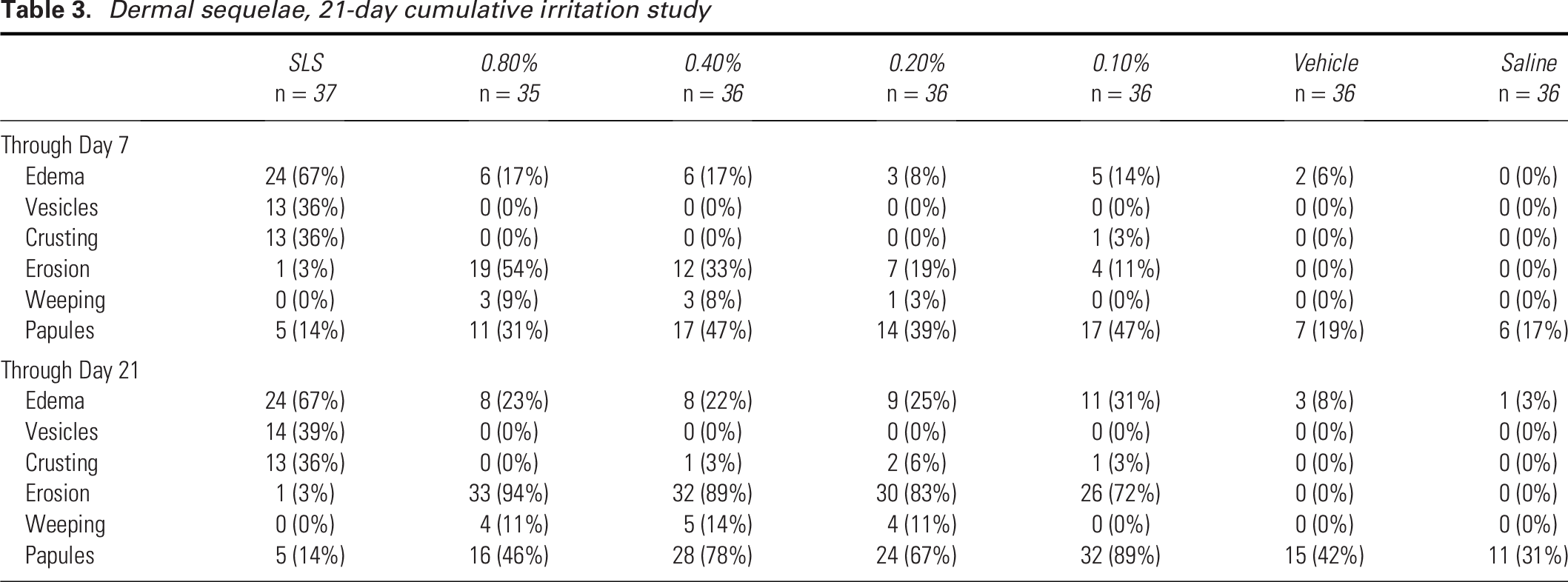
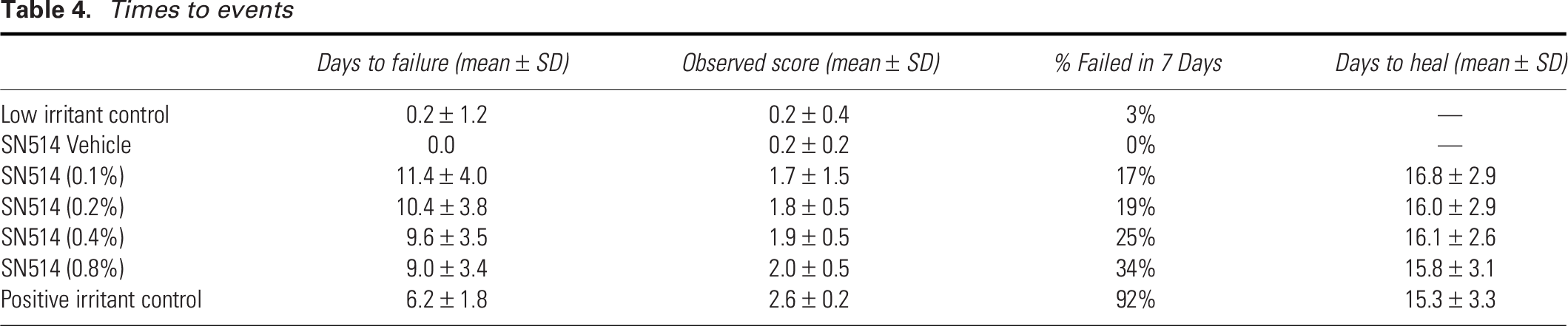
After the first 7 days, average observed erythema scores were 1.7 ± 0.5 for SLS versus 0.6 ± 0.6, 0.6 ± 0.5, 0.5 ± 0.07, 0.4 ± 0.5, and 0.2 ± 0.2 for the 0.80%, 0.40%, 0.20%, 0.10%, and vehicle preparations, respectively. Individual total observed scores showed similar separation (Fig. 6). The positive control produced edema, vesicle formation, and crusting. The enzyme showed less edema but dose-dependent erosions, dose-independent papule formation, and some weeping (Table 3). The mean number of days required for the erythema observed score to reach 3 or greater (“failure”) showed a dose response, as did the proportion failed in the first 7 days (Table 4). After 21 days, nearly all enzyme patches had developed strong erythema. All patch sites returned to their pretest condition (“healed”) in 15–17 days on average. The trial completed as planned.
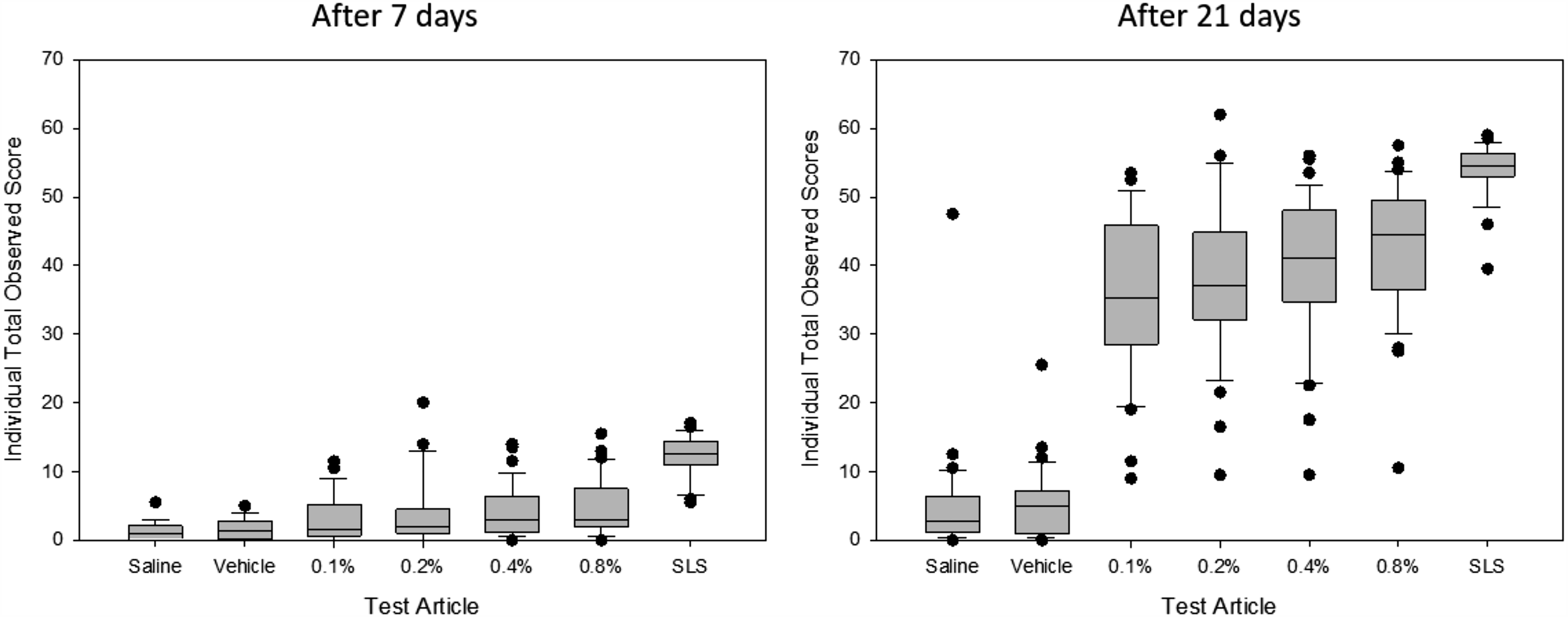
The categorization result of the 21-day cumulative irritation test derives from the sum of scores, as shown in Table 2. Individual scores after 7 and 21 days are shown here to illustrate the spread of data and the relative intensities after 1 and 3 weeks of continuous exposure.
Dermal sequelae, 21-day cumulative irritation study
Times to events
DISCUSSION
The adequacy of debridement has been found to be an independent predictor of wound healing. 14 Many surgical and mechanical techniques are in use with good effect but at the cost of potential blood loss and pain in sensate wounds. Adequate enzymatic debridement can involve a trade-off between aggressiveness and tolerability, with the fastest acting enzymes causing considerable discomfort. SN514 is a thermolysin enzyme under development as a burn and wound debrider. Enzymes used for debridement should ideally be broadly reactive (“nonspecific”), rapidly acting, stable under ambient storage conditions in a ready to use formulation, and tolerable in clinical use. Preclinical testing of SN514 in various formulations and concentrations has shown that when properly formulated, it fulfills the desired attributes for broad reactivity, rapid action, and shelf stability. The animal studies found rapid debridement with minimal periwound irritation, consistent with the findings of Stone et al. 15 and supporting advancement to the first in human clinical study.
The testing of immediate and cumulative skin irritancy potential remains a standard component of the safety evaluation of topically applied products. Since the late 1960s, a 21-day cumulative irritation patch test based on the work of FDA’s John Draize 16 has been utilized for evaluating cumulative irritation, with additional modifications incorporated over time. A sample size of 30 evaluable subjects conforms to industry and regulatory standards for the determination of dermal cumulative irritation potential. The approach to classification of irritancy is based on empirical experiences of previously tested products, rather than using more sophisticated statistical approaches. 17 The progressive increase seen in cumulative irritation scores followed with increasing concentration of enzyme, while the vehicle gel showed no apparent potential for worsening wounded tissue, a hypothetical concern that arises in vehicle-controlled efficacy studies. 18 The development of papules and erosions shows that the enzyme gel is not entirely benign when kept in contact with healthy skin continuously over several weeks. Erosions are defined as disruptions of the epithelium only, with the basement membrane remaining intact. Such breaks in the protective outer surface of the skin can increase the risk of bacterial or fungal invasion. While the magnitude of risk compared with an open burn wound or chronic cutaneous ulcer may be negligible, dosing regimens must be explored, which limit contact time to only what is necessary for effective debridement.
A limitation of the clinical study with respect to planned clinical use of SN514 is that the intent of the CIT study design is to categorize the test article, rather than fully characterize it. Once a maximum erythema score of 3 was reached, application of the patch stopped, and the score and letter grade were carried forward. For example, if strong erythema (score 3) was observed together with edema, cessation of application limited further potential skin damage (e.g., erosion).
Studies in patients with wounds will help to determine the timeframes necessary for debridement, and whether any special measures must be taken to protect the periwound during debridement. A Phase 1 exploration of tolerability in burn wounds is planned, which will examine tolerabilvity in wounds and also inform the ability to use this enzyme on large body surface areas. Additional aspects of use will need to be addressed in future efficacy studies.
KEY FINDINGS
SN514 rapidly digests wound-relevant proteins in vitro SN514 debrides animal model burn wounds with minimal irritation of intact skin SN514-066b hydrogel applied to human skin was mildly irritating over 21 consecutive days under occlusion
AUTHORS’ CONTRIBUTIONS
L.S. conducted the preclinical studies, authored the methods and results, and edited the article. E.D.R. conducted the pig study, authored the methods and results, and edited the article. A.V.J. developed the drug formulation and edited the article. H.B.S. and B.T.R. designed the study and carried it out with N.D.M. Each contributed to writing the article. N.J. supervised preclinical and nonclinical work and edited the protocol and article. There are no undeclared authors.
Footnotes
ACKNOWLEDGMENTS AND FUNDING SOURCES
The authors wish to acknowledge the outstanding contributions of Amie Smith, BS, Reliance Clinical Testing Services/ALS Global, in coordinating the site conduct of the human study. The preclinical studies were funded and carried out by Healthpoint Biotherapeutics or Smith & Nephew; the human clinical trial was funded and carried out by SERDA bv.
AUTHOR DISCLOSURE AND GHOSTWRITING
H.B.S., N.J., and N.D.M. provide services to SERDA under contract. None of the authors are employees or shareholders of or hold any financial interest in SERDA.
