Abstract
Electrolyte imbalance management is crucial in diverse clinical scenarios, with intravenous potassium repletion often required. High-concentration infusions can pose severe complications if extravasation occurs, leading to phlebitis, local tissue damage, or in severe cases, cutaneous necrosis. This risk is elevated in geriatric patients due to factors like reduced tissue elasticity and sensitivity. We report a case of phlebitis and skin necrosis in an elderly woman after peripheral KCl infusion (6% [800 mmol/L]). A woman in her early 70 s presented with obstructive jaundice and critically low potassium levels. A peripheral intravenous administration of 6% KCl was initiated to rectify hypokalemia. Due to superficial venous site selection and the patient's reduced sensitivity, phlebitis developed unnoticed, leading to cutaneous necrosis. Management involved medication discontinuation, cannula removal, application of a magnesium sulfate dressing, limb elevation, and a hydrogel dressing. Despite initial necrosis, wound debridement, ongoing dressings, and moderate hand exercises led to a complete wound resolution. This case underscores the importance of careful selection and monitoring of infusion sites during administration of irritant solutions like concentrated KCl, particularly in geriatric patients. Patient-specific factors, pharmacological implications, and the necessity for adequate vascular assessment are emphasized. Further, the case highlights the necessity for prompt and multifaceted management strategies to handle complications, including patient and caregiver education, careful wound management, and proactive nursing care. The event underscores the need for established protocols regarding the administration of high-risk drugs to prevent severe sequelae.
Introduction
Electrolyte imbalances, particularly hypokalemia, play a significant role in patient outcomes across a wide range of clinical scenarios. Their management, thus, remains a cornerstone in patient care, influencing prognosis and recovery. 1 Various therapeutic interventions are available, with choices typically based on the severity of the imbalance, patient's clinical condition, and underlying etiology. Oral and intravenous routes are the primary modalities for potassium repletion. While the former is preferred for its safety, the latter becomes indispensable in certain situations such as severe hypokalemia or when oral intake is contraindicated. 2 However, intravenous repletion comes with its set of challenges, particularly the risk of extravasation.3,4
Potassium chloride (KCl) infusions, used in severe cases, pose particular risks due to their high concentration and irritative properties. Extravasation, the unintentional leak of this irritant solution into surrounding tissues, can lead to a cascade of complications ranging from simple phlebitis to severe cutaneous and deep tissue necrosis.5,6 In the elderly, the risks associated with intravenous therapy, especially with irritant solutions like KCl, become magnified. Factors such as reduced tissue elasticity, altered vascular structures, and diminished sensitivity contribute to this increased vulnerability. Early detection of complications like extravasation is often hindered in this population, making them more susceptible to severe consequences.
Current medical guidelines recommend the use of Peripherally Inserted Central Catheters (PICC) for administering potassium chloride infusions in patients with hypokalemia to mitigate the risks of extravasation and other complications associated with peripheral intravenous administration. The concentration of potassium chloride typically ranges from 0.3% (40 mmol/L) to 3% (400 mmol/L). For geriatric patients and those requiring fluid restriction, higher concentrations of potassium chloride may be used, such as 6% (800 mmol/L).7,8 However, practical challenges often necessitate deviations from these recommendations. Economic constraints, patient preferences, and the inherent inconveniences associated with PICC lines contribute to a continued reliance on superficial vein injections for potassium chloride administration in some patients. This approach, although common, does not eliminate the potential for adverse effects on the veins, especially given the irritative nature of potassium chloride solutions.
The present case report delves into a practical scenario involving a woman in her 70s, illustrating the inherent challenges in managing older patients, the amplified risks with high-concentration potassium infusions, and the multidimensional approach required in care—from early detection to comprehensive management including patient and family education. The reporting of this study conforms to CARE guidelines. 9
Case presentation
This case report has removed the specific information of identifiable patient. A woman in her 70s was admitted to the Liaoning Cancer Hospital in Shenyang, Liaoning Province, China in February 2023, presenting with cutaneous yellowing coexistent with pruritus, in conjunction with diagnostic imaging indicative of biliary duct obstruction. Initial evaluation by the outpatient department revealed a diagnosis of obstructive jaundice. Patient had no special medical, family, and psychological-social history including relevant genetic information. The patient maintained hemodynamic stability. The clinical examination disclosed pronounced icteric sclera and a generalized pruritus, without any other noteworthy findings.
On day 1, at 11:40, laboratory findings reported a critically diminished potassium level of 2.5 mmol/L. Following the detection of the low potassium level, healthcare professionals recommended the placement of a PICC for the administration of potassium chloride solution. However, the patient declined this recommendation, opting instead for the infusion through a dorsal hand vein. After obtaining informed consent from the patient, and pursuant to medical directives, the patient was administered a 10% solution of potassium chloride (30 mL) combined with sodium chloride (20 mL), which was diluted to prepare a 6% (800 mmol/L) potassium chloride injection solution, at a rate of 5 mL/h via a peripheral micropump. An hourly examination by the nursing staff at 21:00 identified a region of the patient's right hand, proximal to the injection site, exhibiting erythema along the venous pathway, measuring approximately 9cm × 1 cm (Figure 1). Considering her advanced age and reduced sensitivity, the patient reported only minor discomfort, quantified by a numerical rating scale pain score of 1. The patient developed grade 3 phlebitis according to the 2021 Infusion Therapy Standards of Practice. 10

On day 1, at 21:00, nurse identified a region of the patient's right hand, proximal to the injection site, (a) exhibiting erythema along the venous pathway, (b) measuring approximately 9cm × 1 cm.
After discussing the treatment plan with the patient and obtaining consent, the nursing interventions executed involved discontinuation of the medication infusion, removal of the cannula, implementation of a central seal using dexamethasone (10 mg), lidocaine (5 mg), and normal saline (20 mL), immediate application of a 50% magnesium sulfate dressing on the right hand, limb elevation and immobilization, and the administration of a hydrogel dressing, replaced bi-daily. Upon completion of the jaundice treatment on day 9, the patient was discharged, with a directive to return for dressing replacements. However, due to the logistical challenge of a significant commute to the hospital, the patient opted to self-manage the dressing changes at home. During the postdischarge period, regular nursing follow-ups were conducted. On day 13, the patient's dermatological presentation appeared to have intensified, with the manifestation of an ulceration, approximately 2 cm by 1 cm in dimension, evident at the periphery of the venous catheter insertion site on the dorsal aspect of the patient's hand. Additionally, purulent exudate was observed emanating from the ulcer, as depicted in Figure 2. Nevertheless, the patient denied experiencing any significant discomfort at the site of the lesion. The nurse advised the patient to return to the hospital for further treatment. However, the patient declined, expressing a preference to continue with cold compresses at home.

On day 13, a lesion measuring 2 cm by 1 cm surfaced at the location of the venous catheter insertion on the dorsal aspect of the patient's hand, with purulent discharge noted from the ulcerated site.
On day 27, the patient returned to the hospital for intervention. Upon observation, an ulceration, approximately 3 cm by 1 cm in size, was noted over the peripheral venous catheter insertion site on the dorsum of the patient's hand, revealing underlying necrotic vessels (Figure 3).

On day 27, a discernable ulceration, approximately 3 cm by 1 cm in dimension, manifested at the peripheral venous catheter insertion site on the patient's dorsal hand surface, exposing subjacent necrotic vasculature.
A wound care specialist was consulted, and after obtaining the patient's informed consent, wound debridement was performed. As depicted in Figure 4, on day 28, an intricate procedure of wound debridement was executed. Initially, the affected area was meticulously cleansed with isotonic saline solution. Following this, the extremities of the necrotic vessels were fastidiously ligated utilizing medically approved surgical sutures. Subsequently, surgical excision of the necrotic vessels was accomplished. The wound was then purged anew with isotonic saline, supplemented with a 0.5% povidone-iodine solution. This culminated in the installation of a drainage system. For wound coverage, an alginate dressing was meticulously applied, superimposed with a layer of sterilized gauze, and finally secured with a progressively wrapped elastic bandage. The patient was instructed to moderately exercise the affected hand to maintain functionality. Throughout the therapeutic intervention, the patient demonstrated exemplary adherence and reported an absence of adverse experiences.

On day 28, (a) initial cleansing with saline, (b) ligation, and (c) excision of necrotic vessels, (d) subsequent sterilization employing isotonic saline and a povidone-iodine solution, (e) implementation of a drainage system and an alginate dressing, and (f) final envelopment with gauze and an elastic bandage.
In the subsequent days, the wound on the patient's right hand commenced contraction, with the surrounding erythema and edema gradually subsiding and the skin temperature returning to normal. By day 48, the skin on the dorsum of the patient's right hand had nearly completely re-epithelialized, displaying no signs of erythema or edema around the digits. Figure 5 elucidates the progression of cutaneous wound restitution occurring on the dorsal aspect of the patient's right hand. The wrist demonstrated free movement, exhibiting unhindered flexion, extension, internal rotation, external rotation, and gripping action. The patient expressed high satisfaction with both the treatment outcomes and rehabilitation results, particularly noting the restoration of hand functionality. Figure 6 illustrates the timeline of all medical diagnoses and treatments for the patient's phlebitis and skin necrosis.
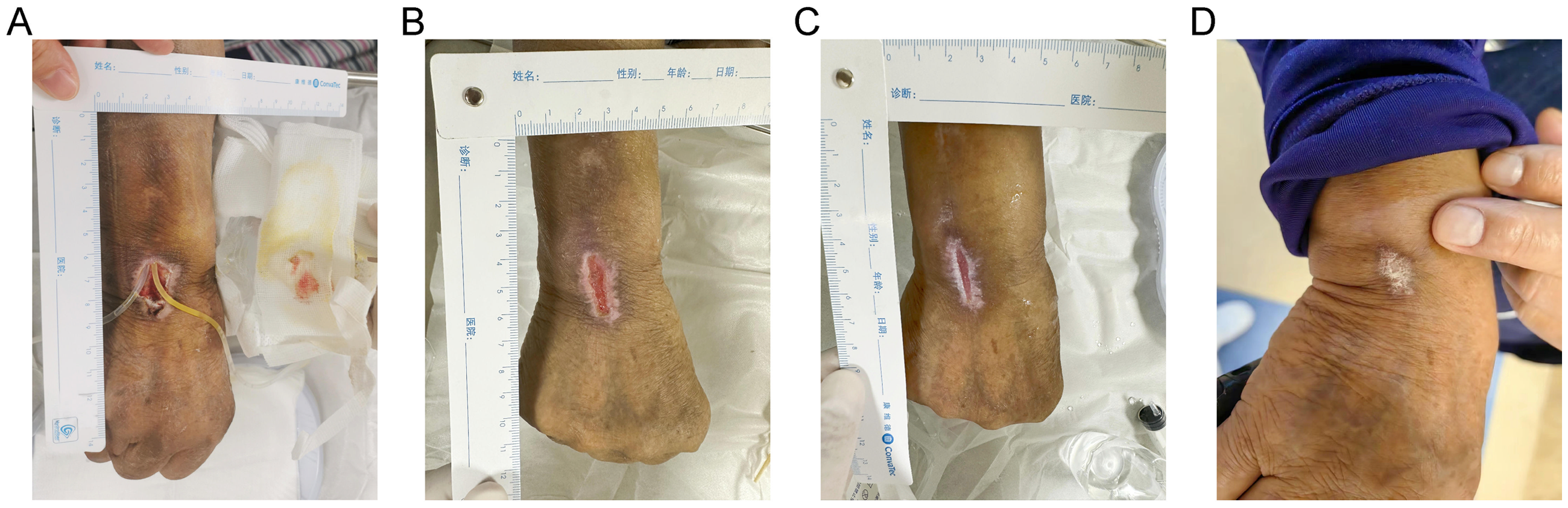
The respective statuses of the patient's wound recuperation on (a) day 34, (b) day 38, (c) day 43, and (d) day 48.

Patient's medical progression timeline.
Insights and lessons learned
Causal factors of the case
Several key factors contributed to the development of complications in this case. A significant patient education gap led to the refusal of PICC placement, highlighting the need for enhanced communication about KCl administration risks.11,12 The inherent properties of potassium chloride played a crucial role, as its corrosive nature increases vascular endothelial permeability and leads to fluid extravasation when administered peripherally at high concentrations. 13 Patient vulnerability was another contributing factor, as advanced age and reduced sensitivity masked early warning signs and delayed intervention.14,15 Finally, the use of superficial peripheral access for concentrated KCl administration, rather than the recommended deep or central venous access, further contributed to the adverse outcome. 16
Reflective thoughts on peripheral venous administration of potassium chloride
Patient education and monitoring protocols were established to ensure safe potassium administration. Healthcare providers emphasized the risks of potassium extravasation to patients and caregivers. 12 For intravenous potassium administration, deep veins or peripheral veins with good elasticity (median cubital, basilic, or cephalic) were recommended, avoiding compromised limbs and superficial veins. 11 Enhanced nursing surveillance was crucial, particularly for patients with decreased sensitivity, advanced age, or communication difficulties, as localized pain could indicate medication leakage even with positive blood return.17–19
Management of potassium chloride extravasation
Immediate management of potassium chloride extravasation requires stopping the infusion, aspirating residual medication, elevating and immobilizing the affected limb, and applying local treatments including 0.25% lidocaine block and 50% magnesium sulfate dressing.20–22 For cases progressing to skin necrosis, treatment involves careful debridement of necrotic tissue, followed by wound care with saline cleansing, povidone-iodine disinfection, and alginate dressing application.23–25 Key aspects of follow-up care include regular wound assessment, monitoring for infection, and implementing early functional exercises to prevent complications.26,27 Nutritional support and patient education are essential for optimal wound healing outcomes.
Discussion
Current medical guidelines advocate for the use of PICC when administering potassium chloride infusions in hypokalemic patients to reduce the risks of extravasation and other complications linked with peripheral intravenous administration. Nevertheless, practical considerations frequently necessitate deviations from these guidelines. Financial limitations, patient preferences, and the logistical challenges associated with PICC lines often result in a continued reliance on superficial vein injections for potassium chloride delivery. While this approach is relatively common, it does not completely prevent the potential adverse effects on veins due to the irritative nature of potassium chloride solutions. The present case study, involving a septuagenarian female suffering from acute hypokalemia who underwent treatment via peripheral venous administration of a 6% potassium chloride solution, underscores the essentiality of swift identification and strategic management of associated complications like extravasation and phlebitis. Furthermore, this report confers a wealth of practical knowledge and enlightening lessons for medical practitioners confronted with analogous clinical circumstances.
The foremost lesson gleaned from this case is the absolute necessity of judicious venous site selection and rigorous monitoring of intravenous infusions. The phenomenon of extravasation in the present scenario could be largely attributed to the utilization of a superficial peripheral vein as the conduit for a hypertonic irritant. The perils associated with infusing concentrated potassium chloride solutions into peripheral veins are widely recognized and include inflammation and phlebitis, cutaneous necrosis, and protracted periods of recuperation.5,6,28
In retrospect, the choice of a central venous catheter or PICC line would have been more suitable for this patient, considering the higher concentration and irritant quality of the solution being infused.10,29 In scenarios where the use of a PICC line or central venous catheter is not practicable, it is of paramount importance to verify that the chosen peripheral vein is sufficiently deep and robust to withstand the infusion. Regular evaluation and meticulous monitoring of the infusion site are also integral to early detection of complications.
Our case highlights the complications following peripheral infusion of 6% KCl in a woman in her 70s leading to severe phlebitis and cutaneous necrosis. At the same time, the case described in the previous literature pertains to a 69-year-old male patient who experienced KCl-induced central vein phlebitis after infusion via a malpositioned central venous catheter. 30 Both patients were of advanced age, which may have predisposed them to certain complications due to age-related changes in physiology and vascular structures. Both cases underscore the risks associated with intravenous potassium repletion, albeit via different venous access points. In both scenarios, complications arose shortly after the administration of the KCl solution, emphasizing the need for vigilant monitoring during such infusions. The adverse events in both cases underscore the potential hazards of intravenous KCl administration, irrespective of its route—be it peripheral or central. It is evident that vascular assessments are crucial before initiating such infusions.
The treatment of potassium chloride extravasation presented in this case report has several limitations. Firstly, while the interventions are effective in the specific context described, they might be perceived as routine. These interventions could share similarities with treatments for other types of extravasation events. Although the described methods are recommended for potassium chloride extravasation, their effectiveness may vary when applied to other agents. Secondly, this study largely relies on well-established methods in the field of medicine extravasation management. These methods, while rooted in patient safety and proven effectiveness, may not offer the innovative approaches that some practitioners desire. Lastly, the case report describes specific instance and may not be universally applicable. The unique characteristics of the patient and specific clinical conditions may limit the generalization of our findings to a broader patient population. Recognizing this limitation, this case report should be viewed as a detailed account of an individual patient's response to a particular set of interventions. It contributes to a larger mosaic of clinical evidence but should not be considered definitive. Further research involving larger sample sizes and diverse patient demographics is necessary to validate and extend our findings, enhancing their relevance and applicability to varied clinical settings.
Conclusions
This case study furnishes essential lessons concerning the prevention and management of complications resulting from the peripheral intravenous administration of potassium chloride solutions. Careful selection and monitoring of infusion sites, amplified patient education, and prompt, multidisciplinary management of complications are crucial in averting severe outcomes like skin necrosis. This report confers crucial insights that can bolster the safety and efficacy of potassium supplementation in patients with acute hypokalemia.
Footnotes
Authors’ contributions
HL and HJ contributed to the conceptualization, project administration, supervision, and validation of this work. The tasks of data curation, formal analysis, investigation, methodology, visualization, and the drafting of the original manuscript were performed by FL. The tasks of data curation and investigation were performed by TW, LW, SZ, YZ, and YR.
Data availability statement
The data that support the findings of this study are included within the article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval and consent to participate
This study was reviewed and approved by the Ethical Committee of the Liaoning Cancer Hospital & Institute, Shenyang, Liaoning Province, China, with the approval number: KY20230703. The patient provided written informed consent to participate in the study.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Empowerment Program—Scientific Research Fund Project (KC2023-JX-0288-FQ08) from Beijing Science and Technology Innovation Medical Development Foundation.
Informed consent
Written informed consent was obtained from the patient for publication of this case report, including all clinical details, images, and associated data. The patient was thoroughly informed about the purpose and implications of the study. The patient was assured that all possible steps would be taken to ensure anonymity and confidentiality. The written consent included permission for the use, analysis, and publication of the images and clinical data for research and educational purposes.
