Abstract
Abstract
Introduction:
Near-infrared fluorescence imaging has emerged as a promising modality in parathyroid localization, as parathyroid glands emit natural autofluorescence when excited by near-infrared lasers.1–5 Two devices have been approved by the Food and Drug Administration for performing real-time identification of parathyroid glands during surgery: Fluobeam (Fluoptics) and PTeye Parathyroid Detection System (Medtronic). 1 The Fluobeam system utilizes an optical head that emits a near-infrared laser as the light source and subsequently detects the signal in surgical tissue with a near-infrared sensitive camera, reproducing a real‐time grayscale image enhanced by autofluorescing tissues on a computer monitor. Conversely, the PTeye System includes a sterile probe, which is used in proximity to the tissue to analyze its optical properties, producing distinct audio and visual signal. The practical utility of these autofluorescent devices and ultimate impact on clinical outcomes remain controversial.
Materials and Methods:
As part of a multicenter study of autofluorescence in thyroid and parathyroid surgery, the authors utilized the FluobeamLX (Fluoptics) system for ~100 thyroid surgery cases. 5 To utilize the FluobeamLX autofluorescent system, both the surgeon headlights and overhead operating room lights must be turned off or away from the surgical field. The camera is placed over the surgical site or specimen, and the live autofluorescent video is shown on an adjacent computer monitor. Six unique patients undergoing thyroid surgery with varying pathology reports using parathyroid autofluorescence are illustrated, juxtaposing both live surgical video and autofluorescent imaging using Fluobeam (Fluoptics).
Results:
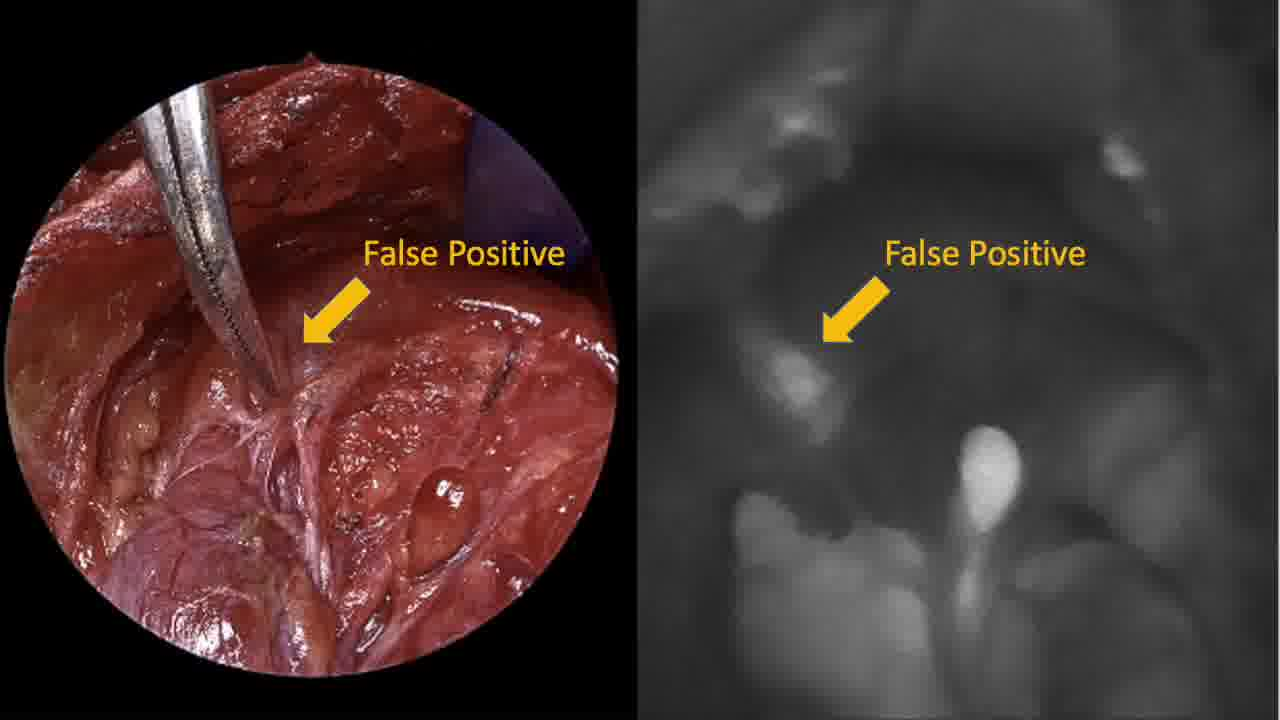
Autofluorescent imaging of the thyroid surgical site is demonstrated for all six patients, exhibiting the autofluorescent signal intensity of parathyroid tissues. In some patients, autofluorescent imaging of the specimen is utilized to identify parathyroid tissue adherent to or encased within thyroid tissue, thyroid cancer, or lymphatics. Specific examples of false positive and false negative autofluorescence are illustrated in select cases. Operative time was increased by ~15 to 20 minutes for each surgery because of the use of this parathyroid autofluorescence technology. Specifically, for each instance of use of the Fluobeam system in any particular surgery, about 5 minutes was needed to set up the camera (e.g., removing lights from the surgical field), perform the autofluorescence evaluation, and reset the lights to the surgical field.
Conclusion:
In this unique video demonstration juxtaposing live surgical images with autofluorescent imaging, both the promise and limitations of this technology can be appreciated. In its current form, the Fluobeam (Fluoptics) system can effectively identify parathyroid glands through autofluorescence, both in the surgical field and in the surgical specimen. 5 However, there are several limitations that will likely limit its routine use in thyroid surgery, including increased operative time and interruption to the normal flow of surgery (e.g., necessity of turning off lights and looking away from the surgical field to evaluate potential parathyroid glands), false positive autofluorescence (e.g., brown fat, lymph nodes, thyroiditis, and cancer), and false negatives (uncovered parathyroid glands).
Disclosure Statement:
J.M.: none. M.Z.: no conflicts of interest/disclosures relevant to this publication. For clarity I participated in a multicenter prospective study (PI, Eren Berber) using Fluobeam (Fluoptics), for which neither I nor my institution received research funding or other compensation. The Fluobeam system was purchased commercially by MD Anderson Cancer Center. Disclosures not relevant to this publication: Clinical trial funding (to institution, MD Anderson Cancer Center) from Merck and Eli Lilly.
Runtime of video: 8 mins 1 sec
IRB Approval:
This case series review was conducted after approval by the MD Anderson institutional review board. As this is an anonymized observational study, patient consent was waived.


Get full access to this article
View all access options for this article.
