Abstract
Epiphytic yeasts are promising biocontrol agents of plant diseases but preserving and transferring them to the field is challenging. Here, we studied six cost-effective lyophilization protective agents to preserve seven strains of Amazonian yeast species isolated from the phyllosphere of native cacao (Theobroma cacao) in Peru. We evaluated the viability of yeasts at 30 and 90 days post-lyophilization in vitro, and their survival after controlled inoculation on cacao fruits in the field. The best protective agents were maltodextrin, honey + skim milk, and honey. Wickerhamomyces anomalus KLG-014 and Wickerhamomyces sp. EGZ-38 showed higher than 97.3% viability after 30 days when lyophilized with maltodextrin. Additionally, Candida sp. KLG-103 showed a viability greater than 50% after 30 days when lyophilized with honey + skim milk. At 90 days, W. anomalus KLG-014, Hannaella theobromatis KLG-063, and Kwoniella heveanensis EGZ-07 showed a viability greater than 20%, with the latter showing an outstanding 100% viability, when lyophilized with honey + skim milk. Conversely, sodium alginate was the least protective agent, as yeast showed 0% viability. In the field, W. anomalus KLG-014, K. heveanensis EGZ-07, Debaryomyces hansenii EGZ-31, and Wickerhamomyces sp. EGZ-38 were successfully re-isolated from the surface of cacao fruits under all treatments after 30 days, except for sodium alginate. This was corroborated via morphological and molecular evidence. This study demonstrates that maltodextrin, honey, and skim milk are suitable for ensuring the in vitro viability of biocontrol yeasts up to 90 days after lyophilization, and their survival up to 30 days after inoculation on cacao fruits in the field. This is a first step toward the development of a biocontrol alternative to mitigate cacao pathogens using native microorganisms from the Amazon in Peru.
Keywords
Introduction
Fungicides and other agrochemicals are effective for controlling crop diseases. 1 However, these chemicals contribute to pollution and can negatively impact human health.2,3 The use of microorganisms, such as epiphytic yeasts, as biocontrol agents of crop pathogens is a sustainable alternative to agrochemicals.3–5 Many biocontrol products are stored and commercialized as dry powder, so various methods for dehydrating them have been developed, simplifying the procedure and increasing their storage period while maintaining biocontrol characteristics.6,7 Several studies indicate that lyophilization is one of the most effective methods to accomplish this, 8 and can be used for preserving yeasts.9,10 Lyophilization allows the conversion of liquid yeast cultures into a solid, dry, biologically active, and stable form adequate for storage. After storage, it can then be easily rehydrated in a solution, increasing its shelf life and facilitating further applications in agriculture.
Lyophilization is more stable than conventional air-drying methods.11,12 However, during lyophilization, microbial cells may suffer deadly damage to their cellular structure due to ice formation, excessive dehydration, increased concentration of solutes inside cells, and osmotic imbalance.9,13,14 Cells can tolerate high levels of dehydration. However, if tolerance limits are exceeded, viability may be lost.15,16 To reduce these effects, protective agents or microencapsulants are commonly used, which help maintain cell viability. Also, the use of protectants in formulations is an effective strategy to enhance yeast activity after lyophilization. 17 Various compounds, including saccharides, antioxidants, and emulsifiers, are frequently used as protectants to maintain yeast viability during the drying process.6,18–20 Studies have obtained positive results in maintaining the viability of yeasts and other fungi after lyophilization. 6 After lyophilization and storage, cells may die but some may survive, and the survival of 0.1% of the original cell population is sufficient to ensure the continued survival of the microorganism. 21
Recent studies in Peru have revealed the presence of epiphytic yeasts from the phyllosphere of cacao (Theobroma cacao), 22 and cacao-related hosts. 23 These yeast species have shown biocontrol potential against pathogens such as Moniliophthora roreri, 22 the main cacao pathogen in Peru and Latin America. 24 These yeasts are currently used in the development of a new biocontrol package that can be applied in the field to mitigate this pathogen. However, an effective way to lyophilize, store, and transfer them to the field is missing. Therefore, the objectives of this study were: (1) to evaluate the most efficient protective agents to improve the viability of native yeasts from Amazonas, Peru during the lyophilization process, (2) to determine the viability of lyophilized yeasts after lyophilization, and (3) to determine the survival of lyophilized yeasts in the field after inoculations on cacao fruits.
Materials and Methods
Yeast strains and protective agents evaluated
The yeast strains used in this study are from the native phyllosphere of fine-flavor cacao grown in Amazonas, Peru, and the Ichigkat Muja-Cordillera del Cóndor National Park.23,25 All these strains are maintained in the collection of the Plant Health Research Laboratory at the National University Toribio Rodríguez de Mendoza of Amazonas (UNTRM), Peru (Table 1, Fig. 1). The yeasts were selected based on their high in vitro biocontrol potential against M. roreri previously evaluated by researchers from the institution.23,25
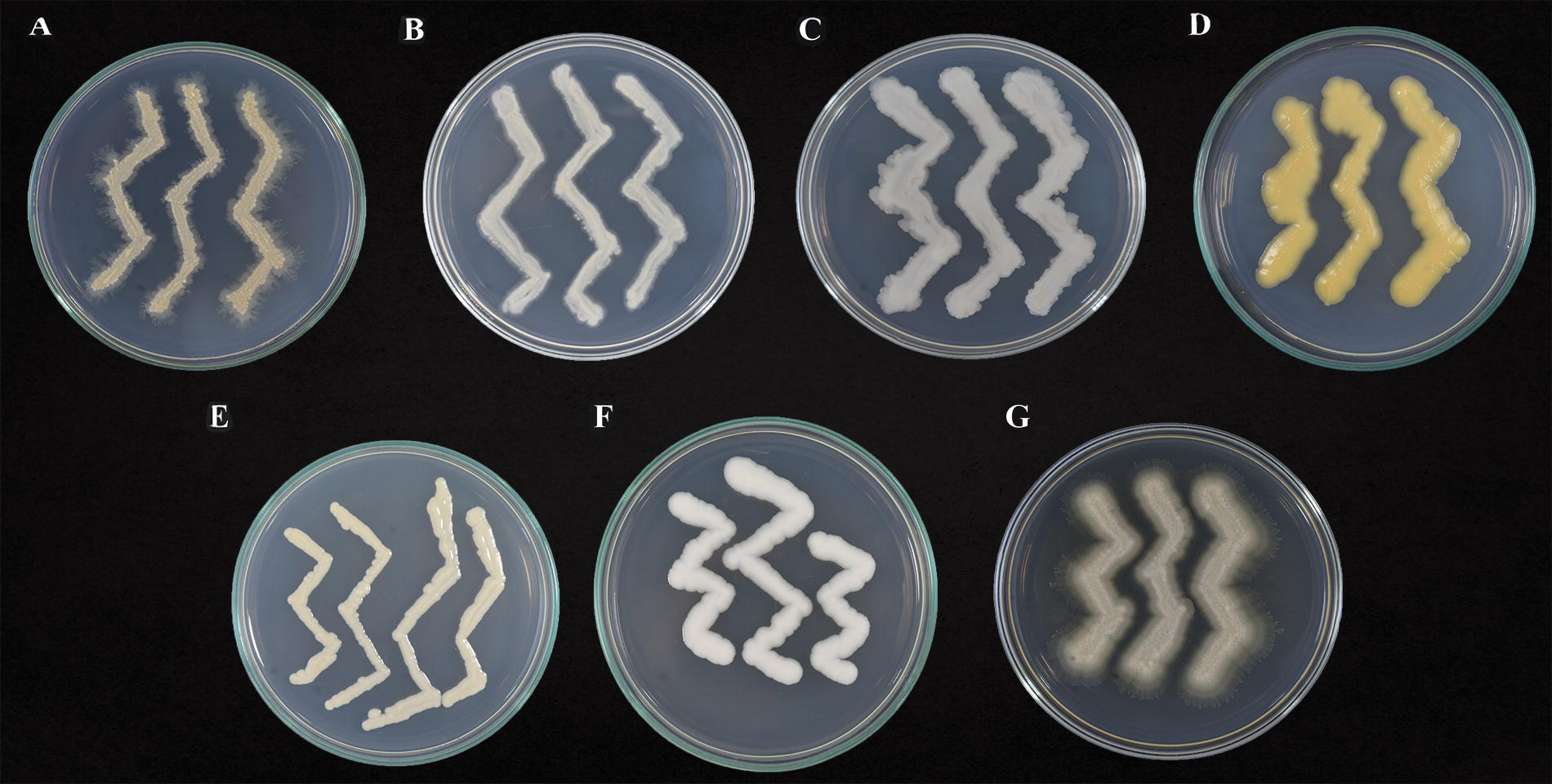
Yeast strains used in this study after 2 weeks of growth on potato dextrose agar medium.
Yeast Strains and Species Used in This Study

ITS, internal transcribed spacer region of the rDNA (ITS1-5.8-ITS2) sequence; LSU, large sub-unit or 28S region of the rDNA partial sequence.
Six suspension media were evaluated as protective agents. These included 15% (w/v) maltodextrin (Vasuman International, Gujarat, India), 15% gum arabic (Vasuman International), 15% sodium alginate (Spectrum Chemical MFG Corp., New Brunswick, NJ, USA), 15% skim milk (NESCAFÉ®, Nestlé, Vaud, Switzerland), 15% honey (ALTA MONTAÑA, Amazonas, Peru), and 7% honey (ALTA MONTAÑA) + 7% skim milk (NESCAFÉ®). These cost-effective protective agents were selected based on previous studies on other microorganisms.9,11
Preparation of containers for lyophilization
Containers for lyophilization were prepared according to Ref. 26 with some modifications. Pure yeast cultures were transferred to 5 mL of liquid Luria-Bertani (LB) medium contained in 50 mL Falcon tubes. Tubes were incubated at 28°C for 12 hours in an incubator shaker (TOU-50/120 SERIES, MRC, Harlow, UK) at 6 g, then centrifuged for 30 minutes at 3,800 g. The supernatant was discarded. The biomass pellet was then suspended in 5 mL of each of the protective agents tested and incubated at 28°C for 12 hours at 6 g. These agents had been sterilized at 120°C for 45 minutes to ensure they were completely sterile. The samples were pre-frozen at −20°C for 12 hours. Afterward, they were frozen for 24 hours at −80°C prior to lyophilization.
Lyophilization
We used the lyophilizer [LABCONCO, 117 (A65312906), MO, USA] at the Food Engineering and Postharvest Research Laboratory of UNTRM. Falcon tubes with frozen samples were lyophilized at 0.008 mbar and −84°C for 48 hours. Samples were stored at room temperature.
Viability test
Viability was determined right before lyophilization and after 30 and 90 days. Viability was expressed as the number of colony-forming units per milliliter (CFU/mL). 5 Serial dilutions of 10−5 and 10−6 were performed for each protective formulation at each time. The dilutions were plated in triplicate by applying 80 µL onto Petri dishes containing Potato Dextrose Agar and incubated for 48–72 hours. Once the colonies grew, a manual CFU count was performed, and the concentration per milliliter (CFU/mL) was calculated as in the previous study. 27 Lyophilized samples stored for 30 and 90 days were rehydrated in 5 mL of sterile distilled water at room temperature for 3 hours, and the same procedure described above was repeated to calculate the CFU/mL. All results presented in this study are based on three independent trials. For this experiment, we included a lyophilization treatment without any protective agent.
Survival of yeasts inoculated onto cacao fruits in the field
To determine field survival, the rehydrated lyophilized yeasts were inoculated onto T. cacao fruits in the field. A plot of approximately 100 × 100 m in Cajaruro district, Utcubamba province, Amazonas department, Peru (5°44’20.4”S; 78°21’14.4”W) was identified for this experiment, where 30–40-day old fruits in good phytosanitary condition were selected for inoculation. Fruits were disinfected with 70° ethanol, and using a sterile sprayer and surgical gloves, 1.6 μL of each yeast-protectant suspension was inoculated into all experimental units (cacao fruits). After inoculation, each fruit was protected with a Raschel mesh to reduce contact with insects or other potential field contaminants. A completely randomized factorial design was used with three repetitions: factor A (six protective agents, the same used in the viability experiment) and factor B (six yeast strains and the control, i.e., no yeast). A total of 144 cacao fruits were inoculated.
To evaluate whether the lyophilized yeasts survived in the field, re-isolations were performed 30 days after inoculation. For this, the inoculated cacao fruits were washed with sterile ultrapure water. Samples were stored in thermal coolers and transported to the laboratory for yeast re-isolation following the procedures of Ref. 27 Once the isolates were in pure culture, they were analyzed both morphological and molecularly to ensure that the re-isolated yeasts were the same as those inoculated. The sequences of the isolates were analyzed and compared to ensure they were the same species inoculated in the field. For this, we followed the molecular procedures of Ref.22 Sequences obtained were deposited in GenBank (Table 2).
Molecular and Morphological Characteristics of the Re-Isolated Strains Retrieved 30 Days After the Inoculation of Original Strains on Cacao Fruits
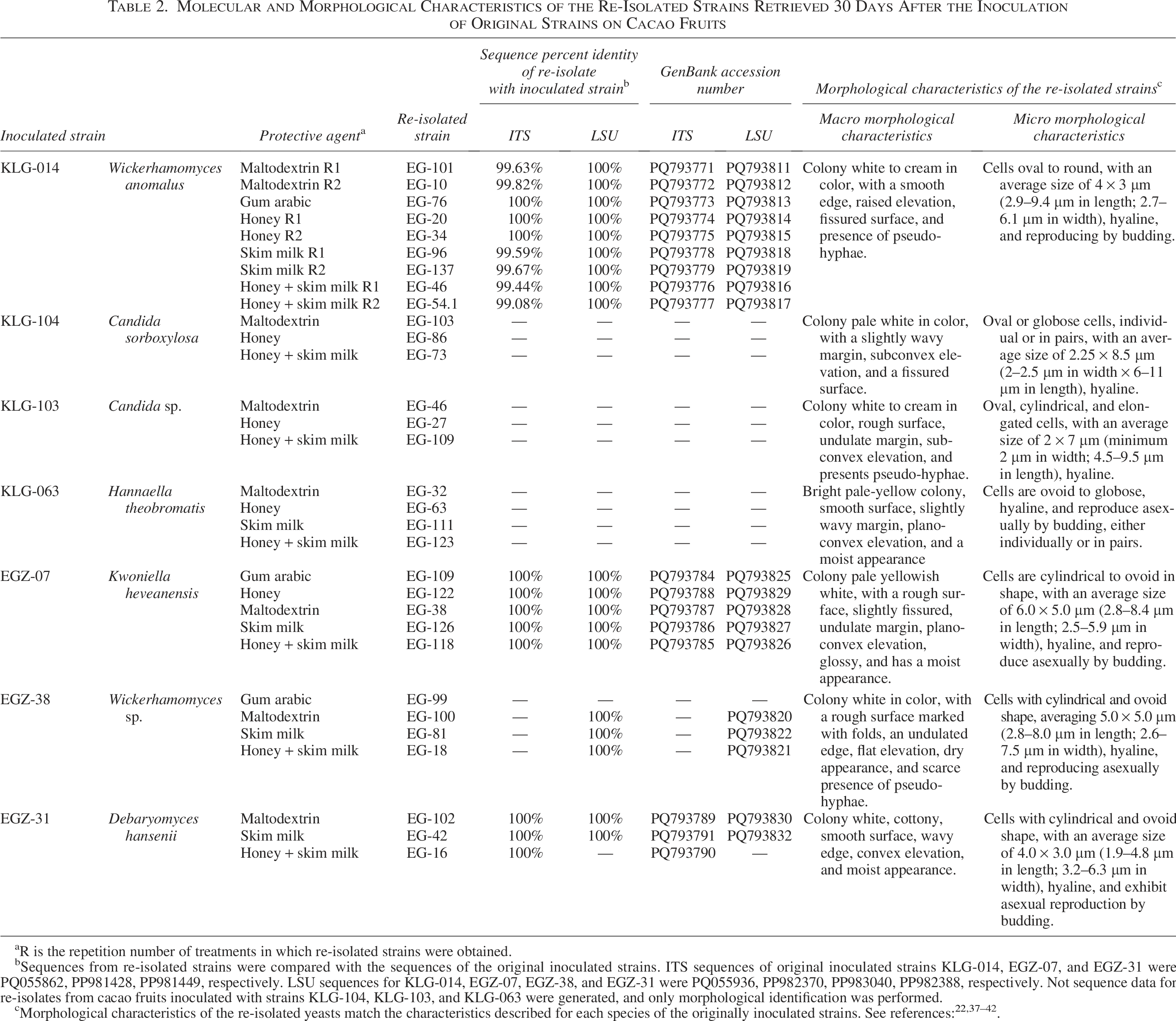
R is the repetition number of treatments in which re-isolated strains were obtained.
Sequences from re-isolated strains were compared with the sequences of the original inoculated strains. ITS sequences of original inoculated strains KLG-014, EGZ-07, and EGZ-31 were PQ055862, PP981428, PP981449, respectively. LSU sequences for KLG-014, EGZ-07, EGZ-38, and EGZ-31 were PQ055936, PP982370, PP983040, PP982388, respectively. Not sequence data for re-isolates from cacao fruits inoculated with strains KLG-104, KLG-103, and KLG-063 were generated, and only morphological identification was performed.
Statistical analysis
Viable cells were determined by calculating the proportion of CFUs compared with the values obtained before lyophilization, and the viability over time was evaluated as a percentage. For statistical analysis, the Shapiro-Wilk test was performed to verify the normality of the groups. An analysis of variance (ANOVA), 28 was performed to evaluate the differences between treatments. When normality was not observed, data were log-transformed and checked for normality before conducting ANOVA. To compare the treatments, the Duncan test 29 was applied to identify significantly different agents.
Results
Viability and stability after lyophilization process
For W. anomalus KLG-014, the most effective protective agent was honey + skim milk, with 50% viability at 30 days and 24.2% at 90 days. This freeze-dried yeast with the other agents had less than 1% viability, maintaining this average up to 90 days. On day 30, maltodextrin was significantly different from the other agents, maintaining a viability of 97.3% (Fig. 2A). For C. sorboxylosa KLG-104 and Candida sp. KLG-103, the most efficient protective agents were honey and maltodextrin (Fig. 2B and C). In the case of Candida sp. KLG-103, honey + skim milk stood out at 30 days with a viability of 57.1%, while honey was the most effective agent at 90 days with a viability of 15.8% (Fig. 2C). For Hannaella theobromatis KLG-063, the best protective agent was honey + skim milk, maintaining its viability at 66.7% and 22.2% at 30 and 90 days, respectively. These agents were significantly different from the others. The other agents showed a rapid decline, with reduced viability at 90 days (Fig. 2D). For Kwoniella heveanensis EGZ-07, the best protective agent was honey + skim milk, consistently maintaining 100% viability at both 30 and 90 days, followed by gum arabic and skim milk, which were significantly different from the other agents (Fig. 2E). For Debaryomyces hansenii EGZ-31, most agents showed a notable decrease in viability at both 30 and 90 days. However, maltodextrin maintained the viability of this strain at 60.5% and was significantly different from the other agents at 30 days. At 90 days, skim milk showed significant differences from the other agents, making these two agents the most effective for this strain (Fig. 2F). For Wickerhamomyces sp. EGZ-38, maltodextrin was the best protective agent, maintaining its viability at 100% at 30 days and 5% at 90 days. The other agents show a drastic decrease in viability at both 30 and 90 days (Fig. 2G).
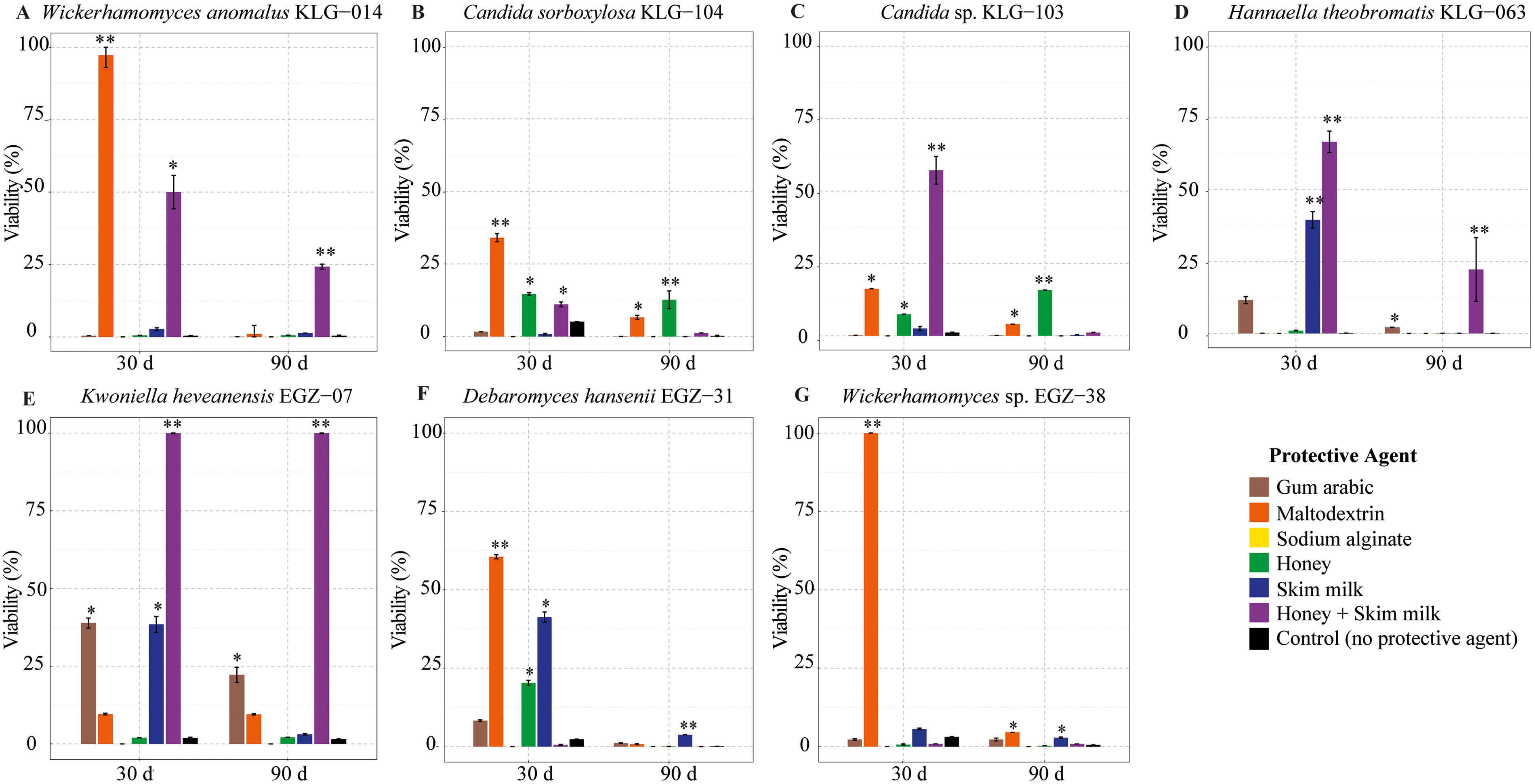
Viability of yeast strains after 30 and 90 days after lyophilization.
In general, viability decreased significantly at 90 days. The most pronounced reduction occurred between day(s) 0 and 30. Some agents were able to maintain a higher percentage of viability during this period, with maltodextrin, honey + skim milk, and honey being the most effective in maintaining viability across all species. These agents were statistically different from the others. Conversely, the worst performing protective agent was sodium alginate, which had 0% viability at both 30 and 90 days, demonstrating that this agent is not suitable for these yeast species (Fig. 2). The effectiveness of the evaluated agents varied depending on the species, highlighting the importance of adjusting the protective agent according to the microorganism under evaluation.
Survival of yeasts inoculated in the field
The inoculated fruits were processed 30 days after inoculation to recover the originally inoculated yeasts. The re-isolation process revealed the growth of mainly the inoculated yeasts with occasional growth of microorganisms not deliberately inoculated, including other yeast species, bacteria, and fungi. The presence of these organisms was attributed to exposure to a natural environment, although they were detected in minimal quantities within the experimental units. To ensure the specific recovery of the inoculated yeasts, the growing colonies were characterized, and successive subcultures were performed to obtain axenic cultures. The yeasts recovered in pure cultures were subjected to morphological and molecular characterization to confirm their identity and activity. The results were compared with the original strains to verify their survival and ensure the integrity of the experiment (Table 2).
After 30 days of inoculation of cacao fruits in the field, lyophilized yeasts showed survival in most of the evaluated treatments (Table 2). The species W. anomalus KLG-014, K. heveanensis EGZ-07, D. hansenii EGZ-31, and Wickerhamomyces sp. EGZ-38 survived and were successfully recovered in all protectant agents, except for sodium alginate. On the other hand, C. sorboxylosa KLG-104 and Candida sp. KLG-103 showed survival only in three protectant agents: maltodextrin, honey, and honey + skim milk. Similarly, H. theobromatis KLG-063 survived in three protective agents: maltodextrin, skim milk, and honey + skim milk. None of the evaluated strains survived when lyophilized with sodium alginate. In all cases, the controls (which consisted only of the inoculation of lyophilized protectant agents without yeast) did not show yeast growth. All re-isolations were the same as the original inoculated strains based on morphological and molecular corroboration (Table 2).
Discussion
We evaluated the efficacy of six protective agents in the lyophilization of Amazonian native yeast from the cacao phyllosphere in Peru. These protective agents were chosen because they were cost-effective and/or readily available. The viability of the strains was analyzed 30 and 90 days after lyophilization, showing variations in the results depending on the species and strain studied. The analysis revealed that honey + skim milk, and maltodextrin were the most effective protective agents, as they showed higher viability in most yeast species evaluated. Conversely, sodium alginate was the least effective, showing 0% viability, both at 30 and 90 days after lyophilization, revealing that this agent is not suitable for preserving these yeasts. No studies have reported the use of lyophilized microorganisms with sodium alginate. However, it has been widely studied as a micro encapsulant for essential oils. 30 Skim milk and gum arabic showed very low viability in most strains. Literature suggests that the minimum survival rate of the original cell population to maintain the microorganism’s viability after lyophilization is 0.1%. 21 Based on this criterion, all the protectant agents evaluated, except for sodium alginate, can be considered suitable for the preservation of these yeasts.
Previous studies have evaluated different protective agents for the lyophilization of yeasts. For example, the use of 10% skim milk powder as a protective agent of Candida sake cells yielded 85% viability right after lyophilization. 18 They also evaluated the use of skim milk combined with other sugars as a protective agent of C. sake, finding that viability without protectors was extremely low, while the combination of protectants was more effective. 6 In our study, a similar pattern was observed when lyophilization was conducted with skim milk alone and combined with honey after 30 days of lyophilization. We obtained viability values around 40% in H. theobromatis KLG-063, K. heveanensis EGZ-07, D. hansenii EGZ-031 when lyophilized with skim milk, and between 50% and 100% in W. anomalus KLG-014, Candida sp. KLG-103, H. theobromatis KLG-063, K. heveanensis EGZ-07 when lyophilized with honey + skim milk. In another study, the combination of sucrose, monosodium glutamate, and skim milk was the most effective lyophilization protective medium for the yeast Pichia membranifaciens, achieving a viability of 76.80%. 6 Moreover, this formulation maintained its biocontrol efficacy against citrus pathogens, highlighting its potential for practical applications in postharvest fruit protection. 6 Therefore, the findings in this study are promising to preserve and transfer the studied yeasts to the field in future bio control experiments against cacao pathogens.23,25
In this study, we also evaluated the long-term stability of the different protective agents on yeast viability. In general, we found that yeast viability after 90 days is reduced compared with 30 days in all treatments. The reduction of viability during storage after lyophilization over time has been commonly reported in previous studies.31–33 Remarkably, the viability of K. heveanensis EGZ-07 when lyophilized with honey + skim milk was not affected (100% viability). We highlight the need to identify the limits of the storage period with the different protective agents and yeasts. Additionally, the conditions of storage should be considered in future experiments. It has been shown, for example, that storing lyophilized C. sake cells at 4°C reduces death of cells over time. 6 Therefore, adjusting the temperature conditions during storage may positively impact the viability of the studied yeasts.
Yeasts can survive the dehydration process by anhydrobiosis, cytoplasmic vitrification, or membrane fluidity. During dehydration, some yeasts can enter a state of anhydrobiosis, reducing their metabolism to a minimum and preserving their viability until rehydration. 34 Another dehydration survival mechanism is cytoplasmic vitrification, in which molecules, such as trehalose, replace intracellular water, forming an amorphous matrix that protects proteins and cellular structures from osmotic and oxidative stress damage. 35 Additionally, membrane fluidity plays a fundamental role in cellular adaptation, as modifications in lipid composition help maintain membrane integrity during dehydration and rehydration. 36 Together, these mechanisms may explain how the yeasts studied retained their viability after lyophilization, which is crucial for their application in biotechnology and biocontrol.
In the field experiment, all the evaluated yeasts demonstrated the ability to survive 30 days after being inoculated onto cacao fruits. This contributes to the understanding of the effectiveness of protective agents during lyophilization and their impact on the viability of yeasts with biocontrol potential in the field. The fact that yeasts were successfully retrieved from the cacao surface after 30 days of inoculation also confirms their epiphytic role in nature. 22
This study not only strengthens our knowledge of yeast preservation strategies but also opens new possibilities for their application in the sustainable management of agricultural diseases, focusing on long-term stability and effectiveness under natural environmental conditions. This study represents the first thorough analysis of the survival of lyophilized yeasts under laboratory and field conditions in Peru, achieving positive results in the survival of strains with biocontrol potential after lyophilization. Finally, this is a first step toward the transferring of epiphytic yeasts to the field so they can be used as potential biocontrol agents of cacao pathogens.
Conclusions
Lyophilized yeast viability decreased after 30 and 90 days, but effective protective agents were identified. The most effective protectants were maltodextrin, honey + skim milk, and honey, which best preserved viability in most strains. In contrast, sodium alginate was the least effective, showing 0% viability in all instances. The effectiveness of protective agents varied by species, highlighting the importance of proper lyophilization agent selection. After 30 days on cacao fruits, most freeze-dried inoculated yeasts survived and were successfully recovered, confirming their epiphytic ecological role. Some strains, such as W. anomalus KLG-014, K. heveanensis EGZ-07, D. hansenii EGZ-31, and Wickerhamomyces sp. EGZ-38, showed a greater survival rate, while C. sorboxylosa KLG-104, Candida sp. KLG-103, and H. theobromatis KLG-063 survived only with certain agents. Thus, freeze-drying with appropriate protective agents is a promising strategy for maintaining yeast viability and functionality, with potential applications in disease management, particularly in cacao cultivation.
Authors’ Contributions
E.G.-Z. Conducted viability tests and field survival experiments, collected and analyzed data, wrote original version of the article, and revised the article. K.J.L.-G.: Study conception, design of field experiment, collected data, revised article, approved final version of article. S.M.O.-C.: Study conception, funding acquisition, field logistics, and revised article. J.R.D.-V.: Study conception, supervised all research activities and data analyses, and revised and critically proofread the article.
Footnotes
Acknowledgments
The authors thank Dr. Juan Carlos Guerrero Abad for providing access to the lyophilizer in the Food Engineering and Postharvest Research Laboratory, UNTRM. The authors also thank Mr. Teobaldo Sanchez Zocon, owner of the cacao plot where the field experiment was conducted. Finally, they thank the staff of the Plant Health Research Laboratory at UNTRM for their support during the execution of this research.
Author Disclosure Statement
The authors declare no conflicts of interest.
Funding Information
This study was funded by the Project “Creación e Implementación del Centro de Investigación e Innovación Tecnológica en Cacao de la Universidad Nacional Toribio Rodríguez de Mendoza de Amazonas” (CEINCACAO), CUI N° 2315081, and Project “Exploiting the bacterial, fungal and entomological resources to fight off the principal production problems of cacao in the heart of its center of diversity” (FBI-CACAO) funded by the International Centre for Genetic Engineering and Biotechnology, agreement N° CRP/24/010. Finally, the APC was covered by the Vicerrectorado de Investigación of the Universidad Nacional Toribio Rodríguez de Mendoza de Amazonas, Perú.
