Abstract
Pulmonary arterial hypertension (PAH)–approved vasodilators improve right ventricular (RV) function in patients with PAH. However, whether PAH-approved drugs ameliorate RV morphology and function in lung disease–associated pulmonary hypertension (lung-PH) remains unclear. We aimed to prospectively evaluate the changes in RV volume and ejection fraction (RVEF) in 14 consecutive severe lung-PH patients treated with PAH-approved vasodilators. Severe lung-PH was defined as a mean pulmonary arterial pressure (MPAP) of ≥35 mmHg or an MPAP of ≥25 mmHg with a cardiac index (L/min/m2) of <2. Right heart catheterization and cardiac magnetic resonance (CMR) imaging were performed at baseline and at 3 months after starting sildenafil with or without other PAH-approved drugs. Follow-up was conducted at 3 months in 11 participants; compared with baseline values, MPAP and pulmonary vascular resistance (PVR) decreased by 18% and 37%, respectively. Baseline CMR imaging revealed an elevated RV end-diastolic volume index (RVEDVI; mL/m2) of 117.5 ± 35.9 and a below-average RVEF of 25.2% ± 7.2%; after 3 months, RVEDVI decreased by 23.7% (P = 0.0061) and RVEF increased by 32.9% (P = 0.0165). Among the 11 patients, 3 were thought to be a stable and homogenous subset in terms of background lung disease and medical management administered. These 3 patients exhibited similar ameliorations in PVR and RVEF, compared with the other 8 patients. PAH-approved drug treatment may improve RV dilatation and systolic function among patients with severe lung-PH. This study was approved by University Hospital Medical Information Network Clinical Trials Registry (UMIN-CTR) on September 1, 2013 (UMIN000011541).
Keywords
Pulmonary hypertension (PH) is classified into five subgroups, with group 3 representing the subset of PH associated with lung disease (lung-PH). 1 Supplemental oxygen is typically used to treat lung-PH. Although drugs approved for use in pulmonary arterial hypertension (PAH) are available, they are generally not recommended because of a lack of evidence regarding their efficacy and possible deterioration of ventilation-perfusion mismatch and hypoxia. 2
Guidelines for PH published in 2013 proposed the following definition for severe lung-PH: a mean pulmonary arterial pressure (MPAP) of ≥35 mmHg or an MPAP of ≥25 mmHg with a cardiac index (CI; L/min/m2) of <2.0. 2 These guidelines recommend referral of such patients to centers with expertise in managing PH and chronic lung disease, because of the uncertain pathogenesis and the need for randomized controlled trials targeting this distinct population. The degree of PH is usually modest in most patients with lung-PH, but a small subset can have severely elevated pulmonary arterial pressures (PAPs).3–5 This population may present with distinct vasculopathy and a different response to vasodilating treatment, compared to typical patients with group 3 PH and mild PAP elevation.6–9
Recent studies have shown a prognostic role of right ventricular (RV) indices measured by cardiac magnetic resonance (CMR) imaging in the assessment of PAH.10–12 For example, a right ventricular ejection fraction (RVEF) of <35% has been shown to have an independent negative prognostic impact in PAH, 13 and CMR-derived RV size has been introduced as a clinically useful variable in assessing PAH. 12 A decline in RVEF was an independent prognostic factor in patients treated with PAH-approved drugs, 13 but the prognostic significance of RV morphology and function has not been addressed in patients with lung-PH. We aimed to prospectively investigate the impact of PAH-approved drugs on RV morphology and function in patients with severe lung-PH.
METHODS
This single-arm prospective study was approved by the Ethics Committee of Hokkaido University Hospital, and written informed consent was obtained from all patients. Between April 2010 and August 2015, we consecutively recruited patients who received a diagnosis of lung-PH based on results of blood testing, pulmonary function tests (PFTs), chest X-ray, high-resolution computed tomography (HRCT) of the chest, and right heart catheterization (RHC). The diagnosis of the background lung disease was based on relevant guidelines for interstitial lung disease or chronic obstructive pulmonary disease (COPD)14,15 or on the pivotal clinical reports for combined pulmonary fibrosis and emphysema (CPFE).16,17 All patients were required to fulfill the definition of severe lung-PH (MPAP ≥ 35 mmHg or MPAP ≥ 25 mmHg with CI < 2.0). 2 Exclusion criteria were as follows: underlying pulmonary disease deemed clinically unstable on the basis of symptoms/signs, chest X-ray, chest HRCT, and PFT in the 6 months before enrollment; inability to perform electrocardiographygated CMR imaging; the use of PAH-approved drug(s) before entry to the study; and the presence of comorbid cardiovascular disorders that affect RV morphology and function. Eligibility was assessed during hospitalization. During hospitalization, patients were placed on salt restriction of less than 6 g/day and received diuretics when suspected of water retention. Supplemental oxygen was given to maintain pulse oxygen saturation (SpO2) of >90%. After this conservative management for at least 1 week, patients underwent RHC for measurement of systolic PAP, diastolic PAP, MPAP, pulmonary arterial wedge pressure, and right atrial pressure using standard methods. Cardiac output (CO) and CI were assessed by thermodilution. Within 1 week from RHC, patients underwent the following investigations: blood testing, which included plasma brain-type natriuretic peptide (BNP) concentration and arterial blood gas analysis; PFTs, which included measurement of the diffusion capacity of the lung for carbon monoxide (DLCO) and DLCO divided by alveolar volume (DLCO/VA); chest X-ray; 6-minute walk distance (6MWD) test; computed tomography; and CMR. During this assessment period, the patients remained in stable condition without any modifications to their PH-related medications.
CMR studies were performed with a 1.5-T Philips Achieva magnetic resonance imaging system (Philips Medical Systems, Best, Netherlands) equipped with master gradients (maximum gradient amplitude: 33 mT/m; maximum slew rate: 100 mT/m/ms). The imaging protocol has been previously described. 18 RV images were analyzed with commercially available software (Extended MR Work Space, ver. 2.6.3, Philips Medical Systems, Amsterdam). We then computed RV end-diastolic volume (EDV) and end-systolic volume (ESV), and the values were corrected for body surface area (RV EDV index [RVEDVI] and RV ESV index). The stroke volume (SV) was calculated as EDV – ESV, and the ejection fraction (EF) was calculated as SV/EDV × 100 (%). CMR analysis was performed by an experienced radiologist (NO-M) who was blinded to the condition of the patients and the modifications in medical treatment, including pulmonary vasodilators, oxygen, or diuretics, if any.
Sildenafil (20 mg, 3 times/day) was used as the first-line vasodilating agent; the addition of another PAH-approved drug was considered when PH was severe and progressive upon baseline assessment. When sildenafil was not tolerated, it was replaced by tadalafil.
A follow-up assessment, including CMR and RHC, was scheduled at 3 months after modifications of PAH-related drugs. Patients followed up for more than 3 months underwent extended assessments that were similar to those for the 3-month assessment. Additional PAH-approved drug(s) were considered at and after the 3-month follow-up when it was deemed necessary to intensify treatment, according to the clinical course and RHC/CMR data.
Descriptive statistics were presented as mean ± SD or as absolute numbers. Clinical parameters obtained at baseline and at 3-month follow-up were compared by paired t test or Wilcoxon signed-rank test, as appropriate. Analyses for the extended follow-up were conducted descriptively. JMP, version 11 (SAS Institute, Cary, NC), was used for statistical analysis. A value of P < 0.05 was considered statistically significant.
RESULTS
Patient characteristics
Sixteen patients received diagnoses of lung-PH during the study period. Among these, 2 did not fulfill the criteria for severe lung-PH on RHC; therefore, 14 patients were enrolled (Table 1). CPFE was the most common underlying lung disease (n = 6), followed by interstitial pneumonia (IP), which included idiopathic pulmonary fibrosis (IPF; n = 2), nonspecific IP (n = 1), and rheumatoid arthritis–associated IP (n = 1); COPD (n = 3); and cystic lung disease (n = 1). All 6 CPFE patients exhibited both emphysematous and fibrotic lesions on chest HRCT, with severely reduced %DLCO (23.2% ± 5.3%, n = 5) and relatively preserved lung function (vital capacity [VC]: 100.8 ± 14.2 mL; forced expiratory volume in 1 second [FEV1]/forced VC [FVC]: 61.2% ± 9.6%; n = 5). In patients with IP, PFT showed low %VC (58.3% ± 15.7%, n = 3) with reduced %DLCO (27.0% ± 11.5%, n = 3). Also, 3 COPD patients showed a slight decrease in %VC (79.3% ± 15.9%) with reduced FEV1/FVC (52.9% ± 12.6%) and %FEV1 (53.2% ± 20.3%). Representative HRCT images of patients with CPFE, IP, or COPD are shown in Figures 1–3.
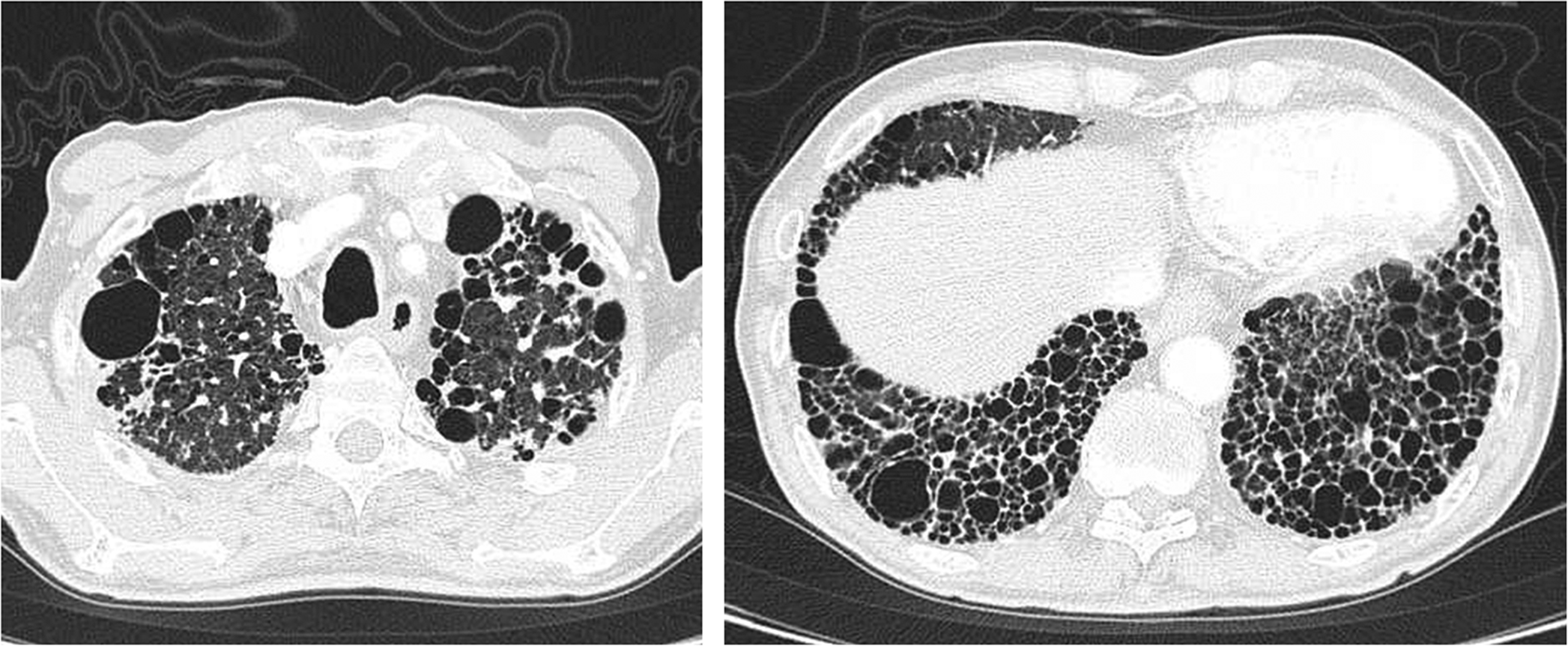
High-resolution computed tomography images of a 66-year-old man with idiopathic pulmonary fibrosis. Fibrotic changes with honeycomb formation are present in the bilateral upper and lower lobes. The patient was noted to have fine crackles on chest auscultation, with reduced VC (1,970 mL), %VC (59%), and %DLCO (13%). VC: vital capacity; DLCO: diffusing capacity of the lung for carbon monoxide.
High-resolution computed tomography images of a 69-year-old man with combined pulmonary fibrosis and emphysema. Emphysematous changes are seen in both upper lobes, along with fibrotic changes in both lower lobes. The patient was noted to have relatively preserved lung function (VC: 4,000 mL; %VC: 103%; FEV1/FVC: 67%; %FEV1: 84%) with reduced %DLCO (34%). VC: vital capacity; FEV1: forced expiratory volume in 1 second; FVC: forced vital capacity; DLCO: diffusing capacity of the lung for carbon monoxide.
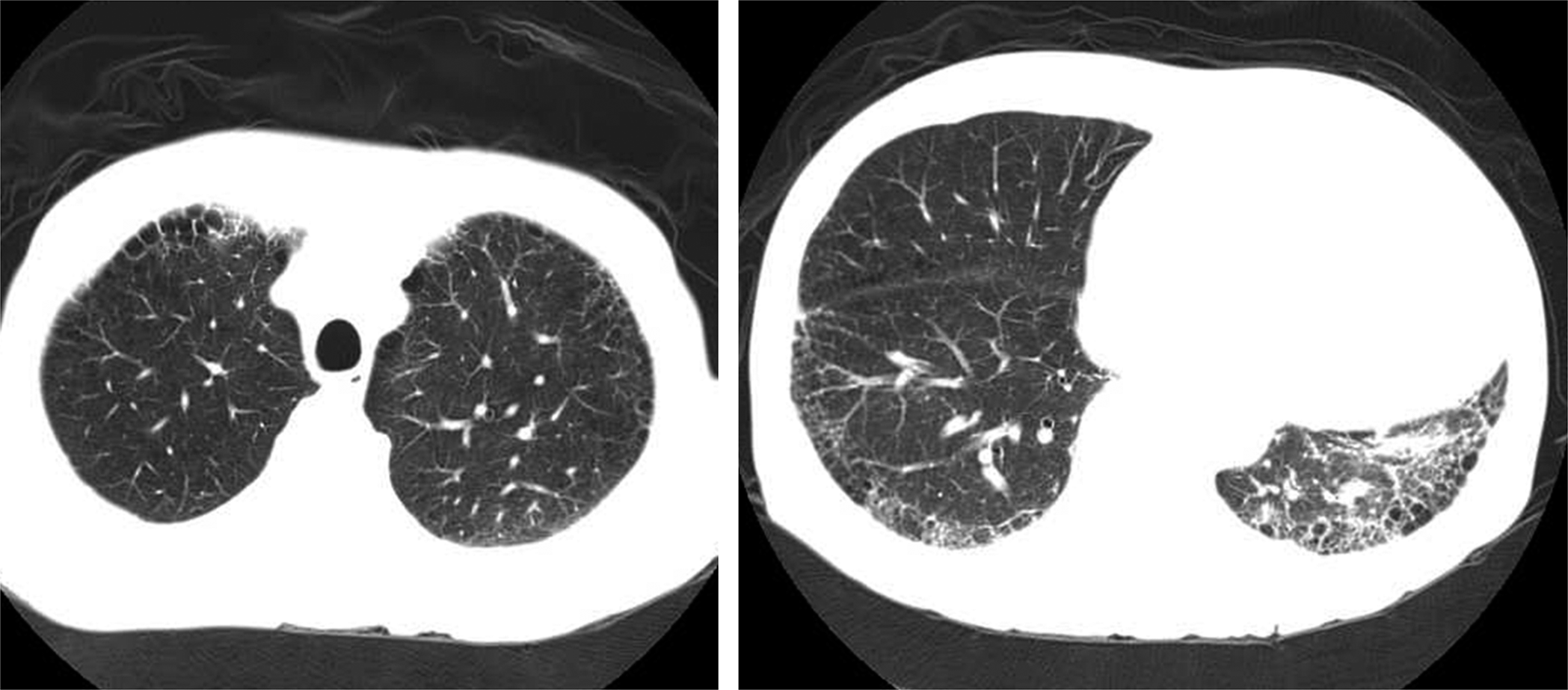
High-resolution computed tomography images of a 68-year-old man with COPD. Emphysematous changes are noted in both upper lobes. The patient had a smoking history of 110 pack-years and had reduced FEV1 (1,820 mL; 64% predicted) and FEV1/FVC (56%) and an obstructive pattern in the flow-volume curve on spirometry. COPD: chronic obstructive pulmonary disease; FEV1: forced expiratory volume in 1 second; FVC: forced vital capacity.
Characteristics of patients with pulmonary hypertension associated with severe lung disease (n = 14)

Note: WHO FC: World Health Organization functional class; CPFE: combined pulmonary fibrosis and emphysema; COPD: chronic obstructive pulmonary disease; VC: vital capacity; FVC: forced vital capacity; FEV1: forced expiratory volume in 1 second; DLCO: diffusing capacity of the lung for carbon monoxide; VA: alveolar volume; BNP: brain-type natriuretic peptide; PaO2: partial pressure of oxygen; PaCO2: partial pressure of carbon dioxide; MPAP: mean pulmonary arterial pressure; PAWP: pulmonary arterial wedge pressure; MRAP: mean right atrial pressure; CI: cardiac index (L/min/m2); PVR: pulmonary vascular resistance; RVEDVI: right ventricular end-diastolic volume index (mL/m2); RVESVI: right ventricular end-systolic volume index (mL/m2); RVEF: right ventricular ejection fraction.
The dose of oxygen supplementation varied in each patient, ranging from 0 to 8 L/min.
All 14 patients received continuous or on-demand supplemental oxygen for at least 2 weeks before RHC; 11 had been receiving oxygen treatment for 3 months or longer, whereas the remaining 3 had received oxygen for less than 3 months. All patients presented with short breath, edema, and/or hypoxia upon hospitalization, whereas the background lung disease was deemed stable, as specified in the exclusion criteria. CMR study showed increased RVEDVI and reduced RVEF, compared with the normal values reported previously.18,19
PAH-approved drug regimens
All patients started to receive sildenafil for the treatment of PAH 7–24 (14 ± 7) days after hospitalization, with dosages increased to the standard maximum of 60 mg daily within 1 week. Three patients were treated with additional PAH-specific vasodilators for symptoms/signs of severe and progressive right heart failure with high pulmonary vascular resistance (PVR; >10 Wood units) or reduced RVEF (<10%): 2 patients received additional bosentan, 62.5 mg twice daily, and 1 patient received additional beraprost, 120 μg three times/day, which were added 7–8 days after the start of sildenafil treatment. In the 2 other patients, sildenafil was changed to tadalafil 40 mg once daily because of muscle pain in one and hepatic dysfunction in the other.
Follow-up at 3 months
Before the assessment at 3 months, 1 patient with CPFE died suddenly and another with CPFE was hospitalized for pneumonia; therefore, we canceled their assessments. A patient with IPF was found to have advanced left renal cancer on follow-up admission; therefore, he did not undergo RHC. Finally, 11 patients were assessed at 3 months; PAH-approved drug regimens were not changed during the 3-month follow-up period.
Compared with the baseline data, the World Health Organization functional class (WHO FC) and PFT results did not change significantly, whereas plasma BNP concentration decreased and the 6MWD increased at 3 months (Table 2). In addition, MPAP and PVR decreased, and CO increased on assessment by RHC (Table 2; Fig. 4). Figure 5 shows the changes in the partial pressure of oxygen (PaO2). One patient needed an increased dose of nasal oxygen supplementation, from 8 L/min at baseline to 12 L/min at 3 months; however, the degree of hypoxia did not deteriorate in the other 10 patients, with 6 of them receiving the same dose of oxygen and 4 requiring less oxygen. CMR imaging showed that RVEDVI decreased by 23.7% (P = 0.0061; Fig. 4) and RVEF increased by 32.9% (P = 0.0165; Fig. 4). The changes in 3 patients who had homogenous clinical backgrounds, had fibrotic lung lesions (CPFE or IP), received oxygen treatment for at least 3 months, and were treated with phosphodiesterase type 5 (PDE-5) inhibitor alone are shown with bold lines in Figure 4.
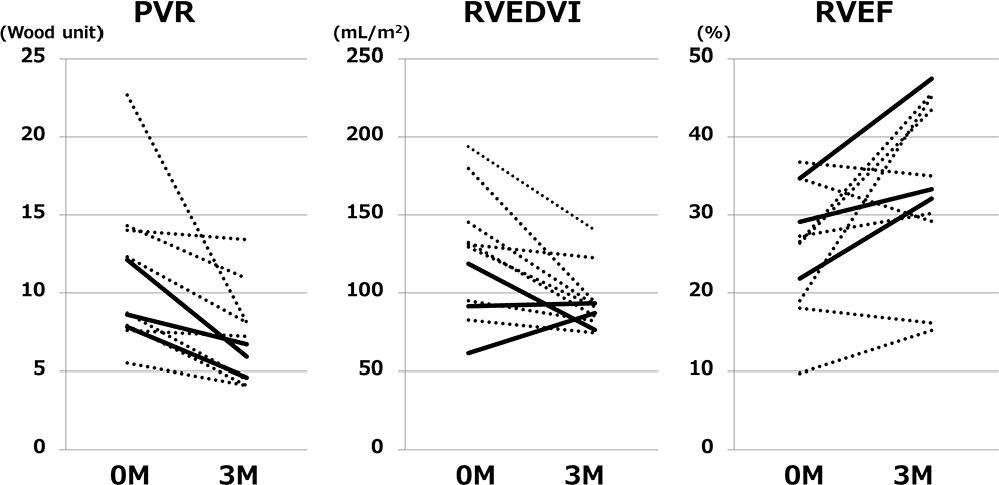
Changes in pulmonary vascular resistance and magnetic resonance indices during the 3-month follow-up. PVR and RVEDVI decreased and RVEF increased at 3-month follow-up in 11 patients with severe group 3 pulmonary hypertension treated with pulmonary arterial hypertension–approved drug(s). Bold lines indicate the changes in the 3 CPFE or IP patients who received at least 3 months of oxygen treatment and were treated with a phosphodiestrerase 5 inhibitor alone. PVR: pulmonary vascular resistance; RVEDVI: right ventricular end-diastolic volume index; RVEF: right ventricular ejection fraction; M: months; CPFE: combined pulmonary fibrosis and emphysema; IP: interstitial pneumonia.
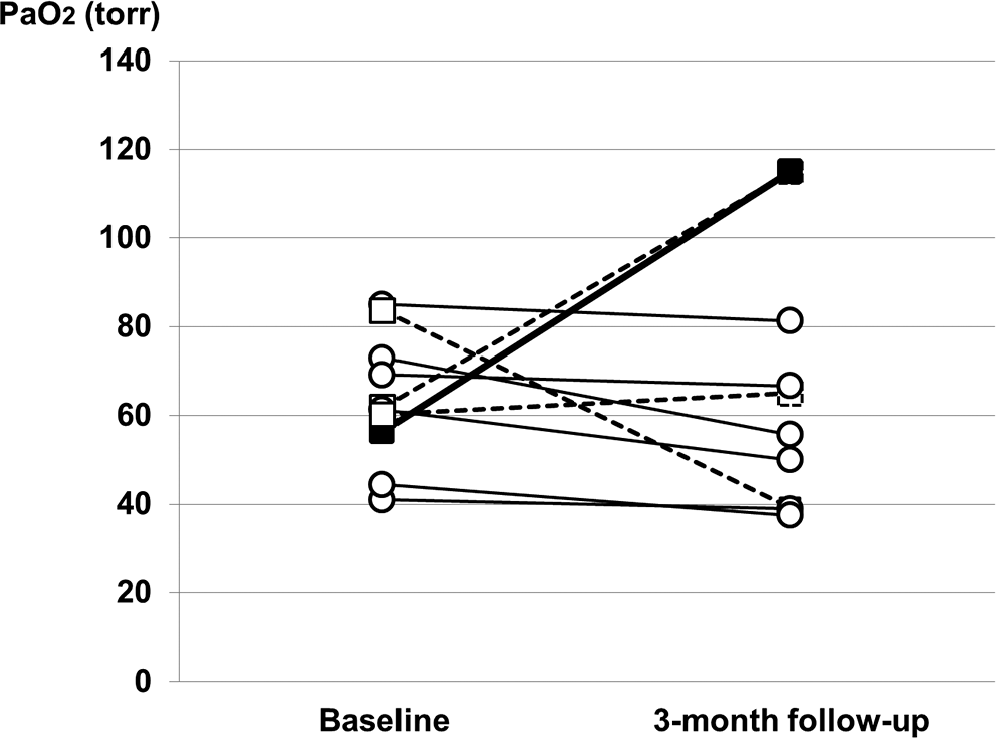
Changes in PaO2 during the 3-month treatment with PAH-approved drugs. Six of the 11 patients had arterial blood gas analysis on the same oxygen level (open circles), with a slight decrease in PaO2, from 62.3 ± 17.0 Torr at baseline to 55.0 ± 16.8 Torr at follow-up. Three patients were noted to have improved PaO2 and decreased oxygen supplementation (open squares), from 7 to 5 L/min, from 1 to 0.5 L/min, and from 4 to 0 L/min. In contrast, 1 patient had worsening hypoxia and needed an increase in oxygen supplementation from 8 to 12 L/min to maintain SpO2 at ≥90% (filled squares). Data for arterial blood gas analysis were missing for 1 patient, but the SpO2 levels remained at ≥90%, and he did not need oxygen supplementation at baseline or at 3 months. PaO2: partial pressure of oxygen; PAH: pulmonary arterial hypertension; SpO2: pulse oxygen saturation.
Changes in the clinical indices of patients with severe lung disease–associated pulmonary hypertension after treatment with pulmonary vasodilators (n = 11)
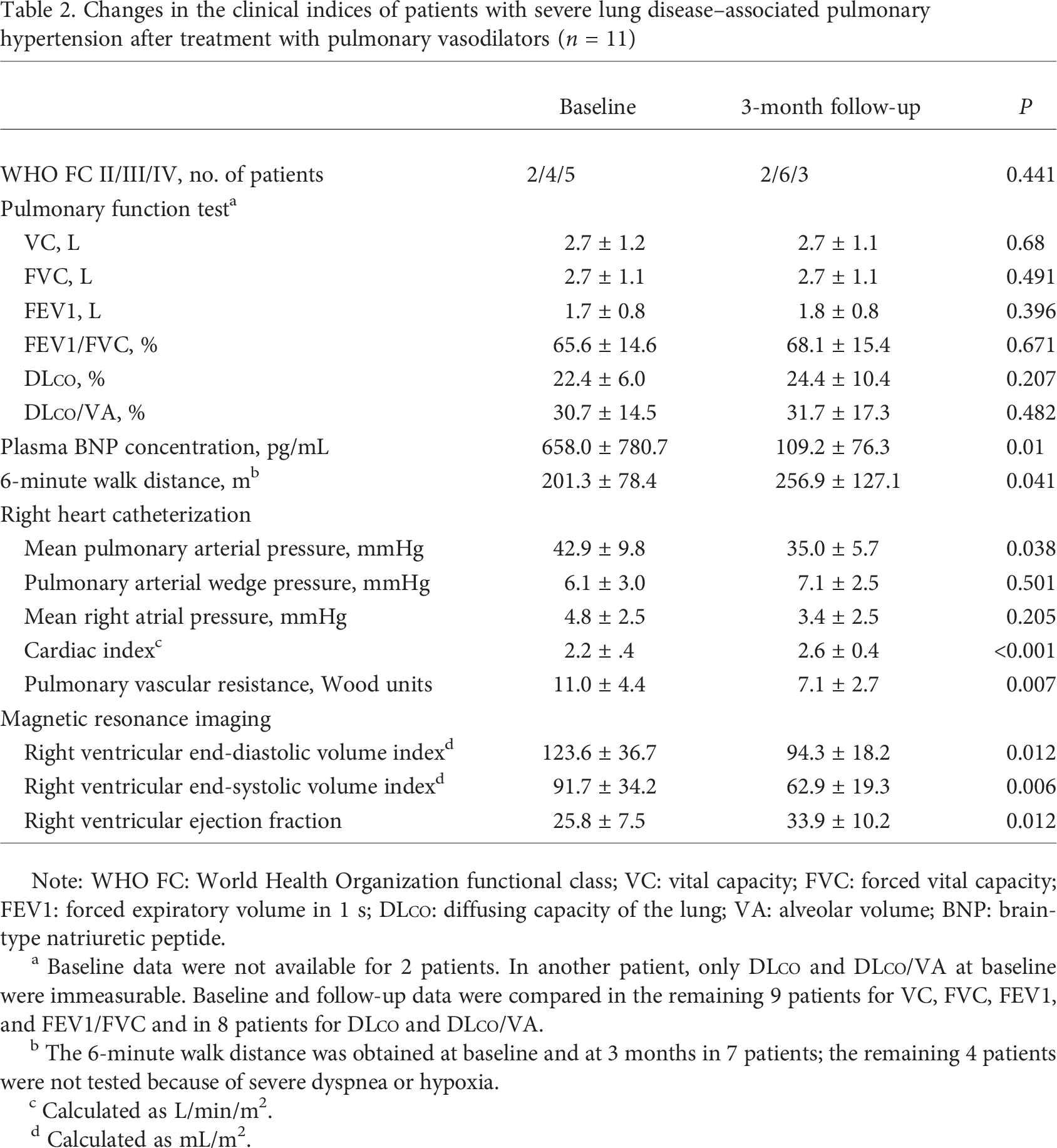
Note: WHO FC: World Health Organization functional class; VC: vital capacity; FVC: forced vital capacity; FEV1: forced expiratory volume in 1 s; DLCO: diffusing capacity of the lung; VA: alveolar volume; BNP: brain-type natriuretic peptide.
Baseline data were not available for 2 patients. In another patient, only DLCO and DLCO/VA at baseline were immeasurable. Baseline and follow-up data were compared in the remaining 9 patients for VC, FVC, FEV1, and FEV1/FVC and in 8 patients for DLCO and DLCO/VA.
The 6-minute walk distance was obtained at baseline and at 3 months in 7 patients; the remaining 4 patients were not tested because of severe dyspnea or hypoxia.
Calculated as L/min/m2.
Calculated as mL/m2.
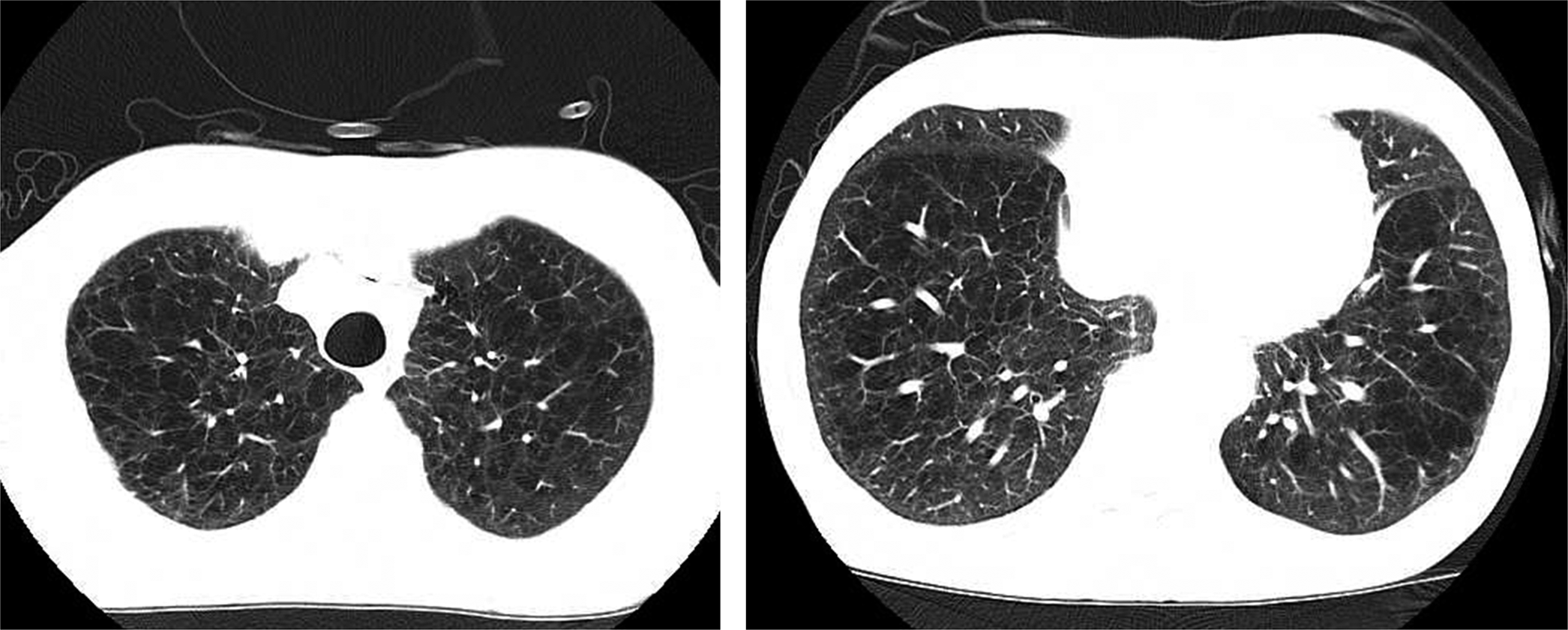
Figure 6 shows representative CMR images of an IPF patient treated with sildenafil alone as a pulmonary vasodilator. RVEDV did not change significantly (130 mL at both baseline and follow-up), whereas the bowing of the interventricular septum slightly improved after 3 months of treatment. RVEF also increased from 29.1% at baseline to 33.3% at 3 months.
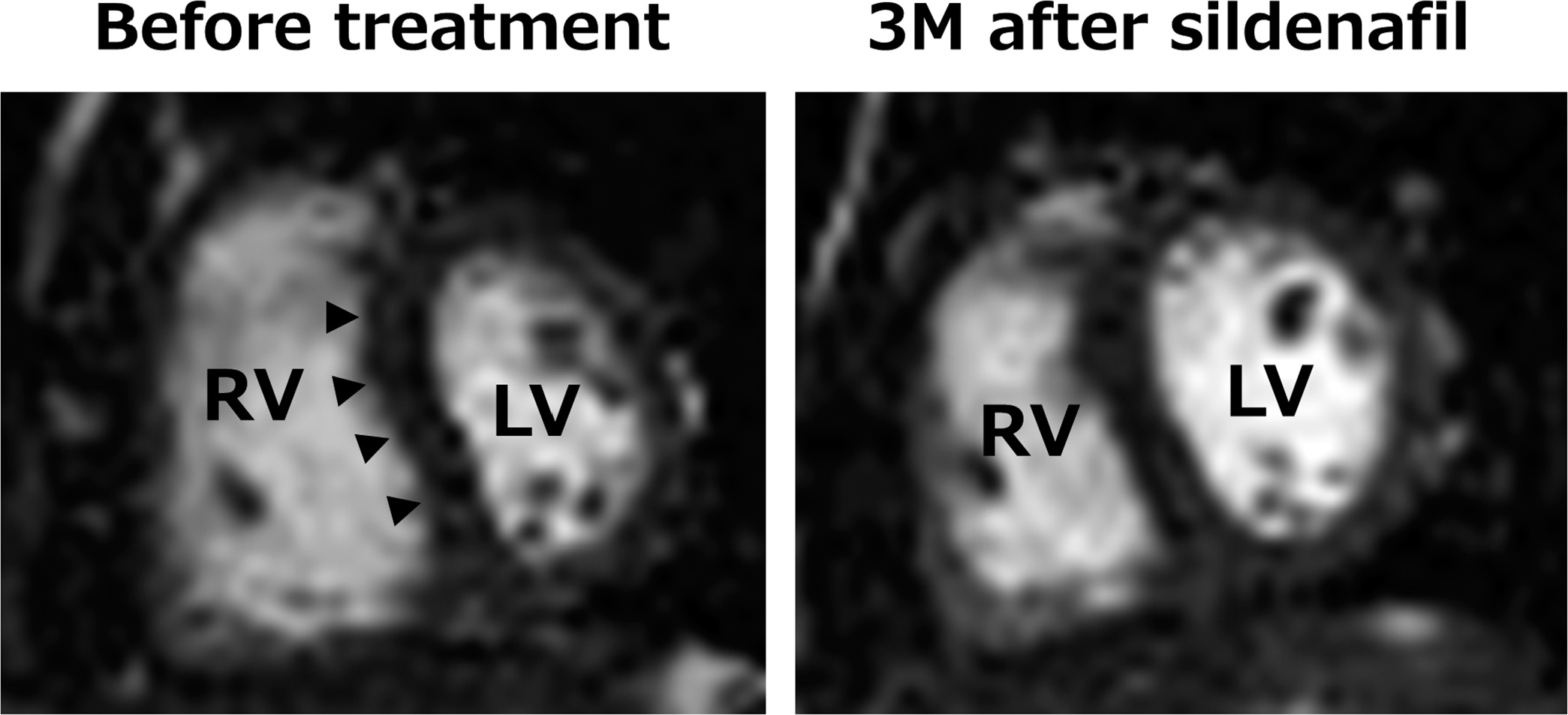
Cardiac magnetic resonance images of a 66-year-old man with idiopathic pulmonary fibrosis treated with sildenafil for 3 months. Right ventricular end-diastolic volume did not change significantly (130 mL at both baseline and 3-month follow-up), but the bowing of the interventricular septum at end diastole (arrowheads) slightly improved after 3 months of treatment with sildenafil 20 mg 3 times/day. Right ventricular ejection fraction increased from 29.1% at baseline to 33.3% at 3 months (3M). RV: right ventricle; LV: left ventricle.
Extended hemodynamic and cardiac evaluation
RHC and CMR follow-up beyond 3 months were conducted over 4 years in 6 patients. As shown in Figure S1, available online, improvements in CI, PVR, RVEDVI, and RVEF mostly persisted beyond 3 months. Three of the 6 patients received additional PAH drugs for increased PVR during follow-up, but they showed a decrease in PVR and an increase in RVEF after the addition of bosentan (n = 1) or ambrisentan (n = 2).
DISCUSSION
This pilot prospective study demonstrated that treatment with PAH-specific drugs for 3 months significantly decreased the RV volume and increased RVEF in 11 patients with severe lung-PH. Three patients with homogenous clinical background in terms of underlying lung disease and treatment regimen responded similarly to the other 8 patients. Extended follow-up over 4 years showed sustained improvement in RV size and RVEF in 6 patients. These results suggested the potential benefit of PAH-approved drugs for RV dilatation and RVEF in patients with severe lung-PH.
The efficacy of PAH-approved drugs for lung-PH patients has been investigated in several randomized trials; however, the results have been disappointing. For example, in a randomized prospective study, bosentan did not improve functional capacity or pulmonary hemodynamics in patients with PH due to fibrotic idiopathic IP. 20 Ambrisentan was not effective in treating IPF and IPF-associated PH and was shown to be associated with a possible increased risk for disease progression and hospitalization. 21 Conversely, some basic and observational data suggested that beneficial effects existed for PAH-approved drugs, particularly sildenafil, in this group.22,23 Consequently, the use of PAH-approved drugs in patients with lung-PH has been allowed in Japan since 2009, but only when clinical data suggest that severe PH cannot be explained by the lung disease(s) alone. This unique situation in Japan has enabled further examination of the effects of these PAH drugs on the CMR-derived RV morphology and function in patients with severe lung-PH under the current Japanese medical insurance system.
The prognostic value of RV morphology and function in PAH has been documented.10,11,24 A large RV volume and low SV strongly predict mortality and treatment failure in patients with idiopathic PAH, 10 and an increase in RV area and a decline in RVEF have been shown to indicate poor prognosis.13,24 However, the prognostic value of RV assessment among patients with lung-PH has not been addressed. In this study, patients with severe lung-PH had increased RVEDVI and reduced RVEF at baseline, compared with previously reported normal values,18,19 and therapy with PAH-approved drugs over 3 months significantly decreased the RVEDVI (by 23.7%) and increased the RVEF (by 32.9%). Importantly, there were no significant changes in the WHO FC, whereas 6MWD and serum BNP levels improved significantly at the 3-month follow-up. These results showed the potential utility of RV indices as markers and predictors of functional capacity and prognosis in patients with lung-PH, despite the differences in characteristics of PAH patients, including higher age, male predominance, prevalent smoking history, and higher hypoxia rates.
There are several possible explanations for the amelioration of RV size and function observed in our study. First, basic and clinical studies have shown that PAH-approved drugs can decrease PAP even in group 3 PH patients, contributing to reduced RV afterload.25,26 In our study, PVR mostly decreased, and this might have led to the improvement in the RV dilatation and RVEF. Another possibility was a direct effect of PAH drugs on the cardiomyocytes. This is plausible because PDE-5 has been reported to be present in hypertrophied RV myocardium in humans, 27 and sildenafil has been shown to reverse hypertrophy and improve contractile function of the cardiomyocyte in rats.27–29
Worsening of hypoxia is a critical concern when considering pulmonary vasodilator treatment in patients with lung-PH. In fact, previous basic and clinical studies have reported an unfavorable impact on ventilation-perfusion mismatch that result in worsening hypoxia.30,31 Therefore, we conducted this study with great caution to avoid these potential deleterious effects of PAH drugs in aggravating hypoxia. Although 1 patient with COPD experienced decreased PaO2 and needed an increase in oxygen supplementation, the other patients had no worsening of hypoxia and were given the same (n = 7) or less (n = 3) oxygen supplementation after 3 months. Considering the single-arm observational design of this pilot study, any effect of the PAH drugs on hypoxia must be thoroughly evaluated in larger, prospective studies.
During the extended follow-up period, 3 patients received bosentan or ambrisentan plus PDE-5 inhibitors because of insufficient or deleterious response to the initial PDE-5 inhibitor. All 3 of these patients exhibited improvements in PVR, similar to those reported in previous studies on patients with PAH.32,33 The efficacy and safety of this sequential combination in patients with severe group 3 PH should be addressed in prospective, double-armed trials.
The limitations of our study include the small number of patients and the lack of a control arm. Moreover, this study included COPD, IP, and CPFE as background lung diseases, which have different impacts on cardiac morphology and function and may respond differently to PAH-specific treatment. Another limitation was that medical treatments other than vasodilators may have affected RV size and function, because these were not controlled. Indeed, all patients underwent rest, restriction of salt intake to less than 6 g/day, and oxygen therapy, and we conducted RHC at least 1 week after initiating supportive care before starting PAH drug(s). Therefore, the changes in the CMR-derived indices at the 3-month follow-up might have been significantly affected by the comprehensive medical management in addition to vasodilator therapy. Such medical management might have also affected the RHC and CMR data observed during the extended follow-up. In particular, because hypoxia is thought to be a critical mechanism for development of RV dysfunction,34,35 oxygen treatment might have improved RV size and function, especially in patients who were receiving oxygen treatment for a short period before vasodilator administration. More strictly controlled prospective studies are needed to examine the pure efficacy of PAH-approved drug(s). To overcome these limitations, larger prospective studies on well-controlled patients with lung-PH will be needed. Nevertheless, we think that pilot studies like ours will help determine the optimal end point(s) and a suitable number of participants for future studies.
In conclusion, this pilot study indicated that there is a potential role for PAH-specific vasodilators in improving RV dilatation and dysfunction in patients with severe lung-PH. Possible mechanisms of action include reduced RV afterload and the direct impact of PAH-approved drugs on the RV myocardium. The safety and efficacy of PAH-approved drugs for the subset of patients with severe lung-PH and the clinical relevance of the changes in RV assessment should be further addressed in larger prospective, double-blind clinical trials.
Footnotes
Acknowledgments
We genuinely thank Professor Paul M. Hassoun for his valuable suggestions on data interpretation, presentation, structure, and English writing of this article.
