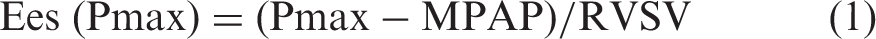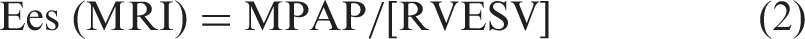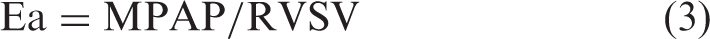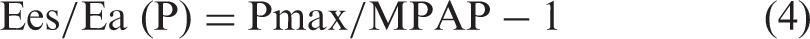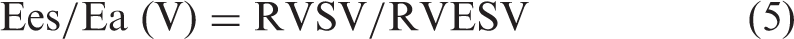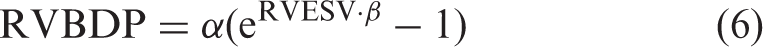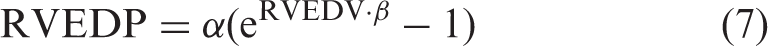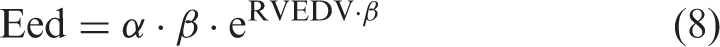Abstract
Right ventricular function critically affects the prognosis of patients with pulmonary arterial hypertension. We aimed to analyze the prognostic value of right ventricular indices calculated using magnetic resonance imaging and right heart catheterization metrics in pulmonary arterial hypertension. We retrospectively collected data from 57 Japanese patients with pulmonary arterial hypertension and 18 controls and calculated six indices of right ventricular function: two indices of contractility (end-systolic elastance calculated with right ventricular maximum pressure and with magnetic resonance imaging metrics); two indices of right ventricular–pulmonary arterial coupling (end-systolic elastance/arterial elastance calculated with the pressure method (end-systolic elastance/arterial elastance (P)) and with the volume method (end-systolic elastance/arterial elastance (V)); and two indices of right ventricular diastolic function (stiffness (β) and end-diastolic elastance). We compared the indices between controls and patients with pulmonary arterial hypertension and examined their prognostic role. In patients with pulmonary arterial hypertension, end-systolic elastance (right ventricular maximum pressure) was higher (pulmonary arterial hypertension 0.94 (median) vs control 0.42 (mmHg/mL), p < 0.001), end-systolic elastance/arterial elastance (V) was lower (pulmonary arterial hypertension 0.72 vs control 1.69, p < 0.001), and β and end-diastolic elastance were significantly higher than those in the controls. According to the log-rank test, end-systolic elastance/arterial elastance (P) and end-diastolic elastance were significantly associated with the composite event rate. According to the multivariate Cox regression analysis, decreased end-systolic elastance/arterial elastance (P) was associated with a higher composite event rate (hazard ratio 11.510, 95% confidence interval: 1.954–67.808). In conclusion, an increased right ventricular contractility, diastolic dysfunction, and a trend of impaired right ventricular–pulmonary arterial coupling were observed in our pulmonary arterial hypertension cohort. According to the multivariate outcome analysis, a decreased end-systolic elastance/arterial elastance (P), suggestive of impaired right ventricular–pulmonary arterial coupling, best predicted the pulmonary arterial hypertension-related event.
Keywords
Introduction
Studies have shown that right ventricular (RV) function critically determines the prognosis of patients with pulmonary arterial hypertension (PAH).1,2 RV function can be assessed by indices such as cardiac index and RV ejection fraction (RVEF); however, these parameters are affected by RV preload/afterload and are not ideal indicators of intrinsic RV function.
Ventricular elastance calculated from pressure–volume loop analysis is the gold standard index of RV function. For example, RV end-systolic elastance (Ees) and end-diastolic elastance (Eed) represent intrinsic RV systolic and diastolic function, respectively.3–5 Additionally, Ees divided by arterial elastance (Ea) is a parameter of RV systolic function in relation to afterload, or right ventriculo–pulmonary arterial (RV–PA) coupling. 5 Furthermore, β is an index of RV stiffness, which is an important element of RV diastolic function. 6 Ideally, these indices may be applied to evaluate RV function; however, they are not widely used because dedicated pressure and conductance catheters and invasive procedures are required.
Recently, less invasive approaches to evaluate intrinsic RV function have been introduced. Studies using cardiac magnetic resonance imaging (MRI) have revealed increased Ees, β, and Eed and decreased Ees/Ea in PAH and other types of pulmonary hypertension (PH).6–8 Additionally, the outcome predictability of the indices of RV–PA coupling and RV diastolic function, i.e. Ees/Ea, β, and Eed, has been documented.8–11 Importantly, however, only some RV indices were selected for the analyses in most studies. Therefore, it is unknown which of them better predicts the outcome of patients with PAH. Moreover, no prior studies have reported indices of RV systolic/diastolic function and of RV–PA coupling in a comprehensive manner for Asian patients with PAH, although ethnic differences in RV morphology and function have been reported. 12
In this study, we aimed to calculate MRI and right heart catheterization (RHC)-derived indices of RV function in our PAH cohort and compare them with those of control participants. We also sought to clarify which of the RV indices best predicts PAH-related outcomes. Of note, the present study was conducted in a limited number of patients with PAH and of clinical events and, thus, is regarded as a preliminary study rather than a study that draws robust conclusions.
Methods
Study participants
We retrospectively collected data from patients with PAH hospitalized in our University Hospital between January 2010 and April 2018. All patients underwent blood tests, echocardiography, cardiac MRI, and RHC within three weeks without significant change(s) in clinical status and modifications in PAH drug(s). RHC measurements included pulmonary arterial pressure (PAP), pulmonary arterial wedge pressure, RV end-diastolic pressure (RVEDP), right atrial pressure, cardiac output, and pulmonary vascular resistance (PVR). Cardiac output was measured by a thermodilution method. We also calculated the ratio of pulmonary pulse pressure (systolic PAP – diastolic PAP) to RV stroke volume (SV) (cardiac output divided by heart rate), as an index of pulmonary arterial compliance.
Patients with PAH were treated according to the guidelines published by the European Society of Cardiology and European Respiratory Science for PH. 13 In the present study, the following drugs were regarded as PAH drugs: phosphodiesterase 5 inhibitors (sildenafil and tadalafil), endothelin receptor antagonists (bosentan, ambrisentan, and macitentan), and prostacyclin I2 and its derivatives (epoprostenol, intravenous and treprostinil, intravenous or subcutaneous). Beraprost was not included as a PAH drug due to lack of proven sustained efficacy 14 and limited recommendations in the guidelines. 13 Patients with portopulmonary PAH and congenital heart disease-associated PAH were excluded because of their different homodynamic properties compared with idiopathic/heritable/drug-induced PAH (I/H/D-PAH) or connective tissue disease-associated PAH (CTD-PAH).
Control participants underwent cardiac MRI and RHC within two weeks. The exclusion criteria for the control group were a mean PAP (MPAP) of ≥21 mmHg, PVR of ≥3 Wood units, or the presence of cardiac disease(s) identified by echocardiography or MRI.
Assessment of RV morphology and function using MRI
Cardiac MRI was performed using a 1.5-T Philips Achieva MRI system (Philips Medical Systems, Best, Netherlands) with a five-channel coil and Master gradients (maximum gradient amplitude, 33 mT/m; maximum slew rate, 100 mT/m/ms). The protocol for image acquisition and analysis has been reported in previous studies, with high intra- and inter-observer reproducibility.15–17 In brief, 12 axial slices were acquired using a steady-state free precession pulse sequence (repetition time, 2.8 ms; echo time, 1.4 ms; flip angle, 60; acquisition matrix, 192 × 256; field of view, 380 ms; slice thickness, 10 mm; 0 mm inter-slice gap; and 20 phases/cardiac cycle). Images were analyzed using commercially available analysis software (Extended MR Work Space ver. 2.6.3; Philips Medical Systems, Amsterdam, Netherlands). In axial datasets, the endocardial contours of the RV were manually traced, and RV and left ventricular end-diastolic volume (EDV) and end-systolic volume (ESV) were computed. RV and left ventricular SV and EF were calculated as SV = EDV − ESV and EF = SV/EDV × 100%, respectively.
Calculation of Ees, Ees/Ea, β, and Eed
Ees was calculated using the single beat method
10
and another method that applies MPAP and MRI-derived RV end-systolic volume (RVESV), as reported by Sanz et al.
7
Fig. 1 shows the rationale of the two methods. The RV maximum pressure (Pmax) used in the single beat method was determined using commercially available software (GetData Graph Digitizer 2.26, MATLAB R2018a program, The Math Works, Natick, MA, USA) as has been reported by Brewis et al.
10
In short, printed pressure recordings obtained with a Swan–Ganz catheter were scanned, saved as bitmap image files, and the files were reread using GetData Graph digitizer. We then put plots overlaying the lines at the early systolic phase (from the end-diastolic phase up to the site where the slope is the positively steepest (dP/dtmax)) and at the early diastolic phase (from the site where the slope is the negatively steepest (dP/dtmin) to the last part of isovolumic relaxation). The interval between the two adjacent points was 6–7 ms (∼150 Hz). The plots with their time- and pressure coordinates were then transferred to MATLAB, in which we performed a non-linear extrapolation curve fitting and determined the Pmax (Supplemental Figure 1).5,18 We calculated the Pmax five times and used the mean value as representative data. Ees was then calculated as follows
18
Diagram of the rationale for the calculation of Ees, Ea, and Eed. Ees can be calculated by the single beat method (Ees (Pmax)) and the method that uses MPAP and RVESV measured by MRI (Ees (MRI)). In the single beat method, Ees is calculated using the following formula: ((Pmax – MPAP)/RVSV), where Pmax is calculated based on the RHC-derived RV pressure curve as explained in Supplemental Figure 1. In the second method, Ees is calculated using the following formula: MPAP/(RVESV – V0). In this method, V0 can be neglected and the formula is simplified to MPAP/RVESV. End-diastolic elastance (Eed) is the slope of the tangent line at the RVEDV of the non-linear end-diastolic pressure–volume relationship. Ea is calculated as MPAP/RVSV.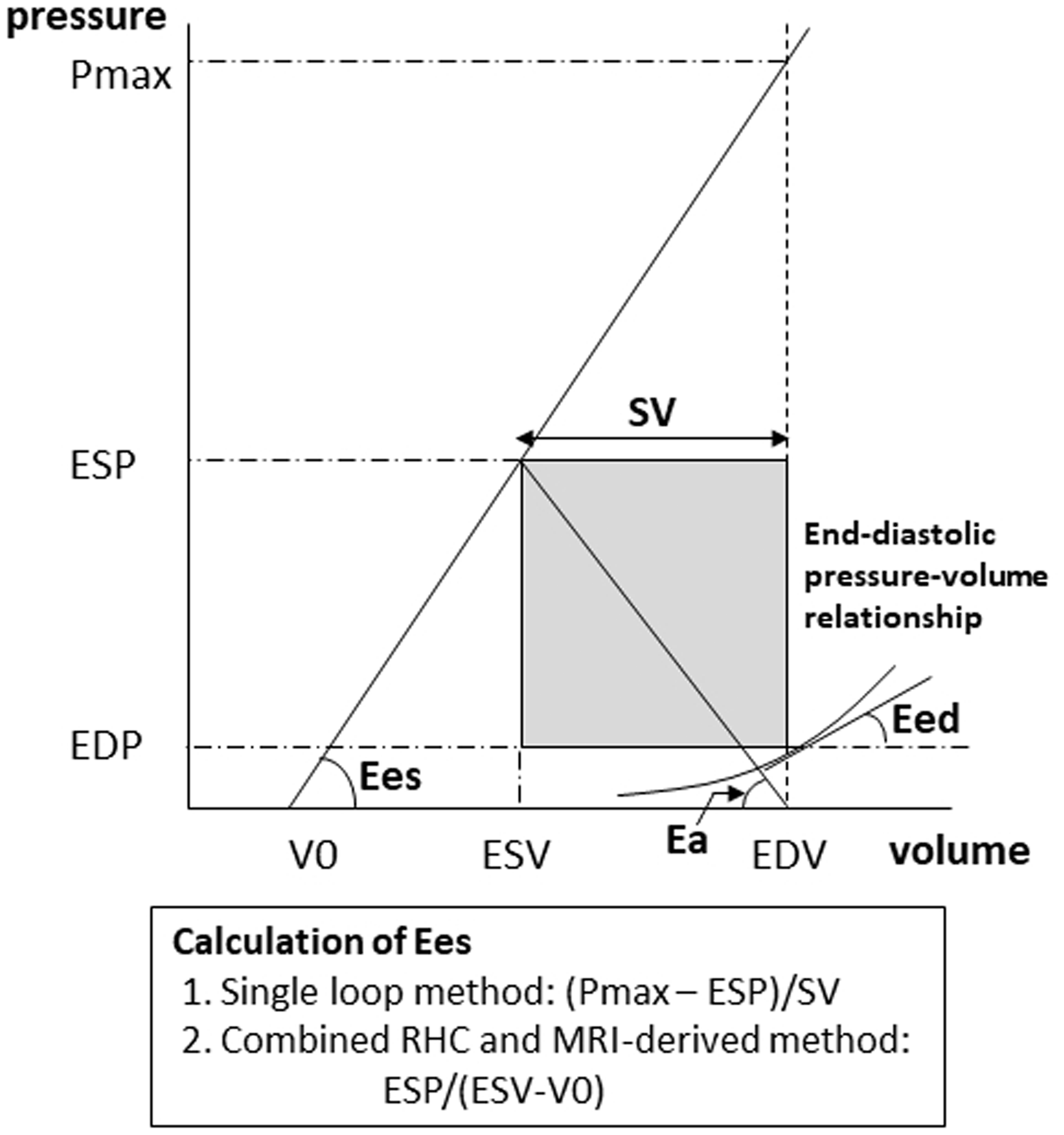
Here, RVSV represents the RV stroke volume.
In Sanz et al.’s study, Ees was calculated as the ratio of MPAP to (RVESV – V0).
7
V0 is the volume when the RV pressure is zero; to derive Ees/Ea (V), it is assumed herein to occur at zero volume as well
7
Ees/Ea is the ratio of Ees to the Ea, where Ea was estimated by the ratio of MPAP to RVSV5,19
Ees/Ea was then calculated by two equations: equations (4) and (5). The former is the “pressure method” and the latter is the “volume method.”
Here, of note is that Ees/Ea (V) is a mathematical transformation of RVEF as represented by the following formula: Ees/Ea (V) = RVEF/(1 – RVEF).
β was calculated as a solution of the following two simultaneous equations as reported by Rain et al.
6
RV begin-diastolic pressure (RVBDP) is the pressure present at the end of isovolumic relaxation (or the lowest pressure point during the diastolic phase), and RVEDP was measured at the maximal diastolic filling pressure point. RVBDP was normalized at 1 mm Hg, and RVEDP was calculated as 1 + (RVEDP – RVBDP) to avoid measurement errors. 6
Eed was calculated as the slope of the diastolic pressure–volume relationship at end-diastole using the formula by Trip et al.
8
Data analysis
Categorical values are expressed as absolute numbers (percentage) and continuous variables as medians (interquartile range), unless otherwise stated. Demographics and clinical parameters, including the six RV parameters (Ees (Pmax), Ees (MRI), Ees/Ea (P), Ees/Ea (V), β, and Eed) were compared between control and PAH groups using Fisher’s exact or Wilcoxon rank-sum tests. The same parameters were compared among the three groups of PAH: I/H/D-PAH, non-systemic sclerosis (SSc)-CTD-PAH, and SSc-CTD-PAH using Fisher’s exact or Kruskal–Wallis tests. The associations of the six RV indices with other PAH-related parameters were assessed using Wilcoxon rank-sum or Kruskal–Wallis tests for categorical data or Spearman’s coefficient (ρ) for continuous variables.
To examine the reproducibility of the Pmax in our study, we performed an analysis examining the interobserver variation. For this purpose, RV pressure curves of 10 participants (two controls and eight patients with PAH) were randomly selected and two observers (T.N. and I.T.) independently put plots in the early systolic and diastolic phases using GetData Graph digitizer without any information regarding the patient’s clinical situation and the other examiner’s interpretation. The 10 sets of Pmax obtained by the two examiners were compared using Bland–Altman’s analysis and intraclass coefficients.
In the outcome analysis, we applied a composite endpoint of hospitalization due to PAH worsening, lung transplantation, or PAH-related death. In this analysis, we aimed to compare the prognostic value of Ees/Ea (P), Ees/Ea (V), β, and Eed because these four RV indices reportedly have prognostic value. We conducted a receiver operating characteristic (ROC) curve analysis and determined the optimal cut-off values for these four RV indices. 20 Patients with PAH were divided into two groups using these cut-off values, and the outcome was compared using Kaplan–Meier curves and log-rank test. Subsequently, we conducted univariate and age-adjusted Cox regression analysis examining the associations of the PAH-related parameters with the outcome. We conducted multivariate analysis for the variables, including age and the following four RV indices: Ees/Ea (P), Ees/Ea (V), β, and Eed. We also included these four RV indices and other PAH-related clinical parameters with proven prognostic value (i.e. WHO functional class, plasma brain natriuretic peptide concentration, six-minute walk distance, cardiac index, and right atrial pressure). 1 Additionally, to further clarify the superiority among these four RV indices, we performed multivariate Cox regression analysis and compared the outcome predictability of the four indices using forest plot analysis.
A two-sided p value <0.05 was considered significant. JMP Pro version 12 (Japanese version, SAS Institute Inc., Cary, NC, USA) was used for statistical analyses.
Results
Characteristics of the study populations
Comparison of demographics and PAH-related parameters in controls and PAH patients.
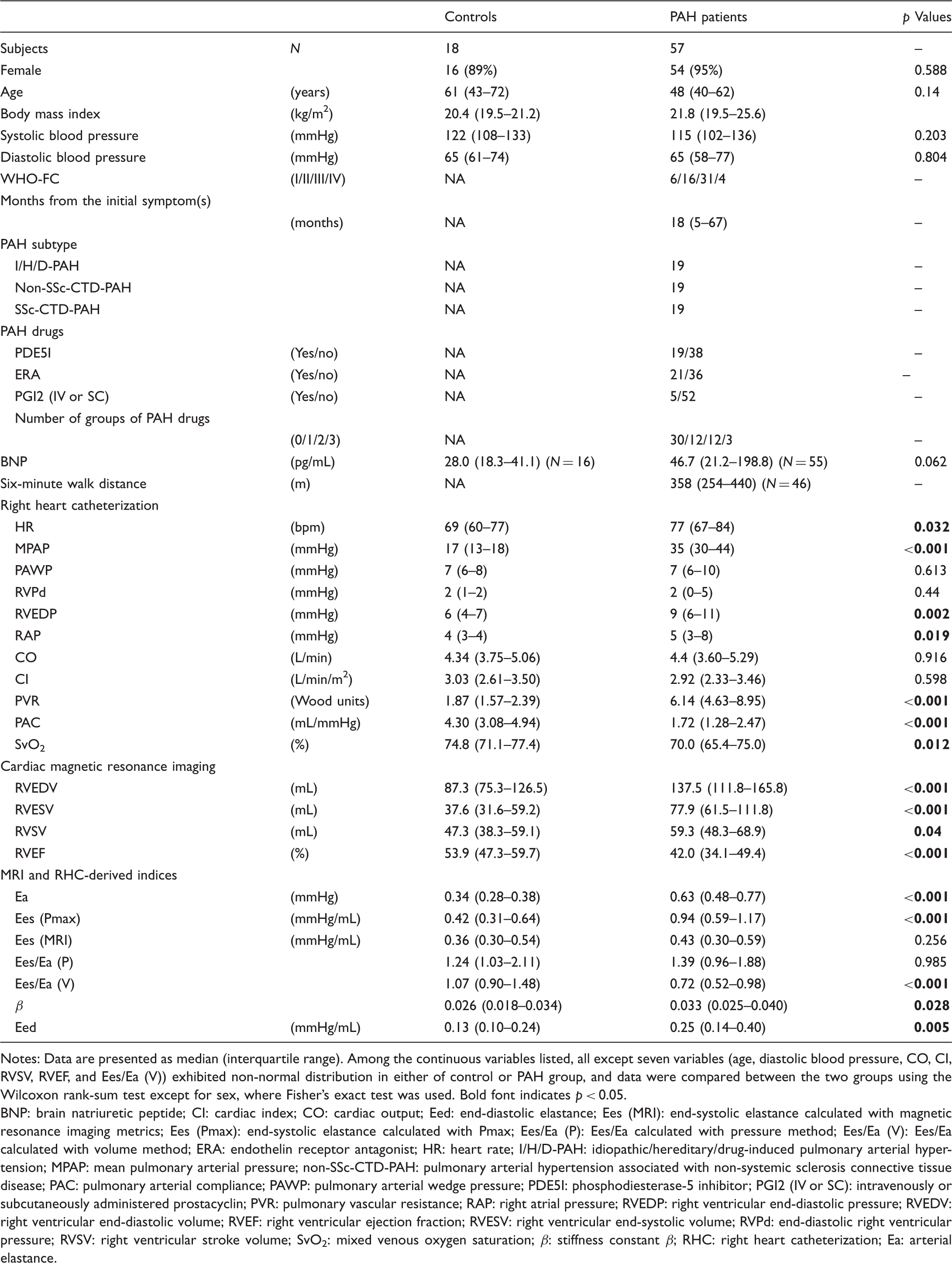
Notes: Data are presented as median (interquartile range). Among the continuous variables listed, all except seven variables (age, diastolic blood pressure, CO, CI, RVSV, RVEF, and Ees/Ea (V)) exhibited non-normal distribution in either of control or PAH group, and data were compared between the two groups using the Wilcoxon rank-sum test except for sex, where Fisher’s exact test was used. Bold font indicates p < 0.05.
BNP: brain natriuretic peptide; CI: cardiac index; CO: cardiac output; Eed: end-diastolic elastance; Ees (MRI): end-systolic elastance calculated with magnetic resonance imaging metrics; Ees (Pmax): end-systolic elastance calculated with Pmax; Ees/Ea (P): Ees/Ea calculated with pressure method; Ees/Ea (V): Ees/Ea calculated with volume method; ERA: endothelin receptor antagonist; HR: heart rate; I/H/D-PAH: idiopathic/hereditary/drug-induced pulmonary arterial hypertension; MPAP: mean pulmonary arterial pressure; non-SSc-CTD-PAH: pulmonary arterial hypertension associated with non-systemic sclerosis connective tissue disease; PAC: pulmonary arterial compliance; PAWP: pulmonary arterial wedge pressure; PDE5I: phosphodiesterase-5 inhibitor; PGI2 (IV or SC): intravenously or subcutaneously administered prostacyclin; PVR: pulmonary vascular resistance; RAP: right atrial pressure; RVEDP: right ventricular end-diastolic pressure; RVEDV: right ventricular end-diastolic volume; RVEF: right ventricular ejection fraction; RVESV: right ventricular end-systolic volume; RVPd: end-diastolic right ventricular pressure; RVSV: right ventricular stroke volume; SvO2: mixed venous oxygen saturation; β: stiffness constant β; RHC: right heart catheterization; Ea: arterial elastance.
Comparisons of RV indices between control and PAH groups
Regarding the indices of RV function, Ees (Pmax), β, and Eed were significantly higher (p <0.001 for Ees (Pmax), p = 0.028 for β, and p = 0.005 for Eed), whereas Ees/Ea (V) was significantly lower in patients with PAH than in controls (p <0.001) (Table 1, Fig. 2). There were no differences in any of the six RV indices among the three PAH subgroups (Supplemental Table 1). Among the variables listed in Table 1, age, diastolic blood pressure, cardiac output, cardiac index, RVSV, RVEF, and Ees/Ea (V) exhibited normal distribution in both control and PAH groups, and unpaired t-test for these variables exhibited the same results as those obtained by non-parametrical analysis.
Comparison of Ees (Pmax), Ees (MRI), Ees/Ea (P), Ees/Ea (V), β, and Eed between controls and patients with PAH. Ees (Pmax), β, and Eed were higher, whereas Ees/Ea (V) was lower in patients with PAH than in controls.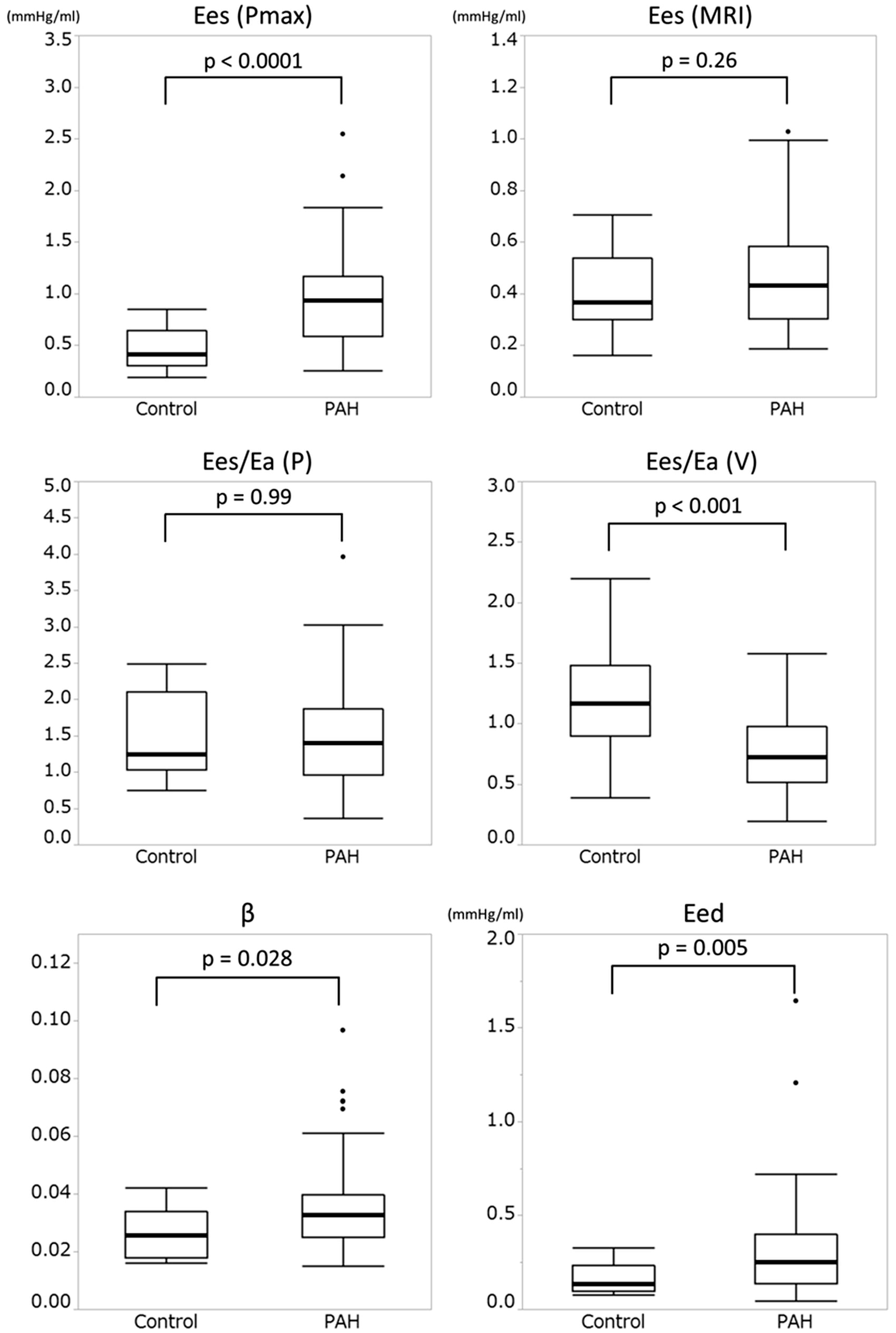
Associations of RV indices with other parameters
Supplemental Tables 2 and 3 and Supplemental Figures 2 and 3 show the associations of the six RV indices with other clinical parameters. The association was relatively strong (p <0.001 and |ρ| ≥ 0.50) between Ees/Ea (V) and plasma brain natriuretic peptide concentration, between β and RVSV, and between Eed and RVEDP. There were also significant associations between Ees (MRI) or Ees/Ea (V) and RV volume-related indices (RVESV, right ventricular end-diastolic volume (RVEDV), and RVEF), which were mathematically natural because Ees (MRI), Ees/Ea (V), and RVEF included RVESV and/or RVEDV in the formula for the calculation. Of note, β was lower in patients with PAH when they received one or more PAH drugs.
Analysis of Pmax reproducibility
In the reproducibility analysis of the variation of Pmax, Bland–Altman analysis indicated a high observer agreement (mean ± limits of agreement: intraobserver, −3.1 ± 3.2; interobserver, 7.4 ± 7.7). Intraclass coefficients were also high at 0.989 for intraobserver reproducibility and 0.949 for interobserver reproducibility.
Survival analysis
During the follow-up period of 45 (24–85) months, 13 patients met the composite endpoint (hospitalized due to PAH worsening, 11; lung transplantation, 0; PAH-related death, 2). Furthermore, the endpoint was reached in 2/19 (11%) in the I/H/D-PAH group, 8/19 (42%) in the SSc-PAH group, and 3/19 (16%) in the non-SSc-PAH group.
The cut-off values calculated based on the ROC analysis were 0.727 for Ees (Pmax), 0.450 for Ees (MRI), 1.12 for Ees/Ea (P), 0.59 for Ees/Ea (V), 0.0346 for β, and 0.252 for Eed. Area under the curve values for each variable were as follows: Ees (Pmax), 0.51; Ees (MRI), 0.53; Ees/Ea (P), 0.62; Ees/Ea (V), 0.58; β, 0.57; and Eed, 0.6.
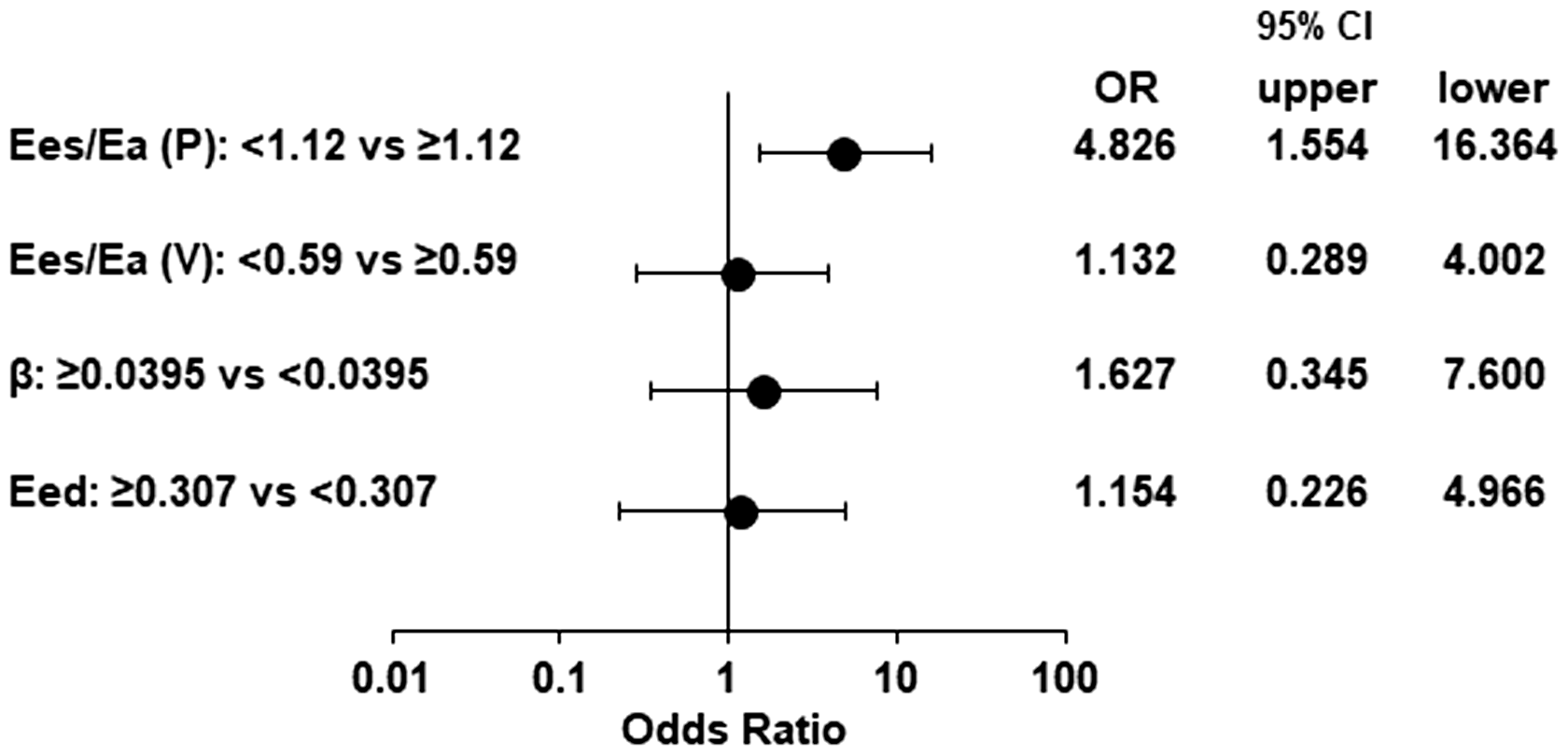
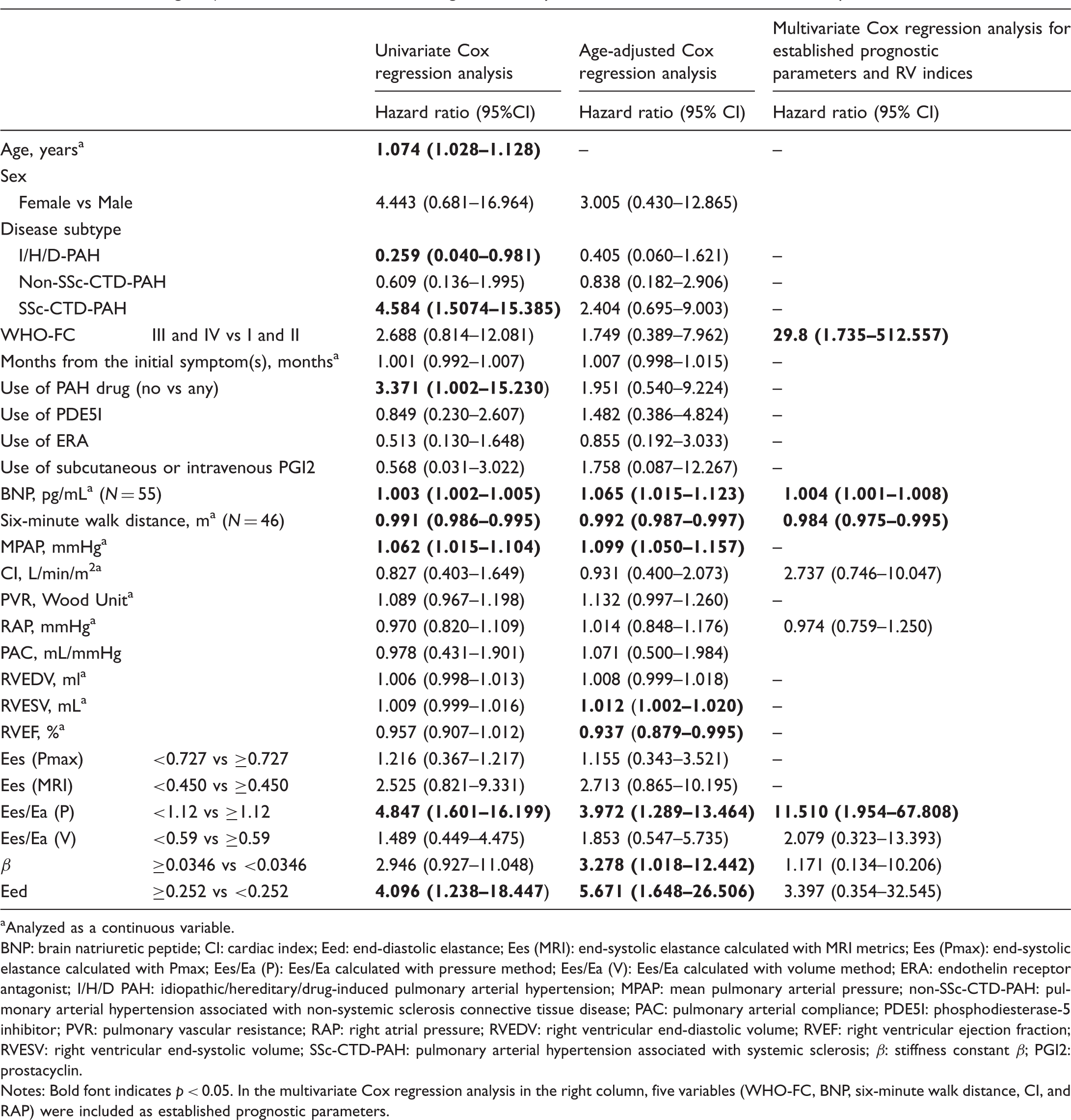
Kaplan–Meier curves and log-rank tests demonstrated a significant difference in survival between the two groups divided by an Ees/Ea (P) of 1.12 and those divided by an Eed of 0.252 (Fig. 3). In the univariate Cox regression analysis, significant associations with the composite outcome were observed for age, PAH subtype, use of PAH drug, brain natriuretic peptide, six-minute walk distance, MPAP, Ees/Ea (P), and Eed (Table 2). In the age-adjusted Cox regression analysis, statistical significance was observed for brain natriuretic peptide, six-minute walk distance, MPAP, RVESV, RVEF, Ees (Pmax), and Ees/Ea (P). In the multivariate Cox regression analysis and in the forest plot analysis, among the four RV indices, only Ees/Ea (P) had a statistically significant association with the outcome (Table 2, Fig. 4).
Kaplan–Meier curves of two PAH groups classified by the ROC analysis-derived cut-off values of Ees/Ea (P), Ees/Ea (V), β, and Eed. There was a significant difference in the survival rate between the two groups divided by Ees/Ea (P) and those divided by Eed. Forrest plot of the results of multivariate Cox regression analysis for Ees/Ea (P), Ees/Ea (V), β, and Eed. Odds ratios for the composite outcome, i.e. hospitalization due to PAH worsening, lung transplantation, or PAH-related death, are compared among the four RV indices. Univariate, age-adjusted, and multivariate Cox regression analysis on the association of PAH-related parameters with the outcome. Analyzed as a continuous variable. BNP: brain natriuretic peptide; CI: cardiac index; Eed: end-diastolic elastance; Ees (MRI): end-systolic elastance calculated with MRI metrics; Ees (Pmax): end-systolic elastance calculated with Pmax; Ees/Ea (P): Ees/Ea calculated with pressure method; Ees/Ea (V): Ees/Ea calculated with volume method; ERA: endothelin receptor antagonist; I/H/D PAH: idiopathic/hereditary/drug-induced pulmonary arterial hypertension; MPAP: mean pulmonary arterial pressure; non-SSc-CTD-PAH: pulmonary arterial hypertension associated with non-systemic sclerosis connective tissue disease; PAC: pulmonary arterial compliance; PDE5I: phosphodiesterase-5 inhibitor; PVR: pulmonary vascular resistance; RAP: right atrial pressure; RVEDV: right ventricular end-diastolic volume; RVEF: right ventricular ejection fraction; RVESV: right ventricular end-systolic volume; SSc-CTD-PAH: pulmonary arterial hypertension associated with systemic sclerosis; β: stiffness constant β; PGI2: prostacyclin. Notes: Bold font indicates p < 0.05. In the multivariate Cox regression analysis in the right column, five variables (WHO-FC, BNP, six-minute walk distance, CI, and RAP) were included as established prognostic parameters.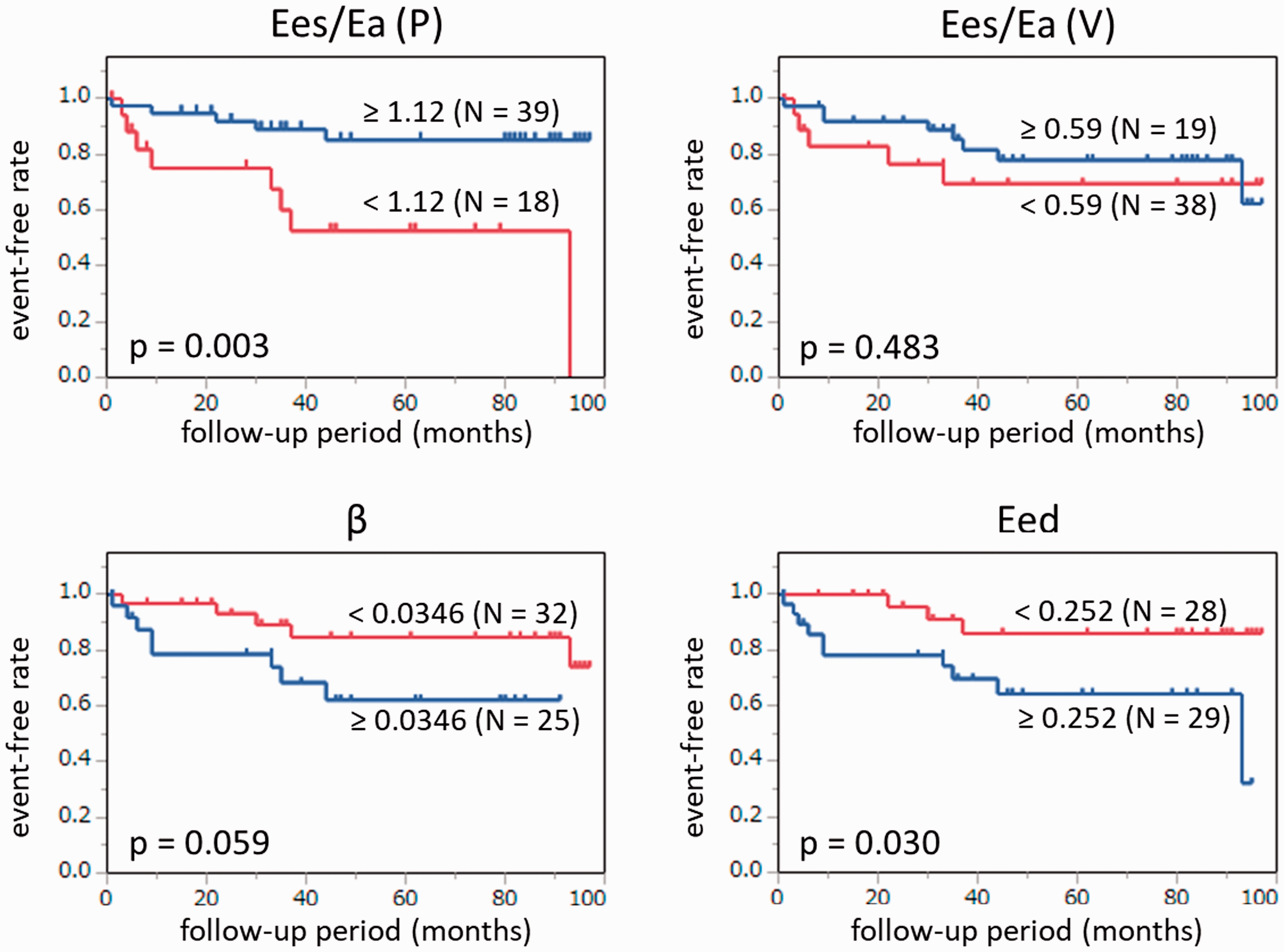
Discussion
Summary
This retrospective observational study revealed that patients with PAH had higher Ees (Pmax), β, and Eed, and lower Ees/Ea (V) than did controls, regardless of PAH etiology. Log-rank test and Cox regression analysis indicated that a decrease in Ees/Ea (P), an indicator of RV–PA coupling, predicts a poor outcome in our PAH cohort better than other RV indices. To our knowledge, this study is the first to report the four indices of RV–PA coupling and RV diastolic function, i.e. Ees/Ea (P), Ees/Ea (V), β, and Eed, and comparably examine their prognostic value in patients with PAH.
RV contractility (Ees)
To date, eight studies have reported RV Ees in PH,7–11,18,21,22 and three of them have reported an elevated Ees in patients with PH compared with that in controls.7,8,10 In the present study, Ees (Pmax) was higher in patients with PAH than in controls, verifying an increased RV contractility in our PAH cohort. Ees (Pmax) correlated positively with MPAP, which suggests that RV contractility increases in response to a sustained elevation of RV afterload.
RV–PA coupling
Ees/Ea (P) and Ees/Ea (V) are indices that are considered to reflect RV–PA coupling.7,9,11 Importantly, however, recent reports have claimed that Ees/Ea (P) is a reliable indicator of RV–PA coupling, whereas Ees/Ea (V) is not.18,23 A recent study by Richter et al. has directly verified the greater accuracy of Ees/Ea (P) over Ees/Ea (V). 24 One reason for this is that V0 was assumed to be zero for the calculation of Ees/Ea (V), which leads to an underestimation of RA-PA coupling, particularly when V0 is increased as in PH. In the present study, the Ees/Ea (P) of patients with PAH was similar to that of controls. In contrast, Ees/Ea (V) was lower in patients with PAH than in controls, which could be because of the above-mentioned underestimation. Taken together, RV–PA coupling was largely preserved or, if anything, mildly impaired in our PAH cohort.
RV stiffness (β) and Eed
An increased β and/or Eed has been observed in patients with PAH compared with that in controls in prior studies,6,8,11 which is consistent with our observations. We thus verified, in our Japanese PAH cohort, that the RV is stiffened. The mechanisms of the RV stiffening in our PAH cohort cannot be clarified in this study. However, in the correlation analysis, patients with PAH who were taking PAH drug(s) had a lower β than those who were not. Thus, PAH-specific drugs may reduce RV stiffness and sustain its diastolic function. This hypothesis can be supported by a basic study where PAH drugs confer anti-fibrotic effects in animal models. 25 Further basic and dedicated clinical studies are needed to investigate the pathobiology of RV diastolic function and if it could be affected by PAH drugs in PH.
Associations of RV indices with the outcome
Multivariate outcome analysis demonstrated more PAH-related hospitalization or death events in patients with PAH with decreased Ees/Ea (P), suggesting that impaired RV–PA coupling is predictive of poor outcome in PAH. This result is consistent with a recent study that reported that Ees/Ea (P) was significantly lower in patients with PAH who survived <5 years compared with those who survived >5 years. 8
In contrast, two prior studies by Brewis et al. and by Vanderpool et al. have reported no association between Ees/Ea (P) and the outcome.10,11 We do not have a clear explanation for this discrepancy; it could be due to different patient characteristics. For example, in the study by Brewis et al., 10 PH due to portal hypertension and congenital heart disease was included and the MPAP was higher (50 ± 13 mmHg) than that in our study (median MPAP: 35 mmHg). Similarly, patients with PH and lung disease or left heart disease were included in Vanderpool et al.’s study, 11 unlike ours. This allows for a speculation that the prognostic value of Ees/Ea may be evident, particularly in patients with I/H/D-PAH or CTD-PAH with a mild PAP elevation as examined in our study, whereas it may not be the same in a population with different PH type and severity. The same explanation can be applied to the lack of outcome predictability of some established prognostic indices such as right atrial pressure, cardiac output/cardiac index, and RVEF, and its mathematical transform, Ees/Ea (V).
Associations of β and Eed with outcome have also been reported.8,11 However, in the present study, no such outcome predictability was observed either for β or Eed in the multivariate analysis. This discrepancy between the previous studies and ours may be a result of the small sample size and low event rate in our study. However, importantly, no prior studies have compared the predictive value among Ees/Ea, β, and Eed in PAH and our study notably indicated the superior predictive value of RV–PA coupling indices over those of RV diastolic function in PAH. Of note, however, MPAP and PVR were lower in our patients with PAH than those in prior studies that demonstrated a prognostic value of RV diastolic function indices.8,11 Thus, larger prospective studies are warranted to further examine the prognostic value of RV–PA coupling and RV diastolic function indices in patients with different types/severity of PAH.
Cardiac involvement in SSc
Cardiac involvement in SSc is well-recognized, 26 and RV myocardial involvement has also been reported in SSc. 27 Additionally, advanced RV diastolic dysfunction in SSc-CTD-PAH has been reported, 21 which led us to hypothesize that SSc negatively affects RV function in our PAH cohort. Contrary to our expectation, all six indices of RV function did not differ between SSc-CTD-PAH and other PAH subgroups. Importantly, however, there were significant differences in age, disease duration, and PAH drug use among the three groups. Additionally, both β and Eed were higher for patients with SSc-CTD-PAH than for the other two groups, although the difference was not statistically significant. Furthermore, RV–PA uncoupling was reported to occur exclusively during exercise in a recent study. 28 Thus, we cannot deny that the SSc-CTD-PAH subset in our study might have early-phase SSc-related RV dysfunction masked at rest. Future research is needed to investigate the presence and nature of RV involvement in SSc.
Limitations
This study has several limitations. First, the sample size was insufficient for robust analyses, such as multiple comparisons among the (sub)groups. Additionally, only 13 patients met the composite endpoint during follow-up, which precluded detailed statistical analyses. Thus, as stated in the introduction, the present study is regarded as preliminary and the obtained findings need to be tested in larger prospective studies. Second, we applied a single beat method or used MRI-derived RV measures to calculate RV indices based on previous studies.5,7,10,11,18,29,30 However, measurement errors in estimating Pmax and multiple assumptions in the equations might have caused some inaccuracy. For example, the use of MPAP for end-systolic pressure might cause an underestimation of end-systolic pressure and, as a result, overestimation of Ees (Pmax) and Ees/Ea (P). 23 However, MPAP in our study cohort was relatively low and such inaccuracy seemed limited. Third, controls were not without comorbidities. In total, 14 of 18 control participants had CTD, which might have influenced the comparison between the PAH and control groups. Fourth, RHC and MRI were not performed simultaneously. All patients were stable; however, any change that occurred between the two examinations might have confounded the results. Fifth, we excluded beraprost from the list of approved PAH drugs; however, its vasodilating effect may have partially affected the pulmonary/systemic hemodynamics. Finally, this was a retrospective observational study; thus, the cause–effect relationship and time course of RV indices could not be investigated.
Conclusions
Altered RV systolic and diastolic function with RV–PA coupling in patients with PAH was observed. Furthermore, our results indicated a higher prognostic value of an index of RV–PA uncoupling than those of RV diastolic function in patients with relatively mild PAH.
Supplemental Material
sj-pdf-1-pul-10.1177_2045894020957223 - Supplemental material for Right ventriculo–pulmonary arterial uncoupling and poor outcomes in pulmonary arterial hypertension
Supplemental material, sj-pdf-1-pul-10.1177_2045894020957223 for Right ventriculo–pulmonary arterial uncoupling and poor outcomes in pulmonary arterial hypertension by Toshitaka Nakaya, Hiroshi Ohira, Takahiro Sato, Taku Watanabe, Masaharu Nishimura, Noriko Oyama-Manabe, Masaru Kato, Yoichi M. Ito and Ichizo Tsujino in Pulmonary Circulation
Footnotes
Ethical approval
Based on the Japanese legislation, the need for written informed consent was waived as all data were obtained through usual clinical work-up. This study was conducted following the Declaration of Helsinki and approved by the ethics committee of Hokkaido University Hospital (#016-0461).
Guarantor
Ichizo Tsujino, MD, PhD.
Contributorship
T.N., H.O., T.W., M.K., and I.T. collected clinical data; N.O.-M. was in charge of CMR image acquisition and analysis; M.K. provided data of patients with connective tissue disease; Y.M.I. supervised the statistical analysis; T.N. analyzed data and wrote the manuscript draft; T.S. and I.T. reviewed the statistical data and edited the draft; M.N. approved the entire content of the manuscript and submission of the paper.
Conflict of interest
The author(s) declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.