Abstract
Diabetes complicates management in a number of disease states and adversely impacts survival; how diabetes affects patients with pulmonary hypertension (PH) has not been well characterized. With insulin resistance having recently been demonstrated in PH, we sought to examine the impact of diabetes in these patients. Demographic characteristics, echo data, and invasive hemodynamic data were prospectively collected for 261 patients with PH referred for initial hemodynamic assessment. Diabetes was defined as documented insulin resistance or treatment with antidiabetic medications. Fifty-five patients (21%) had diabetes, and compared with nondiabetic patients, they were older (mean years ± SD, 61 ± 13 vs. 56 ± 16; P = 0.02), more likely to be black (29% vs. 14%; P = 0.02) and hypertensive (71% vs. 30%; P < 0.001), and had higher mean (±SD) serum creatinine levels (1.1 ± 0.5 vs. 1.0 ± 0.4; P = 0.03). Diabetic patients had similar World Health Organization functional class at presentation but were more likely to have pulmonary venous etiology of PH (24% vs. 10%; P = 0.01). Echo findings, including biventricular function, tricuspid regurgitation, and pressure estimates were similar. Invasive pulmonary pressures and cardiac output were similar, but right atrial pressure was appreciably higher (14 ± 8 mmHg vs. 10 ± 5 mmHg; P < 0.001). Despite similar management, survival was markedly worse and remained so after statistical adjustment. In summary, diabetic patients referred for assessment of PH were more likely to have pulmonary venous disease than nondiabetic patients with PH, with hemodynamics suggesting greater right-sided diastolic dysfunction. The markedly worse survival in these patients merits further study.
Diabetes has undergone unprecedented growth in the United States, with the number of diagnosed cases having increased from 1.6 million in 1958 to 21.1 million in 2010. 1 Furthermore, the proportion of those receiving a diagnosis of diabetes has increased regardless of age, sex, race, and education level. It is well known that diabetes results in systemic macro- and microvascular dysfunction, and mechanistic explanations for endothelial dysfunction have been well described. 2 Diabetic individuals are at increased risk of myocardial infarction, stroke, and death, and diabetes complicates the management of multiple medical conditions. 3 However, the impact of diabetes on the pulmonary vasculature is underappreciated. Epidemiologic studies have shown that diabetic patients are at increased risk for developing pulmonary hypertension (PH) independent of coronary artery disease, congestive heart failure, hypertension, or smoking. 4 Patients with pulmonary arterial hypertension (PAH) have significantly higher health care costs and comorbidity than patients without PAH. Thus an argument can be made for improved recognition and management of PH in diabetic individuals.
Despite the growing body of literature showing a link between diabetes and PH, the impact of diabetes on outcome in PH has not been previously well defined. This study aimed to compare the clinical, echocardiographic, and hemodynamic features of diabetic patients with PH to those of nondiabetic patients with PH and to identify the effect of diabetes on survival in patients with PH.
MATERIAL AND METHODS
Patients from two tertiary care academic-affiliated medical centers (Duke University and the Cleveland Clinic) with newly diagnosed PH (incident cases) undergoing detailed evaluation and hemodynamic assessment under the supervision of the same primary operator were prospectively entered into a dual-institution, institutional review board-approved database between 1998 and 2009. Diabetes was identified at entry by documented insulin resistance or the use of antidiabetic medications. Baseline demographic characteristics (including age, sex, and race), medical histories (including Dana Point PH classification, World Health Organization [WHO] function classification, PAH-specific medications, and the presence of other comorbidities) and laboratory test results, including hemoglobin Ale (HbA1c) values, were collected. Patients with PH owing to left ventricular (LV) systolic dysfunction were excluded from the study. Patients were stratified according to Dana Point classification (group I: pulmonary arterial hypertension; group II: pulmonary hypertension owing to left heart disease; group III: pulmonary hypertension owing to lung disease and/or hypoxia; group IV: chronic thromboembolic pulmonary hypertension; group V: miscellaneous). WHO functional class was collected at the time of catheterization (class I: PH without resulting limitation of physical activity; class II: PH resulting in slight limitation of physical activity; class III: PH resulting in marked limitation of physical activity; and class IV: PH with inability to perform any physical activity without symptoms).
Echocardiograms were performed at initial visits and subsequent follow-up visits for all patients in the study. Standard echocardiographic images were obtained in the parasternal long- and short-axis views, apical 2- and 4-chamber views, and subcostal view. In addition to calculation of the left ventricular ejection fraction (LVEF) using Simpson's method, right ventricular function (contractility) was graded on a 4-point scale (from 0 to 3) where 0 = normal, 1 = mildly decreased, 2 = moderately decreased, and 3 = severely decreased. Assessment of right ventricle (RV) size was similarly reported on a 4-point scale, where 0 = normal, 1 = mildly enlarged, 2 = moderately enlarged, and 3 = severely enlarged. The degree of tricuspid regurgitation (TR) was determined using color-flow Doppler and assigned a grade from 0 to 4+ depending on the extent of color flow relative to the right atrial (RA) area. Estimated RV systolic pressures were obtained by applying continuous wave Doppler to the TR jet and then utilizing the Bernoulli equation. This result was then added to the estimated RA pressure (RAP), obtained through assessment of the inferior vena cava size and response to inhalation. If TR velocity could not be adequately visualized by color Doppler, saline microbubbles were injected to improve image quality.
Cardiac catheterization was conducted to obtain baseline hemodynamic measurements, including RAP, mean pulmonary artery pressure (mPAP), mean pulmonary capillary wedge pressure (mPCWP), and cardiac index for all patients. Measures were then repeated during inhalation of 40 ppm of nitric oxide (iNO; Ikaria; Hampton, NJ).
Therapeutic decisions for each individual patient were left to the discretion of the referring physicians. All subsequent use of PH-specific therapies was documented. Patients were contacted by telephone at the completion of the study to verify vital status, which was initially assessed by using the Social Security Death Index. Survival was compared using Kaplan-Meier analysis.
STATISTICAL ANALYSIS
Data are presented as mean ± standard deviation for continuous variables and as percentage for discrete variables. Comparison of dichotomous variables was performed using the χ 2 test or Fisher exact test where appropriate. Comparisons of continuous variables between groups were performed using 2-sided t-tests and one-way analysis of variance. Statistical significance was assumed with P value of <.05. Survival analyses were performed using the Kaplan-Meier and proportional hazards regression methods. Statistically significant differences in the survival functions were assessed with the Wilcoxon test. Univariable analysis was performed for demographic, clinical, and hemodynamic variables. The proportional hazards model was constructed in a forward step-wise manner, investigating the effect of each covariate and potential interactions individually. Covariates examined included the baseline differences with largest statistical significance and those deemed to be clinically important to include in the model (all with P value <.05). The number of covariates included in the model was predetermined to be 8 to limit over fitting. Assumptions of the proportional hazard model were verified graphically. All analyses were performed using JMP, version 7.0 (SAS Institute; Cary, NC).
RESULTS
Diabetes was present in 55 patients (21% of the cohort). The median HbA1c for the diabetic population was 6.7 (range, 4.8–10.1). Compared with nondiabetic individuals, diabetic patients were older (61 ± 13 years vs. 56 ± 16 years; P = 0:02), more likely to be hypertensive (71% vs. 30%; P < 0:001) and more likely to be black (29% vs. 14%; P = 0:02; Table 1. Comparing underlying PH classification, diabetic patients were less likely to be WHO group I (PAH; 36% vs. 62%) and significantly more likely to be WHO group II (pulmonary venous hypertension; 24% vs. 10%; P = 0:01). At the time of referral, the severity of symptoms was similar in both groups; WHO class III or IV in two-thirds of the cohort. Diabetics had lower serum sodium levels (138 ± 3 mg/dL vs. 139 ± 3 mg/dL; P = 0:004) and worse renal function (serum creatinine level, 1.1 ± 0.5 vs. 1.0 ± 0.4; P = 0:03).
Baseline clinical characteristics according to diabetic status
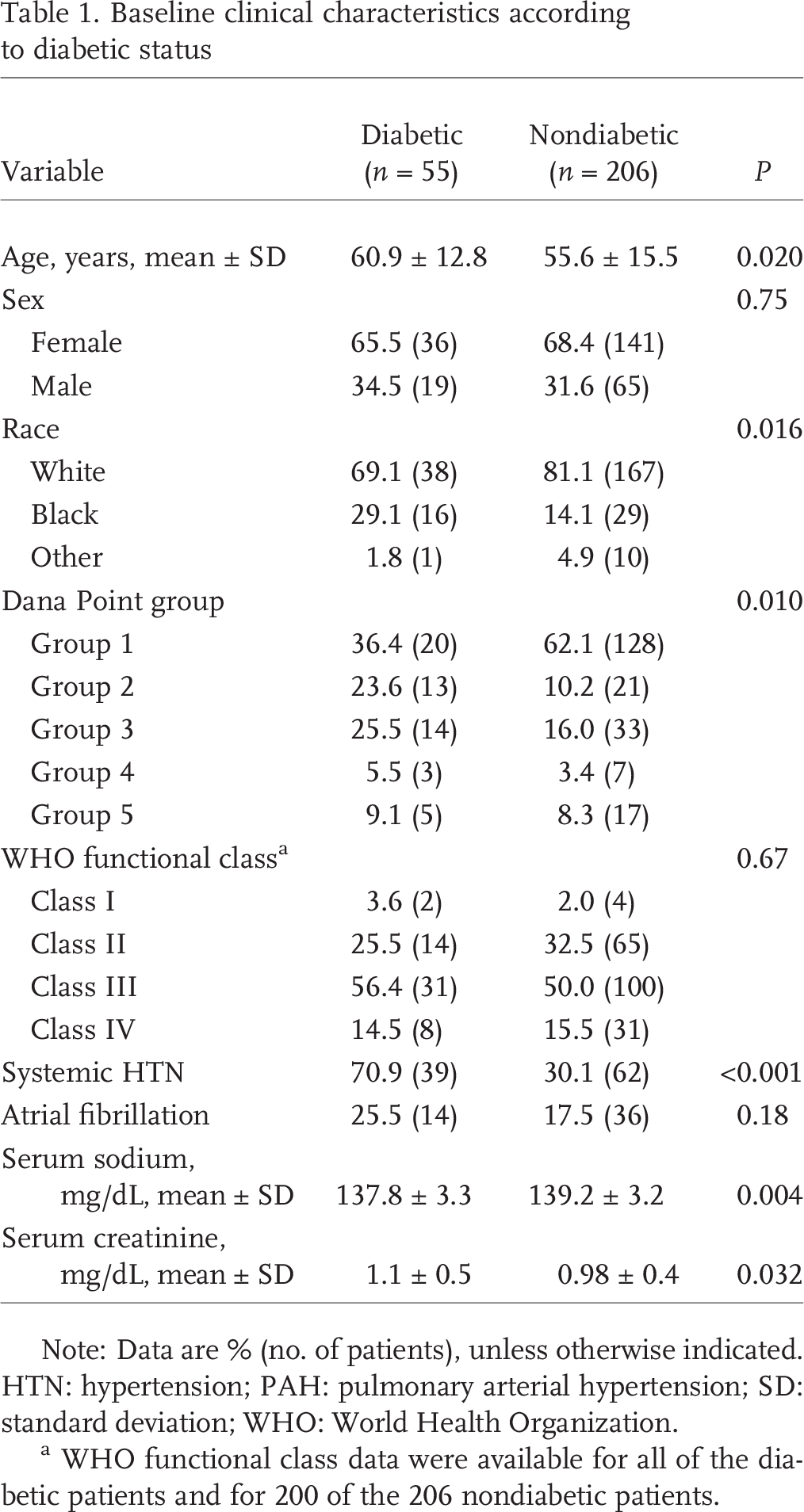
Note: Data are % (no. of patients), unless otherwise indicated. HTN: hypertension; PAH: pulmonary arterial hypertension; SD: standard deviation; WHO: World Health Organization.
WHO functional class data were available for all of the diabetic patients and for 200 of the 206 nondiabetic patients.
Baseline echocardiographic measures
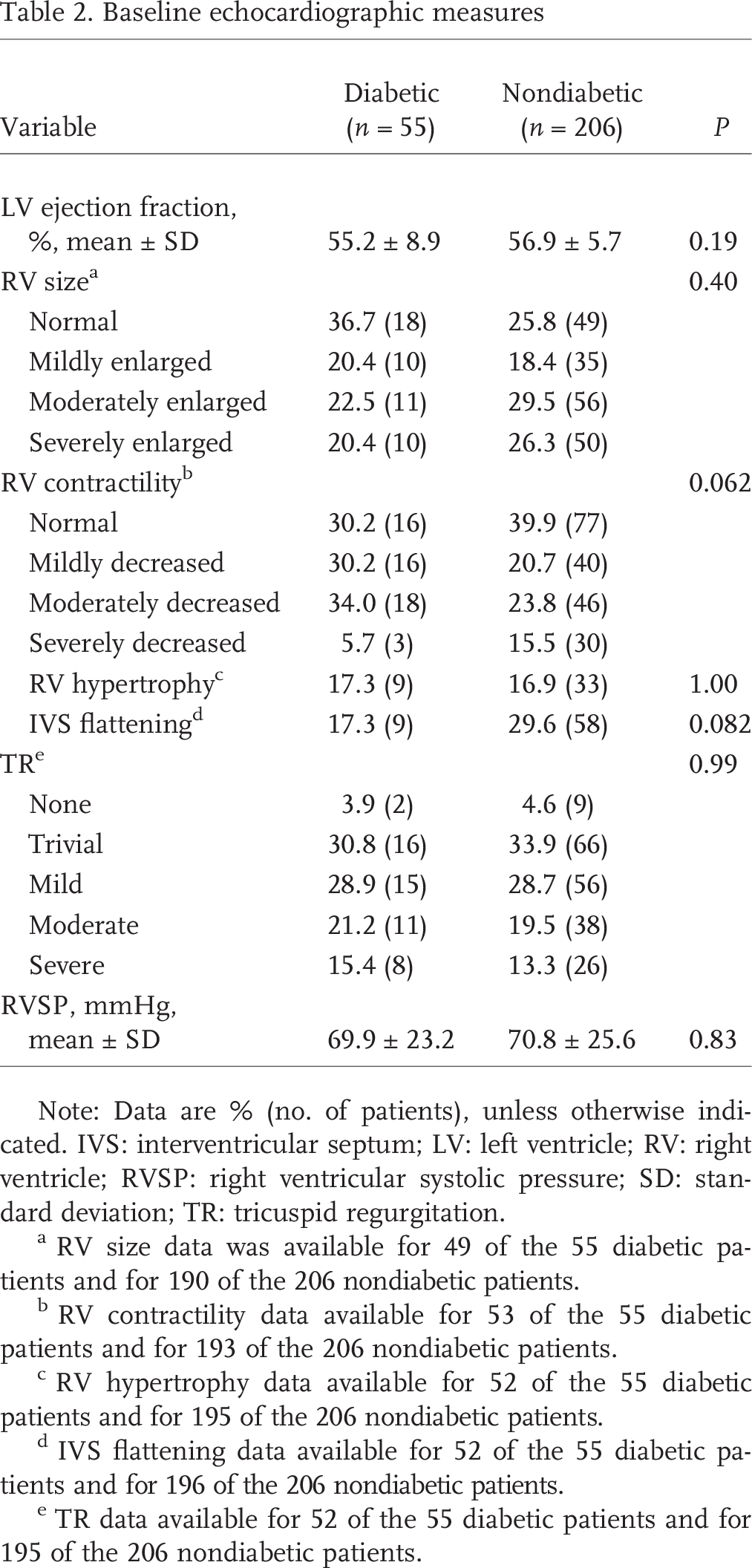
Note: Data are % (no. of patients), unless otherwise indicated. IVS: interventricular septum; LV: left ventricle; RV: right ventricle; RVSP: right ventricular systolic pressure; SD: standard deviation; TR: tricuspid regurgitation.
RV size data was available for 49 of the 55 diabetic patients and for 190 of the 206 nondiabetic patients.
RV contractility data available for 53 of the 55 diabetic patients and for 193 of the 206 nondiabetic patients.
RV hypertrophy data available for 52 of the 55 diabetic patients and for 195 of the 206 nondiabetic patients.
IVS flattening data available for 52 of the 55 diabetic patients and for 196 of the 206 nondiabetic patients.
TR data available for 52 of the 55 diabetic patients and for 195 of the 206 nondiabetic patients.
Echocardiographic measurements, including LVEF, RV size and contractility, TR grade, and estimated right ventricular systolic pressures, were similar in diabetic patients and nondiabetic individuals (Table 2). Invasive hemodynamics were also comparable, except for higher RA pressure (14 ± 8 mmHg vs. 10 ± 5 mmHg, P < 0:001) and mPCWP (17 ± 8 mmHg vs. 12 ± 6 mmHg, P < 0:0001) in diabetic patients (Table 3). Despite a trend toward lesser decrease in pulmonary pressure with iNO in diabetic patients (5 ± 5 mmHg vs. 7 ± 6 mmHg, P = 0:07), no significant difference in positive vasoreactivity (defined by a decrease in mPAP by at least 10 mmHg to less than 40 mmHg) was seen (13% of diabetic patients vs. 15% of nondiabetic patients).
Baseline hemodynamic parameters
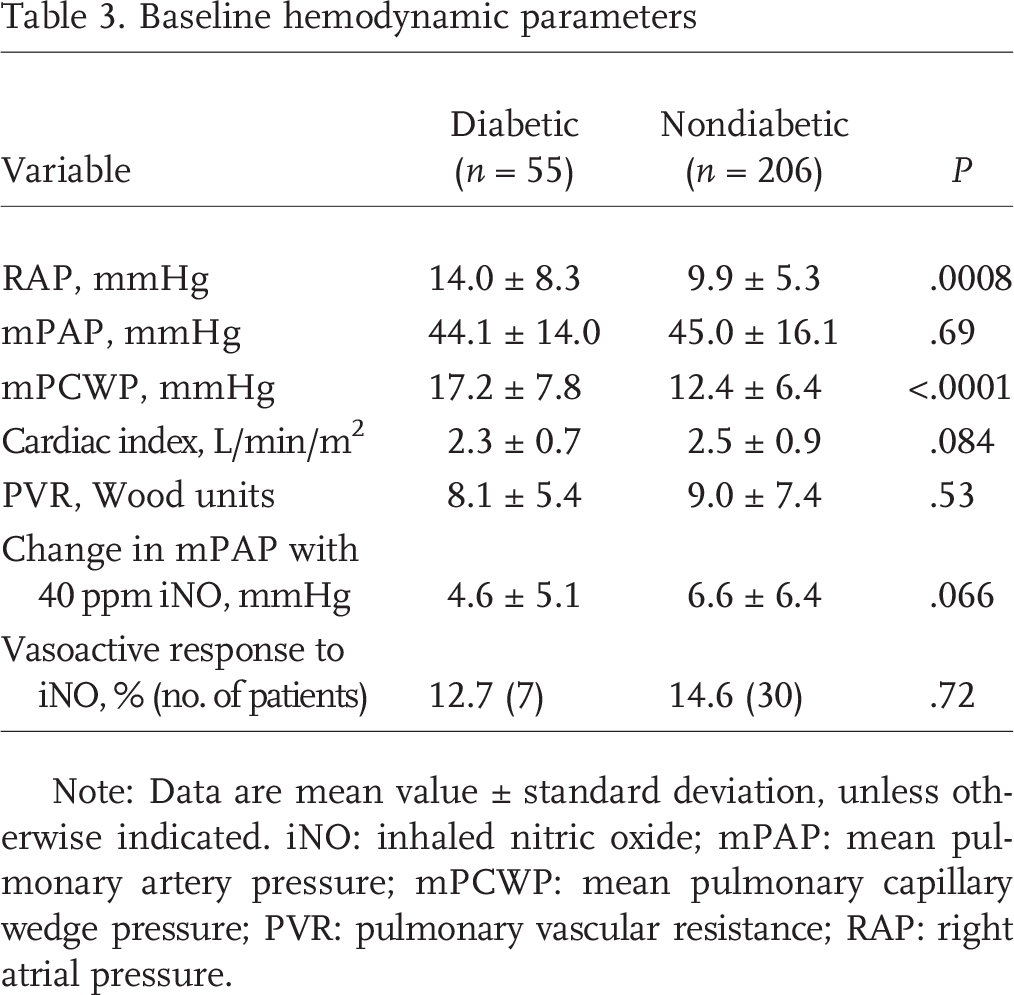
Note: Data are mean value ± standard deviation, unless otherwise indicated. iNO: inhaled nitric oxide; mPAP: mean pulmonary artery pressure; mPCWP: mean pulmonary capillary wedge pressure; PVR: pulmonary vascular resistance; RAP: right atrial pressure.
Both groups were treated similarly in terms of PAH-specific agents: 60% vs. 53% with at least one targeted drug and 13% vs. 16% receiving combination therapy (Table 4). Mortality, however, was clearly higher in the diabetic group. Five years after evaluation for PH, 70% of nondiabetic patients were alive compared with only 25% of diabetic patients (Fig. 1). Univariate predictors of survival included age, WHO functional class, RV size and contractility, TR, RAP, right ventricular diastolic pressure (RVDP), cardiac index and diabetes (Table 5). A proportional hazards analysis controlling for age, WHO functional class, and RV size (Table 6) revealed a hazard ratio for all-cause mortality of 1.7 for diabetic patients with PH compared with patients with PH without diabetes (P = 0:044; 95% confidence interval, 1.01–2.77).
List of medications used to treat pulmonary hypertension and frequency of use
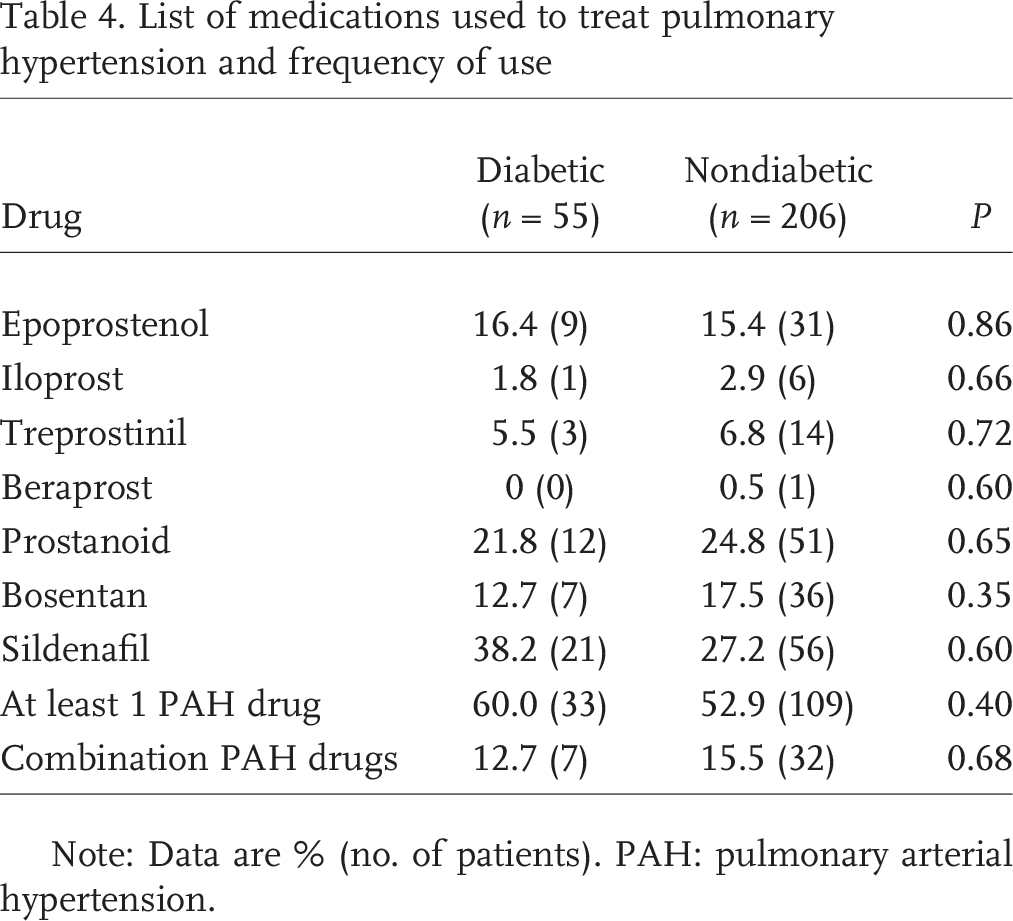
Note: Data are % (no. of patients). PAH: pulmonary arterial hypertension.
Univariate predictors of survival
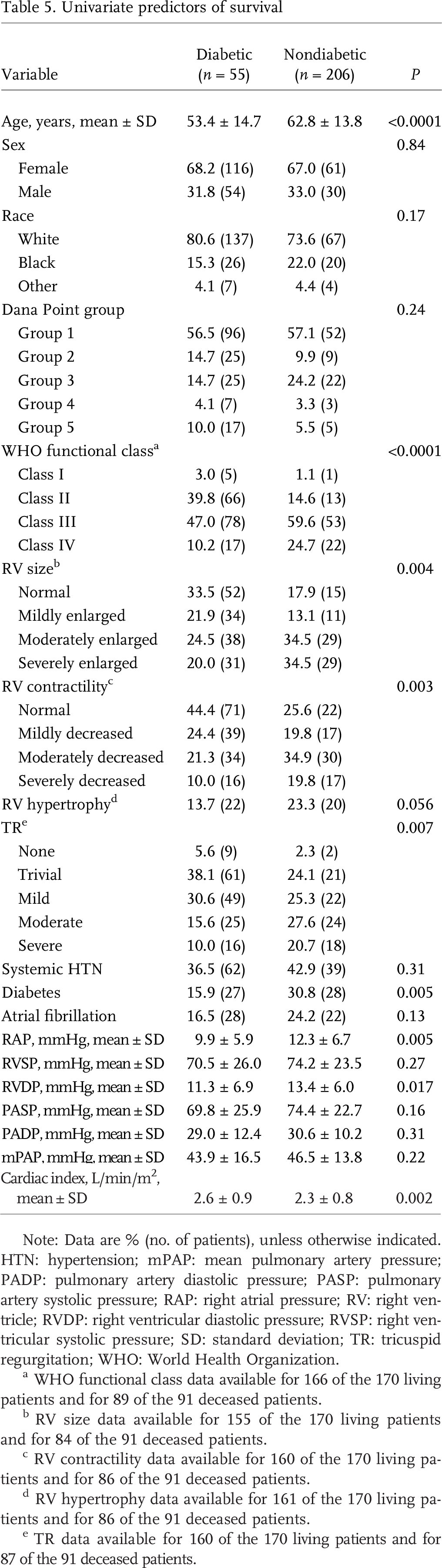
Note: Data are % (no. of patients), unless otherwise indicated. HTN: hypertension; mPAP: mean pulmonary artery pressure; PADP: pulmonary artery diastolic pressure; PASP: pulmonary artery systolic pressure; RAP: right atrial pressure; RV: right ventricle; RVDP: right ventricular diastolic pressure; RVSP: right ventricular systolic pressure; SD: standard deviation; TR: tricuspid regurgitation; WHO: World Health Organization.
WHO functional class data available for 166 of the 170 living patients and for 89 of the 91 deceased patients.
RV size data available for 155 of the 170 living patients and for 84 of the 91 deceased patients.
RV contractility data available for 160 of the 170 living patients and for 86 of the 91 deceased patients.
RV hypertrophy data available for 161 of the 170 living patients and for 86 of the 91 deceased patients.
TR data available for 160 of the 170 living patients and for 87 of the 91 deceased patients.
Univariate predictors of survival that remained significant in a multivariate analysis

Note: RV: right ventricle; WHO: World Health Organization.
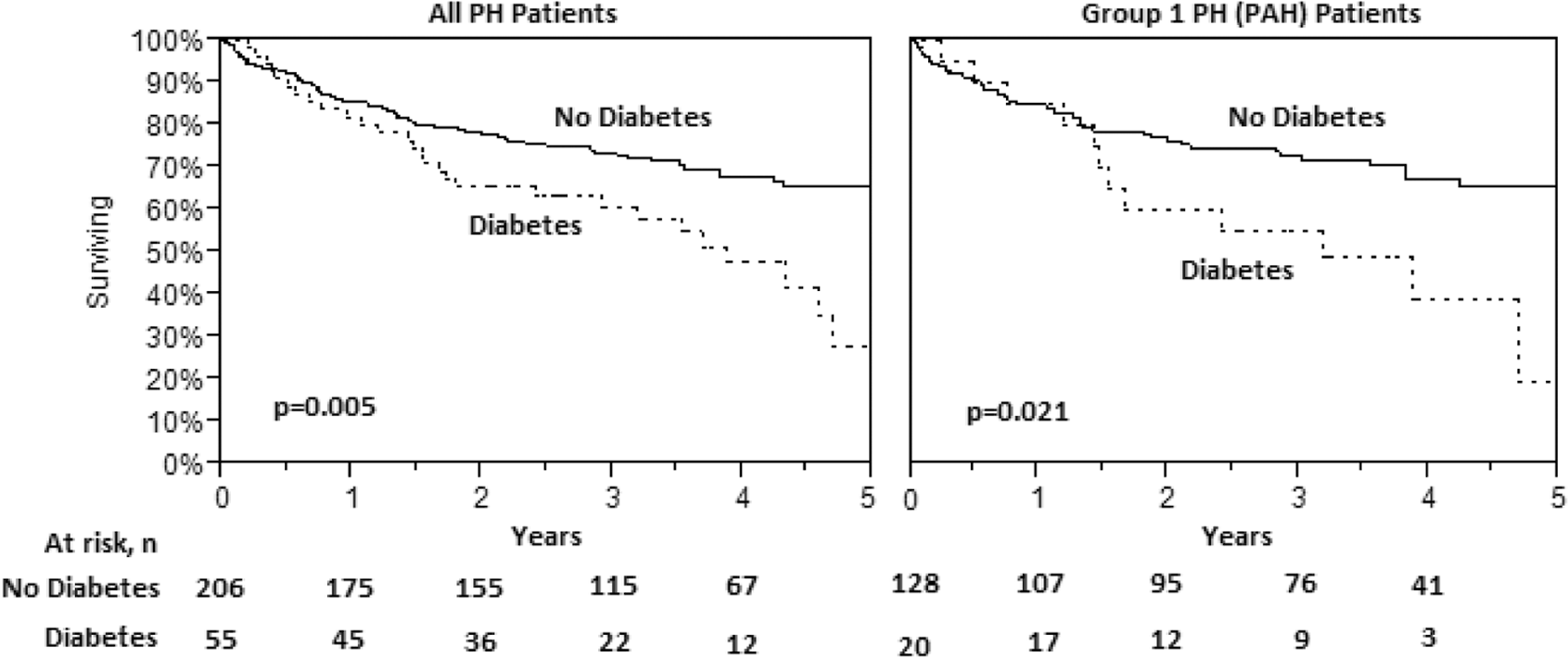
Kaplan-Meier survival curves for patients with pulmonary hypertension (PH) based on the presence or absence of diabetes. The curve on the left represents patients with all types of PH, whereas the curve on the right represents only patients with group 1 PH (i.e., pulmonary arterial hypertension [PAH]).
DISCUSSION
Diabetes was a common comorbidity in our population of patients with PH referred for invasive evaluation, and its prevalence was similar to the estimated prevalence of diabetes in the United States. 1 The diabetic patients were significantly more likely to be hypertensive and to have a pulmonary venous etiology for their condition (Dana Point group 2) than were nondiabetic patients, consistent with earlier studies. 6 Mechanistically, systemic hypertension leads to diastolic dysfunction and elevation of left-ventricular end-diastolic pressure (LVEDP), thus increasing pulmonary venous pressure and resulting in reactive vasoconstriction. Distinguishing pulmonary venous hypertension from PAH is a critical first step in determining the management of these patients, because the therapeutic approach vastly differs. Animal studies have shown that diabetes plays a role in PH not just by causing left-heart dysfunction, but also through direct effects of hyperglycemia on the pulmonary vasculature.7,8 This phenomenon may also explain the observation that patients with chronic obstructive pulmonary disease (COPD) and diabetes have more severe PH than do patients with COPD alone. 9
Although the cellular and molecular biology of endothelial dysfunction leading to PH has been extensively studied,10–18 the biologic and clinical effects of diabetes on the pulmonary arterial bed have been less well recognized. Patients with diabetes in our cohort trended toward lesser response to iNO (a smaller decrease in PA pressure). This finding is consistent with animal models of diabetes demonstrating that pulmonary vascular tone is less responsive to vasodilating substances, 7 and it supports the idea that diabetic patients may have greater intrinsic damage to their vascular endothelium. The difference, however, is subtle enough to not result in an observable difference in vasoreactivity by the commonly accepted definition. Reduced vasoreactivity is therefore unlikely to explain the difference in survival between these two groups. Although similar PA pressure, pulmonary vascular resistance (PVR), and RV systolic function were seen in our diabetic cohort, diabetic patients had significantly higher RAP, which suggests the presence of stiffer right ventricles resulting in more profound diastolic dysfunction, a finding consistent with other studies. 19 This process may be similar to the impact of insulin resistance on the left ventricle, 20 where diastolic abnormalities have been found to be much more prevalent.
A limitation of this study is how we defined diabetic patients; at the initiation of the study, HbA1c was not widely validated as a tool to diagnose diabetes. We defined diabetic patients as those patients who received oral and subcutaneous medications to control their blood sugar level. Of the patients in the diabetic group, the median HbA1c of 6.7 suggests that patients, on average, had well-controlled blood sugar levels. A recent study suggests that HbA1c may be of prognostic significance even in nondiabetic patients with PAH, 21 with an observed increase in all-cause mortality of 2.2-fold for every 1-unit increase in HbA1c. It is interesting to note that insulin resistance and undiagnosed diabetes may be relatively common in patients with PAH.22,23 In fact, a few of the patients in our cohort who were defined as nondiabetic were later found to have HbA1c values high enough to be classified as insulin resistant or even diabetic. Future studies involving diabetic patients with PH should follow the trends of HbA1c to help determine the utility of this bio-marker with regard to prognosis. Our study also demonstrates that some diabetic patients without group I PH (PAH) were treated with pulmonary artery vasodilators (Table 1): only 36% of the diabetic group was classified as having PAH (the category for which these drugs are currently approved), whereas 60% of the patients were treated with PAH-specific therapies. This implies off-label use of these agents in this patient population. Some of these agents have recently been studied as adjunctive therapy for group II PH, 24 mostly with disappointing results. We made no attempt to control medication use over the course of the study. We found use of these medications not to be associated with worsened outcomes, although the study was not powered to assess this properly.
Despite the high mortality in our diabetic cohort, the low absolute number of events limits the multivariate model and prevents more sophisticated statistical analysis, such as propensity matching. Lack of power also prevents comparisons between groups according to Dana Point classification, although inclusion in the multivariate model did not appear to lessen the impact of diabetes on survival. All these limitations are offset by the robustness of data collection and the availability of full hemodynamic and echocardiographic assessment in each of these patients, which allowed us to examine the structural and physiologic differences in patients with diabetes and PH.
In conclusion, despite similar use of disease-specific therapies for PH, long-term survival appears worse in diabetic patients. Future studies should attempt to elucidate the role of aggressive glucose control and the impact of specific antidiabetic therapies in this patient population. The new paradigm of PH and diabetes treatment may lie in modifying endothelial dysfunction. It remains to be seen whether treating systemic endothelial dysfunction (as with statins, thiazolidinediones, and angiotensin-converting enzyme inhibitors) will concurrently treat the complications of both diabetes and PH and will improve survival in this high-risk patient population.
