Abstract
In order to evaluate the therapeutic potential of fluoxetine in pulmonary arterial hypertension, 13 patients with pulmonary arterial hypertension underwent catheterization before and after 12 (N = 5) or 24 (N = 8) weeks fluoxetine therapy. No change was seen in the primary endpoint of pulmonary vascular resistance, other hemodynamic values, or any secondary endpoints.
Serotonin signaling is thought to play a significant role in the pulmonary vasculature and is considered a potential contributor to the pathophysiology of pulmonary arterial hypertension (PAH). Basic science studies suggest that antagonism of serotonin receptors 1 or blockade of the serotonin transporter2,3 may have therapeutic value. Selective serotonin reuptake inhibitor (SSRI) medications block serotonin uptake at the cell surface receptor, and this reduction in serotonin uptake has been shown to reduce pulmonary artery smooth muscle cell growth in vitro.4,5 Additionally, in both monocrotaline 6 and hypoxic 7 models of pulmonary hypertension (PH), SSRIs reduce PH severity. We therefore undertook an open label pilot study evaluating hemodynamic changes in PAH after 12- or 24-weeks of treatment with the SSRI fluoxetine.
Eligible patients had idiopathic, connective tissue disease-associated, or congenital heart disease-associated PAH with a stable PAH therapy regimen for three months and with no change in dose of PAH therapies for one month; all patients were required to have functional class (FC) II–III symptoms, be able to complete a 6-minute walk distance (6MWD) test, and have a pulmonary arterial (PA) PA mean pressure >25 mmHg, wedge <15 mmHg, and pulmonary vascular resistance (PVR) >4 wood units. Under the initial protocol, patients 1–5 received fluoxetine initiated at 20 mg/daily and increased every 2 weeks by 20 mg (as able) to a maximum of 80 mg, with down-titration allowed if necessary and follow-up testing after 12 weeks. Because some patients had difficulty with the relatively rapid SSRI uptitration, a protocol modification was then made and patients 6–13 underwent a slower uptitration with increases every 4 weeks, and the timepoint for follow-up testing was changed to 24 weeks. Outcome measures included right heart catheterization, 6MWD, the short-form 36 quality of life survey (SF-36) and the quick inventory of depressive symptomatology self-report (QIDS-SR) depression questionnaire. The study was powered around an anticipated baseline PVR of 9 Wood units, a change in PVR of 20% (1.8 Wood units), and a standard deviation of 1.8 Wood units. Planned enrolment was 16 patients (16 completions), resulting in 80% power (alpha 0.05) using the above assumptions; additional patients could be enrolled if dropouts occurred in order to achieve the target sample size. The primary endpoint was change in PVR between baseline and follow-up. Secondary endpoints were changed in other clinical variables including other hemodynamic results (right atrial pressure, pulmonary artery pressure, cardiac output, cardiac index, and pulmonary artery oxygen saturation), 6MWD, proportion of patients in FC 1 or 2, and change in SF-36 and QIDS-SR depression questionnaire. The Wilcoxon rank sum test was used to evaluate changes between baseline and follow-up data for hemodynamic results, the QIDS-SR and SF-36, while the proportion of patients with FC 1 or 2 symptoms was compared using the McNemar test. Results from the 12- and 24-week studies were analyzed together for the primary analysis; the potential for interaction by treatment duration (12 vs. 24 weeks) and by fluoxetine dose achieved (maximum vs. less than maximum) were analyzed using analysis of variance. P < 0.05 was considered significant. Studies were registered on clinicaltrials.gov: NCT00942708 (12-week study) and NCT03638908 (24-week study), the study received institutional review board (IRB) approval (study numbers STU 082013 and 052009-009) from our University IRB, and informed consent was obtained from all subjects.
A total of 13 subjects were formally screened for eligibility, and all 13 entered the study. This included five initiating the 12-week protocol and eight initiating the 24-week protocol. Eleven patients completed all end of study testing; one patient discontinued early due to medication-related side effects (anxiety which persisted despite down-titration to 10 mg), and one patient completed 24 weeks of therapy but declined to return for testing at the end of the study. Enrolment was halted prior to achieve the targeted sample size of 16 study completers, due to slow recruitment.
Mean age was 44 ± 9 years, most subjects were females (92%) and Caucasian (69%), and all patients were FC 2 (N = 5) or 3 (N = 8). The trial maximum dose of fluoxetine 80 mg daily was reached by eight patients (62%); the remainder were receiving 40 mg (N = 3), 20 mg (N = 1), and 10 mg (N = 1) at end of study or last follow-up. Adverse events limiting uptitration included anxiety, nausea, and diarrhea. Overall, there was no significant change between baseline and follow-up in any measured tests including hemodynamics, 6MWD or QIDS-SR depression scale (Table 1). There was also no significant change in the percentage categorized as FC I/II (5 at baseline, 7 at end of study) or SF-36 results. PVR, the primary endpoint, increased from 6.5 ± 2.6 to 6.8 ± 2.6 Wood units, with a median change in PVR of 0.3 (95% confidence interval (CI) −0.83 to 1.3) Wood units overall, and there was no heterogeneity seen by maximal fluoxetine dose achieved or 12 versus 24 week duration of the study. 6MWD was stable, measuring 389 ± 105 meters at baseline and 401 ± 85 meters at follow-up (median change 2.75 meters, 95% CI −78 to 116.5).
Study measures at baseline and following 12–24 weeks fluoxetine.
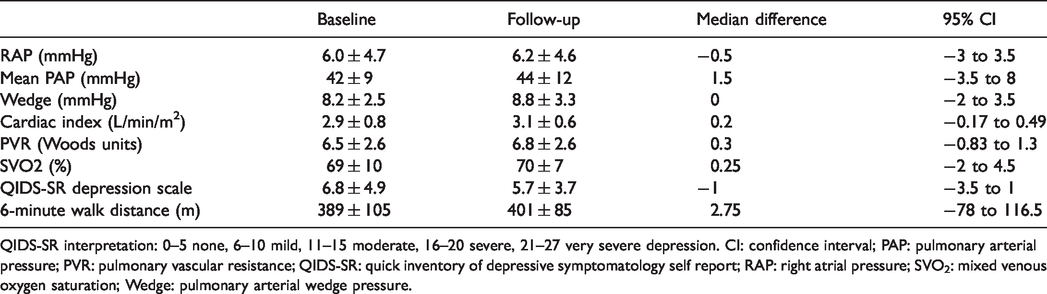
QIDS-SR interpretation: 0–5 none, 6–10 mild, 11–15 moderate, 16–20 severe, 21–27 very severe depression. CI: confidence interval; PAP: pulmonary arterial pressure; PVR: pulmonary vascular resistance; QIDS-SR: quick inventory of depressive symptomatology self report; RAP: right atrial pressure; SVO2: mixed venous oxygen saturation; Wedge: pulmonary arterial wedge pressure.
As far as clinical worsening events, there were no deaths, hospitalizations, or other episodes of clinical worsening during the study.
In summary, treatment with fluoxetine did not lead to improvement in PVR or other clinical endpoints between baseline and end of study, and we found no evidence for clinical improvement with the use of fluoxetine in PAH. Limitations of the current study include the lack of randomization, the small sample size which was below the targeted sample size used in power calculation, and the relatively short duration of follow-up. Although these design features are common to many early phase studies in PAH, these features could increase the risk of a false negative study—although we think this is unlikely in this case.
The cause of the discrepancy between these findings versus more promising animal studies is unclear, but serotonin signaling in the lungs is known to be complex and to have considerable differences across species and disease states. Additionally, animal models do not accurately replicate many features of PAH. Other medications targeting the serotonin pathway have also had disappointing trial results in human subjects, including ketanserin, a serotonin 2 A receptor antagonist that led to significant systemic hypotension, 8 and terguride, a serotonin 2 A/2B antagonist that was ineffective in a 16 week clinical trial. 9 Despite this, new studies targeting the serotonin pathway continue to be developed. Potential future targets include the serotonin 1B receptor, a potent mediator of vasoconstriction in human pulmonary arteries, combined antagonists of both the serotonin transporter and one or more receptor(s), shown to be a more effective strategy in animal models, and global reductions in serotonin signaling via inhibition of serotonin synthesis. 1
Finally, although we found no evidence to support the use of SSRIs as a potential therapy in PAH, we also found no evidence of worsening PAH. This is important, as the prevalence of anxiety, panic attacks, and depression is increased in PAH, particularly in those with more severe PAH symptoms, 10 and SSRIs remain a key medical component of therapy. Aside from their mental health effects, SSRIs have been considered both potentially beneficial as well as potentially harmful for patients with PAH in the past, and this uncertainty has led some clinicians to recommend against their use even in treating anxiety or depression. Epidemiologic studies looking at SSRI’s effect on PAH in adults have had mixed findings, with some suggesting a reduced risk of PAH development or improved outcomes in those with established PAH,11–13 and others suggesting harm.14,15 Somewhat stronger evidence has been put forward for persistent PH of the newborn after maternal SSRI use during late pregnancy16,17; however, this also has been questioned due to inconsistent results,18,19 and SSRI use during pregnancy is not considered to be contraindicated when clinically indicated. 19
In conclusion, among patients with group 1 PAH receiving fluoxetine for 12 to 24 weeks, we found no evidence of hemodynamic or other improvement versus baseline. No clinical worsening or serious adverse events occurred during treatment, but side effects associated with fluoxetine did limit dose uptitration in 5 of 13 patients.
Footnotes
Conflicts of interest
KC has received consulting fees from Actelion, United Therapeutics, Gossamer Bio and Bayer (through a grant to UCSD), fees for work as an editor from the American Heart Association, and funding for research from Actelion, Ironwood and the NIH.
Funding
Sources of support included the NIH (NHLBI) through grant K23HL105784.
Contributorship
Adetoun Sodimu and Oluwatosin P. Igenoza acquired data, assisted with analysis and interpretation and drafted key portions of the manuscript. Sonja Bartolome contributed to the design of the study and data acquisition and made critical revisions to the manuscript. Dr. Kelly M. Chin designed the study, completed the analyses, completed the first draft of the paper and is responsible for the overall work. All authors approved the final version.
