Abstract
Selexipag is an oral prostacyclin receptor agonist; it was recently approved for use in adults with pulmonary arterial hypertension. The safety and efficacy of selexipag has not yet been determined in the pediatric population. We describe short-term hemodynamic and clinical data with selexipag therapy in four pediatric patients with pulmonary hypertension. We reviewed clinical, echocardiographic, and hemodynamic data. One patient was transitioned from subcutaneous treprostinil to selexipag, and in three patients, selexipag was added as a third agent. Drug dosing was attained empirically based on patient body size. A follow-up catheterization was performed 12–18 months after initiation of selexipag therapy. All four patients tolerated selexipag well, without significant side effects. One patient transitioned successfully from subcutaneous treprostinil to selexipag. None of the four patients had clinical deterioration. In three patients who were able to perform a 6-minute walk test, pre and post selexipag distances were 350 and 400, 409 and 390, and 300 and 360 m, respectively. Echocardiograms showed no significant changes. Catheterization showed a variable change in pulmonary vascular resistance (small decrease in three patients and increase in one patient). Brain natriuretic peptide levels before and after selexipag in the four patients were 38 and 49, 33 and 54, 29 and 25, and 12 and 14 pg/mL, respectively. Selexipag use for 16–28 months was safe in four pediatric patients; none of them had clinical deterioration. Larger number of patients and longer follow-up intervals are necessary before further recommendations can be made.
Introduction
Abnormal vascular smooth muscle cell contractility, proliferation, and migration are important processes in the development of pulmonary arterial hypertension (PAH). Three molecular pathways have been the target of PAH-specific medications, including nitric oxide—cyclic guanosine monophosphate—phosphodiesterase, endothelin receptors, and prostacyclin receptors. The prostacyclin receptor (IP) is a membrane receptor coupled to a Gs type protein, which leads to an increase in cAMP, resulting in vasodilatory and anti-aggregatory effects. IP is also coupled to Gi and Gq proteins, which causes a reduction in vascular smooth muscle cell contraction, proliferation, and migration. Despite a reduction in IP receptor levels in end-stage PAH, the antiproliferative effects of IP receptor agonists appear to be preserved, with concurrent activation of both the EP4 receptor and the PPARy pathway. Data suggest that both IP receptor-dependent and -independent effects are responsible for the antiproliferative effects of prostaglandin analogs.1–4
Selexipag, a selective IP receptor agonist, was approved in December 2015 by the Food and Drug Administration for the treatment of PAH World Health Organization (WHO) Group 1 patients with functional class II or III to delay the disease progression and reduce the risk of hospitalization. Selexipag has a similar mechanism of action as prostacyclin (PGI2); however, selexipag is a nonprostanoid IP receptor agonist. Selexipag works as a pro-drug. Its active metabolite (ACT-333679) has a 130-fold higher selectivity for the IP receptor than other prostanoid receptors. This high selectivity is probably responsible for the low side effect profile of selexipag compared with the PGI2 analogs. Moreover, it appears that selexipag does not cause IP receptor desensitization and internalization, which avoids the tachyphylaxis observed with PGI2 analogs.5,6
There are few published reports on the use of selexipag in the pediatric population.7,8 We describe preliminary and short-term hemodynamic (12–18 months) and clinical (16–28 months) data with selexipag use in four pediatric patients, one a former premature infant with chronic lung disease and three with idiopathic PAH.
Case 1
Summary of hemodynamic and clinical data for patient no.1.
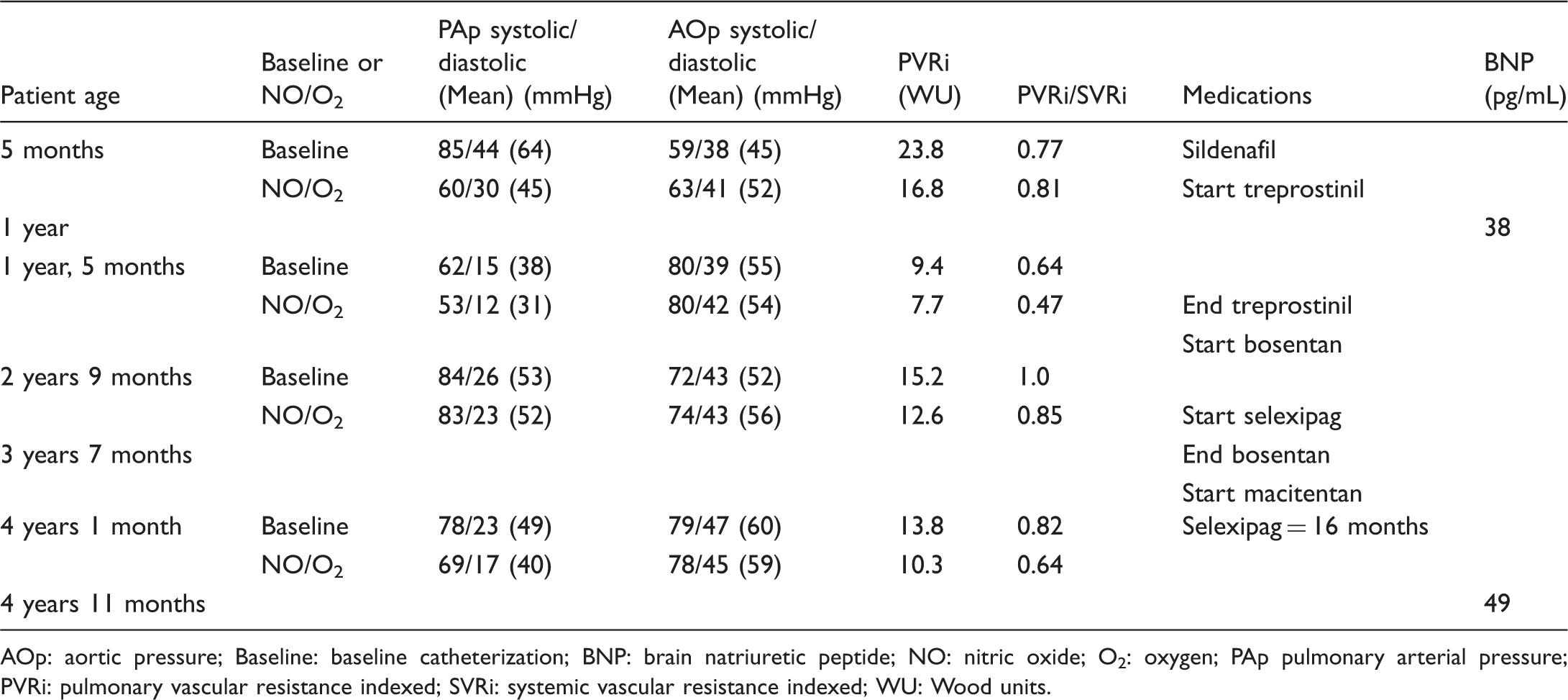
AOp: aortic pressure; Baseline: baseline catheterization; BNP: brain natriuretic peptide; NO: nitric oxide; O2: oxygen; PAp pulmonary arterial pressure; PVRi: pulmonary vascular resistance indexed; SVRi: systemic vascular resistance indexed; WU: Wood units.
At 3.5 years of age, his liver enzymes were elevated, and he was therefore switched from bosentan to macitentan. Sixteen months after starting selexipag, his weight was 13.4 kg and he was on nightly oxygen, sildenafil 10 mg three times a day, selexipag 600 mcg twice a day, and macitentan 3 mg daily. A cardiac catheterization showed systemic level pulmonary arterial pressure with a PVRi of 13.8 WU. Twelve months later, he continued to do well clinically without adverse effects from the medications. His brain natriuretic peptide (BNP) level was 49 pg/mL, compared with a BNP level of 38 pg/mL prior to starting selexipag. His echocardiogram was unchanged showing good left ventricular (LV) function, moderate right ventricular dilatation with good function, moderate flattening of the interventricular septum in systole and trace tricuspid regurgitation.
Case 2
Summary of hemodynamic and clinical data for patient no.2.
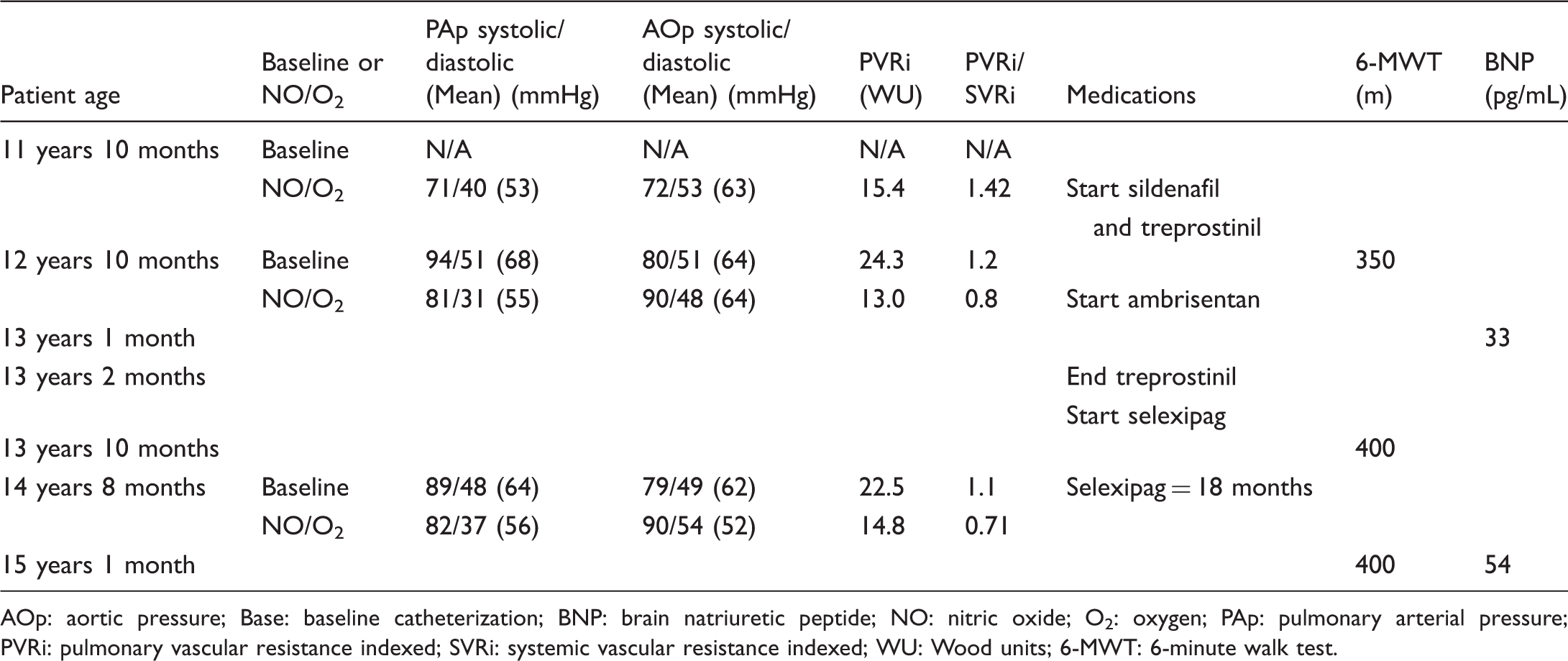
AOp: aortic pressure; Base: baseline catheterization; BNP: brain natriuretic peptide; NO: nitric oxide; O2: oxygen; PAp: pulmonary arterial pressure; PVRi: pulmonary vascular resistance indexed; SVRi: systemic vascular resistance indexed; WU: Wood units; 6-MWT: 6-minute walk test.
She was started on ambrisentan. Four months later, the patient was suicidal and requested discontinuing treprostinil (which was at a dose of 40 ng/kg/min). She was transitioned to selexipag over a 4-week period, starting with a dose of 200 mcg twice a day, increasing weekly until she reached 1000 mcg twice a day. Simultaneously, the subcutaneous treprostinil was weaned by 25% a week. The patient reported significant improvement in energy level and exercise tolerance in the ensuing weeks. Eighteen months later, she weighed 58 kg and was on nightly oxygen, sildenafil 20 mg three times a day, selexipag 1000 mg twice a day, and ambrisentan 10 mg daily. At cardiac catheterization, the pulmonary arterial pressure was slightly suprasystemic, with a PVRi of 22.5 WU. Oxygen and nitric oxide administration resulted in subsystemic pulmonary arterial pressure; the PVRi decreased to 14.8 WU. Six months later, the patient was doing well clinically with good exercise tolerance and no symptoms. She carried her backpack at school without difficulties. Her BNP level was 54 pg/mL compared with a BNP level of 33 pg/mL prior to starting selexipag. Her 6-minute walk test (6-MWT) distance 2 years after starting selexipag was 400 m compared with 350 m before starting selexipag.
Case 3
Summary of hemodynamic and clinical data for patient no. 3.
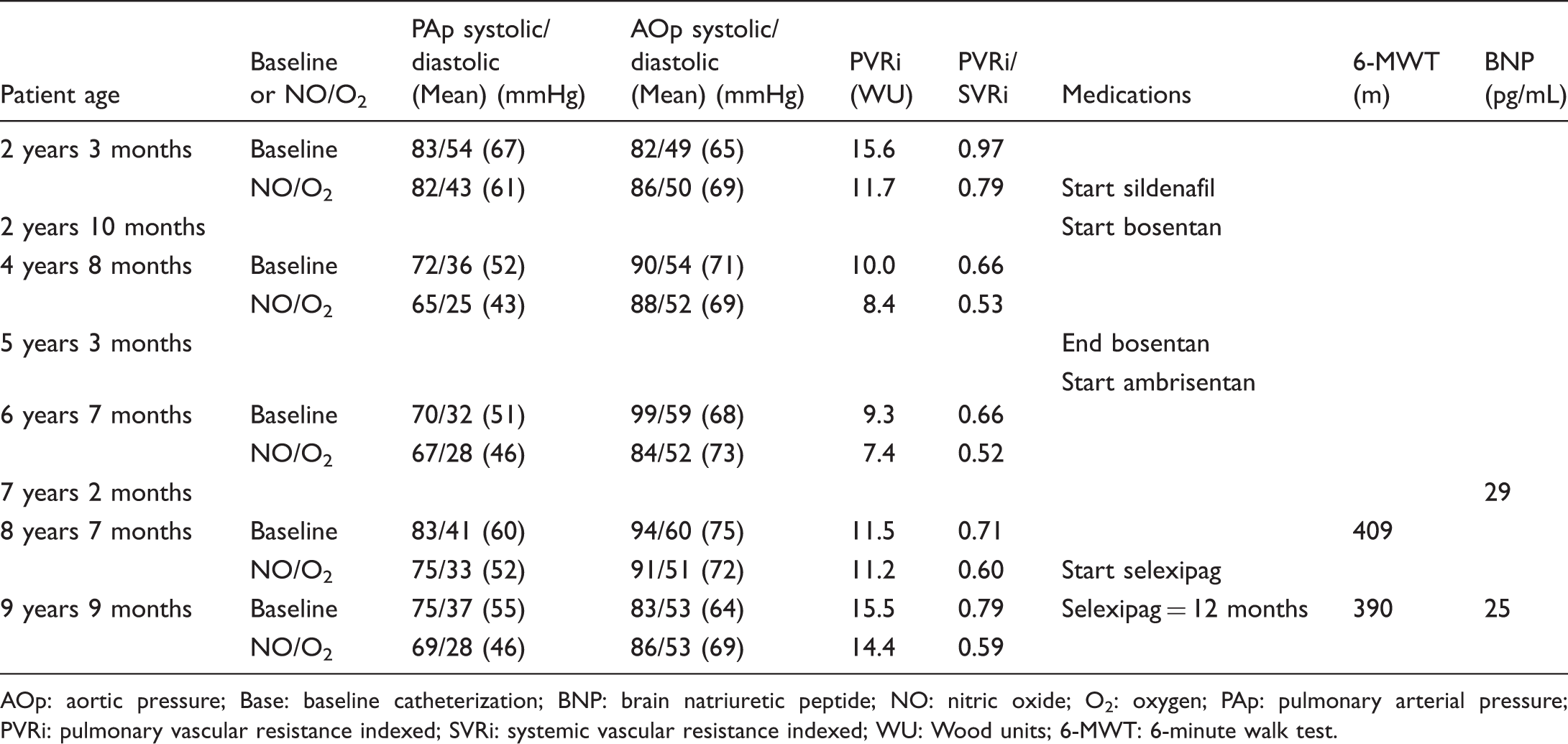
AOp: aortic pressure; Base: baseline catheterization; BNP: brain natriuretic peptide; NO: nitric oxide; O2: oxygen; PAp: pulmonary arterial pressure; PVRi: pulmonary vascular resistance indexed; SVRi: systemic vascular resistance indexed; WU: Wood units; 6-MWT: 6-minute walk test.
Six months later, she was switched from bosentan to ambrisentan. A cardiac catheterization at 6.5 years showed pulmonary pressures at 72% of systemic level with a PVRi of 9.3 WU. A repeat cardiac catheterization at 8.5 years of age showed slightly subsystemic pulmonary arterial pressure, with a PVRi of 11.5 WU. She was started on selexipag, increasing the dose up to 600 mcg twice a day over 2 weeks. Due to dizziness and decreased activity level, unclear if related to the new medication, the dose was transiently decreased to 400 mcg twice a day, and a few weeks later increased back to 600 mcg twice a day. Twelve months after starting selexipag, she weighed 29 kg and was on nightly oxygen, sildenafil 20 mg three times a day, ambrisentan 5 mg a day, and selexipag 800 mcg twice a day. A catheterization showed subsystemic pulmonary arterial pressure with a PVRi of 15.5 WU. Her BNP level was 25 pg/mL compared with 29 pg/mL prior to starting selexipag. Her 6-minute walk distance was 390 m compared with 409 m prior to starting selexipag. Four months later, the patient was doing well clinically, without significant limitations.
Case 4
Summary of hemodynamic and clinical data for patient no. 4.
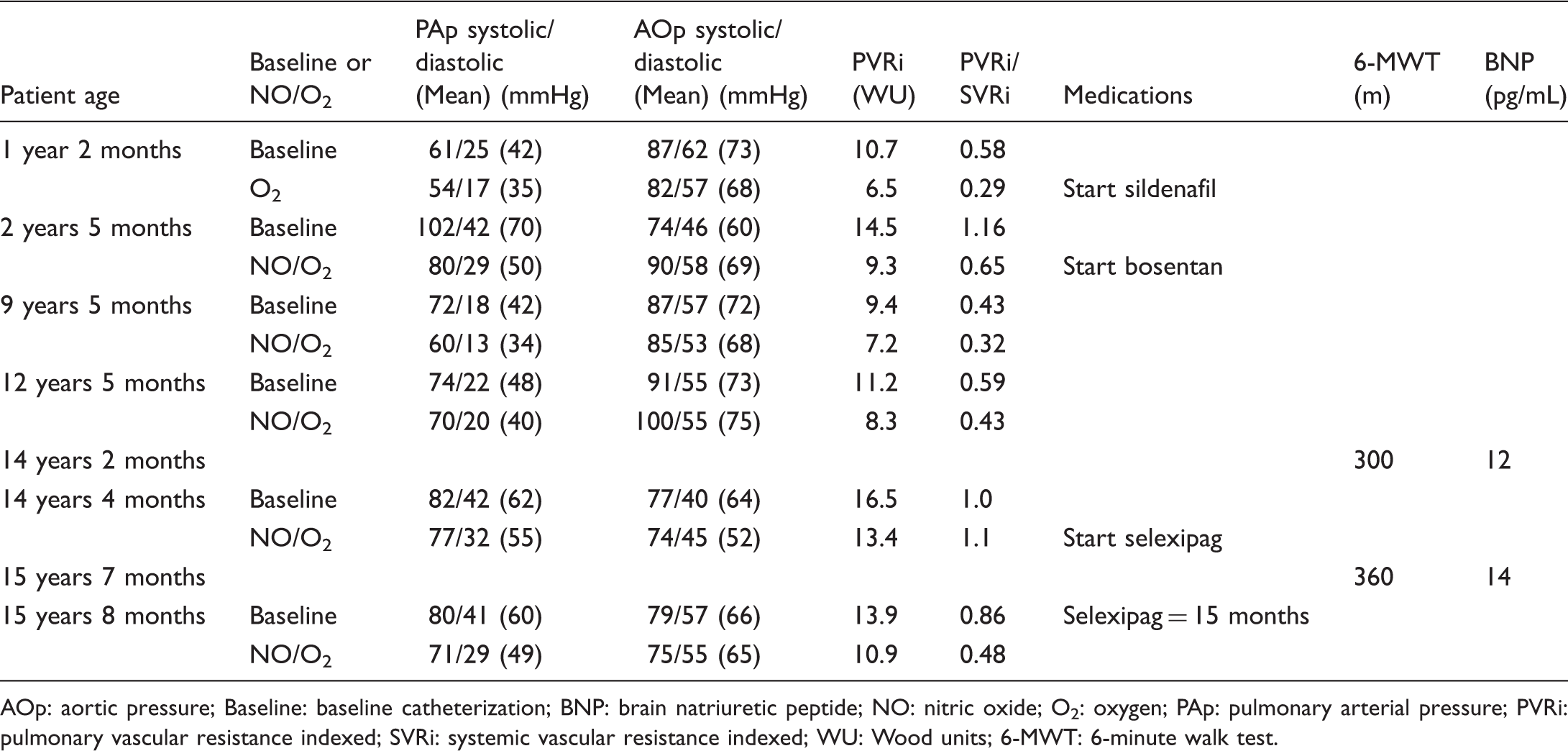
AOp: aortic pressure; Baseline: baseline catheterization; BNP: brain natriuretic peptide; NO: nitric oxide; O2: oxygen; PAp: pulmonary arterial pressure; PVRi: pulmonary vascular resistance indexed; SVRi: systemic vascular resistance indexed; WU: Wood units; 6-MWT: 6-minute walk test.
She did well clinically for several years and underwent catheterizations at 9.5 and 12.5 years of age revealing subsystemic pulmonary arterial pressures. A cardiac catheterization performed at 14 years of age showed slightly suprasystemic pulmonary arterial pressure with a PVRi of 16.5 WU. Her WHO functional class was II. Selexipag was started at a dose of 200 mcg twice a day, increasing the dose weekly up to 1000 mcg twice a day; however, she developed diarrhea and headaches. The dose was decreased transiently to 800 mcg twice a day with resolution of side effects. Fifteen months later, she weighed 67 kg and was on nightly oxygen, sildenafil 20 mg three times a day, bosentan 62.5 mg twice a day, and selexipag 1000 mcg twice a day. A repeat catheterization showed systemic level pulmonary arterial pressure with a PVRi of 13.9 WU. After the administration of oxygen and nitric oxide (20 ppm), the PVRi decreased to 10.9 WU. Her BNP level was 14 pg/mL compared with 12 pg/mL prior to starting selexipag. Her 6-minute walk distance was 360 m compared with 300 m prior to starting selexipag. Four months later, the patient was on selexipag at a dose of 1200 mcg twice a day and was doing well clinically.
Discussion
Pediatric patients with PAH have traditionally been started on one or two drugs, often a phosphodiesterase 5 inhibitor and/or an endothelin receptor antagonist. Patients with severe PAH or disease progression have been treated with intravenous (IV) or subcutaneous prostacyclin analogs. However, the difficulties with drug storage and delivery and significant side effects and complications of the latter have limited their use. The recent approval of an oral prostacyclin receptor agonist, selexipag, has provided a newer option to target this molecular pathway.
The GRIPHON study (NCT01106014) 9 was a multicenter, phase 3 study, in adult patients (selexipag: n = 574; placebo: n = 582) with symptomatic (WHO class II and III) PAH. Selexipag treatment duration was up to 4.2 years, with a median of 1.4 years. Selexipag reduced the risk of time to first morbidity or mortality event by 40% (p < 0.0001). The most frequent adverse effects of selexipag in that and other studies were headache, myalgia, nausea, pain in the jaw, arthralgia, skin irritation, diarrhea, pain in the upper abdomen, and dizziness. Compared with PGI2 analogs, selexipag did not cause significant platelet dysfunction and was noted to cause stronger relaxation of the pulmonary arteries. Bioavailability of selexipag after oral administration was demonstrated to be approximately 50%.5,6,9–16
The first report of a pediatric patient treated with selexipag was a 12-year-old girl with WHO functional class III, right ventricular failure, recurrent syncope, dizziness, and progressive fatigue. 7 The patient had been previously treated with bosentan and sildenafil with no improvement for 9 months. Selexipag was started and advanced to the maximal dose of 1600 mcg within 10 days. Six months later, the patient had a decrease in PVR, right atrial pressure, right ventricular end-diastolic pressure, right atrial and right ventricular size, recovery of vasoreactivity and improved cardiac index, 6-minute walking distance, functional class, and body weight. Her only side effect was mild to moderate nausea.
The largest reported series of selexipag use in the pediatric age group is from the UCLA Mattel Children’s Hospital, 8 consisting of 10 patients (5 with idiopathic PAH, 4 with congenital heart disease, and 1 with congenital diaphragmatic hernia) with a mean age of 16.5 years. The patients were on a phosphodiesterase 5 inhibitor and an endothelin receptor antagonist; four were on IV treprostinil. The four patients on IV treprostinil were successfully transitioned to oral selexipag. Selexipag was well tolerated, with the most common side effects being headaches, loose stools, and jaw pain. The maximum dose of 1600 mcg was achieved in all but one patient. The patients reported improved energy, stamina, exercise tolerance, and decrease in oxygen requirement. One patient became a candidate for cavopulmonary anastomosis only after starting selexipag. Among 7 of the 10 patients who performed 6-MWT before and after selexipag therapy, 3 had an increase and 4 had a decrease in walk distance. Hemodynamic data were not obtained routinely before or after starting selexipag.
In a recent research letter, Koestenberger and Hansmann 17 briefly summarized their experience using selexipag in nine patients ranging in age from 1.5 to 17 years and weight from 7 to 76 kg. Final doses were 400–1600 mcg twice daily. In three of the nine patients, they noted desaturation of more than 5%. When the dose was decreased by 200 mcg twice daily, the saturations improved, presumably from less intrapulmonary shunting and subsequently lower drug metabolite levels. The most common side effects they observed were nausea, vomiting, flushing, and headache, all of which improved with anti-emetics.
Here, we report hemodynamic data, 6-MWT results, and BNP levels before and 12–18 months after starting selexipag in four pediatric PAH patients. In one patient, selexipag was used as a substitute for subcutaneous treprostinil, in the other three patients it was added as a third agent due to hemodynamic deterioration. Each of the patients did well clinically for 16–28 months after initiating selexipag. Two of the three older patients reported improved exercise tolerance and energy levels in the first few months after starting selexipag; one reported no change. Two patients had transient side effects, which resolved with temporary reduction of the selexipag dose. The maximum dose utilized in each patient was empiric, based on body weight. At cardiac catheterization, PVRi did not change significantly (small decrease in three patients and a small increase in one patient). A 6-MWT could be performed in three of the patients; the walk distance increased slightly in two and decreased slightly in one. BNP levels were in the normal range (<100 pg/mL) in all four patients before and after selexipag therapy; there was a small decrease in one and a slight increase in three in BNP levels after the selexipag course.
We speculate that with added experience, selexipag could be an alternative for patients who are on subcutaneous or IV prostacyclin analogs and have significant adverse effects with administration or tolerance. The other potential use for selexipag could be as a third agent for WHO class II or III patients who have worsening on a phosphodiesterase inhibitor and an endothelin receptor antagonist. Whether selexipag could be the second agent of initial double therapy or the third agent of initial triple therapy for pediatric PH patients remains to be determined by future studies.
There are limitations to our study. One is the small number of patients, which precludes definitive conclusions about the safety and effectiveness of selexipag in the pediatric age group. In case 1, bosentan was replaced by macitentan while on selexipag, and in case 2, ambrisentan was started on the patient 4 months before selexipag. In these two cases, it is not possible to know whether some of the beneficial effect was from the endothelin receptor antagonist and not selexipag. Also, the follow-up period of 16–28 months was relatively short. In addition, the final selexipag dose attained in each patient may have been suboptimal. A clinical phase 2 study, with about 55 PAH participants aged 12–18 years, has the objective of determining more optimal doses of Selexipag in children with PH. It started enrolling in 2018 and has an estimated study completion date of December 2025 [NCT03492177]. 18
In summary, our experience with selexipag for pediatric PH adds to the limited published literature and suggests that selexipag was well tolerated, with minimal side effects that resolved with transient dose reduction. None of the patients showed clinical signs of worsening. Hemodynamic data were generally reassuring. Larger clinical experience and duration of therapy will be necessary to make more definite recommendations regarding the use of selexipag in pediatric patients with PH.
Footnotes
Authors’ contribution
Abraham Rothman: Concept, design, analysis, draft, critical revision, approval of final version. Gabriel Cruz, William N. Evans, and Humberto Restrepo: Data acquisition, analysis, critical revision, and approval of final version.
Conflict of interest
The author(s) declare the following conflicts of interest: Rothman reports speaker fee and educational conference support from Actelion Pharmaceuticals. Evans reports educational conference support from Actelion Pharmaceuticals. The other authors declared no conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
This investigation received approval from the Sunrise Health Institutional Review Board, approval number 8398, and the study protocol conforms to the principles of the Declaration of Helsinki. We obtained parental informed consent for the invasive procedures.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
