Abstract
Venous thromboembolism (VTE) encompasses deep-vein thrombosis and pulmonary embolism (PE). It is the third-most-frequent cardiovascular disease, with an overall annual incidence of 1–2 per 1,000 population. Chronic thromboembolic pulmonary hypertension (CTEPH) is regarded as a late sequela of PE, with a reported incidence varying between 0.1% and 9.1% of those surviving acute VTE. Right ventricular (RV) function is dependent on afterload. The most precise technique to describe RV function is invasive assessment of the RV–to–pulmonary vascular coupling. However, assessments of RV afterload (i.e., steady and pulsatile flow components and their product, the RC-time) may be useful hemodynamic surrogates of coupling. RV load is different in acute and chronic PE. In acute PE, more than 60% occlusion of the crosssectional area of the pulmonary artery within a short period of time leads to abrupt hemodynamic collapse. If the time of occlusion is limited to ∼15 seconds, significant decreases in fractional area change, tricuspid annulus systolic excursion, and RV free-wall deformation (strain) occur, with the latter showing significant postsystolic shortening. These changes have similarities to ischemic stunning, and they recover within minutes. In CTEPH, studies of pulmonary vascular resistance (PVR) and pulmonary arterial compliance demonstrated low RC-times that were further lowered after pulmonary endarterectomy (PEA). Immediate postoperative PVR was the only predictor of long-term survival/freedom from lung transplantation, suggesting that the effect of PEA on opening vascular territories to flow outweighs its effect on proximal stiffness. This review summarizes the current knowledge on vascular and intrinsic RV adaptation to VTE, including CTEPH, and the role of imaging.
Venous thromboembolism (VTE) encompasses deep-vein thrombosis (DVT) and pulmonary embolism (PE). It is the third-most-frequent cardiovascular disease, with an overall annual incidence of 1–2 per 1,000 population. Chronic thromboembolic pulmonary hypertension (CTEPH) is regarded as a late sequela of PE. Right ventricular (RV) function is an important determinant of long-term outcome in patients with acute PE and CTEPH. In these conditions, the right ventricle (RV) is subjected to abnormal and increased loading that varies in timing, magnitude, and duration. Consequently, RV dysfunction and pulmonary hypertension (PH) are variably present at initial presentation of acute PE. After an episode of PE, pulmonary hemodynamics and RV function normalize within a few weeks in the majority of patients. CTEPH results from persistence of thrombotic obstructions in the pulmonary vasculature in the presence of significant positive and obliterative vascular remodeling and chronic elevation in pulmonary pressures. CTEPH leads to a progressive increase in RV afterload, causing RV dysfunction and eventually RV failure and death. The condition is underdiagnosed, and the true prevalence is still unknown. Prognosis of acute PE and development of CTEPH can be predicted by pulmonary artery pressures and RV function at the time of diagnosis of the first episode of PE. 1 While noninvasive diagnostics are performed in acute PE, right heart catheterization and pulmonary angiography remain gold standards for the diagnosis of CTEPH.
INVASIVE ASSESSMENT OF RV AFTERLOAD
RV function is dependent on RV afterload, which consists of pulmonary vascular resistance (PVR; steady flow load of the RV), pulmonary arterial compliance (CPA; oscillatory load of the RV during systole), and characteristic impedance (Z) of the proximal pulmonary artery. 2 RV hydraulic load is determined by the dynamic interaction between PVR and CPA. 3 RC-time, the product of resistance and compliance, represents the exponential pressure decay in the pulmonary artery during diastole. 4 The CPA relates to oscillatory load and has been shown to be of greater prognostic importance than resistance 5 and to be associated with RV dysfunction5,6 in patients with idiopathic pulmonary arterial hypertension (iPAH).
By means of the pulmonary artery occlusion technique, the decay from pulmonary artery pressure level to pulmonary arterial wedge pressure level can be assessed to estimate the pressure in precapillary small pulmonary arteries (POCCL).7–9 With POCCL, PVR can be partitioned into largerarterial (upstream resistance) and small-arterial plus venous (downstream resistance) components7–9 with the pulmonary artery occlusion technique.
INVASIVE ASSESSMENT OF RV FUNCTION
RV function can be directly measured by conductance catheterization and an instantaneous integration of RV pressure, and pulmonary arterial flow (so-called pressure-volume loops) can be derived. Diastolic RV function can be assessed by RV end-diastolic pressure and volume, the minimal rate of RV pressure change (dP/dtmin), the RV stiffness constant (β), and the isovolumetric relaxation time constant (τ).10–12 The adaptation of the RV to its afterload can be assessed by quantification of the coupling of RV systolic function to arterial elastance. Two measures are critical for the assessment of right ventricular–to–pulmonary vascular (RV-PV) coupling: (1) end-systolic ventricular elastance (Ees = end-systolic pressure/end-systolic volume), which is the best possible load-independent measure of contractility, and (2) arterial elastance (Ea = end-systolic pressure/stroke volume [SV]), a measure of afterload that opposes the RV. The Ees and Ea can be graphically derived from pressure-volume loops of the RV. 13 RV-PV coupling can be calculated by dividing Ees by Ea (Ees/Ea ratio). The optimal matching of systolic ventricular and arterial elastances occurs at an Ees/Ea ratio of ∼1.5. An isolated increase in Ea or decrease in Ees leads to a decrease in Ees/Ea ratio, indicating decoupling of the ventricle from its arterial system and a decrease in SV. However, in contrast to that in the left ventricle (LV), measurement of Ees in the RV might be inaccurate because of the complex geometry of the RV and the triangular shape of the RV pressure-volume loop, resulting from the high compliance of the pulmonary vasculature 14 and from the fact that RV ejection continues after end-systole. This limitation can be resolved by measuring pressure-volume loops during preload reduction of the RV by temporary balloon occlusion of the inferior vena cava. However, this method is very invasive and may cause alterations in sympathetic tone, thus leading to alterations in hemodynamics. An alternative method that has been proposed to assess Ees without the need for preload reduction is the single-beat method.15,16 With the single-beat method, the maximal ventricular pressure (Pmax), as encountered in an isovolumetric nonejecting beat, can be estimated by fitting a sinus wave over the RV pressure curve during the isovolumetric contraction-and-relaxation phase (Fig. 1). The Ees can be derived from Pmax by dividing the difference between Pmax and mean pulmonary artery pressure (mPAP) by SV.12,17 The Ea can be estimated as mPAP/SV.12,17 Brimioulle et al. 16 found an excellent correlation between Pmax predicted by the single-beat method and Pmax derived from pressure-volume loops during preload reduction in dogs. Animal studies have shown that RV-PV coupling is preserved in survivors of acute PE, 18 while it is decoupled in models of chronic pressure overload due to recurrent embolization. 19 Reduced ventriculoarterial coupling efficiency has been shown in recent clinical studies in different forms of PH.20–22 Intrinsic RV dysfunction contributing to more severe decoupling could be demonstrated in patients with scleroderma-associated pulmonary arterial hypertension (PAH), compared to patients with iPAH. 22 Recent data suggest that the RV is decoupled from the pulmonary vasculature in patients with CTEPH and in those with chronic thromboembolic pulmonary vascular disease (CTPVD). 23
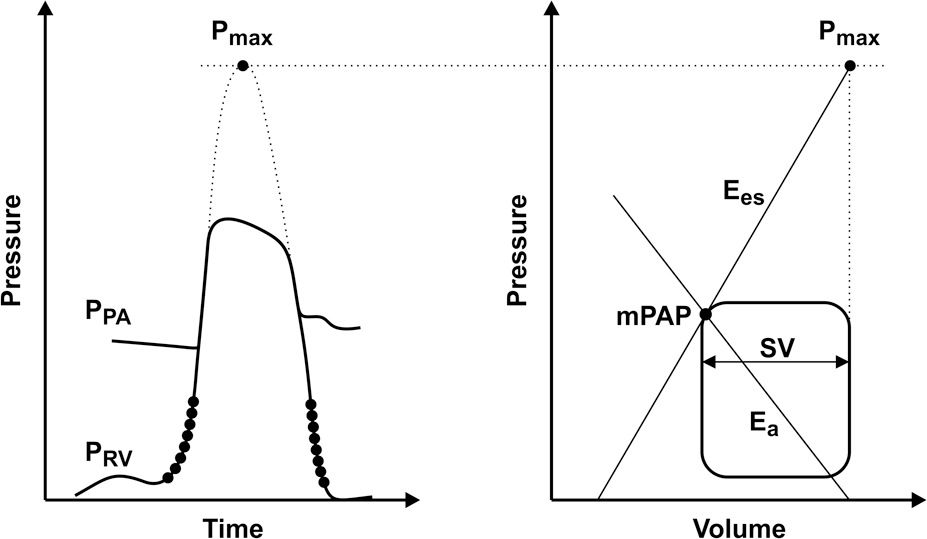
Prediction of right ventricular–to–pulmonary vascular (RV-PV) coupling by the single-beat method. Left, maximal ventricular pressure (Pmax) as encountered in an isovolumetric nonejecting beat, which is estimated by fitting a sinus wave over a right ventricular pressure (PRV) curve during the isovolumetric contraction-and-relaxation phase. PPA: pulmonary artery pressure. Right, end-systolic ventricular elastance (Ees) can be derived from Pmax by dividing the difference between Pmax and mean pulmonary artery pressure (mPAP) by the stroke volume (SV). Arterial elastance (Ea) can be estimated as mPAP/SV. Adapted from Trip et al. 17
NONINVASIVE ASSESSMENT OF RV AFTERLOAD AND FUNCTION
Assessment of RV function by imaging is challenging because of its complex anatomy. While RV ejection fraction (RVEF) is commonly used as an index of RV function, it is highly load dependent and does not reflect RV contractility. 24 This problem affects the clinical assessment and management of patients with RV dysfunction, including those with acute and chronic PE. On that account, other parameters reflecting RV function, such as RV fractional area change (RVFAC) and tricuspid annular plane systolic excursion (TAPSE), have been introduced into clinical routine. 25 More recently, several indexes have been proposed as surrogates of RV function and contractility, including the RV myocardial performance index (RVMPI, or Tei index)26–28 and the acceleration of the myocardium during isovolumetric contraction (IVA). 29 Based on Doppler imaging, RVMPI is an established marker of myocardial performance and ventricular contractility that is independent of ventricular geometry. RVMPI has been evaluated for the assessment of LV and RV function in heart failure and PH.26–28,30–33 Myocardial deformation parameters of the RV free wall, such as strain and strain rate, have been proposed as load- and heart motion–independent measures of RV function.34–36 It has been suggested that echocardiographic indexes of RV function, including TAPSE, RVMPI, RVFAC, and IVA, are related to RV-PV coupling (Ees/Ea) rather than to Ees alone. 19 RV echocardiographic indexes, especially RVFAC and IVA, have been shown to correlate more strongly with Ees/Ea than with Ees in an experimental model of RV chronic pressure overload induced by pulmonary arterial ligation and recurrent embolization. 19
RV-PV coupling can also be determined by combining standard right heart catheterization and measurements derived during magnetic resonance imaging. Studies in healthy individuals and patients with PH have shown a good agreement with conductance catheterization data.20,21
THE RV IN ACUTE PE
Pathogenesis and epidemiology
Acute PE and DVT are part of the spectrum of VTE. Thrombi commonly form in deep veins in the legs. Venous thrombi detach from their formation sites and embolize through the venous system, right atrium, and RV toward the pulmonary circulation. DVT has an incidence of 1.5 per 1,000 person-years. About 79% of patients who present with PE have evidence of DVT. One-half of the patients with proximal DVT experience an episode of PE. The direct consequence of PE is an elevation in RV afterload, followed by an increase in RV wall tension that may lead to dilatation, dysfunction, and ischemia of the RV. Death results from RV failure. RV ischemia in acute PE results from myocyte necrosis and myocardial inflammation by infiltration with macrophages, T cells, and neutrophils and is distinct from the pattern due to epicardial vessel occlusion seen in myocardial infarction.37,38
Noninvasive assessment and risk stratification of acute PE
Electrocardiographic signs of RV strain, such as T wave inversions in V1–V4, QR pattern in V1, the S1Q3T3 pattern, and incomplete or complete right bundle-branch block, are useful but insensitive for the assessment of RV dysfunction in acute PE. However, the presence of RV strain on electrocardiogram has been shown to correlate with the extent of pulmonary vascular obstruction 39 and outcomes of acute PE. 40 At least 25% of patients with acute PE have signs of RV dysfunction on echocardiography. Overall and in-hospital mortality rates have been shown to be higher in patients with echocardiographic signs of RV dysfunction at the time of diagnosis of acute PE.41–44 Patients with systolic pulmonary artery pressures higher than 50 mmHg estimated by echocardiography at the time of diagnosis have a 3-fold risk for persistent PH and RV dysfunction. 45
RV afterload in acute PE
Acute PE leads to a number of pathophysiological changes in pulmonary function. The most important among these alterations is an acute increase in PVR. Abrupt increase in PVR leading to RV failure is the principle cause of death from PE. The mPAP and PVR increase in proportion to the degree of pulmonary vascular obstruction in patients without preexisting pulmonary vascular disease.46,47 In a series of 76 patients with acute PE and without previous cardiopulmonary disease, long-term prognosis was related to the level of mPAP and the presence of RV failure. 1 Patients with mPAP higher than 30 mmHg at initial diagnosis had progressive PH. Survival after 2 years of follow-up was less than 20% when mPAP was higher than 50 mmHg. 1
In contrast, acute PE in patients with preexisting pulmonary vascular disease leads to higher pulmonary artery pressures as a result of RV hypertrophy. It has been shown that there is no correlation between the degree of pulmonary vascular obstruction and mPAP in patients with acute PE superimposed on pulmonary vascular disease. 47 The diastolic pulmonary vascular pressure gradient (DPG), i.e., the difference between diastolic pulmonary artery pressure and mean pulmonary arterial wedge pressure [mPAWP]) has been described as elevated in acute PE. 48 A positive correlation has been found between DPG and the percentage of embolic pulmonary arterial tree obstruction, as assessed by pulmonary angiography.49–51 Furthermore, DPG has been shown to be useful for the differentiation between pulmonary and cardiac causes of acute respiratory failure 52 and PH.53–55
RV function in acute PE
The extent of pulmonary vascular obstruction and the presence of preexisting cardiopulmonary disease determine the increase in RV afterload and the development of RV dysfunction. 56 The sudden rises in pulmonary artery pressure and PVR abruptly increase RV afterload, consequently leading to an increase in RV wall tension, followed by RV dilation and dysfunction. As the RV dilates, the interventricular septum shifts toward the LV. Progressive RV dilation accompanied by LV compression results in diastolic dysfunction and underfilling of the LV. LV underfilling leads to a decrease in systemic cardiac output and systemic blood pressure, potentially impairing coronary perfusion. Elevated RV wall tension itself reduces right coronary artery flow, increases RV myocardial oxygen consumption and demand, and causes ischemia. 57
THE RV IN CTPVD/CTEPH
Pathogenesis and epidemiology
Incomplete resolution of acute PE is frequently observed after acute PE but rarely results in CTEPH. Some symptomatic patients may present with normal pulmonary hemodynamics at rest, despite symptomatic disease, e.g., patients with complete unilateral obstruction. Despite the absence of PH at rest, they are treated as CTEPH patients. Suitable terminology to describe this condition of CTPVD, or chronic PE, has not been accepted. In previous studies, the cumulative incidence of CTEPH after acute PE was reported as 0.1%–9.1% after symptomatic PE.45,58–66 However, one has to take into account that the initial thromboembolic event may have been asymptomatic. For example, in the recent European CTEPH registry, 28% of patients did not have a history of acute VTE. 67 Therefore, the true incidence of CTEPH may be even higher. Pulmonary endarterectomy (PEA) is the treatment of choice for CTEPH, 68 with a periprocedural mortality rate of less than 5% in Europe today, 69 nearly normalized hemodynamics, and substantial improvement in clinical symptoms in the majority of patients.68–70
Diagnosis and definition
A diagnosis of CTEPH can be made only after effective anticoagulation of at least 3 months to discriminate the condition from subacute PE. 71 CTEPH is defined by an mPAP of at least 25 mmHg with mPAWP no higher than 15 mmHg in the presence of at least one (segmental) perfusion defect detected by lung scanning/multidetector computed tomographic angiography or pulmonary angiography.
RV afterload in CTEPH
Chronic obstructions in the pulmonary circulation lead to an increase in RV afterload and eventually to an impairment in RV function. RV afterload is characterized by a steady flow component, expressed by PVR, an oscillatory load component, expressed by CPA, and characteristic impedance (Z) of the proximal pulmonary artery. 2 Despite similar PVRs, mPAP is lower in CTEPH than in iPAH, 72 which has led to the assumption that RV adaptation may be poorer in CTEPH patients than in iPAH patients. 73 This observation may be explained by the generally older age of patients, the long duration of functional impairment, and disease-specific alterations of the pulmonary circulation. 73 RC-time is significantly lower in proximal CTEPH than in distal CTEPH and PAH, which can be explained by structural differences of the pulmonary circulation and implies a higher RV workload. 74 Compliance (CPA) has been shown to be of greater prognostic importance than resistance in patients with iPAH.5,6 The prognostic value of CPA could also be demonstrated in CTEPH patients undergoing PEA. 75 In a study of 110 consecutive patients 76 assessing RV afterload in CTEPH patients before PEA, immediately after PEA, and 1 year after PEA, 84% of the patients had an immediate improvement in CPA after PEA. Immediate postoperative CPA was identified as a univariate predictor of outcome. However, in multivariate analysis, immediate postoperative PVR was the only independent predictor of long-term survival/freedom from lung transplantation after PEA. Patients who had concurrent improvements in CPA and PVR had the lowest likelihood for adverse outcomes (death or persistent/recurrent PH) after PEA (Fig. 2). 76 According to current knowledge, PVR appears to be the most critical measure in CTEPH patients, especially when measured immediately after PEA.
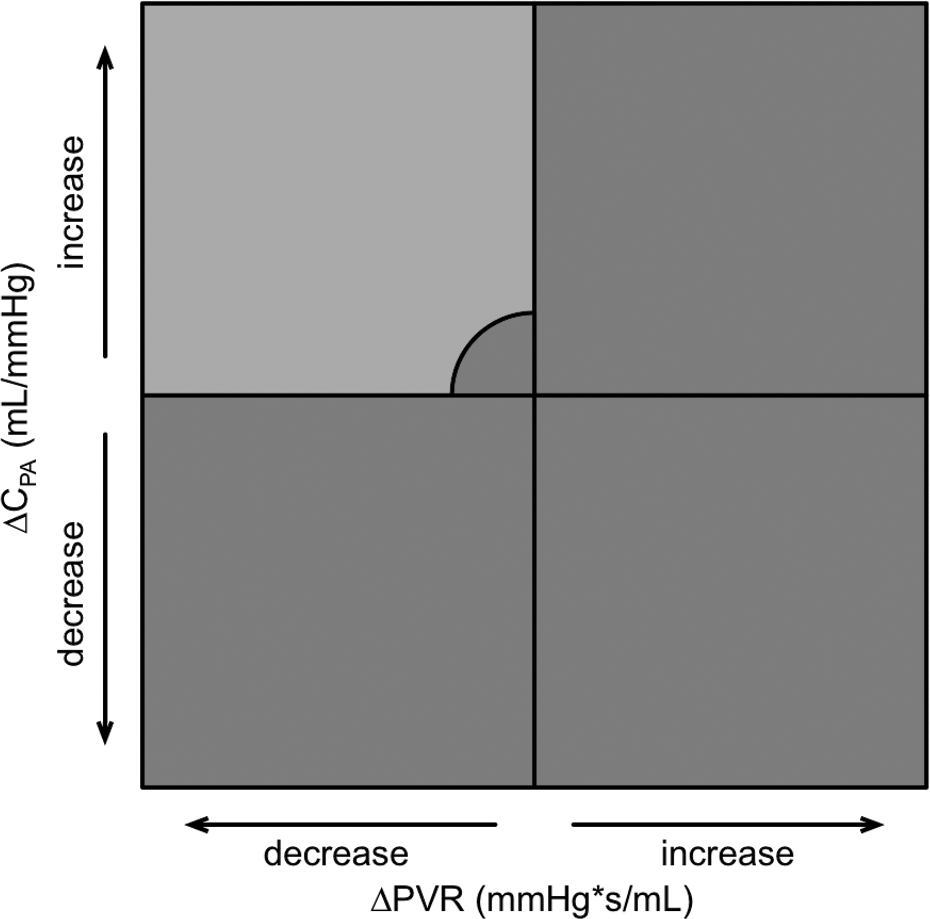
Quadrants summarizing vectors that depict changes in both pulmonary vascular resistance (PVR) and pulmonary arterial compliance (CPA) between catheterizations at baseline and immediately after pulmonary endarterectomy, to discriminate “PVR and CPA responders” from “PVR and CPA nonresponders.” All patients' vectors with persistent/recurrent pulmonary hypertension in the upper-left quadrant are localized within the circled area (representing cases with only a minor improvement in CPA and PVR). Hemodynamic changes that are associated with improved survival are all within the upper left quadrant. A color version of this figure is available online.
PEA has been shown to lead to an immediate decrease in PVR and a concordant increase in CPA, 76 resulting in a mild decrease in RC-time (baseline: 0.72 ± 0.71 seconds, immediately after PEA: 0.60 ± 0.3 seconds; P = 0.13). A significant decrease in RC-time after PEA was reported in a study from the United Kingdom (baseline: 0.49 ± 0.11 seconds, after PEA: 0.38 ± 0.11 seconds; P < 0.05). 77 It appears that patients from whom more and longer thrombus material can be removed have a better improvement in hemodynamics and are less likely to develop persistent/recurrent PH. 76 Specimen assessment takes into account both the technical success of PEA and the anatomical CTEPH type that has previously been correlated with outcomes. 78 It has been reported that patients with persistent exertional dyspnea after successful PEA display an abnormal pulmonary hemodynamic response to exercise, characterized by increased PVR and decreased CPA. 3
Upstream resistance is significantly higher in operable CTEPH than in nonoperable CTEPH and iPAH. 79 Patients with higher downstream and lower upstream resistance before PEA appear to be at increased risk for persistent PH and poor outcomes after PEA. 80 Data indicate that patients with upstream resistance of less than 60% are at highest risk for adverse outcomes after PEA. 80 It has been suggested that CTEPH patients with lower upstream and higher downstream resistance may suffer from concomitant small-vessel disease that resembles PAH, but this has not been proven by histologic evidence.
RV function in CTPVD/CTEPH
Changes in pressure-volume loop morphology have been observed in CTEPH patients as well as in patients with CTPVD. 23 Compared to healthy controls, patients with CTEPH and CTPVD display a positive pressure differential during systolic ejection from pulmonary valve opening to valve closure (−6.3 ± 3.0, 3.2 ± 3.5, and 20.4 ± 18.4 mmHg for controls, CTPVD patients, and CTEPH patients, respectively). 23 The RV has been shown to be decoupled in patients with CTEPH, with an Ees/Ea ratio of 0.6 ± 0.18, compared to controls (Ees/Ea = 1.46 ± 0.3). RV-PV decoupling could be observed in patients with CTPVD (Ees/Ea = 1.27 ± 0.36), suggesting RV dysfunction despite normal hemodynamics. In addition, slower RV relaxation, as indicated by the time constant of isovolumetric relaxation τ, has been found in CTEPH and CTPVD patients, compared to controls. 23
RV remodeling in CTEPH and reverse remodeling after PEA
As a consequence of chronic increase in afterload, the RV undergoes a remodeling process in order to maintain pulmonary blood flow. This process is characterized by an increase in wall thickness and chamber dilatation. According to the Laplace relationship, the thin-walled RV augments thickness in order to cope with a chronic increase in intraluminal pressure and wall stress. 81 This is mainly achieved by hypertrophy. With dilatation, the RV loses its triangular shape, leftward ventricular septal bowing develops, and tricuspid annular dilation results in tricuspid regurgitation. Leftward ventricular septal bowing with LV compression, in combination with low LV preload and underfilling (“LV unloading”), has been shown to cause LV diastolic filling impairment in a significant proportion of patients with CTEPH. 82 Chronic LV unloading leads to atrophic remodeling, with LV diastolic and systolic dysfunction. 83 A recent study has shown that RV failure in CTEPH patients is associated with a reduction in LV free-wall mass, which is reversible after PEA. 83 In addition, the authors showed, in an animal model, that this observation might be explained by myocyte shrinkage due to atrophic remodeling. 83 PEA has been shown to restore RV remodeling by an acute reduction in RV afterload,84,85 with subsequent improvement in biventricular cardiac function and reduction in ventricular septal abnormalities and RV systolic wall stress.86,87 Magnetic resonance imaging studies have shown that reverse remodeling occurs early after PEA. 86 Echocardiographic studies demonstrated improvements in RV volumes and areas, 88 tricuspid regurgitation,89,90 TAPSE, 85 RV strain, 35 and RVMPI,30,85 indicating better RV function after PEA. Reductions in RV mass and septal bowing, accompanied by an increase in RVEF, have been documented by magnetic resonance imaging studies.84,91 Moreover, the magnitude of reverse RV remodeling after PEA has been shown to correlate with changes in hemodynamics.84,85 However, despite significant improvement in RV function after PEA, RVEF remained reduced compared to that in healthy individuals.84,85 The reason why indexes of RV function, including RVEF, RVMPI, and TAPSE, fail to completely recover after PEA is unknown. It has been suggested that RV remodeling is only partly reversible because of diffuse myocardial fibrosis, similar to PAH. 12
OUTLOOK
Mortality in acute PE and CTEPH depends on RV function. One may assume that, in contrast to that in iPAH and scleroderma-associated PAH, the RV in VTE is intrinsically normal, making VTE-dependent RV function an ideal model to study consequences of abrupt (acute PE) and gradual (CTEPH) afterload increase, because RV functional changes are correlated with the degree of mechanical obstruction of the pulmonary circulation. RV functional recovery after PEA in CTEPH is a further attractive human model to study reverse RV remodeling that may provide insights in RV adaptation to increased afterload.
