Abstract
Little is known about the impact of metabolic syndrome (MS) on right ventricular (RV) structure and function. We hypothesized that mice fed a Western diet (WD) would develop RV lipid accumulation and impaired RV function, which would be ameliorated with metformin. Male C57/Bl6 mice were fed a WD or standard rodent diet (SD) for eight weeks. A subset of mice underwent pulmonary artery banding (PAB). Treated mice were given 2.5 g/kg metformin mixed in food. Invasive hemodynamics, histology, Western, and quantitative polymerase chain reaction (qPCR) were performed using standard techniques. Lipid content was detected by Oil Red O staining. Mice fed a WD developed insulin resistance, RV hypertrophy, and higher RV systolic pressure compared with SD controls. Myocardial lipid accumulation was greater in the WD group and disproportionately affected the RV. These structural changes were associated with impaired RV diastolic function in WD mice. PAB-WD mice had greater RV hypertrophy, increased lipid deposition, and lower RV ejection fraction compared with PAB SD controls. Compared to untreated mice, metformin lowered HOMA-IR and prevented weight gain in mice fed a WD. Metformin reduced RV systolic pressure, prevented RV hypertrophy, and reduced RV lipid accumulation in both unstressed stressed conditions. RV diastolic function improved in WD mice treated with metformin. WD in mice leads to an elevation in pulmonary pressure, RV diastolic dysfunction, and disproportionate RV steatosis, which are exacerbated by PAB. Metformin prevents the deleterious effects of WD on RV function and myocardial steatosis in this model of the metabolic syndrome.
Metabolic syndrome (MS) is associated with left ventricular (LV) remodeling, diastolic dysfunction, and an increased incidence of heart failure.1,2 The exact mechanisms underlying these associations are unknown, but recent studies suggest that myocardial lipid accumulation may contribute. 3 Increasing body mass index and worsening insulin resistance are both positively correlated with myocardial triglyceride content. 4 Importantly, these relationships exist even in subjects without overt LV dysfunction or prevalent heart failure.
An association between Western Diet (WD) and the development of pulmonary vascular disease has been described previously by our group and others, but the effects of WD on right ventricular (RV) structure and function have not been reported.5,6 MS is highly prevalent in patients with pulmonary hypertension (PH) due to left heart disease and insulin resistance is found in nearly 60% of patients with pulmonary arterial hypertension (PAH).7,8 Moreover, subjects with MS were recently shown to have worse pulmonary hemodynamics and RV diastolic dysfunction compared with obese and non-obese control subjects without MS. 9 Given the observational nature of these studies, a cause and effect relationship between MS and RV/pulmonary vascular dysfunction cannot be inferred. The effect of myocardial lipid accumulation on RV structure and function in the context of MS and whether it is a modifiable therapeutic target have not been studied.
We hypothesized that mice fed a WD (60% fat), a model of insulin resistance and MS, 10 would develop RV lipid accumulation and impaired RV function. We further hypothesized that these abnormalities would be exacerbated by RV load stress and ameliorated with treatment of insulin resistance. This study provides initial evidence that obesity and insulin resistance have a direct and potentially disproportionate adverse effect on RV structure and function that is associated with lipotoxic cardiac steatosis. RV steatosis can be prevented with metformin, resulting in normalization of RV function and pulmonary hemodynamics.
Methods
All animal procedures were approved by the Institutional Animal Care and Use Committee of Vanderbilt University School of Medicine.
Mice
Male C57/Bl6 mice were fed a WD consisting of 60% lard or nutrient-matched standard rodent diet (SD) for eight weeks (n = 16) beginning at three weeks of age and weighed weekly. Treated mice were given 2.5 g/kg metformin mixed in food (BioServ, San Diego, CA, USA) beginning at three weeks of age and continuing until sacrifice at 11 weeks of age. Fasting glucose and insulin and weight were measured biweekly from week 7. We did not sample blood before sacrifice so as not to alter hemodynamics through volume depletion. Glucose and insulin measurements were performed at the Vanderbilt University Mouse Metabolic Phenotyping Center (Nashville, TN, USA). The Homeostatic Model of Insulin Resistance (HOMA-IR) was calculated as (insulin × glucose)/405.
11
The experimental design is illustrated in Suppl. Fig. 1.
Western diet (WD) is associated with increased right ventricular systolic pressure (RVSP) and negatively impacts RV structure and function. (a) RVSP was increased in WD-fed mice compared to standard diet (SD) at baseline (P = 0.005). (b) Similarly, there was increased RV hypertrophy with WD compared with SD at baseline (P = 0.02), while the effects were less pronounced with PAB though the trend was similar (P = 0.01). (c) Right ventricular ejection fraction (RVEF) was increased in WD compared to SD (P = 0.03). While PAB led to an increase in RVEF, this was reduced by PAB in the WD group (P = 0.02). (d) Invasively measured hemodynamics demonstrated prolonged relaxation time, Tau, in both HFD at baseline (P = 0.047) and with PAB (P = 0.04). *P < 0.05 vs. SD, #P < 0.05 WD-PAB vs. SD-PAB.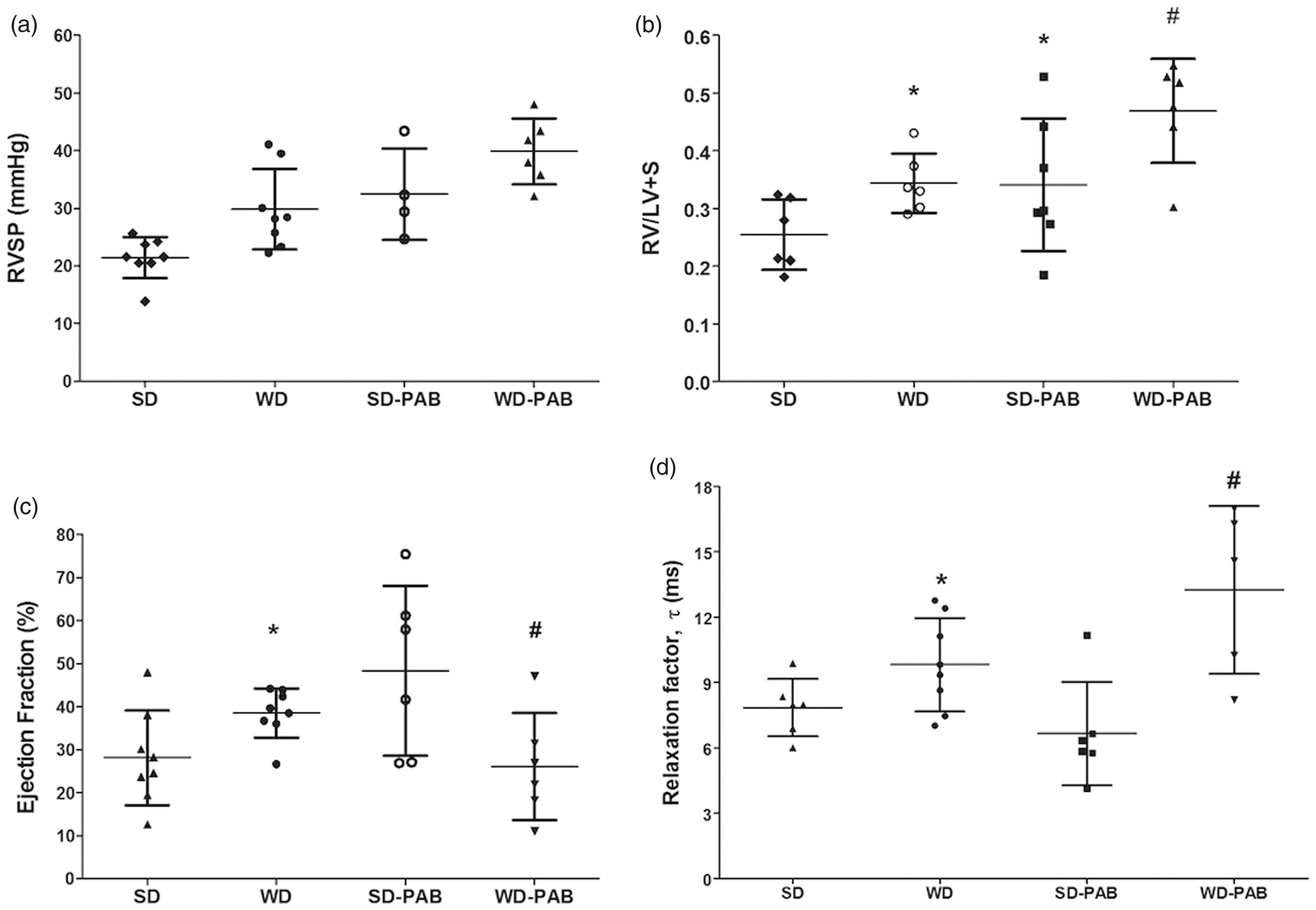
Pulmonary artery banding
A subset of mice (n = 6 in each dietary condition, WD or SD) underwent pulmonary artery banding (PAB) as previously described 12 at 11 weeks of age and remained in place for two weeks, after which animals underwent invasive hemodynamic measurement and were sacrificed, with tissue harvested and preserved in formalin or snap frozen. Fasting glucose, insulin, and weight were measured biweekly through week 12.
In vivo hemodynamics and cardiac function
At the time of sacrifice, invasive hemodynamics were measured through an open-chested technique for measurement of pressure–volume loops in animals after PAB. 12 Hemodynamics were continuously recorded with a Millar MPVS-300 unit coupled to a Powerlab 8-SP analog-to-digital converter acquired at 1000 Hz and captured to a Macintosh G4 (Millar Instruments, Houston, TX, USA). RV hypertrophy was measured by the ratio of RV weight to LV plus septum weight (Fulton index).
Echocardiography
Mice were anesthetized with 3% isoflurane supplied with room air. Depilatory cream was applied to the chest to remove hair before image acquisition. Mice were then placed on a table heated to physiologic temp in a supine position. Isoflurane was administered for the duration of the procedure. Echocardiographic studies were carried out using the Visualsonics Vevo770 platform and with the 707B probe head. Left ventricular outflow tract diameter measurements were traced from frozen B mode frames and velocity time integral measurements of the aorta were recorded from pulsed wave mode. Area was calculated from aortic diameter measurements; stroke volume was calculated, then multiplied times heart rate to obtain cardiac output. Three measurements of each parameter were obtained and averaged for calculations.
Histology
RVs were harvested at the time of sacrifice and snap frozen or placed in 10% formalin. For lipid analysis, RV and LV tissue were embedded in Optimal Cutting Temperature (OCT) medium and flash-frozen in liquid nitrogen. Tissues were stained with Oil Red O as previously described and images acquired by a brightfield slide scanner. 12 Myocardial lipid content was quantified by first setting a threshold red, green, and blue pixel value for lipid stain. Additional thresholds were then set to exclude nuclei and areas stained with eosin. Aperio software as then used to scan each slide pixel-by-pixel and lipid content was expressed as a percent of each slide stained above the RGB lipid value. Paraffin embedded slides were stained with hematoxylin and eosin (H&E) for myocyte counting and analysis via Aperio software. Five sample areas equally distributed over the RV cross-section were chosen for each mouse. Within each of the five sections, ten myocytes were randomly chosen for measurements. Diameter measured at the narrowest width for each myocyte.
Western blot
Mouse RVs were homogenized in RIPA buffer (PBS, 1% Ipegal, 0.5% sodium deoxycholate, 0.1% SDS) with proteinase and phosphatase inhibitor cocktails (Sigma-Aldrich, St. Louis, MO, USA). The protein concentration in the samples was determined by using Bradford assay (Pierce Co., Rockford, IL, USA) and was stored at –70°C until use. Primary antibodies used for Western blot included CD36 (3313, Cell Signaling Technologies [CST], Danvers, MA, USA), β-microglobulin (2101, CST), HSP70 (ab8227, Abcam, Cambridge, MA, USA), and β-actin (ab8227, Abcam).
Polymerase chain reaction (PCR)
RNA was obtained using Qiagen RNeasy Mini Kit (Qiagen, Valencia, CA, USA). First strand complementary DNA (cDNA) was made using QuantiTect® Reverse Transcription Kit (Qiagen) from 1 µg total RNA. Quantitative real-time PCR (qPCR) was performed using a total reaction volume of 25 µL, containing 5 µL of diluted cDNA, 12.5 µL SYBR Green Supermix (Applied Biosystems Foster City, CA, USA), and 0.03 µL of each oligonucleotide primer (250 mM). PCR was carried out in a StepOnePlus Real Time PCR System (Applied Biosystems) using 40 cycles of 950 C for 15 s followed by 600 C for 1 min with a 10-min 950 C initial soak. Each measurement was made in triplicate and expressed relative to the detection of the standard HPRT. PCR was performed for the primer sets HPRT and BNP (Integrated DNA Technologies IDTH Coralville, IA, USA).
Data analysis
Statistical tests were either one-way or two-way analysis of variance (ANOVA) performed using the SPSS version 22 or GraphPad Prism Plus version 5.0 Software (San Diego, CA, USA). Non-normally distributed data were log-transformed. Data are presented as mean ± SD unless otherwise noted.
Results
Effects of Western diet on right ventricular structure and function
Mice fed a WD were significantly heavier and more insulin resistant compared to low-fat controls (HOMA-IR 3.0 ± 0.6 vs. 2.4 ± 0.4, P < 0.001, Suppl. Fig. 2). In unstressed conditions, a WD led to a significant increase in RV systolic pressure (29.8 ± 7.0 vs. 21.4 ± 3.6 mmHg, P < 0.05, Fig. 1a) measured at eight weeks with no difference in cardiac output on echocardiography between groups (12.2 ± 3.4 mL/min vs. 12.2 ± 1.3 mL/min, P = 0.80). WD was associated with an increase RV hypertrophy as measured by the Fulton index (RV/LV + S; 0.34 ± 0.05 vs. 0.25 ± 0.06, P < 0.05) compared with SD mice (Fig. 1b). WD had disparate effects on RV systolic and diastolic function, demonstrating an increase in right ventricular ejection fraction (RVEF; measured via pressure volume loops) but a worsening of diastolic function as measured by the relaxation factor, tau (Fig. 1c and d).
WD is associated with disproportionate RV steatosis. (a) Oil Red O stain for lipid in the RV demonstrates increased lipid droplets (red) in standard image (left) and digitally enhanced (right) in WD-fed mice. (b) Percent lipid stain in (a) is quantified and shown to be increased in the WD RV compared to the SD RV (P < 0.01). WD did not significantly augment LV lipid content. (c) Oil Red O images from RV and LV demonstrating no increase in lipid content in the WD LV. *P < 0.05 WD RV vs. WD RV.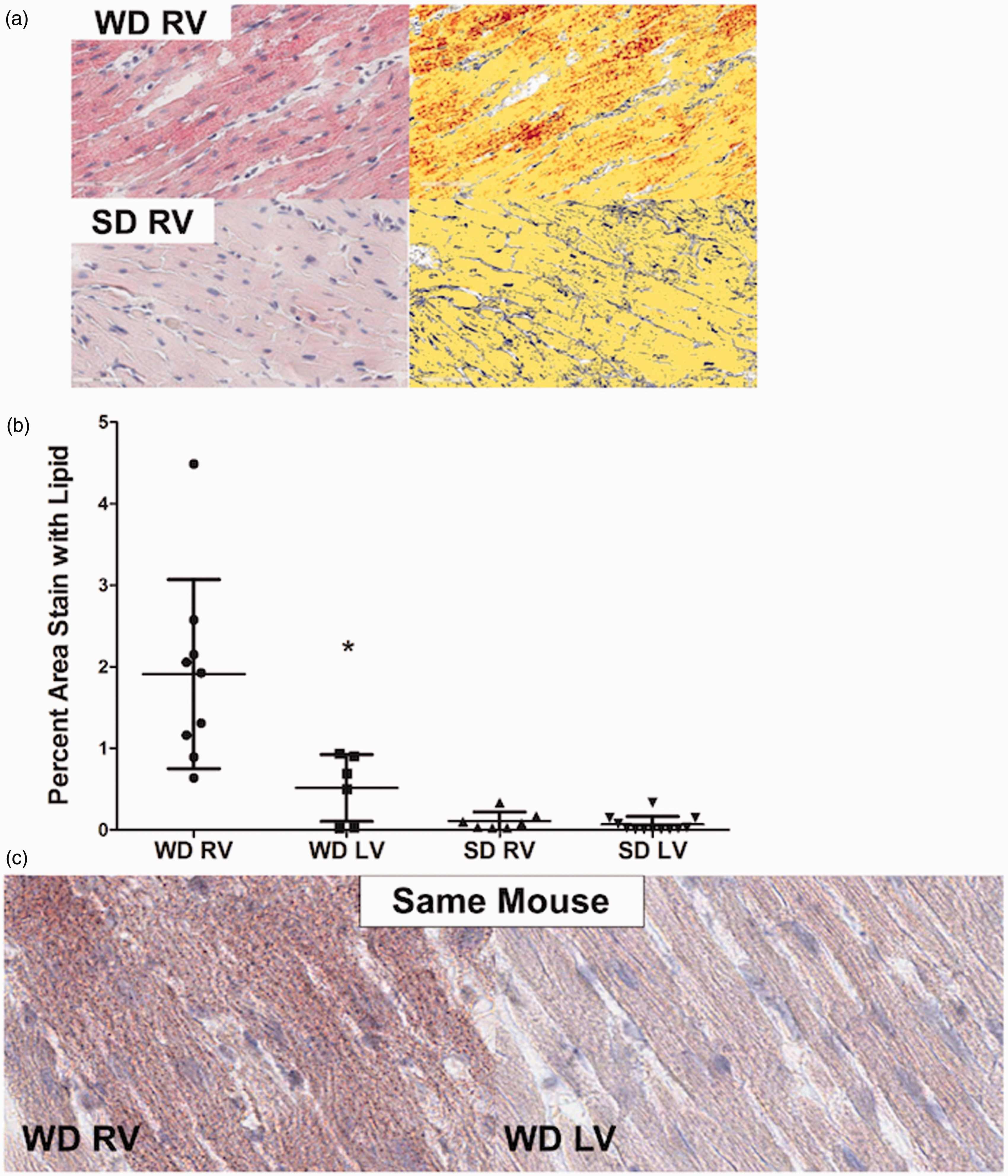
We next tested the hypothesis that RV load stress responses would be impaired in WD-fed mice compared with SD. As expected, all PAB mice had higher right ventricular systolic pressure (RVSP) compared with non-banded mice, likely due largely to mechanism obstruction, but a pulmonary vascular contribution cannot be excluded as these mice were also exposed to WD for eight weeks before banding (Fig. 1a and b). There was no statistically significant difference in RVSP between WD-PAB mice and SD-PAB mice, though there was a non-significant trend to higher pressures in the WD mice (P = 0.13). PAB was associated with increased RV hypertrophy in both the SD and WD groups and was most pronounced in the WD-PAB group. Interestingly, RVEF increased significantly in the SD-PAB group but declined in the setting of WD and PAB. As seen in the unstressed WD group, WD-PAB was associated with a prolongation of the relaxation factor Tau, reflecting worse diastolic function (Fig. 1c and d). These data suggest that WD contributes to both systolic and diastolic RV dysfunction in the setting of elevated afterload.
Western diet causes disproportionate right ventricular lipid deposition
On Oil Red O staining, we found markedly elevated intra-myocyte lipid deposition in the RV in the WD group, which was approximately 15-fold higher than the SD group when quantified using a pixel color detection algorithm (Fig. 2a and b). In comparing RV and LV lipid accumulation, we found that myocardial lipid deposition in the setting of WD was most pronounced within the RV, with a trivial, statistically insignificant increase in the LV (Fig. 2b, P < 0.01). Figure 2c demonstrates in the same mouse the marked difference in steatosis in the RV and LV in the setting of WD. The additional stress of PAB did not further increase lipid content in the RV in WD-fed mice.
Effect of metformin on right ventricular structure and function
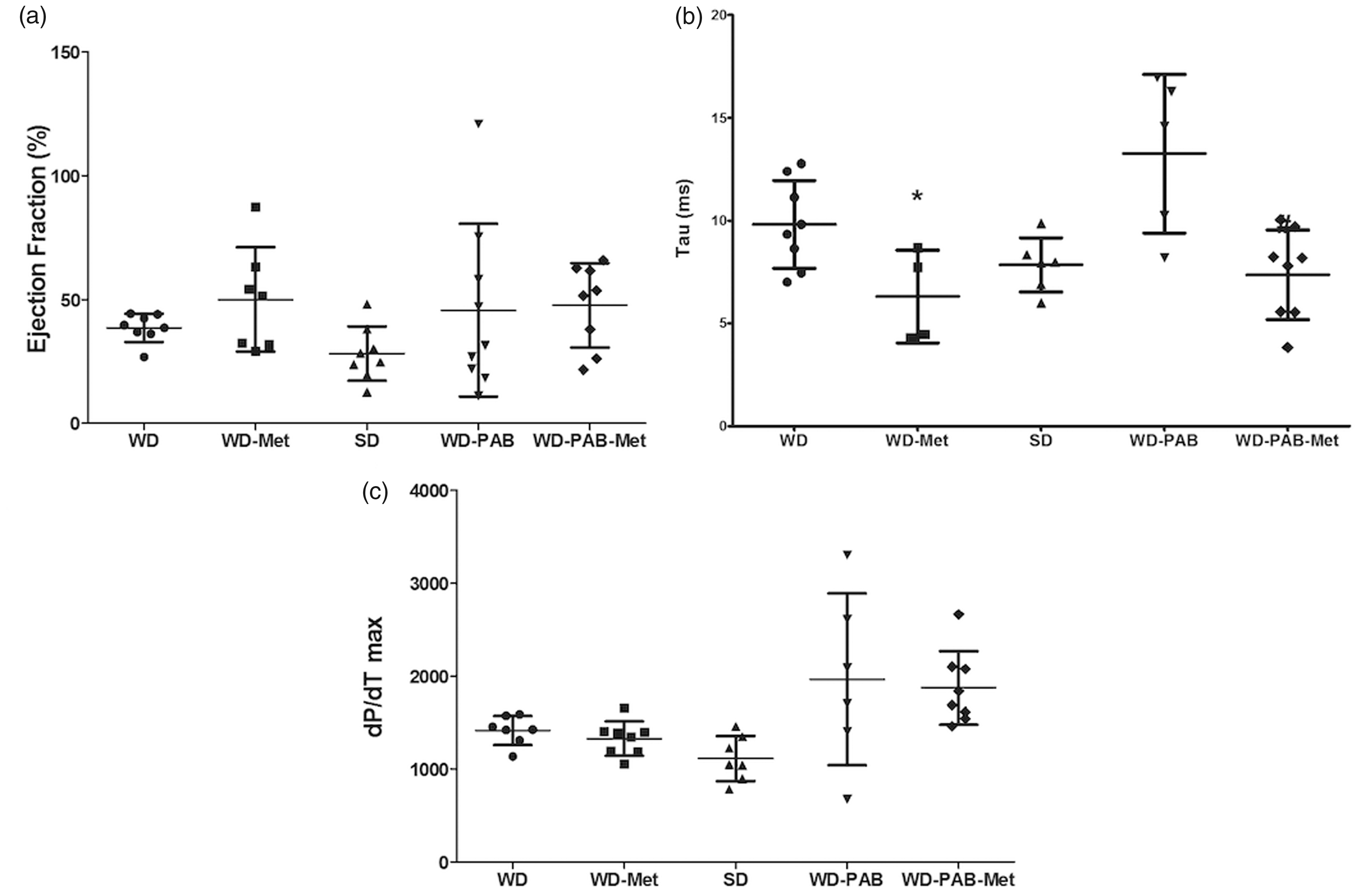
We next sought to test the hypothesis that an intervention to improve insulin resistance would prevent lipid deposition and improve RV function. Mice were fed metformin with SD or WD for eight weeks (non-banded groups) or 10 weeks (with PAB for last two weeks). The addition of metformin resulted in reduced weight gain and prevented elevation in HOMA-IR at seven weeks (Fig. 3a and b). Metformin was also associated with normalization of RVSP in unstressed mice, but no difference was observed in RVSP between WD-PAB mice with or without metformin (Fig. 3c). Metformin prevented RV hypertrophy in the setting of WD in the unstressed RV, but also was associated with reduction in RV/LV + S ratio in the context of load stress (Fig. 3d). There was a small, but significant, increase in myocyte diameter in WD-fed mice both at baseline and in PAB that was attenuated by metformin administration (Suppl. Fig. 3). In addition to these structural changes, metformin resulted in improved RV diastolic function, evident by a shortened Tau to levels similar to SD (Fig. 4b), but no change in RV systolic function (Fig. 4a and c). RV diastolic function also improved dramatically in the WD-PAB group with metformin, with no observed effect on RVEF. These data demonstrate that metformin-mediated improvement in WD-induced insulin resistance is associated with improved metrics of RV hypertrophy and diastolic function in both the unstressed and load-stressed right ventricle.
Physiologic and hemodynamic effects of metformin in the context of A WD. (a) Metformin administration led to a decrease in total body weight (P = 0.005) to a level that was not different from SD-fed mice. (b) There was a similar decrease in HOMA-IR, a measure of insulin resistance (P < 0.001 WD vs. WD-metformin [WD-Met]) to levels that were similar to SD. (c) RVSP was decreased in the WD-Met group compared with the WD alone group. (d) Co-administration of metformin with WD attenuated the WD-associated changes in right ventricular hypertrophy both at baseline (P = 0.001) and with PAB (P = 0.008). *P < 0.05 WD-Met vs. WD, #P < 0.05 WD-PAB Met vs. WD-PAB. Hemodynamic effects of metformin therapy. (a) Conductance catheter measurements demonstrated numerically increased RVEF in WD-Met compared to WD alone; however, no changes met statistical significance (P = 0.16). (b) Metformin therapy lead to improved diastolic filling time (tau) both at baseline (P = 0.025) and with load stress (P = 0.005). *P < 0.05 WD-Met vs. WD, #P < 0.05 WD-PAB Met vs. WD-PAB. (c) RV contractility, as assessed by dP/dTmax, was increased in PAB vs. SD; however, there was no effect of metformin. *P < 0.05 PAB vs. SD.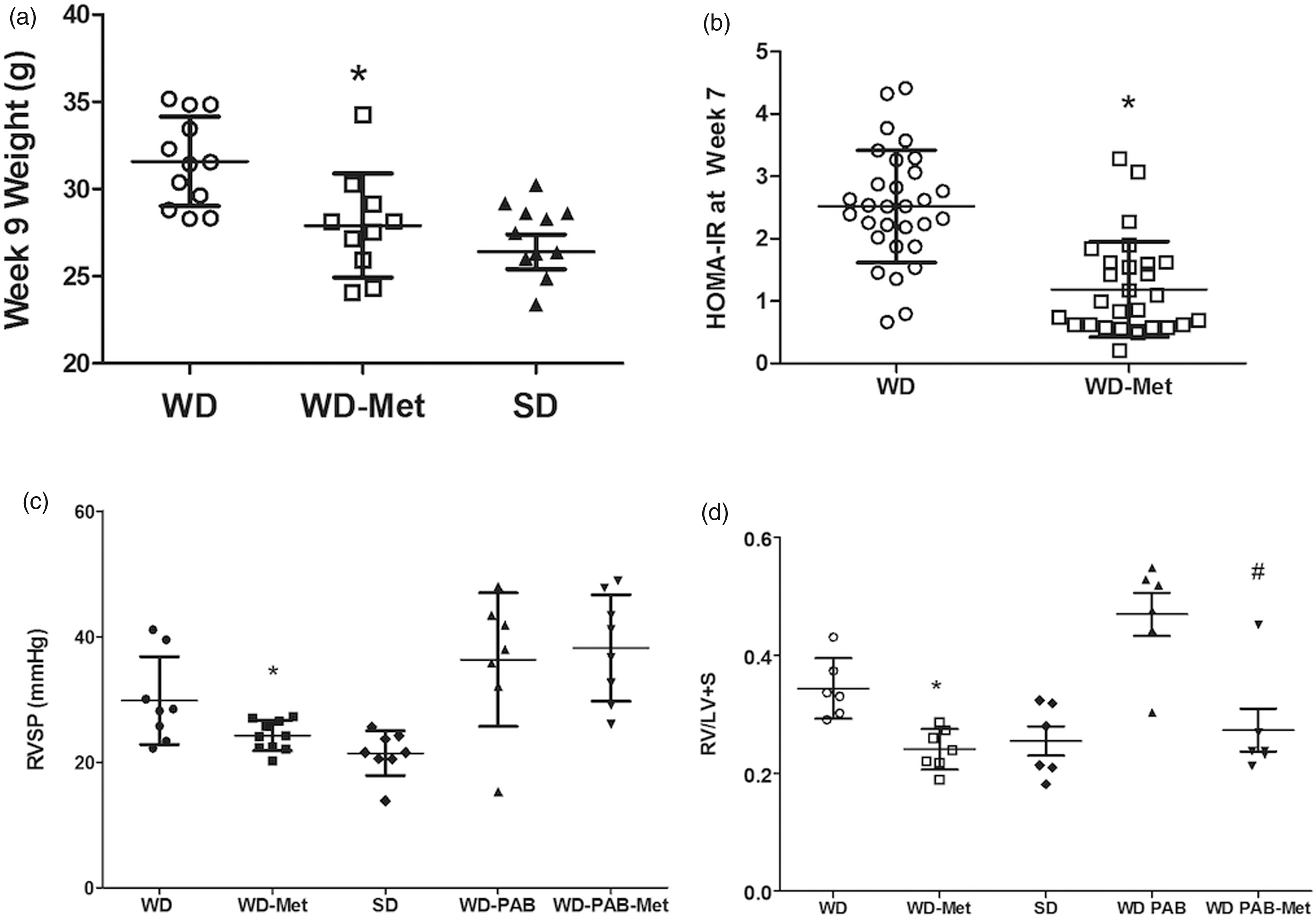
Histopathological and molecular effects of metformin on right ventricular lipid deposition
We next examined whether the positive effects of metformin on RV structure and function were associated with changes in RV lipid content. In both unbanded and PAB mice, metformin reduced RV lipid deposition in the WD mice to levels that were indistinguishable from the SD group demonstrated both visually and quantitatively (Fig. 5a and b). We have previously demonstrated that both humans with PAH and a corresponding rodent model demonstrate features of lipotoxicity with ceramide accumulation.13,14 We tested the hypothesis that WD would be associated with elevated levels of ceramide and that this increase would be further exacerbated by load stress through PAB. WD alone was not associated with higher levels of ceramide compared with SD. However, PAB in the context of WD was associated with elevated levels of ceramide staining (P < 0.05, Fig. 5d) that was attenuated by metformin therapy (P < 0.05), suggesting that WD may promote lipotoxicity in the stressed RV that can be prevented by metformin therapy.
Metformin prevents lipid and ceramide accumulation in the WD RV. (a) Measurement of lipid content in the RV using Oil Red O stain on the left with digitally enhanced image on the right demonstrates reduction of RV lipid content that is quantified in (b). WD-Met is associated with normalization in RV lipid staining in both baseline conditions and load stress, PAB. (c) Immunohistochemistry for ceramide, quantified in (d), demonstrates increased ceramide content in PAB compared to unstressed conditions (P = 0.03) that is attenuated with metformin therapy WD-PAB compared to WD-PAB (P = 0.01). (e) RV CD36 expression was decreased by metformin therapy in unstressed conditions (P = 0.008). In PAB, there was a trend to decreased expression of CD36 with metformin exposure, though it did not meet statistical significance. *P < 0.05 WD-PAB vs. WD, #WD-PAB Met vs. WD-PAB.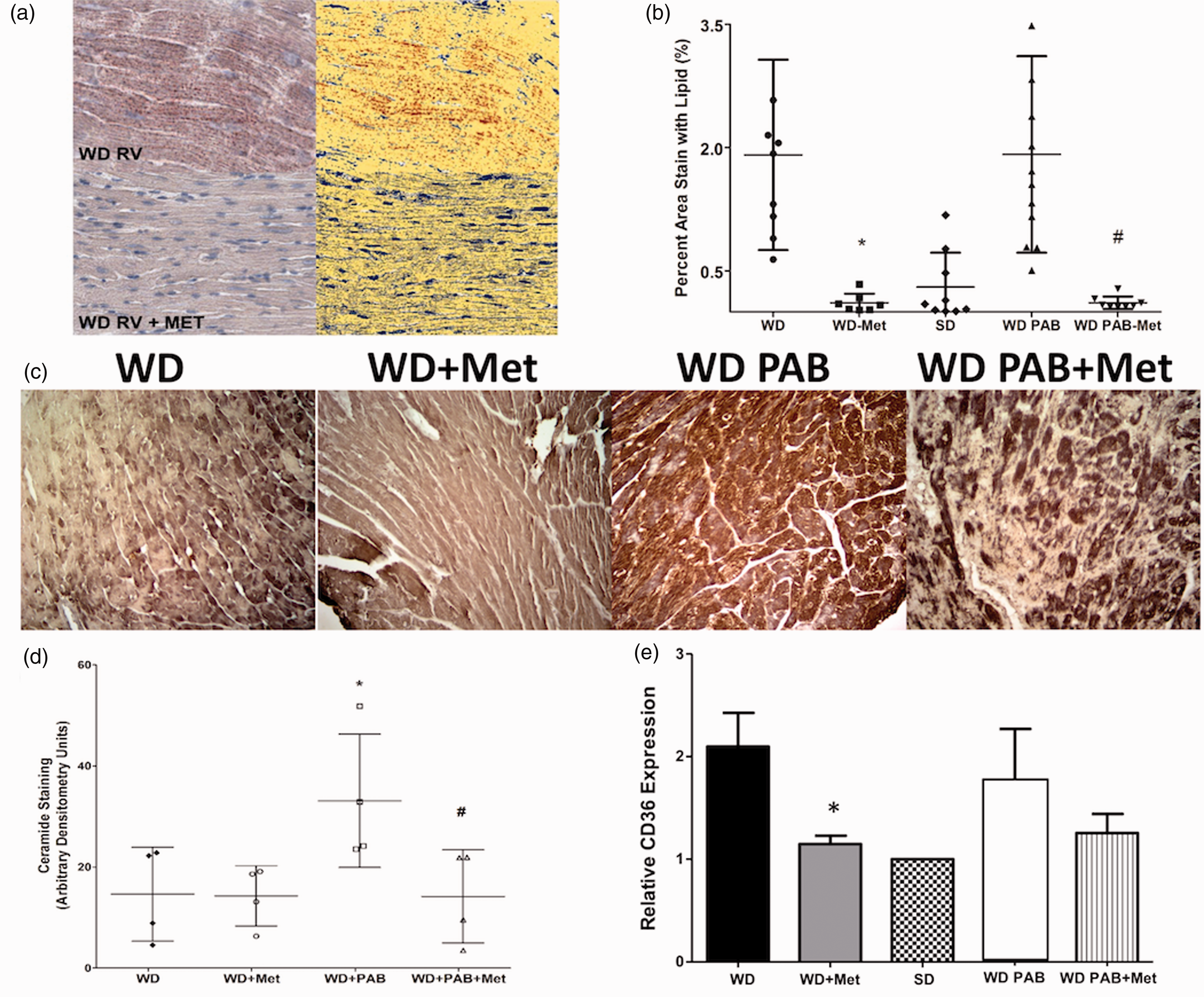
We performed preliminary experiments to determine the mechanism of reduced myocardial lipid in the setting of metformin treatment and focused on our prior work demonstrating enhanced expression of the lipid transporter CD36 in the context of RV failure. 13 RV CD36 expression by Western more than doubled in the WD group and was normalized in WD-fed mice treated with metformin (Fig. 5e), suggesting that, at least in part, metformin may attenuate CD36 mediated lipid transport to reduce RV lipotoxicity.
Discussion
This study sought to determine the effects of a fat-enriched WD on RV structure and function in mice and whether metabolic therapy would modify those effects. Our primary finding was that a WD leads to RV dysfunction and lipid accumulation. Our data suggest that RV steatosis was driven by increased transport of fatty acids into the myocardium by the CD36 transporter and RV dysfunction association with lipotoxic ceramide. Metformin prevented RV remodeling and steatosis in both the unstressed condition and in a model of elevated RV afterload with PAB. Moreover, WD alone led to a significant increase in RVSP, which normalized with metformin. The impact of obesity and insulin resistance on the RV has not been well described in humans or experimental models. This study suggests that the potential detrimental effects of metabolic syndrome on RV function may in part be mediated by toxic myocardial lipid deposition. The importance of this finding is that RV lipid accumulation was highly modifiable in our study with a safe, commonly used diabetes drug, which may have implications for human disease.
This study provides additional evidence from us and others that insulin resistance has direct effects on the RV.9,10,14 Zamanian et al. and Pugh et al. made the clinical observation that insulin resistance is highly prevalent and out of proportion to body weight in patients with PAH, a primary disease of the pulmonary vasculature.7,8 We have also shown that diabetes is associated with worse RV compensation and survival among patients with PAH. 15 These clinical findings are further supported by evidence that insulin resistance contributes to pulmonary vascular disease in experimental models of reduced PPAR-gamma and BMPR2 activity. 16 Further, Kelley et al. previously demonstrated that exposure to WD is associated with pro-inflammatory pulmonary vascular remodeling in wild-type C57BL/6 mice. 5 Our group recently showed that the pulmonary vascular response to WD is driven by hyperinsulinemia rather than hyperglycemia. 6 Although the focus of the present work was to assess the effects of WD on RV structure and function, we observed a 39% increase in invasively measured RVSP in wild-type mouse without any other PH-promoting intervention. Moreover, this WD-induced increase in RVSP was reduced with metformin therapy. These data support the hypothesis that insulin resistance may promote pulmonary vascular disease, as suggested by our studies in other rodent models.
It is unknown whether therapy for insulin resistance reverses pulmonary vascular disease in humans. We reported a single case of a patient with PAH undergoing Roux-en-Y gastric bypass who had marked improvement in pulmonary hemodynamics and RV function postoperatively that were concurrent with improved insulin sensitivity and preceded any significant weight loss. 17 The DPPV-4 inhibitor sitagliptin was recently shown to reduce LV lipid content and improve LV function. 18 Given evidence that the septum shares a common embryologic origin with the RV, 19 this raises the question of whether the investigators are describing an effect on the RV. 20
We found that WD led to greater effects on RV diastolic function than systolic function. This finding is consistent with clinical studies in humans showing that obesity and insulin resistance correlate more strongly with diastolic dysfunction than systolic function. Indeed, myocardial lipid content correlates with diastolic dysfunction in obese humans even in subjects with normal LV size and function, assessed by myocardial strain. 3 It is possible that decline in systolic function occurs later than eight weeks of exposure to WD or requires additional stress, as suggested by our data showing a decline in RVEF at 11 weeks in PAB mice. Our findings suggest that RVH measured by the Fulton index (a weight measurement) is driven by accumulation of myocardial lipid, not a change in myocyte structure. Myocyte diameter was increased in both PAB groups, as expected, therefore increased steatosis in the WD-PAB group may have accounted for the trend (P = 0.10) toward increased Fulton index compared with the SD-PAB group. The reduction in Fulton index in response to metformin was dramatic in both the unstressed WD and WD-PAB groups, supporting the notion that lipid accumulation was driving the differences with the SD groups.
We performed preliminary experiments to begin to understand our physiologic findings. Administration of WD in our study led to increased expression of RV CD36, which is the predominant fatty acid transporter in cardiomyocytes. It is unclear if WD had a direct effect on CD36 expression or if the receptor was upregulated in response to increased circulating fatty acid substrate. However, we acknowledge that metformin has pleiotropic effects on metabolism including enhanced fatty acid oxidation; the experiments here cannot elucidate the specific mechanisms through which metformin may be beneficial. We observed an increase in the lipotoxic metabolite ceramide in mice exposed to WD in both banded and unstressed conditions, but the banded mice had particularly high levels suggesting there may be a second driver of ceramide accumulation in addition to WD. One speculation is that the increase afterload from banding also contributed to ceramide production. We cannot exclude the possibility that LV diastolic dysfunction contributed to increased RVSP and RV dysfunction rather than a direct effect of WD on the RV. We were unable to accurately measure LV filling pressure due to blood loss associated with directly measuring RV pressure. The echocardiographic measures of cardiac output we present suggest no significant difference in LV systolic dysfunction across groups. Moreover, RV lipid accumulation and the reduction in lipid content in response to metformin were similar in both the banded and unbanded groups suggesting that increased afterload is not the primary driver of lipotoxicity in this model.
An unexpected finding of our study was a markedly differential effect of WD on lipid accumulation in the RV and LV. The RV has a unique embryologic origin and transcriptional patterns compared with the other the cardiac chambers and normally operates under low pressure and low resistance. 20 It is possible that the RV may be less able to compensate in the setting of metabolic and hemodynamic stress from WD. Our findings are particularly interesting in light of recent evidence indicating that the metabolic syndrome is associated with subclinical RV dysfunction in humans.6,15,16 Further studies in humans are needed to determine whether these effects are mediated by increased lipid accumulation. We cannot exclude the possibility that excess RV lipid accumulation was driven by a disproportionate increase in RV afterload versus LV afterload. We did not measure systemic blood pressure in this experiment, so we are unable to index lipid accumulation relative to increases in RV and LV afterload. However, we did not observe increased RV lipid content with a combination of WD and PAB (Fig. 5b) in this study or our prior work showing PAB is not sufficient to increase RV lipid. 12 Although not conclusive, these findings suggest that pressure elevation alone is not sufficient to drive RV lipid accumulation.
Limitations
The open chest method for RV pressure measurements does not allow for concurrent measurement of LV hemodynamics due to rapid bleeding on withdrawal of the pressure catheter. Therefore, we were unable to examine the effect of metformin on LV function invasively. The measures of RVEF inherently contain systematic error due to the assumptions of ventricle shape by the conductance catheter software, though this error applies to all groups in the same way. We did not test the effect of other metabolic therapies (e.g. DPPIV inhibitors or thiazolidinediones) on RV function and steatosis so it is unclear whether the effects observed our study are specific to metformin or simply reflect an improvement in insulin resistance. Our study sought to understand the physiologic effects of WD on the RV and we acknowledge that our molecular studies are preliminary at this time.
Conclusions
A WD in mice leads to an elevation in pulmonary pressure, RV diastolic dysfunction, and disproportionate RV steatosis, which are exacerbated by pulmonary artery banding including augmentation of lipotoxic ceramide. Metformin prevents the deleterious effects of WD on RV function and myocardial steatosis in this model of the metabolic syndrome. Further studies are needed to determine whether myocardial steatosis contributes to RV dysfunction in humans with metabolic syndrome.
Conflict of interest
The author(s) declare that there is no conflict of interest.
Funding
This research was funded by the National Institutes of Health (HL1 P01 HL108800-01A1 [Loyd], R01 122417-01 [Hemnes], and T32-HL007411 [Agrawal and Meoli] and the American Heart Association (13FTF16070002 [Brittain]).
Supplemental Material
Supplemental Material1 - Supplemental material for Adverse physiologic effects of Western diet on right ventricular structure and function: role of lipid accumulation and metabolic therapy
Supplemental material, Supplemental Material1 for Adverse physiologic effects of Western diet on right ventricular structure and function: role of lipid accumulation and metabolic therapy by Evan L. Brittain, Megha Talati, Niki Fortune, Vineet Agrawal, David F. Meoli, James West and Anna R. Hemnes in Pulmonary Circulation
Supplemental Material
Supplemental Material2 - Supplemental material for Adverse physiologic effects of Western diet on right ventricular structure and function: role of lipid accumulation and metabolic therapy
Supplemental material, Supplemental Material2 for Adverse physiologic effects of Western diet on right ventricular structure and function: role of lipid accumulation and metabolic therapy by Evan L. Brittain, Megha Talati, Niki Fortune, Vineet Agrawal, David F. Meoli, James West and Anna R. Hemnes in Pulmonary Circulation
Supplemental Material
Supplemental Material3 - Supplemental material for Adverse physiologic effects of Western diet on right ventricular structure and function: role of lipid accumulation and metabolic therapy
Supplemental material, Supplemental Material3 for Adverse physiologic effects of Western diet on right ventricular structure and function: role of lipid accumulation and metabolic therapy by Evan L. Brittain, Megha Talati, Niki Fortune, Vineet Agrawal, David F. Meoli, James West and Anna R. Hemnes in Pulmonary Circulation
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
