Abstract
It is generally acknowledged that, compared with typical antipsychotics, atypical antipsychotic agents, such as olanzapine and risperidone, are associated with a broader spectrum of clinical efficacy and are better tolerated in patients with schizophrenia [1, 2]. Over the past decade, there has consequently been a general shift towards the use of atypical antipsychotic agents for the treatment of schizophrenia [3]. However, the less expensive typical antipsychotic drugs, such as haloperidol, are still commonly used medications for treating patients with schizophrenia in Asian countries [4, 5].
Cross-cultural and cross-ethnic variations in the effectiveness and tolerability of different agents can occur [6], and trial results conducted in one population are not always indicative of response in different ethnic populations. Variables such as metabolic make-up, body mass, lifestyles, dietary habits and attitudes towards illness and treatment can all potentially influence treatment outcomes in different ethnic groups [6, 7]. Most randomized controlled trials (RCTs) demonstrating advantages of olanzapine and risperidone over haloperidol have been conducted in Western populations [8–11], but the benefits of olanzapine and risperidone over typical agents have also been directly observed in RCTs in Asian populations [12, 13]. The results of these RCTs suggest that the under-utilization of atypical antipsychotic drugs evident in Asian countries may not be in the best interests of patient health.
Although RCTs are essential for establishing the efficacy and safety of new medications, certain design features limit the applicability of RCT results to general clinical practice situations. In contrast to RCTs, observational studies aim to evaluate the effectiveness of treatments in real-world clinical practice settings using clinically relevant outcomes. However, few observational studies assessing the treatment effects of antipsychotic agents across different ethnic groups of patients with schizophrenia have been conducted and questions regarding the relative benefits of prescribing atypical antipsychotic drugs in the diverse populations seen in clinical practice remain to be answered [14].
The Intercontinental Schizophrenia Outpatient Health Outcomes (IC-SOHO) study is an ongoing, prospective, 3-year observational trial designed to evaluate treatment outcomes in a large, diverse population of patients with schizophrenia (study code F1D-SN-HGJR). The study is currently being conducted in 27 countries over four continents. This report compares 12-month effectiveness and tolerability results obtained with olanzapine, risperidone or typical antipsychotic monotherapy in patients enrolled in Asian countries participating in the ICSOHO study.
Method
Study design
Patients could be enrolled in the IC-SOHO study if, at the discretion of the treating psychiatrist, they initiated or changed antipsychotic medication as part of routine clinical practice. Any registered antipsychotic treatment available in the participating country could be prescribed and psychiatrists were instructed to make treatment decisions using their standard clinical practice guidelines. Psychiatrists were asked to alternately enter patients into two treatment cohorts (change to/initiation of olanzapine or change to/initiation of any other nonolanzapine agent) until five patients were enrolled in each cohort. Consequently, by nature of the study design rather than physician preference, half of the patients enrolled in the IC-SOHO study were initially prescribed olanzapine. In order to ensure that the study design did not interfere with the normal course of health care, psychiatrists made decisions to change or initiate antipsychotic medication prior to evaluation of patients for enrolment. The treating psychiatrist was responsible for treatment decisions, and all outcome assessments performed during the course of the study. The IC-SOHO study was carried out in accordance with the principles of the Declaration of Helsinki and met ethics requirements in each participating country.
The current report presents data collected in participating Asian countries (Korea, Taiwan and Malaysia) during the first 12 months of the IC-SOHO study.
Treatment groups
Although all patient data were collected and analysed, post-hoc treatment groups were established to evaluate only those patients who started, or switched to, monotherapy with olanzapine, risperidone or a typical antipsychotic at the baseline study visit. Small sample sizes of other atypical antipsychotic agents precluded their inclusion in the analysis. Patients who were prescribed more than one antipsychotic agent at baseline (n = 289) were also excluded from the analysis.
Patients
Any patient who had a clinical diagnosis of schizophrenia (ICD-10 or DSM-IV) presented within the normal course of care and were ≥18 years of age could be enrolled in the IC-SOHO study. Patients (or their legal representatives) were required to at least provide oral consent for release of personal information. Acquisition of written consent was dependent on the regulations existing in each participating country.
Outcome measures
Data used in this analysis were collected at baseline, 3 months, 6 months and 12 months. To allow outcomes to be attributed to the antipsychotic actually received, statistical comparisons were performed only on the patients who remained on their initially prescribed monotherapy at each treatment visit. Antipsychotic dose could be altered, but patients who were changed to another antipsychotic drug or were prescribed additional antipsychotic drugs during the treatment period were excluded from subsequent analyses.
Clinical status was assessed by the treating psychiatrist using a version of the Clinical Global Impressions-Severity of Illness (CGI-S) rating scale that was adapted to differentiate between separate symptom domains (positive, negative, depressive and cognitive symptoms) [15]. Each domain was rated from 1 (normal) to 7 (severely ill). Patients were classified as responders if their overall CGI-S score improved by ≥2 points from a baseline score of ≥4, or if their overall CGI-S score improved by ≥1 point from a baseline score of 3. Patients with baseline scores of 1 or 2, or with no score recorded were excluded from evaluation of response.
Patient perception of compliance was assessed as ‘almost always compliant’ or ‘not always compliant’. Patterns of antipsychotic drug administration were recorded at each treatment visit, including antipsychotic dosage prescribed at each visit and changes made to antipsychotic drug regimens, such as withdrawal or addition of antipsychotic agents. The proportion of patients taking concomitant anticholinergics, antidepressants, anxiolytics/hypnotics and/or mood stabilizers was also recorded.
Treatment tolerability was assessed by the treating psychiatrist using adverse event questionnaires. The presence or absence of extrapyramidal symptoms (EPS), tardive dyskinesia (TD) and sexual dysfunction (loss of libido, impotence/sexual dysfunction, amenorrhea/other menstrual disturbance, gynecomastia and galactorrhea) were specifically assessed. Body weight was measured at each visit.
Statistical analyses
Differences across treatment groups were tested using anova for continuous variables or logistic regression for categorical variables, and where differences across treatment groups were significant, pair-wise comparisons between the individual treatment groups were performed. Adjusted analyses were used for post-baseline comparisons (≥15 observations per group were required for adjusted analyses). Baseline variables used as covariates in all adjusted anova and logistic regression models were age, duration of diagnosis, gender, overall baseline CGIS score, use of depot typical antipsychotic agents in the 6 months prior to baseline, use of clozapine in the 6 months prior to baseline and hospitalization in the 6 months prior to baseline. Weight and presence/ absence of specific adverse events (EPS, TD and indicators of sexual dysfunction) at baseline were included as additional covariates in the relevant analyses. Given the large number of statistical comparisons undertaken in this study, results should be interpreted with caution. Statistical significance was reported at both the 0.001 and the 0.05 levels to aid in the interpretation of results.
Results
Patient characteristics
Of 1256 Asian patients participating in IC-SOHO, 850 (68%) were from Korea, 300 (24%) were from Taiwan and 106 (8%) were from Malaysia. Among these patients, 898 (71%) started or changed to antipsychotic monotherapy with olanzapine (n = 484; 54%), risperidone (n = 287; 32%) or a typical antipsychotic (n = 127; 14%) at baseline. Of the patients prescribed a typical antipsychotic at baseline, 35 received haloperidol. Baseline characteristics of each monotherapy treatment group are shown in Table 1. Patients prescribed typical agents were significantly older than those receiving atypical agents. Patients prescribed monotherapy with olanzapine or risperidone generally tended to have more severe symptoms at baseline than patients prescribed typical agents. Of the three treatment groups, risperidone was prescribed to the group of patients with the lowest incidence of EPS at baseline.
Characteristics of Asian patients with schizophrenia who were prescribed monotherapy with olanzapine, risperidone or a typical antipsychotic at baseline†
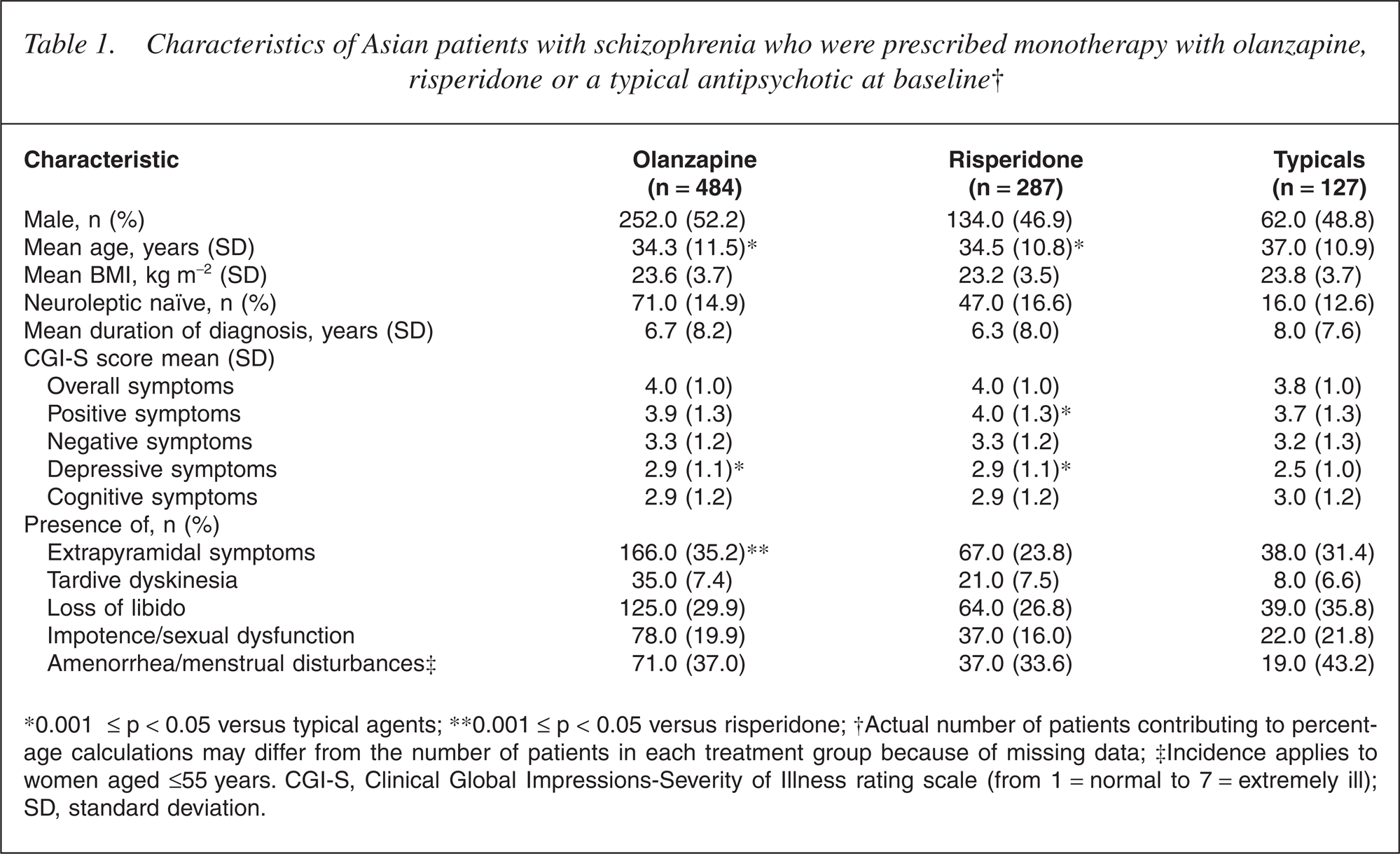
∗0.001 ≤ p < 0.05 versus typical agents; ∗∗0.001 ≤ p < 0.05 versus risperidone; †Actual number of patients contributing to percent-age calculations may differ from the number of patients in each treatment group because of missing data; ‡Incidence applies to women aged ≤55 years. CGI-S, Clinical Global Impressions-Severity of Illness rating scale (from 1 = normal to 7 = extremely ill); SD, standard deviation.
A total of 100 (11%) patients withdrew from this study before the 12-month study visit. Principal reasons were withdrawal of consent (n = 23) or switching to a new psychiatrist (n = 23). Four patients died during the 12-month period: one each from suicide in the olanzapine and risperidone groups, respectively, and two from non-suicide causes in the typical antipsychotic group. Other reasons for discontinuation from the study included prolonged initial stay in hospital (n = 9), participation in another study (n = 1) and other non-specified reasons (n = 40).
Treatment patterns and compliance
Median olanzapine dose increased from 5 mg day−1 at baseline to a stable 10 mg day−1 dose at 3, 6 and 12 months, and median risperidone dose increased from 2 mg day−1 at baseline to 3 mg day−1 at all subsequent assessment points (Table 2). Median typical antipsychotic oral dosage ranged between 250 and 400 mg day−1 chlorpromazine equivalents, and the subgroup of patients receiving haloperidol received median doses ranging between 5.5 mg day−1 at baseline and 10.0 mg day−1 at month 12.
Oral doses (mg day−1) of olanzapine, risperidone and typical antipsychotics prescribed at baseline and 3, 6 and 12 months

Chlorpromazine equivalents. SD, standard deviation.
During the first 12 months of treatment, 72.7% of evaluable patients in the olanzapine treatment group remained on their originally prescribed monotherapy, as did 67.7% of patients in the risperidone group and 62.9% of patients in the typical antipsychotic group. The difference across the treatment groups was not statistically significant (p = 0.37).
High proportions of patients in each of the treatment groups considered themselves to be almost always compliant (ranging from 80.5% in the olanzapine group to 75.0% and 68.1% in the atypical antipsychotic and risperidone treatment groups, respectively; p = 0.13).
Clinical status
Response rates
During 12 months of treatment, a greater proportion of patients in the olanzapine (76.3%) and risperidone (72.7%) groups responded to treatment, when compared with the group of patients who received typical (50.0%) antipsychotics (Fig. 1). Relative to the olanzapine group, the odds ratio for response with a typical agent was 0.38 (p = 0.010).
Proportions (and upper 95% confidence interval limits) of patients receiving olanzapine, risperidone or typical antipsychotic monotherapy who responded to treatment during the 12 month observation period. (∗0.001 ≤ p < 0.05)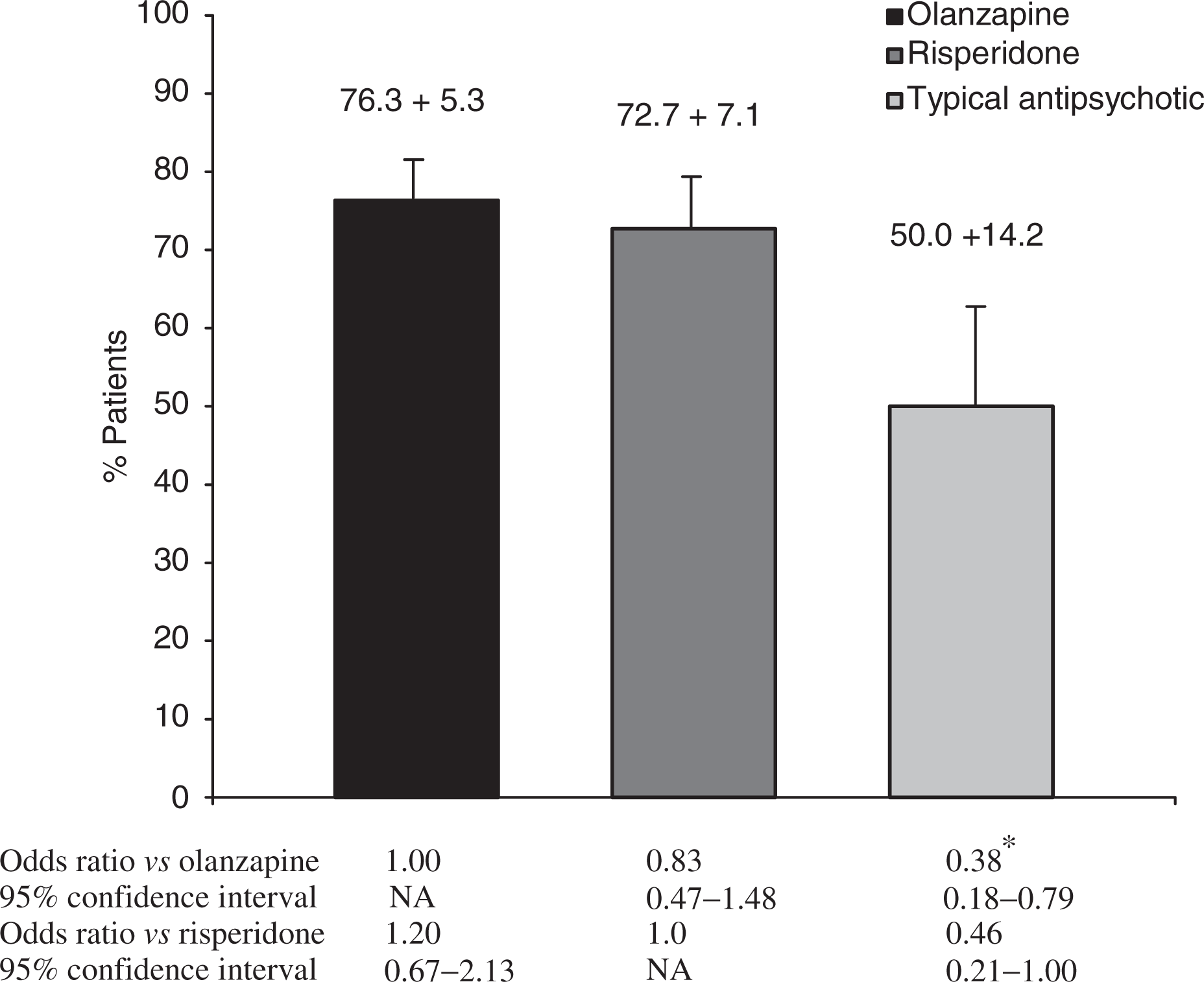
CGI-S scores
Progressive improvements in CGI-S scores were evident for each of the monotherapy treatment groups during the 12-month treatment period (Table 3). At all study visits, patients in the olanzapine and risperidone groups experienced significantly greater improvements from baseline in overall CGI-S score relative to the group of patients prescribed a typical antipsychotic. At the 3-month assessment, a least squares mean reduction in overall CGI-S score of 0.9 of a point was reported for the typical treatment group compared with a 1.2-point reduction in overall score in both the olanzapine (p = 0.004) and risperidone (p = 0.009) groups. In patients still receiving typical antipsychotic monotherapy at the 12-month assessment, improvement of overall score reached the 1.2-point level, while improvements in the atypical treatment groups had progressively increased to 1.8 points with olanzapine (p < 0.001 vs typical antipsychotics) and 1.6 points with risperidone (p = 0.007 vs typical antipsychotics).
Change from baseline in the CGI-S score over 3, 6 and 12 months of treatment, as assessed with the Clinical Global Impressions-Severity of Illness rating scale†
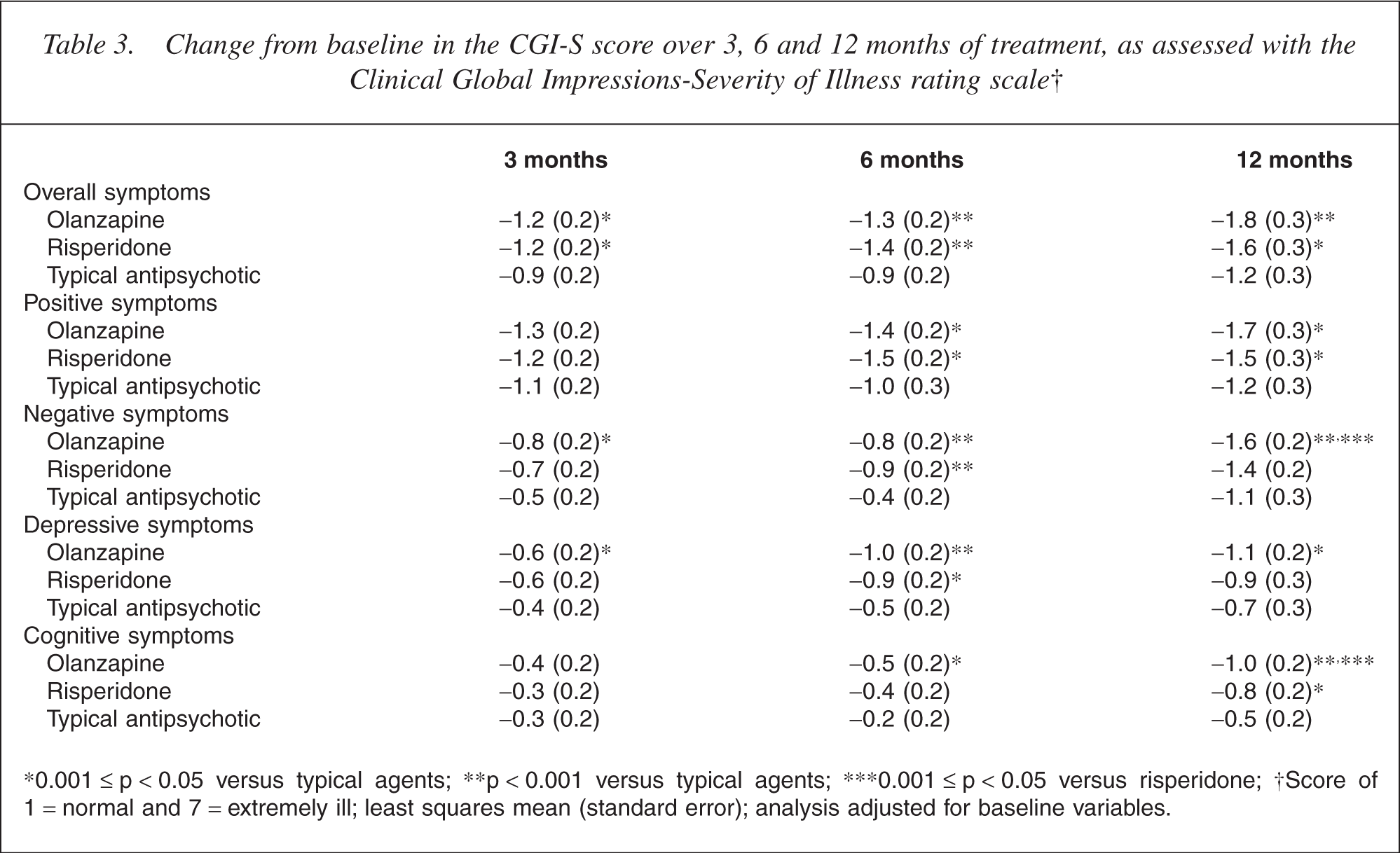
∗0.001 ≤ p < 0.05 versus typical agents; ∗∗p < 0.001 versus typical agents; ∗∗∗0.001 ≤ p < 0.05 versus risperidone; †Score of 1 = normal and 7 = extremely ill; least squares mean (standard error); analysis adjusted for baseline variables.
Reductions in CGI-S positive symptom scores were similar across the three treatment groups at 3 months (p = 0.30). However, the positive symptom score was progressively reduced in the olanzapine and risperidone groups until 12 months, whereas there was no further improvement in the typical treatment group. A 1.2-point reduction from baseline positive symptom score was recorded for the typical group at 12 months, but there was a 1.7-point reduction in the olanzapine group (p = 0.001), and a 1.5-point reduction in the risperidone group (p = 0.032) for the same period.
As early as the 3-month visit, olanzapine demonstrated significantly improved scores, relative to the typical group in the negative (p = 0.024) and depressive symptom (p = 0.030) domains. This was also evident at the 12-month visit (p < 0.001 and p = 0.024, respectively). A statistically significant difference was apparent between the olanzapine and typical treatment groups in the cognitive symptom score at the 6-month (p = 0.012) and 12-month (p < 0.001) visits. Differences between the risperidone and typical treatment groups regarding negative and depressive symptom scores became statistically significant at 6 months, but statistical significance was lost at 12 months. Furthermore, by 12 months, improvements in both negative and cognitive symptom scores observed in olanzapine recipients were significantly greater than those obtained with risperidone (p = 0.036 for negative symptoms, and p = 0.035 for cognitive symptoms).
Tolerability
Extrapyramidal symptoms
During the 12-month observation period, the highest rate of dystonia, akathisia and/or parkinsonism occurred in patients in the typical antipsychotic group (50.9%), followed by the risperidone (31.2%) and olanzapine (19.9%) treatment groups (Table 4). Logistic regression analysis showed that the odds of having EPS were significantly lower in both the olanzapine (p < 0.001) and risperidone (p = 0.010) treatment groups relative to the typical antipsychotic group. In addition, the odds of EPS occurring in patients treated with risperidone were two times greater than for patients treated with olanzapine (p = 0.012).
Likelihood of experiencing adverse events between baseline and 12 months in patients receiving monotherapy with olanzapine, risperidone or a typical antipsychotic agent
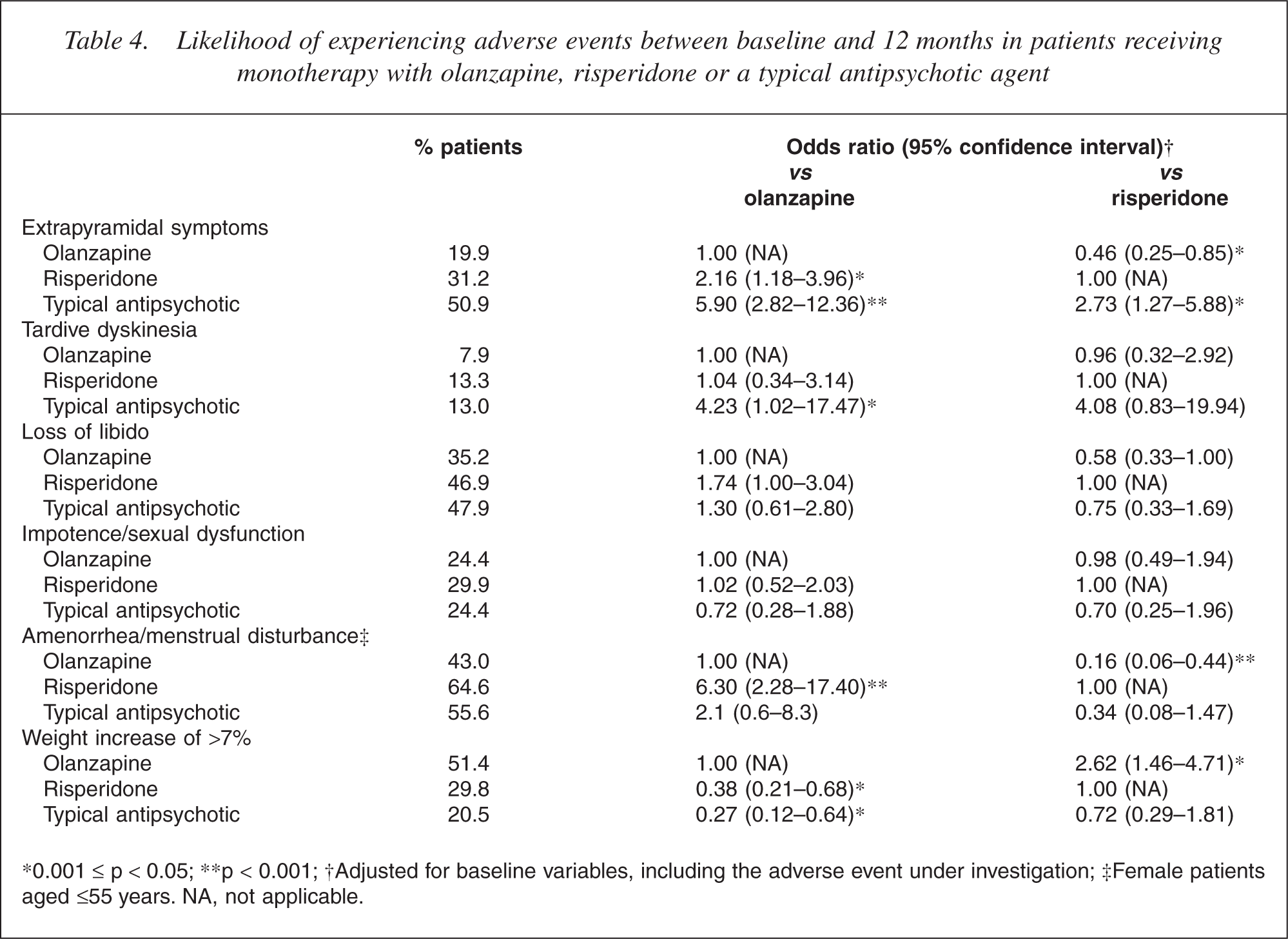
∗0.001 ≤ p < 0.05; ∗∗p < 0.001; †Adjusted for baseline variables, including the adverse event under investigation; ‡Female patients aged ≤55 years. NA, not applicable.
Tardive dyskinesia
During the first 12 months of treatment, TD was present in 13.0% of patients in the typical antipsychotic treatment group, 13.3% in the risperidone group and 7.9% in the olanzapine group (Table 4). Logistic regression showed that the odds of TD occurring during the 12-month period were approximately four times higher in the typical antipsychotic group than in both of the atypical groups (p = 0.046 for olanzapine and p = 0.082 for risperidone). No appreciable difference was found between olanzapine and risperidone groups in the odds of TD occurring during the 12-month treatment period (p = 0.948).
Adverse events related to sexual dysfunction/hyperprolactinemia
The prevalence of amenorrhea/menstrual disturbance was highest in the risperidone treatment group (64.6%) and lowest in the olanzapine group (43.0%) during 12 months of treatment (Table 4). When compared with olanzapine, the odds of amenorrhea/menstrual disturbance were significantly greater in the risperidone group (OR = 6.30; p < 0.001).
There was no significant difference between the treatment groups in loss of libido and impotence/sexual dysfunction (Table 4).
Weight gain
Weight gain was evident in all three treatment groups over the 12-month period. A least squares mean weight gain of 3.5 kg was observed in the olanzapine treatment group by the 12-month visit, while patients receiving typical antipsychotics gained 1.6 kg (p = 0.030 vs olanzapine) and risperidone recipients gained 0.76 kg (p < 0.001 vs olanzapine) in the same period. Clinically relevant weight gain (defined as a weight increase of >7%) during the 12-month observation period was observed in 51.4% of patients in the olanzapine monotherapy group, 29.8% of the risperidone group, and 20.5% of the typical group (Table 4). Logistic regression analysis showed that the risk of a >7% weight gain was significantly increased in the olanzapine group relative to both the typical (p = 0.003) and risperidone (p = 0.001) groups.
Concomitant medication
Patients receiving olanzapine monotherapy were significantly less likely to be prescribed anticholinergic agents than were patients in the typical antipsychotic or risperidone treatment groups (both p < 0.001; Table 5). Compared with the olanzapine treatment group, the odds of anxiolytic/hypnotics prescription were 3.4 times greater in the typical antipsychotic group (p = 0.001) and 1.8 times greater in the risperidone group (p = 0.023). There were no significant differences between the treatment groups for prescription of concomitant antidepressants or mood stabilizers.
Likelihood of being prescribed concomitant medication between baseline and 12 months in patients receiving olanzapine, risperidone or a typical antipsychotic agent
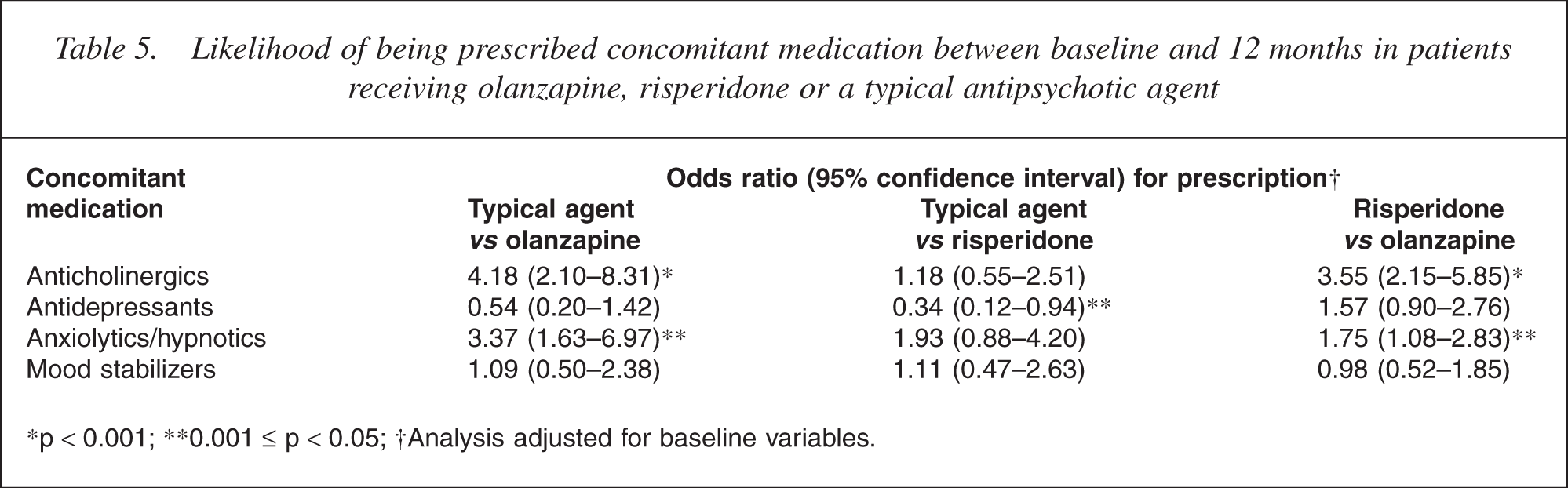
p < 0.001; ∗∗0.001 ≤ p < 0.05; †Analysis adjusted for baseline variables.
Discussion
This is the first analysis from a prospective naturalistic observational study that directly compares clinical outcomes in Asian patients with schizophrenia who were treated with a monotherapy of olanzapine, risperidone or typical antipsychotic. The findings were consistent with the results of RCTs showing that atypical agents have a broader efficacy profile and are better tolerated than typical agents in Asian patients [12, 13]. Results from this subset of Asian patients participating in the IC-SOHO study were also consistent with 12-month results from the overall IC-SOHO study [16].
While typical antipsychotic agents can reduce the most conspicuous positive symptoms of schizophrenia, they often have less effect on other symptoms that are intrinsically linked with the disease, including negative and depressive symptoms and impaired cognition [1, 17, 18]. Compared with the olanzapine treatment group, typical antipsychotic treatment seemed to offer less effectiveness against negative, depressive and cognitive symptoms for the Asian patients participating in the IC-SOHO trial. When compared with the typical group, olanzapinetreated patients experienced earlier improvements in negative, depressive and cognitive symptoms, and these benefits were maintained at 12 months. Further, olanzapine treatment was associated with greater improvements in negative and cognitive symptoms when compared with risperidone over 12 months. Our observations are in line with the results of several RCTs that have shown olanzapine to be superior to risperidone with regard to negative [19] and cognitive symptoms [19–21]. Another RCT reported greater improvements in positive and affective symptoms for risperidone study completers versus olanzapine study completers [22], and a naturalistic study also reported some advantages for risperidone over olanzapine [23].
The risk of developing EPS is a major limitation of typical antipsychotic medications. In Asian patients participating in IC-SOHO, the odds of having EPS during the first 12 months of the trial were significantly greater with typical antipsychotics and risperidone relative to olanzapine. In addition, the likelihood of being prescribed concomitant anticholinergics over the 12-month period was significantly greater in the typical and risperidone groups when compared with the olanzapine group. It has been suggested that Asian patients may be particularly susceptible to developing EPS during antipsychotic drug treatment [24, 25]. This increased susceptibility to developing EPS is concerning in light of evidence that patients with EPS have a significantly greater chance of developing TD when compared with patients without a history of EPS [26, 27].
In this study of Asian patients, the odds of developing TD during the first 12 months of the trial were reduced in both the olanzapine and risperidone groups relative to the typical antipsychotic group. Hyperprolactinemia can occur in association with typical and some atypical drugs and has been associated with adverse consequences with regard to sexual function [28]. Prolactin levels were not measured in our study, but adverse events related to sexual function, particularly amenorrhea/menstrual disturbances, were least likely in patients receiving olanzapine. This may be reflective of the relatively low impact of olanzapine on prolactin levels that has been described in other studies [29–31].
Another adverse event observed with antipsychotic drugs is weight gain, the extent of which seems to vary from drug to drug [32]. Previously published data reporting that weight gain observed with olanzapine is greater than that with risperidone are mainly from studies of patients in Western countries [32, 33]. Lipid metabolism, eating habits and lifestyle can differ between people of different ethnicities, so there is the possibility of differential weight changes in different populations [34]. However, consistent with previously reported patterns, Asian patients participating in IC-SOHO who were in the olanzapine treatment group gained the most weight during our 12-month observation period. Clinicians should take the time to monitor weight during treatment with antipsychotic drugs, and to implement a management plan in the event of potentially harmful weight gain.
The results of several RCTs provide support for the main findings of this observational study. When compared with risperidone treatment, olanzapine treatment has been associated with greater improvements in negative [19], depressive [21] and cognitive [19–21] symptom domains as well as EPS [19]. In contrast, direct comparisons between olanzapine and risperidone revealed similar overall effectiveness and incidence of EPS, while risperidone had advantages with regard to positive and affective symptoms in patients who completed the trial [22]. It has been suggested that these differences between atypical agents, or lack thereof, may be contingent on issues of disproportionate dosing [35–37]. Compared with recommended doses in Caucasian patients [23], dosing was not disproportionately low for Asian patients receiving either olanzapine or risperidone in the ICSOHO trial, suggesting that dosing should not have biased effectiveness or tolerability data in favour of either drug.
The results of the multiple analyses conducted as part of the exploratory, observational IC-SOHO study should be interpreted with caution. Unlike RCTs, selection bias cannot be controlled in observational trials, and differences inherent in treatment groups at baseline may not be completely offset by adjusted analyses. For Asian patients participating in the IC-SOHO study, the olanzapine group had the highest incidence of psychotic symptoms and EPS at baseline. It is possible that there may have been other, unobserved variations within the sample groups that could have influenced the results. Further, prescribing psychiatrists were aware of the treatment received by each patient, and were also responsible for outcome assessments. Although this may potentially bias the results, this methodology would accurately reflect real-life clinical practice, where the clinician may utilize all available patient information to formulate a comprehensive clinical evaluation.
The approach of assessing outcome in patients who remained on the monotherapy prescribed at baseline could also potentially bias results. Patients who remain on a particular therapy over an extended term are likely to be doing well on the treatment, and may not necessarily be reflective of the actual population. However, during our 12-month observation period, the majority of patients did remain on the monotherapy originally prescribed at baseline. Additionally, when outcomes were analysed in terms of baseline treatment group regardless of subsequent changes of therapy during the 12-month treatment period (data not shown), differences between the treatment groups were consistent with the findings in patients who remained on the monotherapy originally prescribed at baseline.
Conclusion
The results of this observational study of Asian patients with schizophrenia indicate that, with the exception of weight gain, olanzapine and risperidone may offer benefits over typical agents. These data also support the existence of some differentiation between olanzapine and risperidone.
Footnotes
Acknowledgements
This study was supported by a research grant from Eli Lilly and Company, IN, US. The independent medical writing assistance was provided by Adis International, funded in part from a financial grant from Eli Lilly Australia Pty Limited, Macquarie Park, NSW, Australia.
