Abstract
Introduction
Schizophrenia is a chronic and devastating mental disorder affecting approximately 0.7 % of populations worldwide (Tandon et al., 2008). Symptoms of schizophrenia are regularly categorized as being either predominantly positive or negative. Positive symptoms − including hallucinations, delusions, and disorganized behaviours − reflect distorted brain functions, whereas negative symptoms − such as anhedonia, alogia, apathy, and avolition − are thought to represent a loss of brain function (Mäkinen et al., 2008; Tandon et al., 2009). Both positive and negative symptoms represent the core clinical manifestation of schizophrenia. Although a variety of antipsychotic drugs have been introduced into clinical use since chlopromazine was first produced in 1950s, the outcome of patients with schizophrenia is still far from satisfactory, especially for those with negative symptoms dominated schizophrenia (Mueser and McGurk, 2004; Jiang, 2007).
The currently available antipsychotic drugs are generally classified into typical (or first-generation) and atypical (or second-generation) antipsychotics according to their pharmacological properties. The typical antipsychotic drugs that primarily work to antagonize D2 receptor in the central nervous system have been shown to be solely effective in treating positive symptoms, whereas they have minimal effects on negative symptoms. Most atypical antipsychotics, which act at multiple receptors in the brain, have been demonstrated to be nearly as effective as typical antipsychotics in treating positive symptoms, but can also ameliorate the severity of the negative symptoms (Mueser and McGurk, 2004; Erhart et al., 2006; Jiang, 2007). Nevertheless, the overall effectiveness of these medications on negative symptoms is still far from satisfactory, contributing to a much poorer outcome in patients with negative symptom dominated schizophrenia. Thus, there is pressing need to search for novel therapeutic strategies for treating the negative symptoms of this condition (Erhart et al., 2006; Jiang, 2007).
Cerebrolysin is a porcine brain-derived neuropeptide preparation that has been demonstrated to have biological properties similar to neurotrophic factors in vivo when administered via intravenous injection (Ubhi et al., 2009; Zhang et al., 2010). Cerebrolysin consists of approximately 15% peptides with a molecular weight not exceeding 10 kD and 85% amino acids (based on the total nitrogen). The solution − prepared for injection or infusion − is free of proteins, lipids, and antigenic properties. One millilitre of cerebrolysin contains 215.2 mg of peptide preparation in aqueous solution. In a series of clinical studies, cerebrolysin has been demonstrated to be effective in treating cognitive impairment in patients with various neurological diseases including Alzheimer’s disease (Xiao et al., 2000; Wei et al., 2007; Alvarez et al., 2011), vascular dementia (Vereshchagin et al., 1991; Plosker and Gauthier, 2009), stroke (Jianu et al., 2010), cerebral haemorrhage (Bajenaru et al., 2010), and traumatic brain injury (Wong et al., 2005). In these studies, the patients suffered from organic brain diseases in which specific neural cells were affected and there were demonstrable structural changes in the central nervous system. It is thought that the therapeutic efficacy of cerebrolysin in these diseases is associated with its neuroprotective effects on the affected neural cells. Schizophrenia, on the other hand, is generally considered to be a functional disorder without definite structural brain abnormalities. This may be why there has been no clinical study investigating the effects of cerebrolysin in schizophrenia. However, with modern brain imaging techniques, there is growing evidence suggesting that the negative symptoms of schizophrenia may be associated with certain pathological abnormalities in the central nervous system (Lahti et al., 2006; Minatogawa-Chang et al., 2009; Takahashi et al., 2009), and there been a controversial debate over whether schizophrenia should in fact be considered to be a neurodegenerative disease (Rund, 2009; Urfer-Parnas et al., 2010). Thus, it would be interesting to investigate whether patients with schizophrenia respond to therapies found to be efficacious in those with neurological diseases.
The aim of the current study was to determine if cerebrolysin would augment the efficacy of risperidone in patients with negative-symptom-dominated schizophrenia over an 8-week, double-blind, placebo-controlled, parallel-group clinical trial. We hypothesized that cerebrolysin would significantly improve negative symptoms and cognitive function in these patients.
Methods
Study design
The trial used a double-blind, placebo-controlled, parallel-group design. The subjects were randomly assigned to one of two treatment groups (cerebrolysin plus risperidone group or risperidone plus placebo). The patients were assessed at baseline, 2, 4, and 8 weeks after the start of treatment. The study period for each subject was 8 weeks and the duration of the entire trial was 2 years.
Both groups received a flexible dosage of risperidone (2−4 mg daily). In addition, the active treatment group received 30 ml of cerebrolysin in 250 ml physiological saline intravenous infusion, while the placebo group received 30 ml of placebo in 250 ml physiological saline intravenous infusion. The infusions were given daily Monday to Friday over 4 weeks. Yellow opaque infusion bottles containing cerebrolysin or placebo were used to ensure blinding.
Subjects
The study enrolled male and female patients aged 18 and over who met DSM-IV diagnostic criteria for schizophrenia and who had a score on the Positive and Negative Symptoms Scale negative symptom scale greater than that on the positive symptom scale. All patients had vision and hearing sufficient for compliance with test procedures. All patients were receiving risperidone monotherapy prior to entering the trial and the dosage of risperidone had been stabilized for at least 1 month prior to the trial.
Patients with evidence of other psychiatric disorders or who had clinically significant or active renal, hepatic, endocrine, or cardiovascular conditions were excluded. Patients with severe heart disease, severe hypertension, and those with severe obstructive pulmonary disease or haematological or oncological disorders were also excluded. Patients were only included if they had not received the investigational drug (cerebrolysin) within the 4 months prior to entering the trial. Patients who were using other nootropics (unless discontinued for at least 4 weeks prior to the trial), hypnotics (short-acting benzodiazepines were allowed), drugs influencing cerebral blood flow, or stimulants were also excluded.
The study was conducted in accordance with the Declaration of Helsinki. Before initiation of the screening activities, the nature and purpose of the investigation were explained and informed consent obtained from both the patients and their legal representatives.
Forty-nine and 52 patients in the placebo and cerebrolysin groups, respectively, completed the follow up, of which the sample size was estimated to detect a difference of 10 in PANSS negative score between the two treatment groups, with 80% power and 5% probability of type I error.
Clinical assessments
The efficacy assessments were performed by experienced psychiatrists, most of whom were attending doctors at the Shanghai Mental Health Center. To assess the clinical response to treatment with cerebrolysin plus risperidone compared to risperidone alone, the following scales were utilized:
The Positive and Negative Symptoms Scale (PANSS) is a 30-item rating instrument evaluating the presence/absence and severity of positive, negative, and general psychopathology of schizophrenia. All 30 items are rated on a 7-point scale (1=absent; 7=extreme).
The Wechsler Memory Scale (WMS) is a comprehensive measure of adult memory functioning which assesses both auditory and visual memory. The scale comprises a number of subtests including information, orientation, mental control, logical memory, digits forwards, visual reproduction, and associative learning.
The modified Chinese version of the Wechsler Adult Intelligence Scale (mWAIS) is a general adult IQ test measuring verbal and performance IQ.
The Clinical Global Impression (CGI) scale is a three-item scale used to assess treatment response in psychiatric patients. The items are severity of illness (1=normal; 7=extremely ill), global improvement (1=very much improved; 7=very much worse), and efficacy index (1=none; 4=outweighs therapeutic effect).
Brief Psychiatry Rating Scale (BPRS) is an 18-item observer-scale measuring positive symptoms, general psychopathology, and affective symptoms. Each item is rated on a seven-point scale (1=not present; 7=extremely severe).
The primary outcome measure was the extent of changes in PANSS negative score and total scores at each follow up. The secondary outcome measures were the extent of change in WMS and mWAIS over the trial, and general improvement as determined by CGI rating at endpoint. The endpoint rating on the BPRS was used as a further secondary outcome measure.
Safety
Adverse events were monitored by structured questions. All events reported by patients, or noticed by caregivers or physicians, were recorded together with the date of onset and resolution, severity, and relationship to trial medication using the standard international Treatment Emergent Side-effects Scale (TESS). Measurements of haemoglobin, haematocrit, erythrocyte count, and total and differential leukocyte and platelet counts were used to monitor haematological effects at baseline and study endpoint. Similarly, clinical chemistry parameters were monitored to assess liver function (alkaline phosphates, aspartate transaminase, alanine transaminase, and total bilirubin), renal function (blood urea nitrogen and creatine), metabolic function (total cholesterol, glucose, total protein, and albumin) and electrolytes (sodium, potassium, calcium, and phosphorus). Electrocardiography and a routine urinalysis (specific gravity, dipstick chemistry, pH, protein, glucose, and ketones) were also performed at baseline and endpoint. Vital signs assessments included blood pressure, heart rate, body temperature, respiratory rate, and body weight.
Data analysis
Continuous demographic variables (e.g. age, height, and weight) were compared between the two treatment groups using Student’s t-tests. Repeated measures analysis of variance was used to compare change in scores on PANSS, mWAIS and WMS between the two groups at each time point. Improvements on the CGI and other categorical variables were compared between groups using the chi-squared test or Fisher’s exact test. All hypothesis tests were two tailed, with p-values ≤0.05 being considered to be statistically significant. All the statistical analyses were undertaken using SPSS version 13.0 (SPSS, Chicago, IL, USA).
Efficacy of treatment was based on the combined results of each patient’s last assessment during the double-blind therapy-defined study endpoint. All hypothesis tests were two-sided, and p-values ≤0.05 were considered to be significant. Continuous demographic variables (e.g. age, height, and weight) were examined and compared between the two groups using the t-test, and categorical demographic variables analysed using the chi-squared test or Fisher’s exact test. The ordinal scaled data or data with multi-model distribution were subject to the Wilcoxon−Mann−Whitney U-test. Analysis of adverse events were conducted by tabulating treatment-emergent signs and symptoms, which were defined as events that occurred during or after administration of the first dose of study medication, or became more severe during treatment. The incidence of treatment-emergent adverse events and treatment-emergent laboratory abnormalities were compared between groups by means of the Fisher’s exact test.
Results
Randomized groups
A total of 109 patients met DSM-IV diagnostic criteria for schizophrenia and the study’s selection criteria for predominant negative symptoms: 54 were randomized to the placebo group (risperidone only) and 55 to the cerebrolysin group (cerebrolysin plus risperidone). Of the 109 patients, 101 completed the study. Reasons for study discontinuation were: (i) changes to antipsychotic drugs due to symptom deterioration (three in the placebo and one in the cerebrolysin group), and (ii) the request for withdrawal by the patients’ legal representatives or caregivers (two in the placebo and two in the cerebrolysin group). The final dataset (Figure 1) consisted of 101 patients (49 placebo and 52 cerebrolysin).
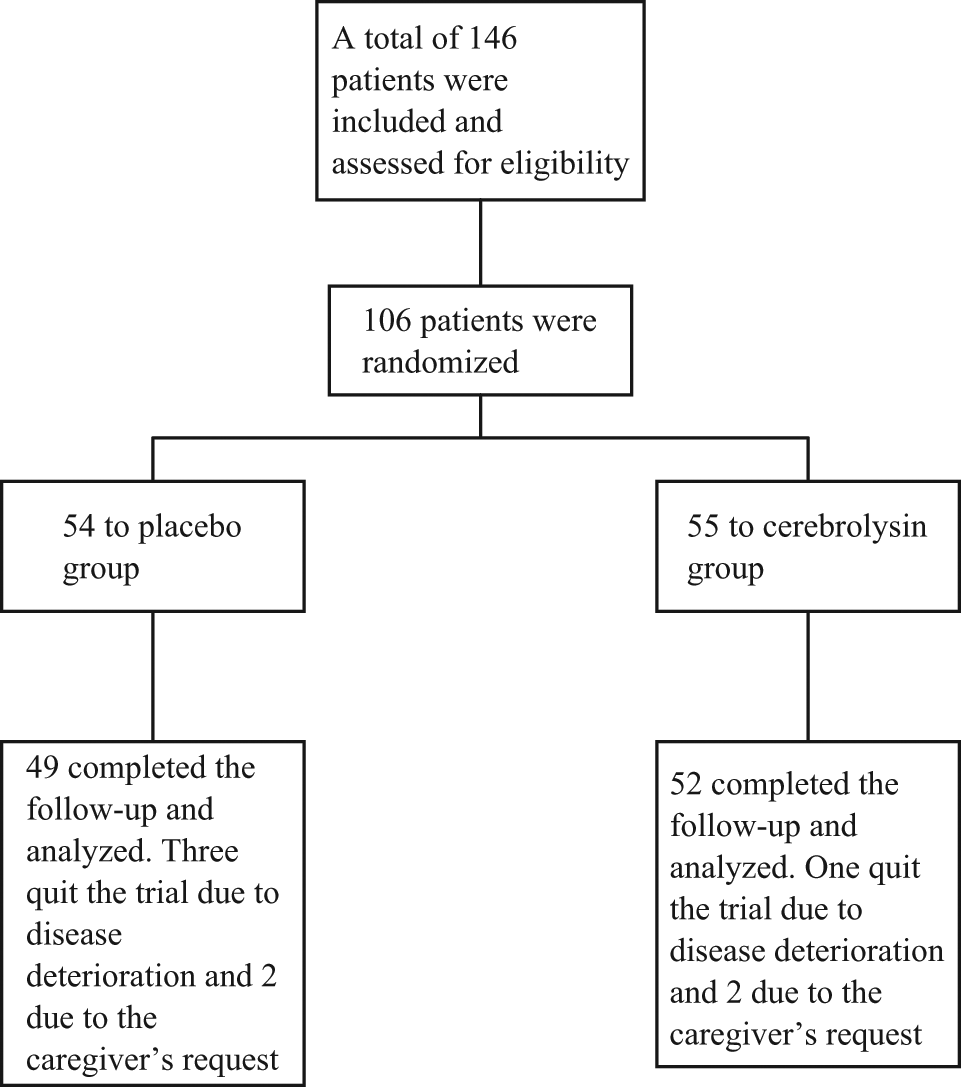
Patient disposition
Demographic data and baseline disease characteristics
The demographic characteristics of patients are presented in Table 1. There were more male than female patients in both groups, but the ratio did not show significant differences between the cerebrolysin and placebo groups. Likewise, no significant differences were observed in other demographic data between the two groups, including level of education, age, marital status, family history of psychiatric illness, duration of illness, age of first onset of illness, and mean dose of risperidone used before the study.
Baseline demographic data
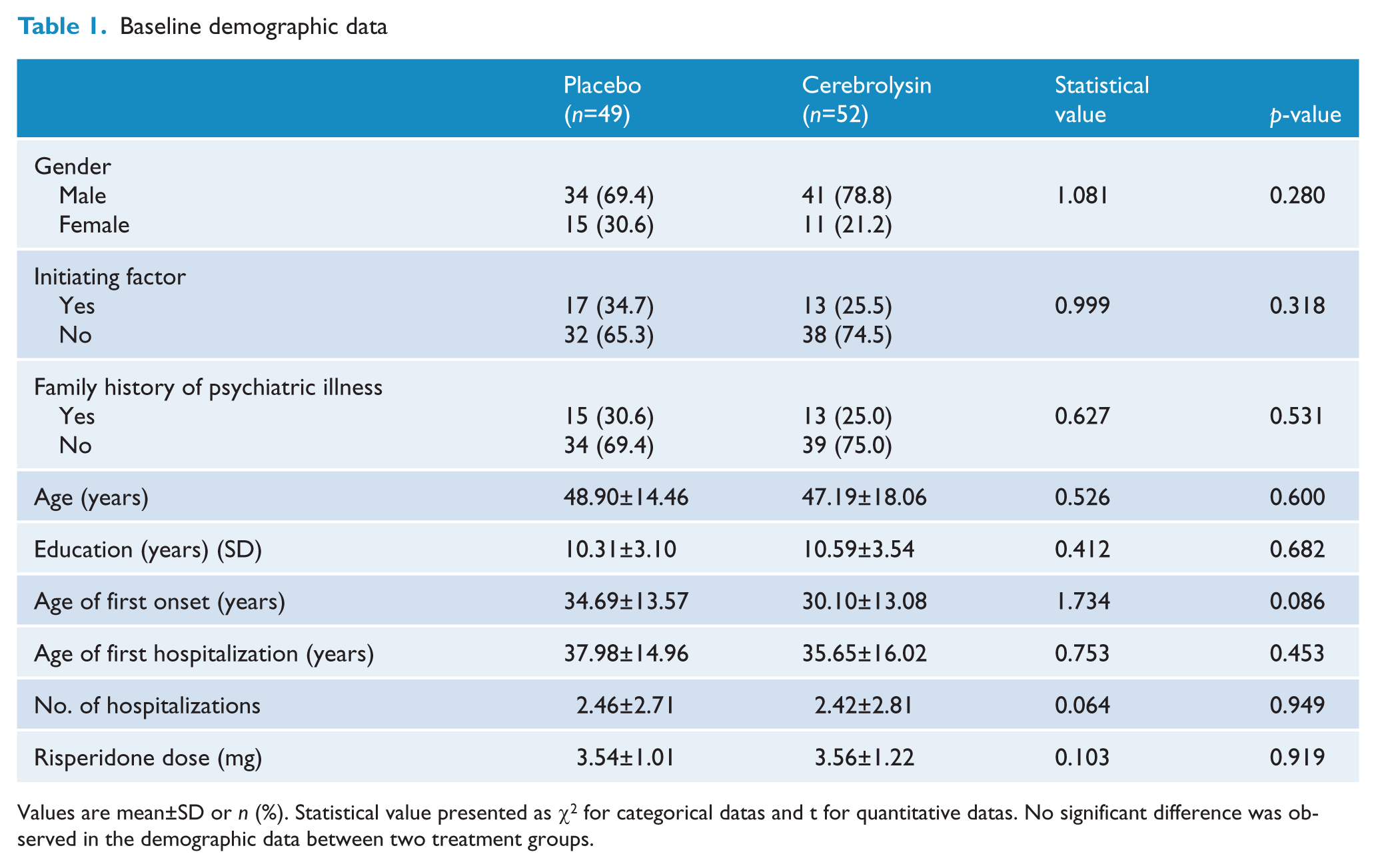
Values are mean±SD or n (%). Statistical value presented as χ2 for categorical datas and t for quantitative datas. No significant difference was observed in the demographic data between two treatment groups.
Baseline measurements of PANSS, mWAIS, and WMS are presented in Tables 2 and 3. Patients in both groups were negative symptoms dominated as shown by the PANSS subtests. There were no significant differences observed in scores on PANSS, mWAIS, and WMS at baseline between groups (data not shown).
Positive and Negative Symptoms Scale (PANSS) score change in the two treatment groups at each follow-up point
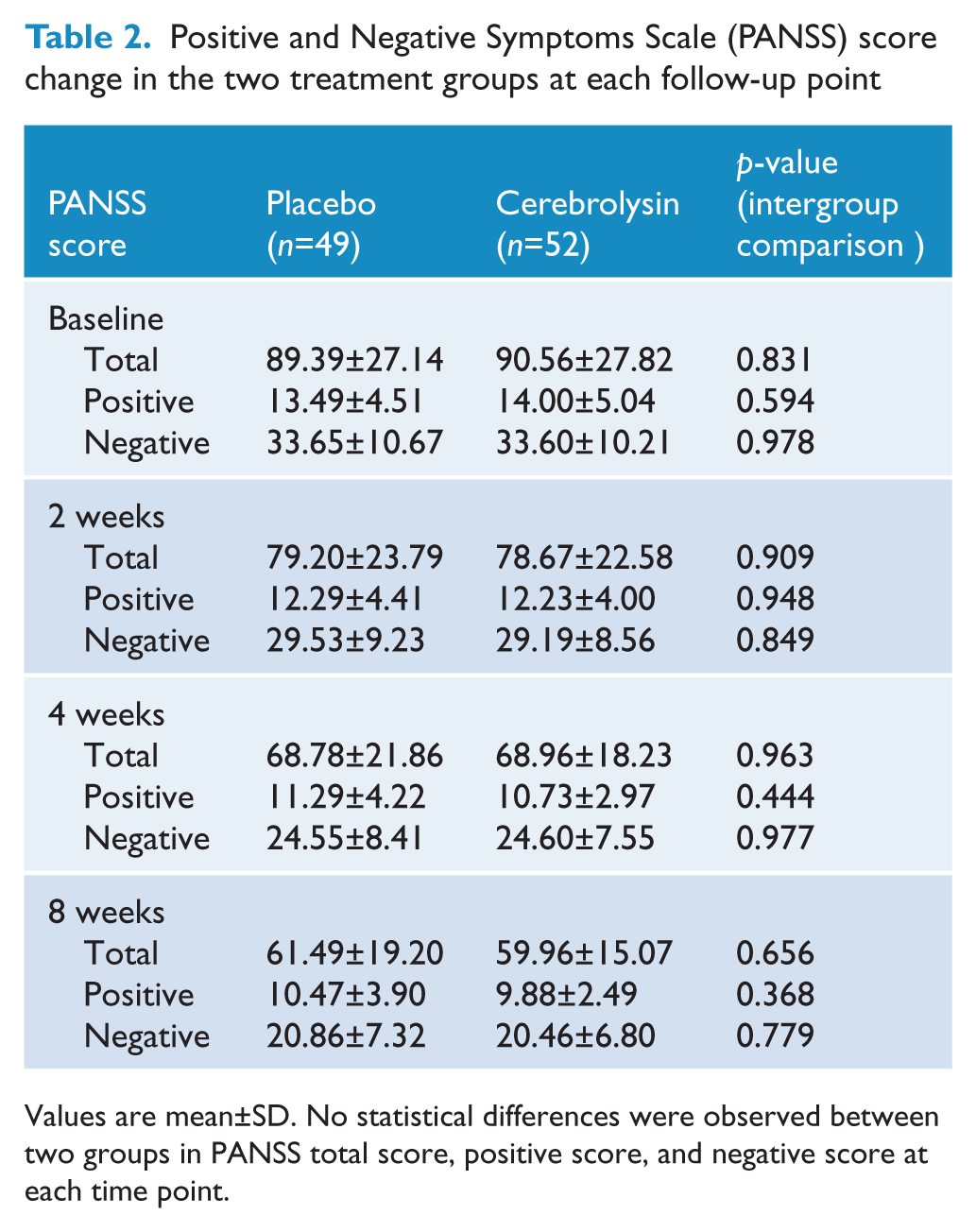
Values are mean±SD. No statistical differences were observed between two groups in PANSS total score, positive score, and negative score at each time point.
Modified Chinese Wechsler Adult Intelligence Scale and Wechsler Memory Scale score changes in the two treatment groups at each follow-up point
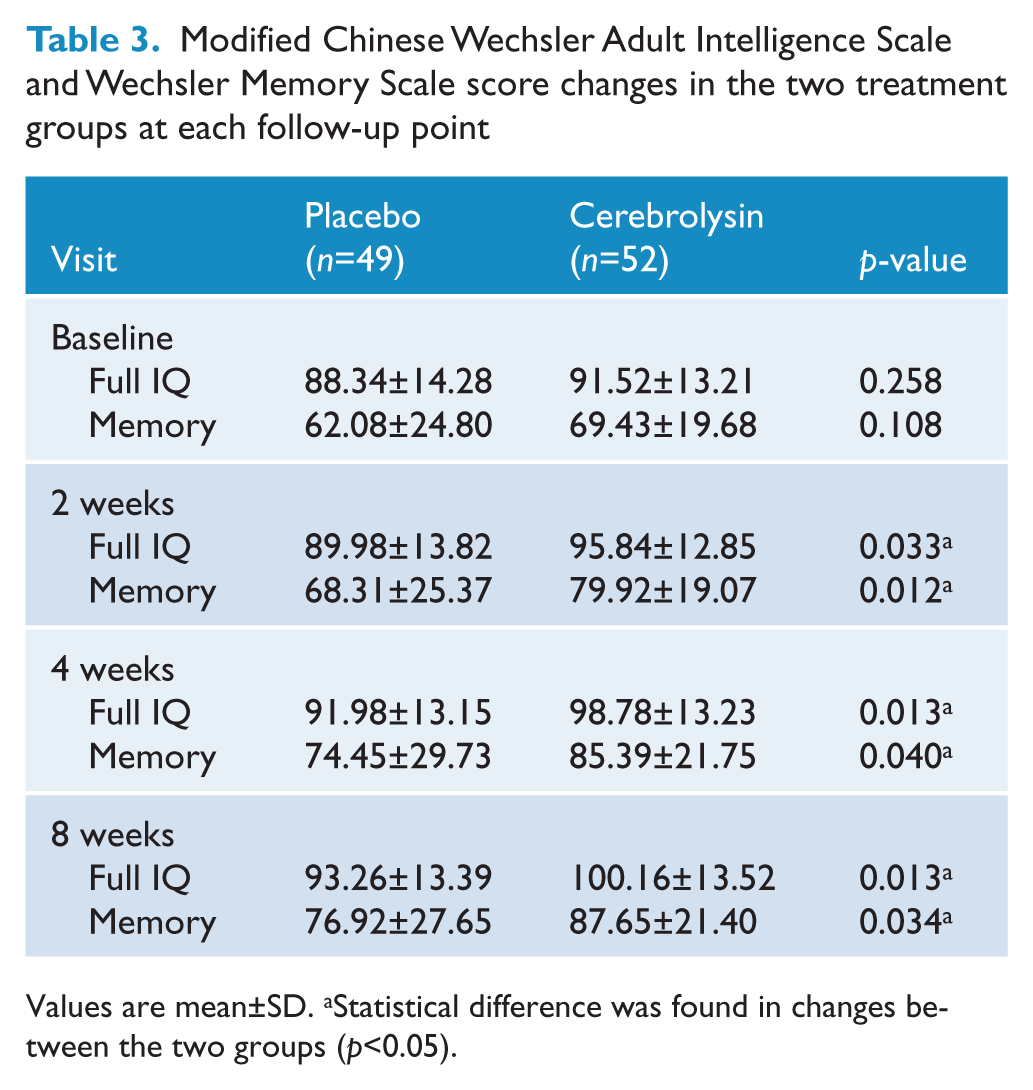
Values are mean±SD. aStatistical difference was found in changes between the two groups (p<0.05).
Primary outcome measures
The outcome of the primary outcome measures as determined by the changes in PANSS negative score and total score at each follow up are presented in Table 2. Patients in both groups demonstrated significant decreases in PANSS total score and negative score after 2 week treatment. Scores continued to decrease over the 8 weeks of the trial. However, there was no statistical difference in the extent of changes in either the PANSS total score or negative score at any follow-up point between the two treatment groups. Greater decreases in PANSS total scores, negative scores, and positive scores were observed in the cerebrolysin group at each interview, but the improvements did not reach statistical significance compared to the placebo group (Table 2).
Secondary outcome measures
Scores on the mWAIS and WMS in the two groups at each follow-up point are presented in Table 3. Both groups showed a significant increase in mWAIS and WMS scores from week 2 to week 8. Furthermore, there was a significantly greater increase in both mWAIS and WMS scores at each follow-up point for the cerebrolysin group compared to the placebo group, indicating that cerebrolysin may augment the efficacy of risperidone in improving cognition and memory.
There was no significant difference between the two treatment groups as determined by the CGI (Table 4).
Clinical Global Impression rating at endpoint
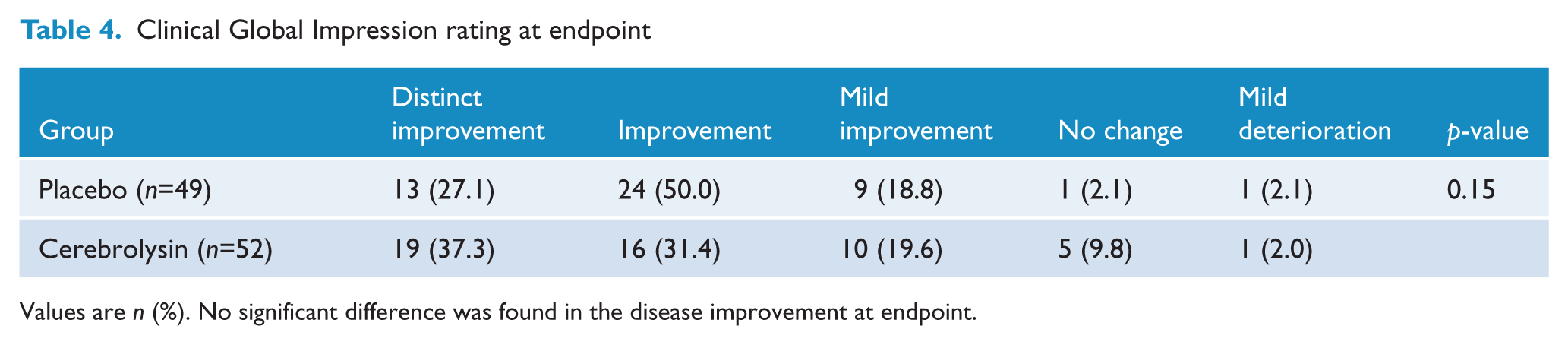
Values are n (%). No significant difference was found in the disease improvement at endpoint.
Safety
The frequencies of treatment-emergent adverse events are summarized in Table 5. There were no severe adverse events observed in either of the groups during the trial. The most common adverse events occurring in at least 5% of any treatment group were abnormal ECG, constipation, abnormal hepatic function, weight gain, weight loss, and abnormal blood profile.
Treatment-emergent adverse events observed in the two treatment groups
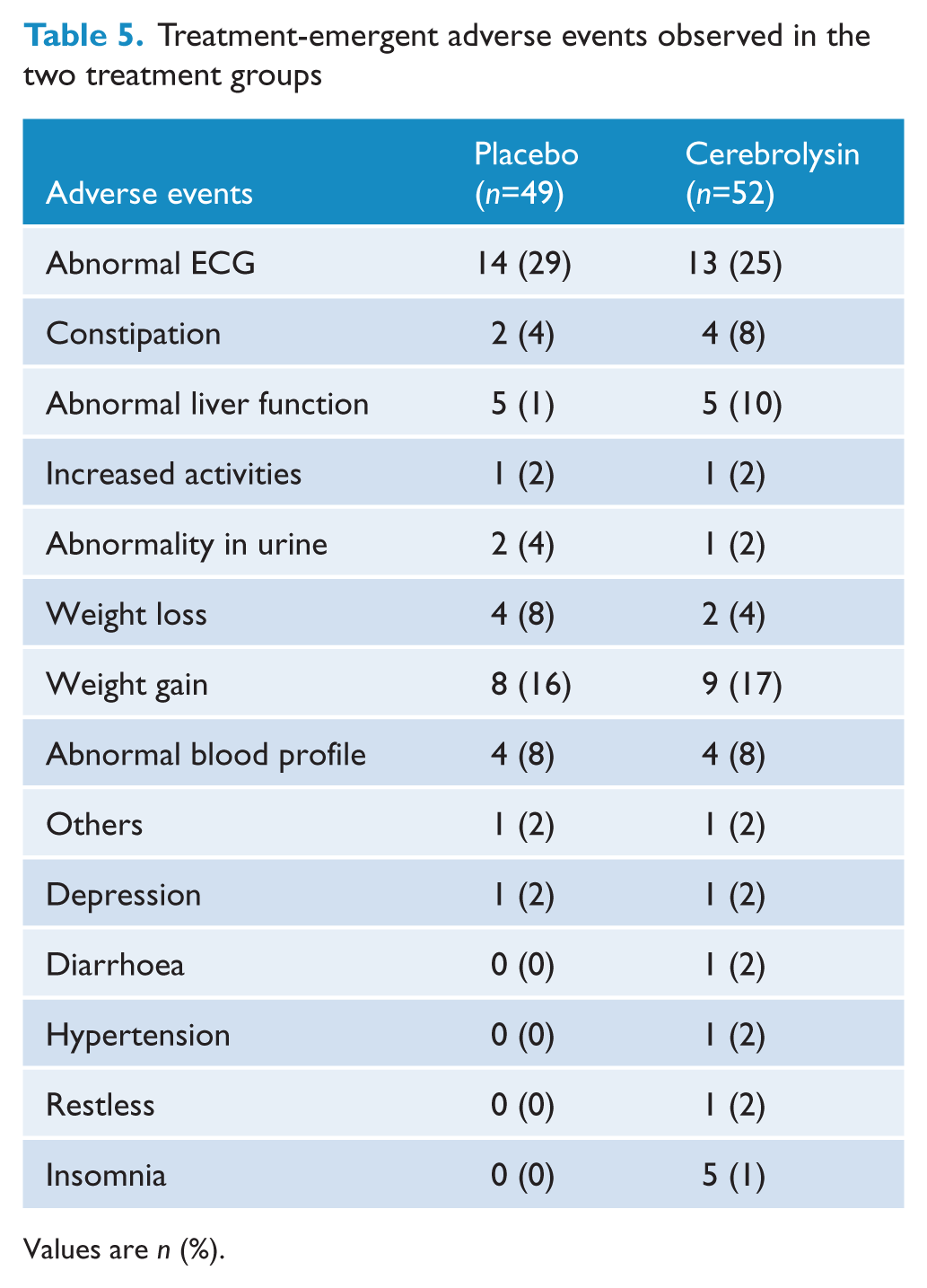
Values are n (%).
Discussion
To our knowledge, this is the first clinical study that has examined the efficacy and safety of cerebrolysin as an adjunctive medication in treating patients with negative-symptom-dominated schizophrenia. First, we found similar improvements between patients treated with cerebrolysin added to risperidone and those treated with risperidone alone in terms of PANSS total and negative scores, particularly from weeks 2 to 8. Consistent with this, the endpoint disease improvement as determined by CGI rating was also comparable between the two treatment groups. Second, however, and of particular interest, we found that patients treated with cerebrolysin showed significantly greater improvements in cognitive and memory function as assessed by the mWAIS and WMS.
Riperidone is an atypical antipsychotic drug used in treating patients with schizophrenia as a first-line medication in many countries. The therapeutic efficacy of risperidone has been proved in a series of clinical trials. It has been demonstrated that risperidone is effective in treating both positive as well as negative symptoms of schizophrenia (Riedel et al., 2007; Foussias and Remington, 2010; Tandon et al., 2010). In a double-blind study comparing the efficacy of risperidone and quetiapine in treating schizophrenia patients with predominantly negative symptoms, Riedel et al. (2007) found that both risperidone and quetiapine significantly improved the positive symptoms and negative symptoms as assessed by PANSS and SANS scores at 12 weeks. Fleischhacker et al. (2003) used long-acting injectable risperidone in patients who had previously received other antipsychotic drugs and found that risperidone significantly reduced the PANSS total, positive, and negative scores. In another study examining the efficacy of selegline added to risperidone in treating negative symptoms of schizophrenia, Amiri et al. (2008) reported that both risperidone in combination with selegline and risperidone alone significantly decreased scores of positive, negative, and general psychopathological symptoms in chronic schizophrenia patients over 12 weeks, indicating that risperidone alone was effective in treating negative symptoms of schizophrenia. In our study, patients in both the cerebrolysin and placebo groups received a stable dose of risperidone ranging 2−4 mg daily. As there was no significant difference in the extent of improvement in the PANSS total, negative. and positive scores between the two treatment groups, we conclude that augmentation with cerebrolysin 30 ml daily does not enhance the efficacy of risperidone in treating the negative symptom of schizophrenia.
Cognitive impairment is not uncommon in schizophrenia patients, especially in those with negative-symptom- dominated schizophrenia (Mäkinen et al., 2008; Galletly, 2009). To determine the effect of cerebrolysin on cognition, we used the mWAIS and WMS as secondary outcome measures. We found that scores on the mWAIS and WMS steadily increased in both treatment groups, but moreover there was a significantly greater increase in these measures in the cerebrolysin group from weeks 2 to 8. Previous studies have demonstrated that cerebrolysin improves cognitive function in patients with various neurological diseases. Xiao et al. (2000) found that cerebrolysin treatment led to improvement in cognition, psychiatric symptoms, and daily activities in patients with Alzheimer disease and vascular dementia. Another recent randomized double-blind controlled trial demonstrated that cerebrolysin significantly improved the global function in terms of cognition, initiation of activities of daily living, and neuropsychiatric symptoms at 10-, 30- and 60-ml doses in patients with moderate to moderately severe Alzheimer’s disease (Alvarez et al., 2011). It has also been reported that cerebrolysin injections of 1.0 ml every other day in combination with antipsychotics was effective in treating behavioural and cognitive disturbances in autism (Radzivil and Bashina, 2006). Recently, Jianu et al. (2010) have reported that cerebrolysin can lead to functional benefit in patients with Broca’s aphasia resulting from ischaemic strokes. In these studies, most patients suffered from specific organic brain injuries, with the therapeutic effects being attributed to the neuroprotective properties of cerebrolysin. At present, there have been no definitively demonstrated pathological changes in the brains of patients with schizophrenia. However, there is some evidence suggesting that a neurodegenerative process may underlie negative-symptom-dominated schizophrenia, i.e., significantly greater enlargement of ventricles, reduced thickness of the cortex, reduced activation as demonstrated by functional imaging techniques, and a relatively more devastating course of disease. Based on the significant findings in our study, in combination with that observed in other reports with a variety of other neuropsychiatric diseases (especially Alzheimer disease), we suggest that cerebrolysin as a adjunctive treatment may further improve (above and beyond any such effect due to risperidone) cognitive function in patients with negative-symptom-dominated schizophrenia. However, the mechanism of this apparent therapeutic effect is unclear. It should be noted that risperidone monotherapy has been reported to improve neurocognition in patients with schizophrenia (Keefe et al., 2007; Sergi et al., 2007).
Safety
No severe adverse events were found during the treatment period nor were there any significant differences in the frequency of adverse events between the two groups. This is consistent with previous studies of doses of cerebrolysin ranging from 10 to 60 ml daily (Bajenaru et al., 2010; Jianu et al., 2010; Alvarez et al., 2011). This suggests that 30 ml of cerebrolysin added to a flexible dosage of risperidone 2−4 mg per day is safe and well tolerated in patients with negative-symptom-dominated schizophrenia.
There were several limitations of our study that must be taken into account when interpreting these results. Firstly, our study enrolled only those patients who had more negative than positive symptoms, indicating that these patients may have been less responsive to antipsychotic medication. Further, whether cerebrolysin will benefit the symptoms of schizophrenia per se should be examined in larger populations with different clinical features. Secondly, this study was conducted over only over the relatively short time period of 8 weeks, which may not be sufficient to allow for the full therapeutic effect of cerebrolysin to emerge. In most previous studies of cerebrolysin, the length of treatment was 12−24 weeks (Vereshchagin et al., 1991; Xiao et al., 2000; Jianu et al., 2010; Alvarez et al., 2011) Although we observed an additional effect of cerebrolysin on cognition and memory from week 2 of the treatment, any effects on negative symptoms may require a longer time to become evident. Finally, only a fixed dose of cerebrolysin was used for all patients. Therapeutic effects may not have been achieved given the variability of pharmacokinetics among different individuals.
In conclusion, although cerebrolysin at the dosage of 30 ml daily did not augment the therapeutic efficacy of risperidone in terms of treating the positive and negative symptoms of schizophrenia in our study, we found that it may improve cognitive function in these patients, similar to that effect observed in the treatment of other neurological diseases. Further research should be conducted using different dosages of cerebrolysin over longer time frames.
Footnotes
Declaration of interest
The authors report no conflicts of interest. The authors alone are responsible for the content and writing of the paper.
Funding
This work was supported by the Stanley Medical Research Institute (grant no. 02I-005).
