Abstract
Schizophrenia involves multiple disturbances of psychological processes and runs an episodic or chronic course. It can cause major disruption to a person's and their family's well-being and lifestyle, and carries considerable risk of premature mortality [1]. Drug treatment of schizophrenia aims to reduce the severity, duration and frequency of psychotic symptoms, and the associated levels of disability and mortality. Because of the nature of the disease, its early onset and low remission rate, the lifetime costs are substantial [2]. Neuroleptic medication is a central element in the treatment of schizophrenia [3]. However, the use of such medications has been linked with disabling side-effects [1]. The longterm management of schizophrenia includes optimal medication, balancing the risk of relapse against the profile of side-effects, together with appropriate psychosocial interventions, rehabilitation and social support. Drugs such as clozapine, risperidone and olanzapine have raised expectations of improved patient outcomes with reduced side-effect profiles, particularly extrapyramidal symptoms, but at much higher cost. Clinical practice guidelines in Australia and overseas [4], [5] recommend the newer drugs as first-line treatment for patients with schizophrenia, relying on both expert opinion and limited trial evidence. However, the question arises as to whether the quality of life benefits of the newer drugs are sufficient to justify their additional cost relative to traditional neuroleptics. This analysis has been undertaken as part of the Assessing Cost Effectiveness – Mental Health (ACE-MH) project, funded by the Australian Department of Health and Ageing, Mental Health and Suicide Prevention Branch and the Department of Human Services, Mental Health Branch, Victoria. The project aim is to assess the options for change that could improve the effectiveness and efficiency of Australia's current mental health services by directing available resources toward ‘best practice’ cost-effective services.
Method
All analyses undertaken in ACE-MH adhere to a detailed economic protocol specifically designed for the study [6]. Briefly, the perspective is that of the health sector. Current practice is the comparator. The time horizon is the remaining lifetime of prevalent cases of schizophrenia and related conditions in Australia in the year 2000. Discounting at 3% for both costs and benefits is incorporated. Benefits are measured in disability-adjusted life years (DALYs), an outcome measure combining years of life lost to premature mortality (YLL) and years lived with disability (YLD).
Schizophrenia is defined by ICD-10 codes and includes paranoid, hebephrenic, catatonic, undifferentiated, schizoaffective, delusional disorder and other non-organic nonaffective psychotic disorders. We model the prevalent cases of schizophrenia by five-year age groups and sex, through a Markov model of annual cycles with relevant hazards and marginal costs, until the whole cohort has died or reached 100 years of age. The main benefits from the drug interventions come from differences in YLD calculated for each cycle by changing disability weights.
Interventions
We model eight alternative interventions for people diagnosed with schizophrenia. Target groups differ depending on the intervention. Risperidone (5 mg/day) is evaluated as a replacement for oral typical neuroleptics, for people with and without significant side-effects, and separately, in comparison to taking low-dose typicals (< 300 chlorpromozine equivalents). Olanzapine (15 mg/day) is evaluated as a replacement for oral typical neuroleptics, for people with and without significant side-effects, and for those taking risperidone. Clozapine (400 mg/day) is evaluated for treatment-resistant cases (those identified as having a chronic course of disorder with either little or clear deterioration), taking typical neuroleptics.
Current practice
The treatment regimens and disability level of the prevalent cases of schizophrenia are extracted from the database of the 1997/98 Low Prevalence (Psychotic) Disorders Study (LPDS). The average length of illness in those surveyed was 15 years (range 0–54 years). A detailed account of the design and methods of the LPDS has been published elsewhere [7]. The LPDS surveyed people in contact with specialist mental health services in Australia in 1997/98, and underrepresented community samples of people with schizophrenia managed solely by general practitioners (GPs). A small community study [8] estimated that the latter comprises about one in three people with schizophrenia in the population. We adjust for the undersampling by expanding the subsample of solely GP-managed schizophrenia cases from 3% to 33% of the total survey records. We estimate there were 37 000 Australians in the year 2000 affected by the condition [2], [7]. We assume 1.5% remission rate based on the median annualized remission rate reported in several longitudinal studies [9–13]. Of the adjusted LPDS respondents, 20% of male and 35% of female respondents were taking oral typical neuroleptics alone. Baseline adherence levels for people taking typical neuroleptics are modelled at 54%, determined from community studies [14], [15]. We derive a raised baseline general mortality risk for schizophrenia, retaining the age and sex pattern, from Australian Bureau of Statistics (ABS) mortality statistics with and without mention of schizophrenia for 1999, while constraining the relative risk of all-cause mortality for ages 15+, to 1.56, as reported from a meta-analysis [16].
The reader is referred to Haby et al. [6] for the full rationale, description and distribution of the disability scores of the LPDS respondents, which are used to indicate the baseline severity levels in the eligible population. In short, severity was measured by interview in the LPDS using six selected items from the WHO Disability Assessment Schedule (DAS), the Lancashire Quality of Life Profile and the Social Contact Questionnaire, plus two questions capturing the respondents' self-rating of satisfaction with independence and with life in general [17]. Assuming a linear relationship between the disability scores in the survey (ranging from 0 best to 16 worst) and extreme values of the four disability weights (DW) used in the Dutch Burden of Disease study [18], we derive a DW for each of the LPDS respondents (ranging from 0.21 for the mildest health state to 0.96 for the worst).
Assessment of benefit
Benefits are calculated using a two-stage process. The first stage involves the estimation of the health gain that could be attributed to each intervention using the disability-adjusted life year (DALY). The second stage involves the assessment of issues that either influence the degree of confidence that can be placed in the cost-effectiveness ratios or broader issues that need to be taken into account in decisionmaking about resource allocation in mental health.
Stage one: measurement of health gain
We scrutinized systematic reviews and more recent randomized controlled trials published in peer reviewed journals up to 2002, for evidence of differences in quality of life, short- and long-term sideeffects of medication, compliance change, symptom reduction and weight gain, under each of the eight intervention scenarios [19–26].
Symptom reduction
Consistent with other ACE-MH analyses, we summarize the impact of each intervention on severity of psychotic symptoms using the effect size (Hedge's g) [27], which relies on the reporting of continuous outcome measures. The outcome measures used to determine the pooled effect size differ by study and most commonly include the Brief Psychiatric Rating Scale (BPRS), Positive and Negative Syndrome Scale (PANSS) and Clinical Global Impression Scale (CGI). First we average effect size within studies, and then pool them across studies using the random effects method of Der Simonian and Laird [27]. The pooled effect size is translated into a DW change by each of the two methods referred to as the ‘Conversion Factor’ (CF) method and the ‘Survey Severity’ (SS) method described elsewhere [6]. The results of the two methods are combined and taken into account in the uncertainty analysis as a reflection of the lack of established practice in translating health outcomes relevant to cost-utility studies of schizophrenia.
Side-effects
The impact of the change in side-effects due to a switch in medication is estimated separately and translated into an additional change in DW. We determine the proportion of people reporting moderate or severe impairment in daily life due to medication in the LPDS from their response to the question: ‘How much has everyday life been affected by these medication-related side-effects?’ We calculate the mean DW based on the disability score as described above, in respondents on typical medication with no or mild (0.32), moderate (0.42) and severe (0.45) problems due to side-effects. We assume these differences in DW reflect the additional disability due to side-effects. The change in health benefit due to side-effects is then determined by the difference in the proportions reporting moderate and severe sideeffects on different drug regimens in the LPDS.
For clozapine, we model no difference in disability due to haematological side-effects as the mandatory frequent blood checks ensure patients are taken off the drug before more serious illness ensues.
Weight gain
We include an estimate of the mortality and morbidity risks associated with weight gain. From a number of studies of the impact of weight gain on ischaemic heart disease, ischaemic stroke, diabetes mellitus and colorectal cancer [28–31] we derive a relative risk of mortality or incidence per kilo of weight gain. The mean relative risks range from 1.03 for colon cancer, 1.04 for ischaemic heart disease and stroke, to 1.11 for diabetes. The disability from diseases associated with weight gain is extrapolated from the ratio of morbidity to mortality ratio for these conditions, in the 1996 Burden of Disease estimates [32].
Suicide
We found evidence for a reduction in suicide risk (by 50%) for patients on clozapine [33], but not for any of the other drugs.
Adherence
From differences in the published rates of leaving trials early, we determine the difference in adherence between drug regimens. If compliance levels improve with the intervention, we model that the comparator for the additional compliers, is no neuroleptic medication, resulting in a greater effect size (as assessed in trials of neuroleptics against placebo) and DW change. The modelled components of DW change and the effect sizes for each intervention appear in Table 1.
Determination of disability weight change by intervention
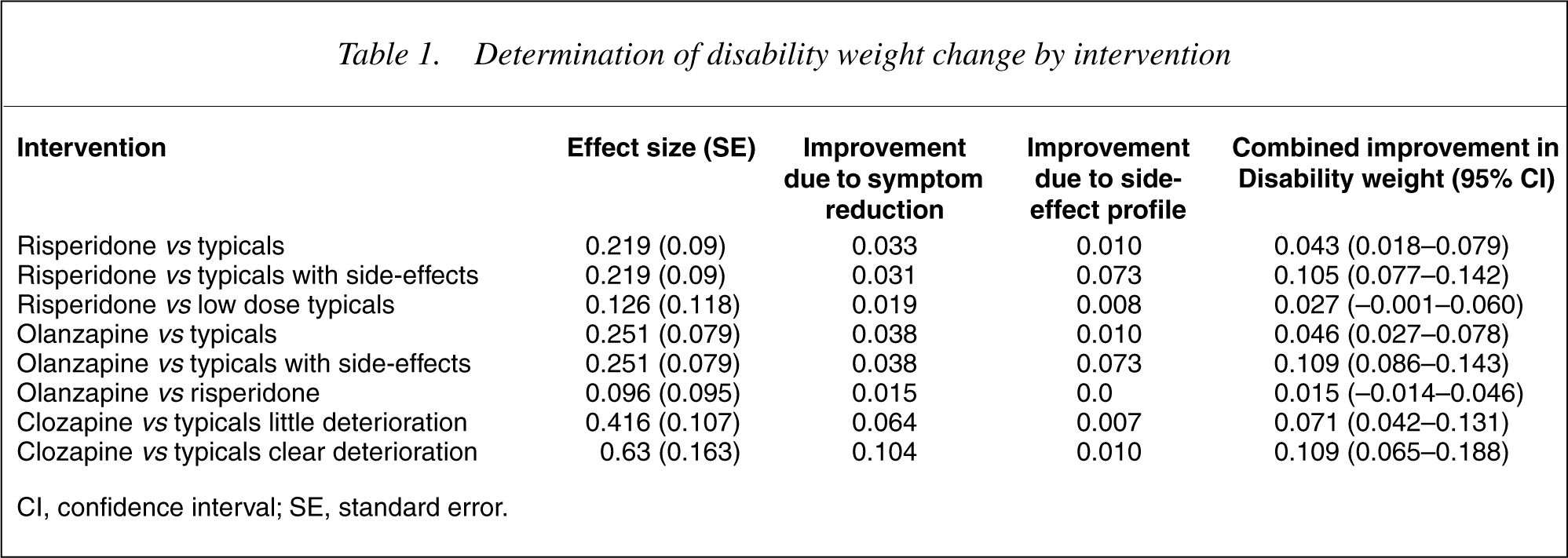
CI, confidence interval; SE, standard error.
Stage two: the second filter criteria
As with other ACE-MH analyses, apart from the cost-effectiveness of results, we take four second filter criteria into account: level of evidence; equity; acceptability to stakeholders; and feasibility after consultation with Steering Committee members [6].
Costs of intervention
The marginal cost of changing from one medication regimen to another is calculated using year 2000 prices. Cost-offsets are not factored into the analyses because we were unable to determine a fall in resource utilization associated with a reduction of illness severity due to a particular intervention. We investigated the potential costs of psychotic relapse versus the costs of people diagnosed with psychosis who do not relapse in Australia in the LPDS by regressing illness severity against resource utilization. None of the proxy variables for illness severity predicts resource utilization. SOFAS scores, DWs and diagnostic categories in the survey, each predict less than 5% of the variation in health service utilization, whether inpatient or outpatient care. The number of GP visits or contacts with mental health teams is assumed constant, except for the clozapine intervention which requires weekly blood tests for the first 18 weeks, and monthly thereafter. Unit cost data sources and assumptions are shown in Table 2. Costs that would have been incurred under current practice are subtracted from the intervention costs to obtain the incremental cost.
Summary of annual unit costs, data sources and assumptions
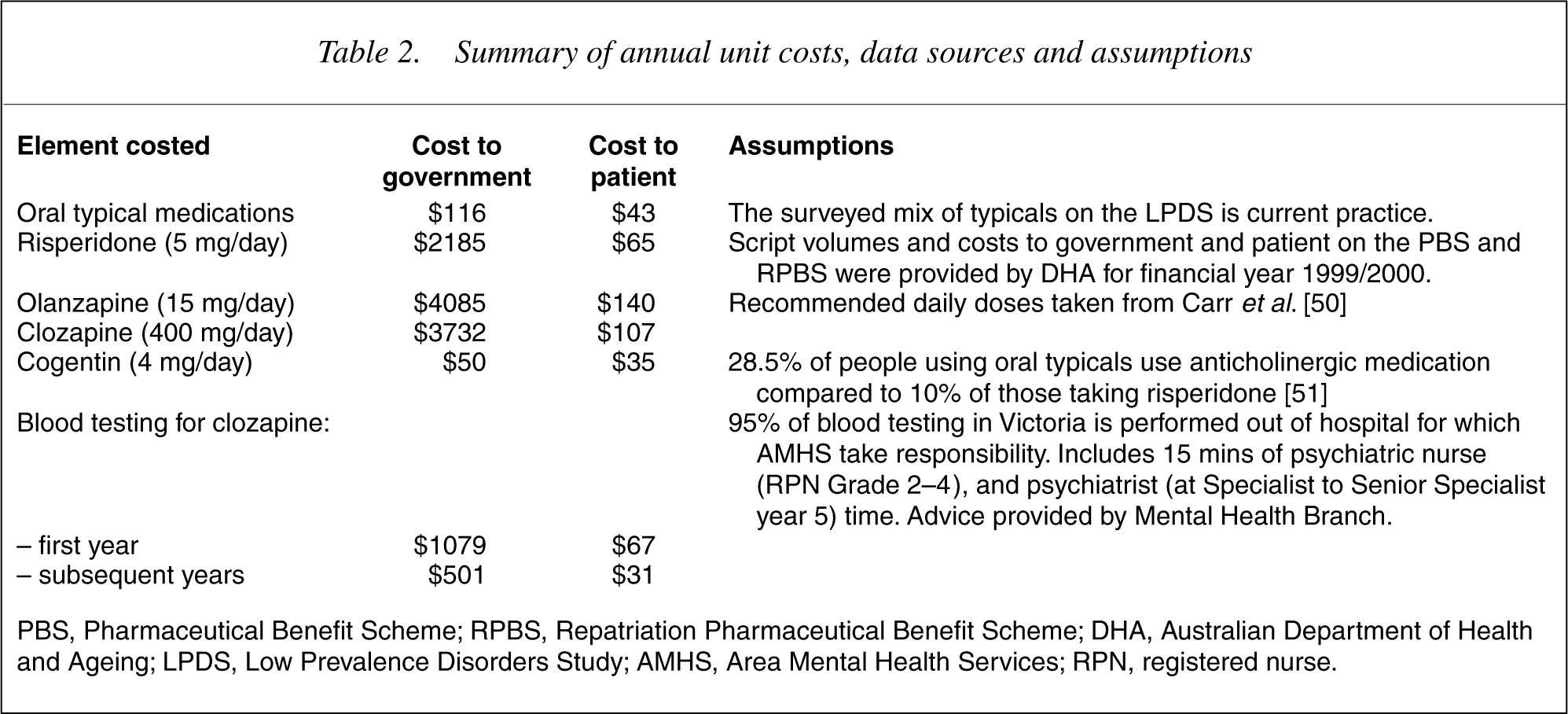
PBS, Pharmaceutical Benefit Scheme; RPBS, Repatriation Pharmaceutical Benefit Scheme; DHA, Australian Department of Health and Ageing; LPDS, Low Prevalence Disorders Study; AMHS, Area Mental Health Services; RPN, registered nurse.
Uncertainty analysis
We perform Monte Carlo simulation-modelling techniques using the @RISK Software (Palisade, 4.0) Newfield, NY and present uncertainty ranges as well as point estimates for benefits and cost-effectiveness ratios that reflect all the main sources of uncertainty in the calculations. Parameters and distributions for the uncertainty assumptions of one intervention as an example, appear in Table 3. The probability distributions around the input variables are based on: (i) standard errors quoted in, or calculated from, the literature; (ii) a range of parameter values quoted in, or calculated from, the literature; or (iii) expert advice. All results are presented to two significant digits.
Uncertainty parameters and sources used in the analysis specific to schizophrenia
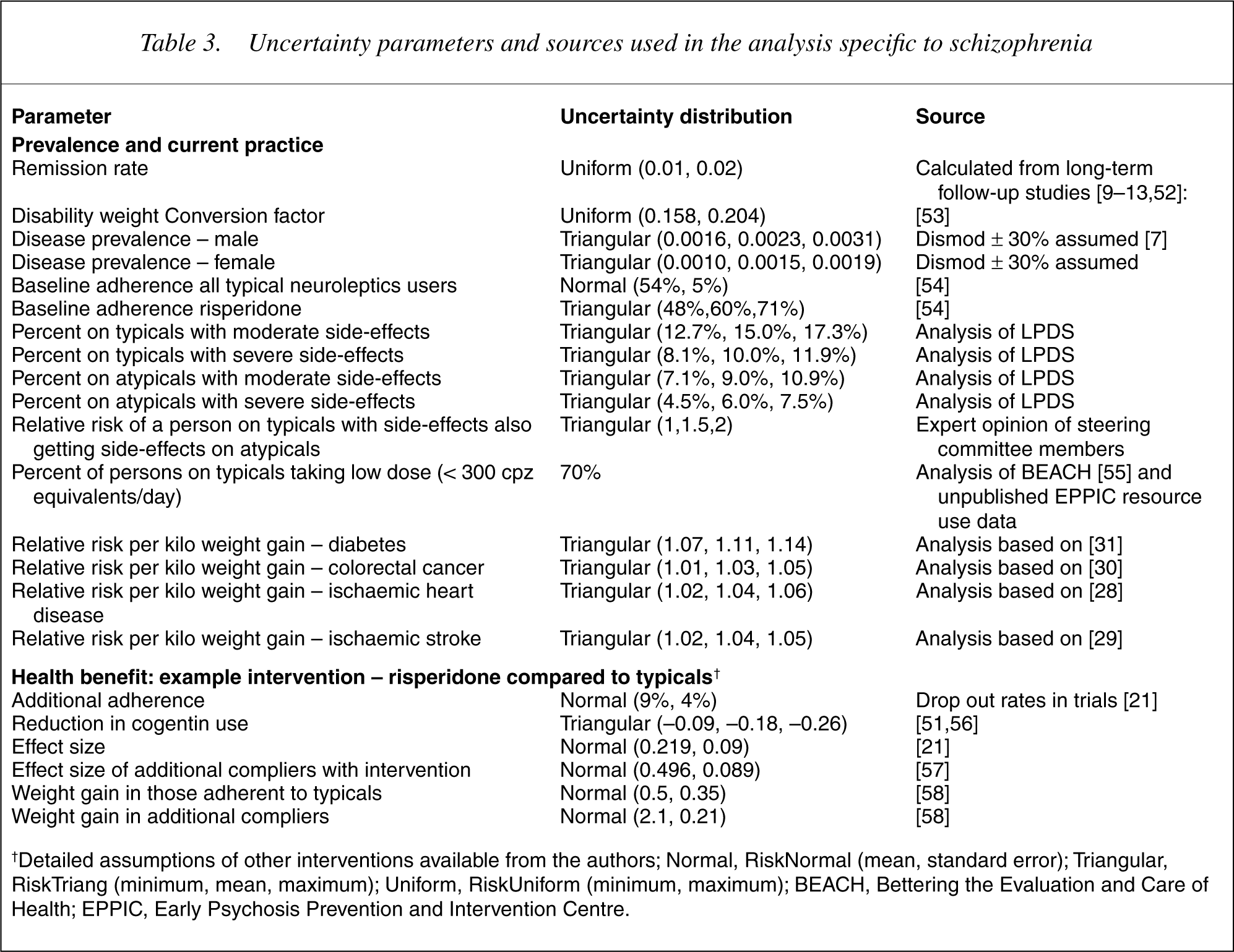
†Detailed assumptions of other interventions available from the authors; Normal, Risk Normal (mean, standard error); Triangular, Risk Triang (minimum, mean, maximum); Uniform, Risk Uniform (minimum, maximum); BEACH, Bettering the Evaluation and Care of Health; EPPIC, Early Psychosis Prevention and Intervention Centre.
Results
To change all people taking oral typicals to risperidone has an incremental cost-effectiveness ratio (ICER) of A$48 000 (Table 4). To change people taking oral typicals to olanzapine has an ICER of A$92 000. Limiting the change to only those people taking typicals with side-effects yields an ICER of A$20 000 for risperidone and A$38 000 for olanzapine. The ICER of changing those people currently taking low-dose typicals to risperidone is A$80 000. Changing the people currently taking risperidone to olanzapine has an ICER of A$160 000. Giving clozapine to people with the worst course of the disorder taking typicals, results in an ICER of A$42 000 and A$23 000 depending on their subclassification in the LPDS on levels of functional deterioration (little or clear), respectively. Consideration of the second stage filters for these interventions is shown in Table 5. Acceptability to stakeholders of a reduction in the use of atypicals emerged as the key concern in this analysis.
Summary of median (95% uncertainty ranges) of benefits, costs, and cost-effectiveness ratios for drug interventions in schizophrenia
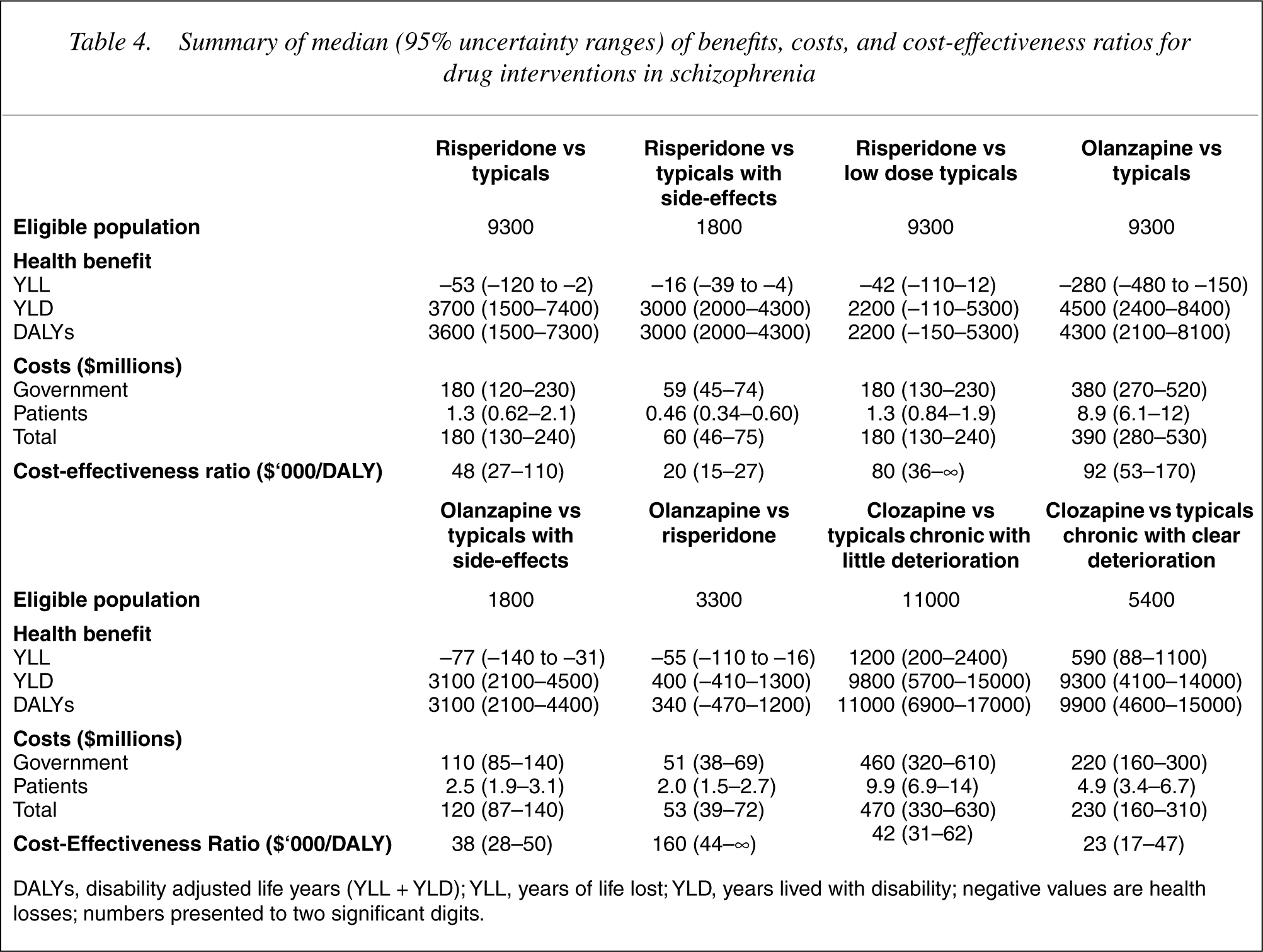
DALYs, disability adjusted life years (YLL + YLD); YLL, years of life lost; YLD, years lived with disability; negative values are health losses; numbers presented to two significant digits.
Consideration of second stage filters

Discussion
Replacing typical neuroleptics with risperidone falls just below the ‘value for money’ threshold of A$50 000 per DALY, which is used in all ACE-MH analyses. If the comparison is made against low doses of typical neuroleptics, a change to risperidone is less cost-effective at A$80 000. For those on typicals with significant sideeffects, risperidone is cost-effective at A$20 000. However, to extend risperidone from those patients on typicals experiencing side-effects to include all the remaining patients on typicals has an ICER of A$120 000. Results for olanzapine are less favourable. While providing olanzapine to patients with side-effects on typicals costs A$38 000 per DALY, the true decision-making issue is what the added benefits are relative to the greater cost of prescribing olanzapine rather than risperidone. As the additional benefits are very small and the cost difference great, olanzapine should never be chosen on cost-effectiveness grounds. Similarly, choosing to switch patients currently on risperidone to olanzapine is not cost-effective (A$120 000) and should not be considered unless there is a compelling clinical indication. The use of clozapine in preference to typical neuroleptics for patients with the more severe course of illness is a cost-effective intervention and this suggests that earlier identification and treatment of these patients with clozapine is warranted. The extensive uncertainty modelling which has provided ranges rather than point estimates for these cost-effectiveness ratios indicates that the above conclusions are justified despite the difficulties in estimation.
We have relied heavily on the evidence provided in Cochrane systematic reviews of drug interventions in schizophrenia because randomized controlled trials are the ‘gold standard’ in comparisons of relative drug efficacy. To this was added more recent trial evidence where available. Where evidence is missing we have chosen to highlight research gaps rather than use lower level evidence for efficacy. This is in contrast to recently developed clinical practice guidelines [4], [5].
The trials reviewed and included in this report suffered in several ways that limit their usefulness for this analysis. First, schizophrenia drug trials are characterized by very high drop-out rates that can severely limit their conclusions. Second, none of the included trials measure patient-rated quality of life. Clinician-rated quality of life measures were undertaken in some trials [34–36]. Hamilton [34] found significant between-treatment differences in favour of olanzapine in some subscales of the QLS instrument, but importantly not in the total scores. A non-randomised observational study [37] comparing the atypicals of interest in this paper to typicals, while showing some advantages in subjective tolerability on some scales for the atypicals, is prone to selection bias and would need to be repeated in well-designed RCTs before informing this type of analysis. The literature on quality of life in schizophrenia identifies that uncertainty remains surrounding the determinants of subjective satisfaction with treatment in schizophrenia [38]. We chose to model disability based on symptoms and treatment adverse effects for reasons of data availability. We recognize that the well-being of people with schizophrenia depends on more than a narrow health focus and our findings are limited by not considering other elements of quality of life such as safety, employment, financial support, family and social relationships.
Third, the included trials are largely conducted on patients with established schizophrenia. Emsley [39] is the only first-episode psychosis trial contributing to the analysis. We can therefore not generalize the findings to newly diagnosed or first-episode psychosis patients. Fourth, trials measuring the longer-term health status or suicide risk of people with schizophrenia on medication are lacking. Fifth, comparison of the levels of sideeffects from risperidone, olanzapine, clozapine or typical neuroleptic drugs in trials is compromised by the fact that many of these trials compare excessively high doses of typicals (e.g. up to 15–20 mg haloperidol) with more moderate doses of these atypicals. Trials report an odds ratio of 0.5 for serious side-effects comparing these atypicals with typicals (in other words, serious sideeffects are twice as common in those taking typicals). In contrast, the observational data we use from the LPDS indicates that there is only about a 70% higher proportion of persons reporting moderate or severe impairment in daily life due to side-effects in those on typicals compared to those on atypicals. Due to the high doses of typicals given to controls in trials we argue that the observational data from the LPDS may be closer to the true difference in side-effects, although we cannot exclude selection bias, recall bias and the potential impact of drug combinations in respondents. We have not considered the possible association with myocarditis and cardiomyopathy as a side-effect of clozapine use [40], [41]. Last, most of the trials exclude people with a history of substance abuse. Treatment outcome with antipsychotic drugs for patients who also abuse alcohol or illicit drugs is poor [42].
The use of a utility measure like the DALY in this analysis highlights the coarse nature of quality of life measurement in schizophrenia in burden of disease studies. The crude categorizations of the DALY DWs do not easily lend themselves to the measurement of change attributed to particular interventions. We have used two untested methods of translating evidence from trials into DALYs. We used the work of Gureje [43] to classify severity by a combination of quality of life indicators (two self-reported) from the survey. Clearly there is room for improvement, particularly in finding more accurate methods of evaluating severity and change in severity in a utility measure. A recent meta-analysis comparing the relative efficacy of typical and several atypical neuroleptics largely confirms our effect size estimates [44].
The main drivers of the differences in cost-effectiveness are the much higher costs of risperidone and olanzapine and the modest effect sizes regardless of the method of translating the effect size into a change in DALYs. The high cost of clozapine however, is combined with a clearly higher effect size.
When the patent period expires, the cost of a drug can change considerably. All the drugs considered in this modelling will come off patent within the next 10 years, but we have not incorporated a change in price to either government or patient. The Australian experience with clozapine suggests that price remains unchanged in generic drug forms in schizophrenia treatment. To the extent that the price of the drugs falls, the ICER results will become more favourable, but such lower ICER results should only inform policy if, and when, the price falls. Other cost-effectiveness studies have reported that the additional cost of the atypical neuroleptics, is offset by cost savings, due to fewer and shorter hospital admissions. However, the majority of these studies was funded by pharmaceutical companies, with a vested interest in a particular atypical drug (45–47] or data from studies that were either not randomised or subject to the previously discussed biases [48]. The cost of hospitalization has not been incorporated into the analysis for two main reasons. We could find no relationship between illness severity and resource use in the LPDS and the decision to hospitalize a person is influenced by many other variables (bed availability for example), apart from the patient characteristics of need.
Cost-effectiveness studies
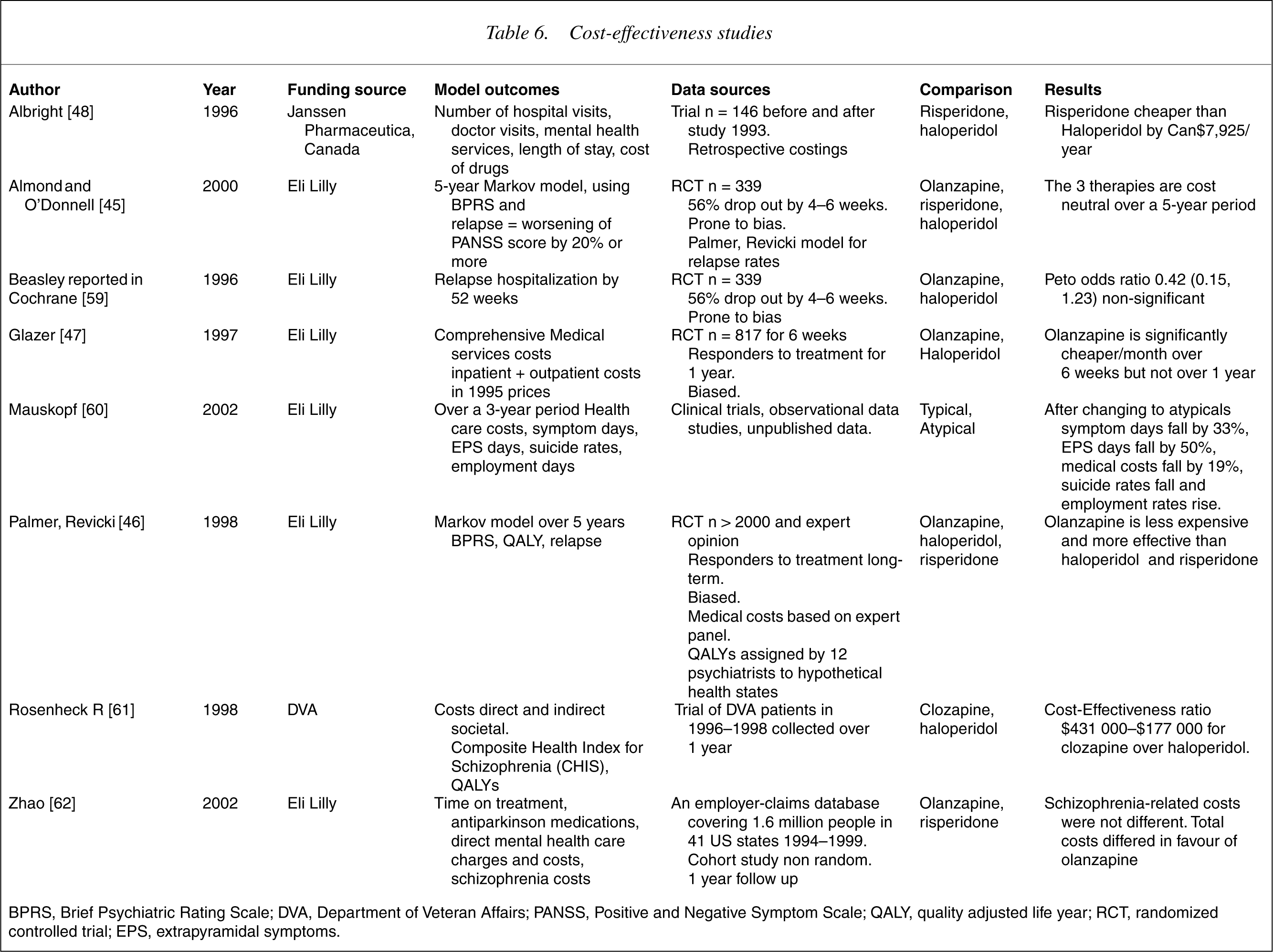
BPRS, Brief Psychiatric Rating Scale; DVA, Department of Veteran Affairs; PANSS, Positive and Negative Symptom Scale; QALY, quality adjusted life year; RCT, randomized controlled trial; EPS, extrapyramidal symptoms.
We base much of our analysis of current practice in Australia on the LPDS. Without the survey these analyses would have been very difficult. However, the survey is limited in a number of ways. It was not a representative community sample, it suffered a low response rate, it included no validated quality of life instrument, nor medication dosage data, duration of medication treatment, or compliance data. Further, people with schizophrenia who were not in contact with specialist mental health services were under-represented. We were able to overcome some of these shortcomings with reference to other smaller Australian community samples, giving greater weight to the GP-managed cases.
This work has drawn upon, but is also limited by the available research and the assumptions necessary to complete the work. Notwithstanding the many shortcomings in data sources and study methods, we conclude that we are able to make valid comparisons of cost-effectiveness results between drug interventions for schizophrenia. Comparisons of results with other ACE-MH interventions are more problematic and it is even more difficult to compare these results with cost per DALY estimates in other health areas where health gain is dominated by extension of life rather than quality of life. However, the absolute value of the ICERs is more influenced by the methodological problems encountered and the untried solutions adopted. For this reason it is advisable to read the range of values for each ICER along with the midpoint values, and to interpret them as indicative rather than definitive. The relative rankings of the drug interventions are clearly identified and would remain constant at any threshold value.
There are no major equity concerns about different choices of drug treatment, as all drugs are available on the Pharmaceutical Benefits Schedule (PBS). Minimal patient contributions occur since most people with schizophrenia have healthcare concession cards. There are more important issues with regards to feasibility and particularly acceptability to stakeholders. We conclude that the high cost of risperidone and very high cost of olanzapine do not weigh up against the relatively modest health benefits, except in the case of those experiencing moderate to severe side-effects on typicals. This conclusion may not conform with values held by clinicians, carers and people with schizophrenia. We may not have adequately estimated all benefits in our analyses and we acknowledge that there is likely to be more variation in the effect of drugs on individuals than we have captured by analysing the mean treatment effects reported in the literature. Hence, these findings cannot be imposed rigidly on clinical practice. However, adopting the A$50 000 threshold, and year 2000 drug prices, current practice seems far removed from cost-effective prescribing.
The market share of atypicals as a group, and the share of olanzapine within the group, has grown since 1997, the year of the LPDS survey. Reversing this trend may not be feasible as pharmaceutical companies are already starting to withdraw some of the typicals. However, it is very difficult to identify economic justification for the two-fold difference in price, minimal differences in efficacy and longer-term side-effect profiles of olanzapine compared to risperidone. Furthermore, the PBS may benefit from a review of drug pricing policies/practices after patent expiration with the high price of generic clozapine as an example.
To overcome weakness in trial evidence, longer-term non-industry-funded trials of existing and new atypical neuroleptics, in comparison with lower dose typical neuroleptics, in both first-episode and established schizophrenia, are desirable. Until then, clinical doubt continues to exist regarding alternative drug therapies. The development of clinical practice guidelines based on lower levels of evidence may inhibit the establishment of trial evidence [49]. Importantly, to enable costeffectiveness to be better evaluated, we recommend that outcome measurement in such studies include quality of life (both patient- and clinician-rated), social and economic functioning, drug tolerability, compliance, satisfaction and side-effects.
Footnotes
Acknowledgements
We thank members of the ACE–MH Steering Committee for their input to the project. The present report has drawn from data collected in the framework of the Collaborative Study on Low-Prevalence (Psychotic) Disorders, an epidemiological and clinical investigation which is part of the National Survey of Mental Health and Well-being, Australia 1997–1998. The views of the authors may not necessarily reflect those of the Low Prevalence (Psychotic) Disorders Study Group.
