Abstract
Objective:
Lithium and valproate are commonly used either in monotherapy or in combination with atypical antipsychotics in maintenance treatment of bipolar I disorder; however, their comparative efficacy is not well understood. This study aimed to compare the efficacy of valproate and lithium on mood stability either in monotherapy or in combination with atypical antipsychotics.
Methods:
We performed a post hoc analysis using data from a 52-week randomized double-blind, placebo-controlled trial, that recruited 159 patients with recently remitted mania during treatment with lithium or valproate and adjunctive atypical antipsychotic therapy. Patients were randomized to discontinue adjunctive atypical antipsychotic at 0, 24 or 52 weeks.
Results:
No significant differences in efficacy were observed between valproate and lithium (hazard ratio: 0.99; 95% confidence interval: [0.66, 1.48]) in time to any mood event. Valproate with 24 weeks of atypical antipsychotic was significantly superior to valproate monotherapy in preventing any mood relapse (hazard ratio: 0.46; 95% confidence interval: [0.22, 0.97]) while lithium with 24 weeks of atypical antipsychotic was superior to lithium monotherapy in preventing mania (hazard ratio: 0.27; 95% confidence interval: [0.09, 0.85]) but not depression.
Conclusion:
Overall, this study did not find significant differences in efficacy between the two mood-stabilizing agents when used as monotherapy or in combination with atypical antipsychotics. However, study design and small sample size might have precluded from detecting an effect if true difference in efficacy existed. Further head-to-head investigations with stratified designs are needed to evaluate maintenance therapies.
Introduction
Bipolar I disorder is a complex lifetime condition that affects 0.6% of the population worldwide (Merikangas et al., 2011) and is characterized by manic and depressive episodes with or without mixed features, sub-syndromal symptoms interspersed with some euthymic periods and high rates of comorbid conditions including anxiety and substance use disorders. Lithium and valproate are mood stabilizers commonly used in either monotherapy or in combination with atypical antipsychotics (AAs) for variable periods of time in the maintenance treatment of patients with bipolar I disorder to prevent relapse of mood episodes (Yatham et al., 2018). There is robust evidence for efficacy of lithium in prophylaxis of mood episodes (Miura et al., 2014). Despite a Cochrane review of various clinical trials revealing limited evidence for the efficacy of valproate in maintenance treatment of bipolar I disorder (Cipriani et al., 2013), valproate remains a commonly utilized agent for this indication in contemporary clinical settings. There is considerable evidence that maintenance therapy in combination with AAs is effective in reducing the risk of relapse or recurrence of mood episodes (Bowden et al., 2010; Buoli et al., 2014; Calabrese et al., 2017; Geddes et al., 2010; Macfadden et al., 2009; Marcus et al., 2011; Suppes et al., 2009, 2013; Vieta et al., 2008). The optimal duration of such strategy appears to be 24 weeks with olanzapine or risperidone adjunct to mood stabilizer (Yatham et al., 2016).
In real-world clinical practice, clinicians use lithium and valproate as monotherapy and in combination with AAs for variable periods of time in the maintenance treatment of bipolar I disorder to prevent relapse of mood episodes. However, there is limited data with regard to whether there are any differences in efficacy between lithium and valproate when given alone or in combination with AAs. Two randomized, double-blind, placebo-controlled trials of lithium and valproate monotherapy reported no differences in time to a mood episode between the two agents (Bowden et al., 2000; Calabrese et al., 2005). However, the Bipolar Affective disorder: Lithium/ANticonvulsant Evaluation (BALANCE) trial found that lithium was marginally superior to valproate monotherapy in preventing relapse of mood episodes (Geddes et al., 2010). Although several studies assessed the efficacy of lithium or valproate when given alone or in comparison to their combination with AAs (Bowden et al., 2010; Calabrese et al., 2017; Macfadden et al., 2009; Marcus et al., 2011; Suppes et al., 2009, 2013; Tohen et al., 2004; Vieta et al., 2008), few reported on their relative efficacy. Of the four studies that reported, two found no differences in efficacy (Calabrese et al., 2017; Tohen et al., 2004), while the other two studies reported results that favoured valproate (Marcus et al., 2011; Suppes et al., 2013). In a study of individuals with inadequate response to lithium or valproate monotherapy, use of aripiprazole with lithium or valproate for 52 weeks significantly increased time to relapse to any mood episode compared with mood stabilizer monotherapy (Marcus et al., 2011). In the lithium subgroup, time to any mood relapse favoured adjunctive aripiprazole treatment vs lithium monotherapy. However, the valproate subgroup did not show a significant difference between adjunct aripiprazole and valproate monotherapy. A pooled analysis (Suppes et al., 2013) of two long-term studies (Suppes et al., 2009; Vieta et al., 2008) comparing quetiapine vs placebo in combination with mood stabilizer (either lithium or divalproex) over a period of 104 weeks found, overall, that the quetiapine + mood stabilizer group significantly increased time to recurrence of any mood episode. No differences were noted in the risk of recurrence of mood event between quetiapine + lithium or quetiapine + divalproex. Interestingly, in patients treated with placebo and lithium, the risk of relapse of a manic episode was significantly greater than in those treated with placebo and divalproex. In participants with an index episode of mania, placebo + lithium treatment was associated with a significantly higher risk of relapse into mania than placebo + divalproex. However, recurrence risk of any mood or depressive episode was similar in the co-treated lithium and divalproex groups. These findings are inconsistent with the results of observational and nationwide register studies (Kessing et al., 2011) and their meta-analysis (Kessing et al., 2018) which found superiority of lithium monotherapy when compared with valproate for the prevention of mood events in patients with bipolar I disorder.
In summary, the current literature for bipolar disorder regarding the comparative efficacy of lithium or valproate when given alone or in combination with an AA in preventing relapse of mood episodes is inconsistent. Therefore, the objective of this post hoc analysis was to examine the comparative efficacy of lithium vs valproate in preventing relapse of mood episodes when given in monotherapy or in combination with olanzapine or risperidone – in maintenance treatment of bipolar I disorder. To this end, we used the Canadian Network for Mood and Anxiety Treatments (CANMAT) randomized trial which enrolled individuals with bipolar I disorder who were taking lithium or valproate in conjunction with placebo or an AA for a variable period of time during the 1-year study period.
Method
Participants
The CANMAT multicentre randomized double-blind placebo-controlled trial was approved by the UBC Clinical Research Ethics Board and detailed methods were previously published (Yatham et al., 2016). In summary, individuals were eligible for participation if they were aged 17 years or above, diagnosed with bipolar I disorder and treated within the previous 12 weeks for an acute manic or mixed episode with mood stabilizer (lithium or valproate) and AA (olanzapine or risperidone) combination. Participants were required to be in remission from manic or mixed episode for at least 2 weeks and up to 6 weeks based on a Clinical Global Impression Severity (CGI-S; Spearing et al., 1997) score of 2 or less for two consecutive weeks, or a Young Mania Rating Scale (YMRS; Young et al., 1978) score of 8 or less and Hamilton Rating Scale for Depression (HAM-D; Hamilton, 1960) 21-item score of 8 or less for two consecutive weeks. Individuals with a history of other axis I disorders and comorbid substance abuse were eligible. Individuals taking other psychotropic medications, except benzodiazepines and anti-Parkinsonian medications, were deemed ineligible. Participants had to be capable of providing written informed consent to be eligible for the study.
Study procedures
During the 52-week double-blind phase, all participants continued taking lithium or valproate. Participants were randomized to discontinue olanzapine or risperidone at study entry (and substituted with placebo for 52 weeks), at 24 weeks (and substituted with placebo for 24 weeks) or continued for the full 52 weeks. Thus, there were three arms in the study: (1) participants taking lithium or valproate with placebo for 52 weeks (‘0-week’ arm), (2) participants taking lithium or valproate and continuing AA for 24 weeks, with the AA substituted by placebo for the remaining 24 weeks (‘24-week’ arm) and (3) participants taking lithium or valproate with olanzapine or risperidone for 52 weeks (‘52-week’ arm). Participants continued the same mood stabilizer and AA taken at study entry, doses and serum levels of each were maintained within therapeutic ranges (0.6–1.2 mmol/L for lithium and 350–830 μmol/L for valproate). Medication dose ranges for the AAs were 1–6 mg/day for risperidone and 5–25 mg/day for olanzapine.
Outcomes
For this post hoc analysis, we compared the outcomes between patients taking lithium and those taking valproate using the primary outcome measure of time to any mood episode defined by any of the following: (1) YMRS score of 15 or greater, (2) HAM-D 21-item score of 15 or greater or HAM-D suicide item score of 3 or greater, (3) CGI-S score of 3 or greater, (4) hospitalization for treatment of mood symptoms or (5) suicide or suicide attempt. Key secondary outcomes included time to a manic episode and time to a depressive episode. Observation and measurement of adverse events were also conducted at baseline and monthly follow-up visits, via biochemical markers, self-report rating scales and clinical observation.
Statistical analysis
Intention-to-treat analyses were conducted on all randomized participants. Individuals who did not experience the primary outcome were censored at time of last follow-up visit. Kaplan–Meier cumulative incidence curves were used to summarize the time to any mood episodes for patients on valproate and lithium. Cox proportional hazards model with adjustment for the AA drug and duration was conducted to compare the treatment effect of valproate and lithium. Similar analyses were performed to compare the two treatment groups on the effect of time to a manic episode and time to a depressive episode. Furthermore, subgroup analyses were conducted for valproate and lithium. The SAS 9.4 statistical package was used for all statistical analyses. Within each treatment group, the three treatment arms (discontinuation of AA at 0, 24 and 52 weeks) were summarized and pairwise compared in terms of time to any mood episode, a manic episode and a depressive episode with use of Kaplan–Meier method and Cox proportional hazards model with adjustment for the AA drug.
Results
Patient demographics and disposition
In total, 74 participants treated with valproate and 85 on lithium were included in the analysis. The baseline characteristics of participants on lithium and valproate are shown in Table 1. There were no significant differences between the two groups on any of the demographic or clinical characteristics except for alcohol or substance use comorbidity, which was present in 28% of lithium users compared with 51% of valproate users (χ2; p = 0.005). Notably, there were no significant differences between valproate and lithium groups in terms of duration of disorder, psychiatric comorbidities, age of onset, type of mood episodes, number of previous episodes or past suicidal behaviours. As well, there were no significant differences between lithium- and valproate-treated patients with respect to dosage of adjunct AA.
Baseline patient characteristics.
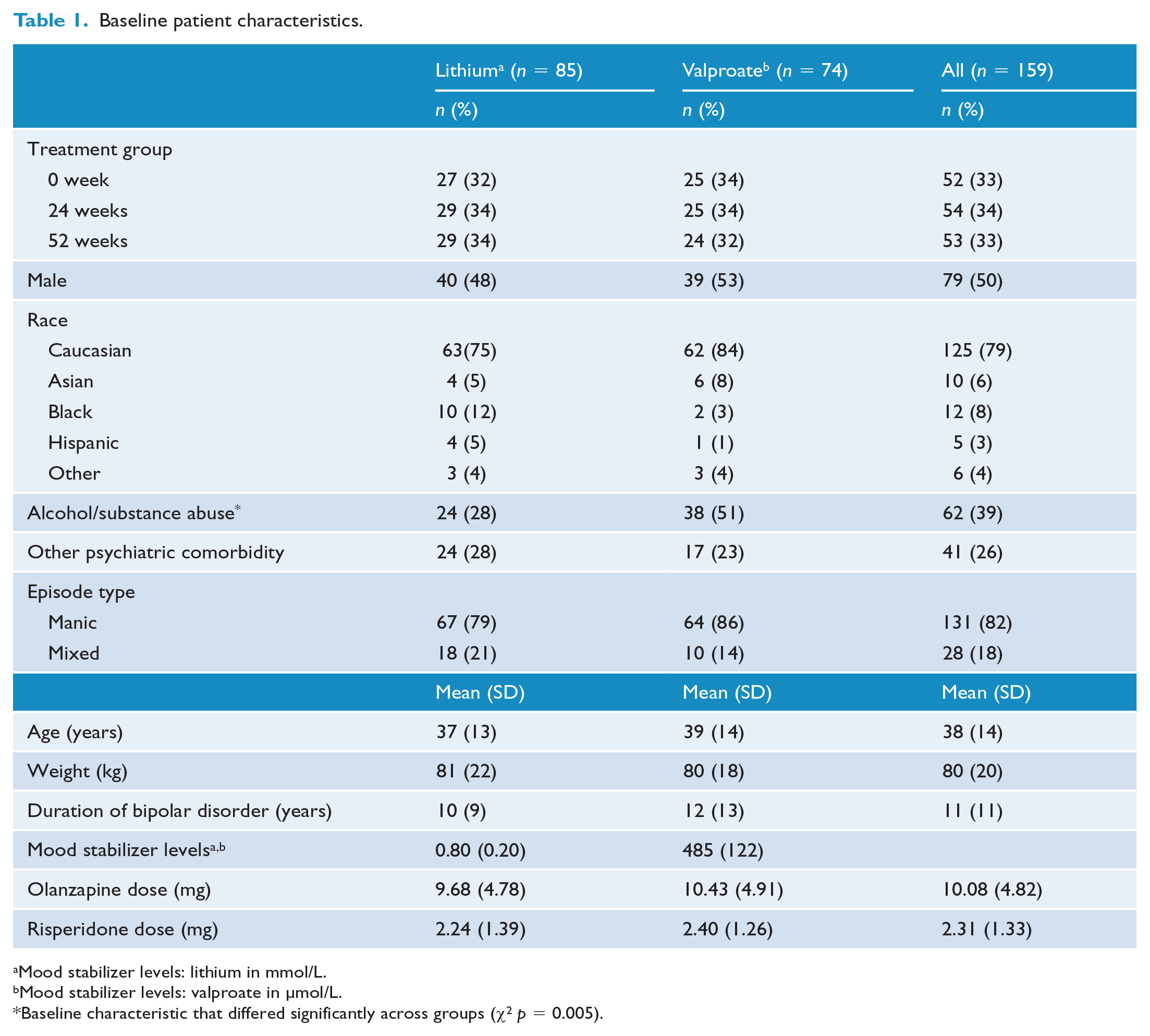
Mood stabilizer levels: lithium in mmol/L.
Mood stabilizer levels: valproate in µmol/L.
Baseline characteristic that differed significantly across groups (χ2 p = 0.005).
Primary and secondary outcomes
Valproate vs lithium – overall results
The median follow-up time was 13.6 weeks (95% confidence interval [CI]: [8.00, 24.14]) for individuals treated with valproate and 15.7 (95% CI: [11.14, 26.71]) weeks for patients on lithium; however, this discrepancy was not statistically significant. In total, 32 (42%) patients discontinued the study in the valproate group compared with 30 (35%) patients in the lithium group.
The Kaplan–Meier cumulative incidence curves for time to any mood episode (Figure 1(a)), manic episode (Figure 1(b)) and depressive episode (Figure 1(c)) over the study period for valproate and lithium groups were similar and do not suggest any significant differences between the two agents. The estimated 52-week event rates in the valproate and lithium groups were as follows: 0.69 (95% CI [0.57, 0.81]) and 0.76 [0.65, 0.86], respectively, for time to any mood episode; 0.24 [0.08, 0.41] and 0.44 [0.27, 0.60], respectively, for time to manic episode and 0.59 [0.46, 0.73] and 0.56 [0.43, 0.70], respectively, for time to depressive episode.
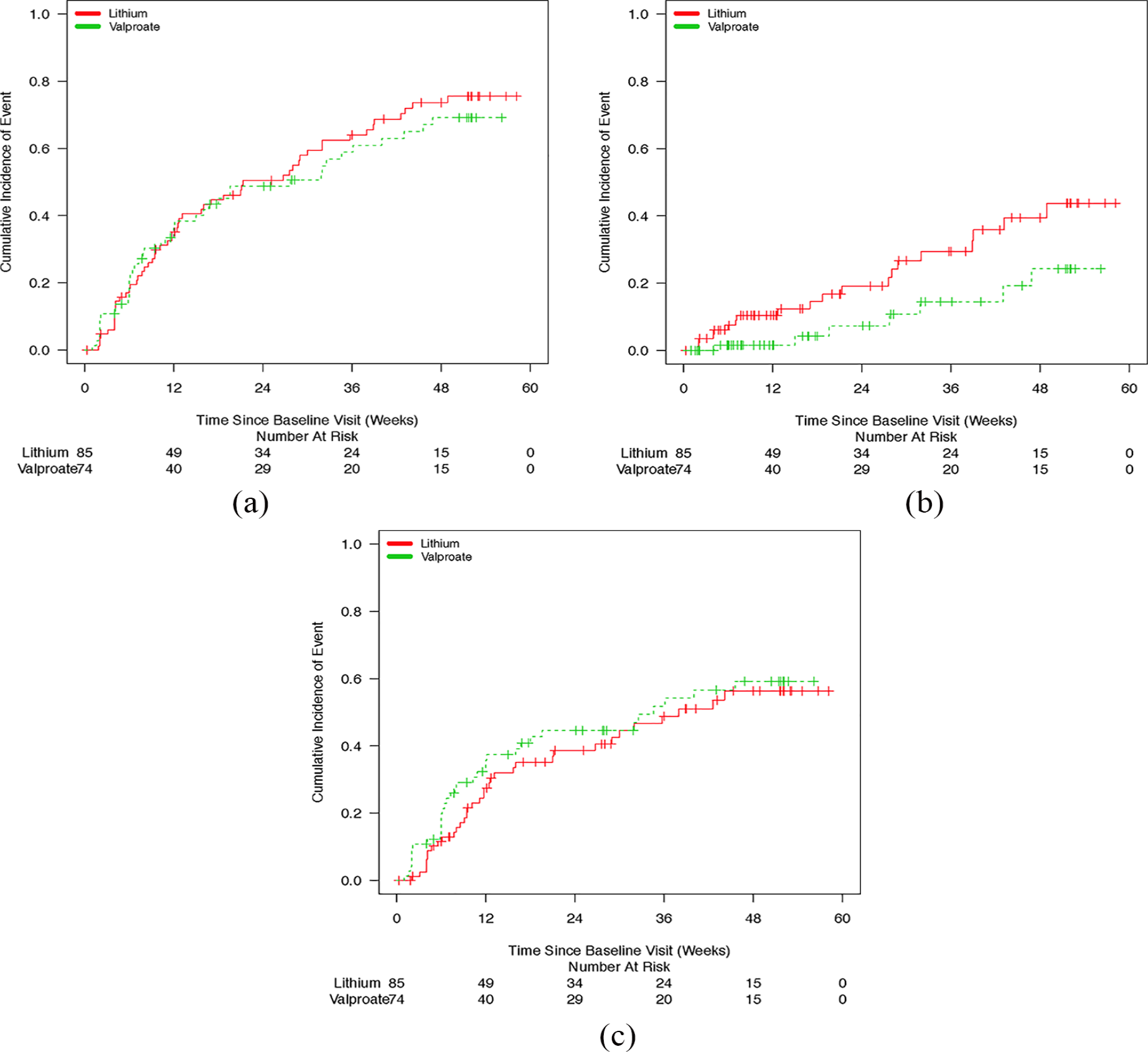
(a) Kaplan–Meier curves depicting time to any mood episode, (b) Kaplan–Meier curves depicting time to manic episode and (c) Kaplan–Meier curves depicting time to depressive episode.
In the adjusted Cox analysis, no differences were noted between valproate and lithium for time to any mood episode (hazard ratio [HR]: 0.99; 95% CI: [0.66, 1.48]; p = 0.95), a depressive episode (HR: 1.31; 95% CI: [0.82, 2.11]; p = 0.26) or a manic episode (HR: 0.42; 95% CI: [0.18, 1.01]; p = 0.05).
Valproate and lithium – subgroup results
For individuals on valproate, 68% of people in the 0-week arm had mood relapses (of which 82.4% were depressive and 17.6% were manic episodes), compared with 48% of people in the 24-week arm (of which 83.3% were depressive and 16.7% were manic episodes) and 54.2% of people in the 52-week arm (of which 84.6% were depressive and 15.4% were manic episodes). For individuals on lithium, 81.5% of people in the 0-week arm had mood relapses (of which 50% were depressive and 50% were manic episodes), compared with 58.6% of people in the 24-week arm (of which 76.5% were depressive and 23.5% were manic episodes) and 55.2% of people in the 52-week arm (of which 68.7% were depressive and 31.3% were manic episodes).
The Kaplan–Meier cumulative incidence curves for time to any mood episode, manic episode and depressive episode are depicted for valproate in Figure 2(a)–(c) and for lithium in Figure 3(a)–(c). The estimated 52-week event rates for any mood episode in the 52-week, 24-week and 0-week arms were 0.66 (95% CI: [0.44, 0.89]), 0.58 [0.36, 0.80] and 0.83 [0.66, 1.00] for valproate and 0.64 [0.45, 0.84], 0.72 [0.53, 0.92] and 0.91 [0.79, 1.00], respectively, for lithium.
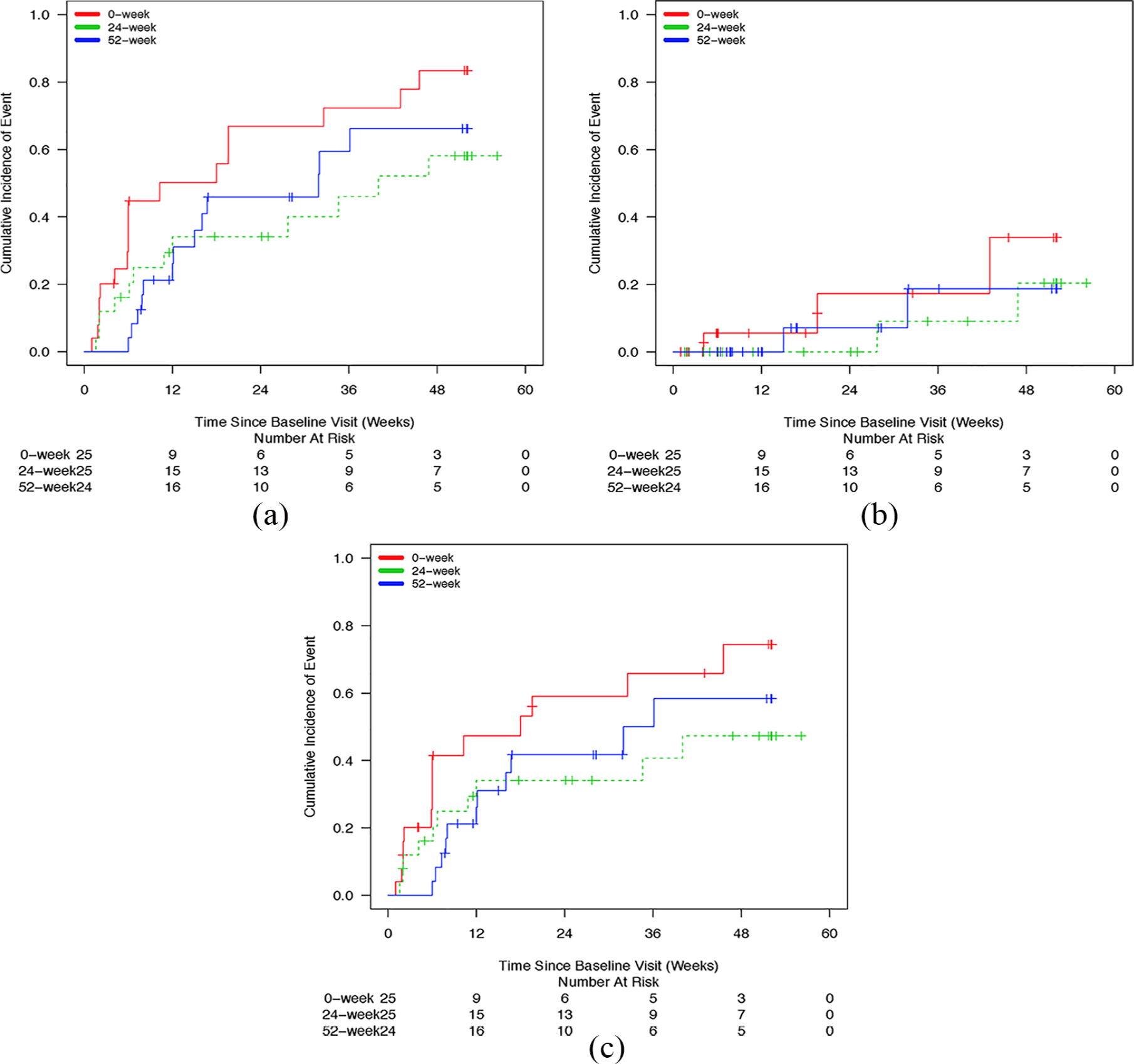
(a) Kaplan–Meier curves depicting time to any mood episode for patients on valproate, (b) Kaplan–Meier curves depicting time to manic episode for patients on valproate and (c) Kaplan–Meier curves depicting time to depressive episode for patients on valproate.
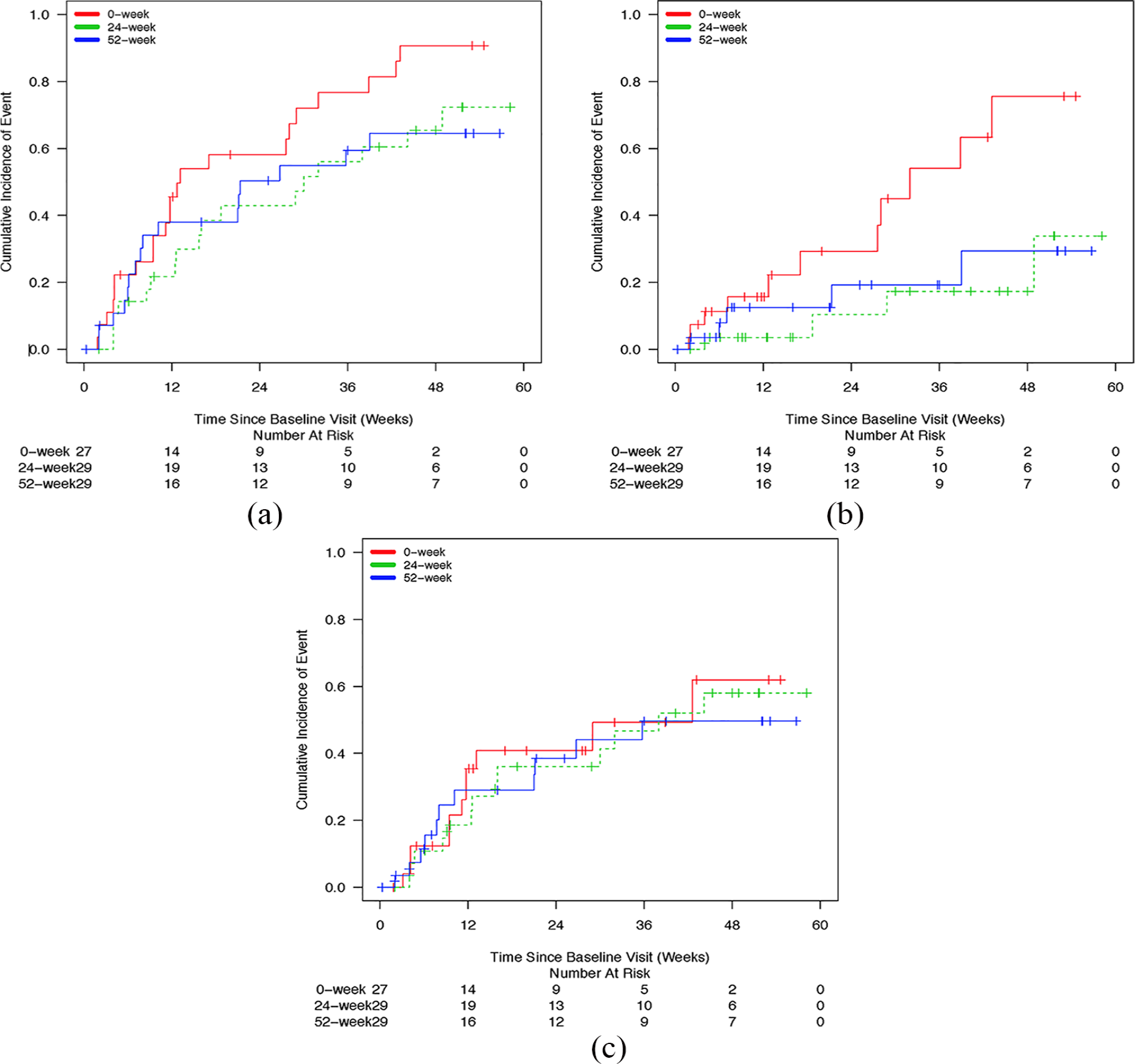
(a) Kaplan–Meier curves depicting time to any mood episode for patients on lithium, (b) Kaplan–Meier curves depicting time to manic episode for patients on lithium and (c) Kaplan–Meier curves depicting time to depressive episode for patients on lithium.
Valproate subgroup
For the valproate subgroup, the time to any mood episode was significantly longer for the 24-week arm relative to the 0-week arm (HR: 0.46; 95% CI: [0.22, 0.97]; p = 0.04), but not for the 52-week arm relative to the 0-week arm (HR: 0.54; 95% CI: [0.26, 1.13]; p = 0.10). No significant difference was observed between the 52-week arm and the 24-week arm (HR: 1.18; 95% CI: [0.53, 2.61]; p = 0.68).
Of the 42 participants (56.8%) in the valproate group that had a mood event, 16.7% of these met criteria for a manic event and 83.3% met criteria for a depressive event. No significant differences were found between valproate 24-, 52- and 0-week treatment arms in terms of risk of either manic (HR for 24 weeks vs 0 week: 0.42, 95% CI: [0.07, 2.56], p = 0.34; HR for 52 weeks vs 0 week: 0.44, 95% CI: [0.07, 2.67], p = 0.37; HR for 52 weeks vs 24 weeks: 1.06, 95% CI: [0.15, 7.59], p = 0.96) or depressive episodes (HR for 24 weeks vs 0 week: 0.47, 95% CI: [0.21, 1.05], p = 0.07; HR for 52 weeks vs 0 week: 0.56, 95% CI: [0.25, 1.24], p = 0.15; HR for 52 weeks vs 24 weeks: 1.20, 95% CI: [0.51, 2.85], p = 0.68).
Lithium subgroup
Within the lithium subgroup, the adjusted Cox analysis indicated that the HR for time to any mood episode was 0.59 for the 24-week arm relative to the 0-week arm (95% CI: [0.31, 1.12]; p = 0.11) and was 0.71 for the 52-week arm relative to the 0-week arm (95% CI: 0.37, 1.37; p = 0.31). The HR for the 52-week arm relative to the 24-week arm was 1.20 (95% CI: [0.60, 2.39]; p = 0.61).
Of 55 participants (67.1%) in the lithium subgroup that had a mood event, 36.4% met criteria for a manic event and 63.6% for a depressive event. The time to a manic episode was significantly longer in lithium users randomized to the 24-week treatment arm compared with participants randomized to the 0-week arm (HR: 0.27; 95% CI: [0.09, 0.85]; p = 0.03). There was also a trend for decreased HR comparing the 52- and 0-week arms; however, this was not statistically significant (HR: 0.43; 95% CI: [0.15, 1.29]; p = 0.13). There was no significant difference between lithium 52- and 24-week treatment arms in terms of time to manic episode (HR: 1.60, 95% CI: [0.42, 6.05], p = 0.49). There were also no significant differences noted between lithium 24- and 52-week treatment arms and 0 week on reducing risk of depressive episode relapse (HR for 24 weeks vs 0 week: 0.93, 95% CI: [0.41, 2.07], p = 0.85; HR for 52 weeks vs 0 week: 0.99, 95% CI: [0.42, 2.32], p = 0.98; HR for 52 weeks vs 24 weeks: 1.07, 95% CI: [0.48, 2.41], p = 0.87).
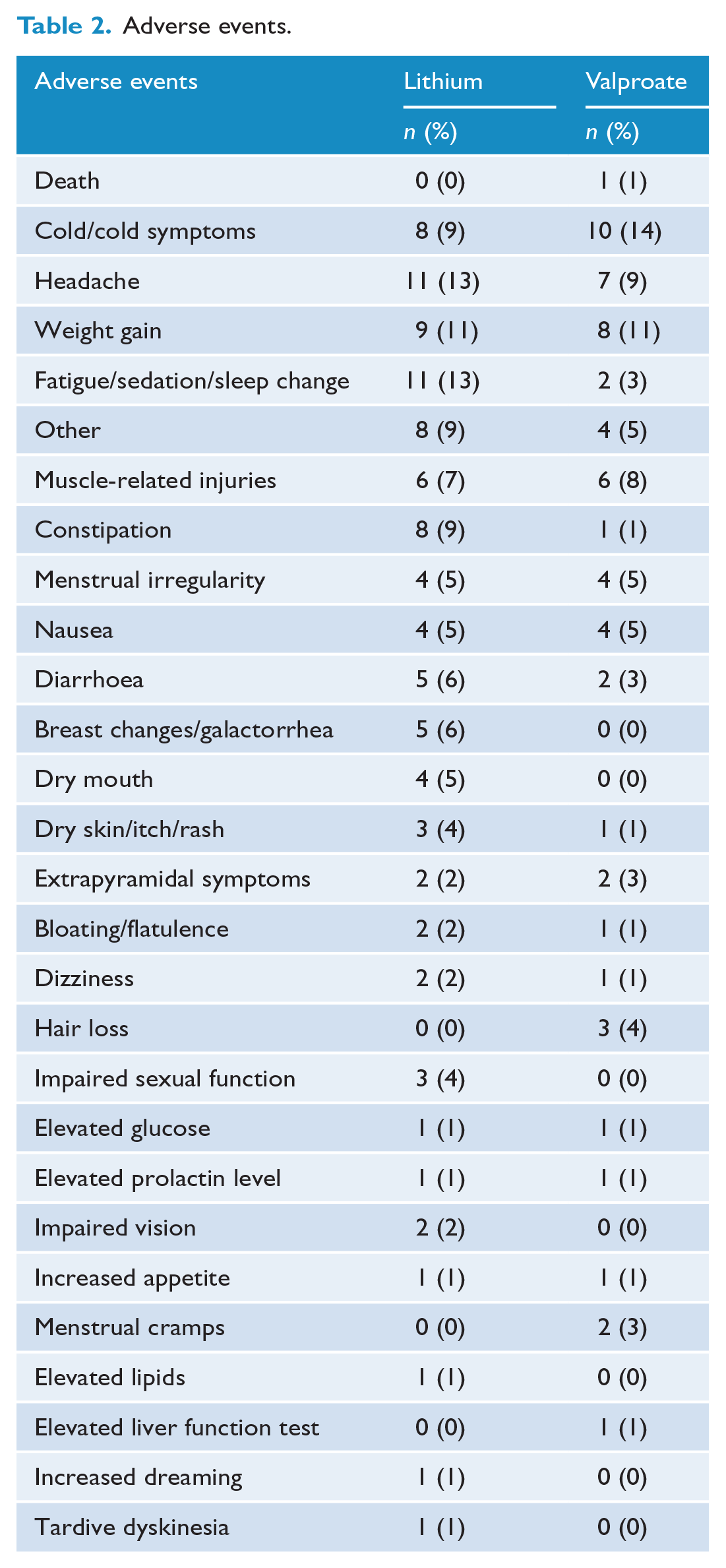
Adverse events
Valproate users appeared to show higher rates of cold symptoms, hair loss and menstrual cramps. Lithium users showed higher rates of headache, fatigue, constipation, diarrhoea, breast changes, dry mouth, skin changes and impaired sexual function. Rates of other adverse events were similar across both groups (Table 2). The single death in the valproate group was determined to be unrelated to study medication by the site investigator. Refer to the original CANMAT trial (Yatham et al., 2016) for detailed description of adverse events and comparison between the three treatment arms.
Adverse events.
Discussion
Study findings
This post hoc analysis compared the efficacy of lithium and valproate alone or in combination with AAs olanzapine or risperidone in maintenance of mood stability following acute manic or mixed episode remission in bipolar I disorder. The main findings of the study are as follows: (1) there were no significant differences on the primary efficacy measure of time to any mood episode between the two mood stabilizer groups, (2) there were also no significant differences in key secondary efficacy measures of time to a manic episode and time to a depressive episode between valproate and lithium groups and (3) analyses within each subgroup showed that time to any mood episode was significantly longer in the 24-week arm relative to the 0-week arm for valproate, while for lithium, time to a manic episode was significantly longer in the 24-week arm relative to the 0-week arm.
The HR for time to any mood episode (0.99) and time to a depressive episode (1.31) for valproate relative to lithium are closer to 1. Thus, it is unlikely that a type II error accounts for a lack of significant difference between the two groups. The results of this study showing no differences in efficacy in time to any mood episode between valproate and lithium are consistent with the data from two previous double-blind trials that compared these agents (Bowden et al., 2000; Calabrese et al., 2005). However, the HR for the time to a manic episode for valproate relative to lithium was 0.42 (CI: [0.18, 1.01]; p = 0.05). Given this, one could argue that this study may have been underpowered to detect a difference in efficacy between valproate and lithium in preventing a manic episode.
Valproate subgroup analysis showed that time to any mood episode was longer in the 24-week arm relative to the 0-week arm, suggesting the additional benefit of an AA in maintenance of mood stability. Although non-significant, the HRs for preventing mania of 0.42 and preventing depression of 0.47 suggest potential benefit of the AA addition for both poles of bipolar I disorder; however, larger confidence intervals raise questions about the validity of this assumption.
The subgroup analysis showed that the time to a manic episode was significantly longer in lithium users randomized to the 24-week treatment arm compared with those randomized to the 0-week arm on lithium monotherapy. This finding is consistent with the notion that adjunctive AA therapy provides additional benefit in preventing mania in those being treated with lithium for maintenance of mood stability. Interestingly, no significant differences were noted between 24- and 52-week lithium treatment groups and the 0-week group on reducing risk of depressive episode (HR = 0.93 for 24 weeks vs 0 week and HR = 0.99 for 52 weeks vs 0 week) suggesting that adding an AA does not provide additional benefit in preventing depressive relapses for participants on lithium.
Valproate users showed higher rates of cold symptoms, hair loss and menstrual cramps whereas lithium users appeared to have more side effects and higher rates of headache, fatigue, constipation, diarrhoea, breast changes, dry mouth, skin changes and impaired sexual function. Rates of other adverse events were similar between lithium and valproate groups. These findings are consistent with previously known safety profiles of these medications (McKnight et al., 2012).
Strengths and limitations
Strengths of this study include large sample size, the enrolment of a clinically representative patient population and the systematic blinded outcome assessment using specified criteria to define relapse of mood episodes. Furthermore, given that clinicians use lithium or valproate monotherapy and that AAs are discontinued at different time points based on efficacy, tolerability, personal practice preferences and so on, the design of this study mirrors real-world clinical practice. As well, subgroup analyses benefitted from the randomized double-blind controlled design and the investigation of outcomes between three treatment arms in which adjunct AA treatment were discontinued at different time points.
However, there are several limitations to be considered. This is a post hoc analysis of a study that was not originally designed to test the comparative efficacy of lithium vs valproate. Furthermore, the mood stabilizer assignment was not random and was chosen by clinicians prior to study entry, perhaps based on their previous experience, clinical features of a manic episode, patients’ previous history of treatment response or clinicians prescribing bias. In addition, the study was not powered sufficiently for analysis of mood stabilizer subgroups. This study only investigated lithium, valproate, olanzapine and risperidone; therefore, results are not generalizable to other medications used in combination therapy. As well, the rates of substance use comorbidity were significantly different between the two groups with 28% of lithium users meeting criteria for any substance use disorder compared with 51% of valproate users (χ2 p = 0.005). This may reflect clinicians’ preference for valproate in patients with substance use comorbidity as there is some evidence that valproate may have better efficacy in such population (Salloum et al., 2005). Sensitivity analyses with additional adjustment for substance use in the models were conducted and the conclusions were still the same. Regardless, given that the hazard ratio was close to 1 in the primary outcome of time to mood episode, it is unlikely this comorbidity would have affected the estimate of true differences in efficacy between the two groups. Results for secondary outcome measures are limited by small sample size and adjustment of confounding variables included in the Cox proportional hazard model. Larger sample sizes would have provided more definitive conclusions between lithium and valproate subgroups, especially for determining secondary outcomes such as time to manic and/or depressive episode. Despite these limitations, several findings are clinically relevant and can impact management decisions of bipolar I disorder by providing insight into which duration and combination of mood stabilizer and antipsychotic maintenance therapy best protects mood stability.
Conclusion
In conclusion, the findings of this study revealed no significant differences in efficacy between valproate and lithium either as monotherapy or combined with olanzapine or risperidone following remission of a manic episode. However, the study design was not fully appropriate to test relative efficacy given that the patients were not randomized to valproate and lithium. Furthermore, the study was not statistically powered to detect significant differences in maintenance efficacy between valproate and lithium. Notwithstanding these limitations, the estimated HR of 0.99 (95% CI: [0.66, 1.48]) suggests that the magnitude of difference between the two treatments in preventing any mood episode may be small. However, given the post hoc nature of the analyses, no firm conclusion regarding the relative efficacy of valproate and lithium on mood stability in patients with bipolar I disorder can be drawn. Subgroup analyses revealed patients are less likely to have a mood relapse if valproate and AA therapy are continued for 24 weeks instead of discontinuing AA soon after remission of mania. Lithium and AA combination therapy for 24 weeks was more effective than lithium monotherapy in preventing mania, but it provided no added benefit in preventing depression or any mood episode. The benefits of combination treatment beyond 24 weeks compared with the mood stabilizer alone are not apparent with either mood stabilizer. Therefore, the potential benefit should be weighed against risks associated with prolonged AA use. Further caution should be exercised in using valproate in women of childbearing age due to teratogenic potential, thus limiting its potential clinical use. Further head-to-head investigations are needed, and studies may employ a similar stratified design to consider duration of AA use as well as evaluate efficacy of maintenance therapies with other mood stabilizer medications.
Footnotes
Declaration of Conflicting Interests
L.N.Y. has received research support from or served as a consultant or speaker for Alkermes, Allergan, CANMAT, CIHR, Dainippon Sumitomo, Forest, Janssen, Lundbeck, Otsuka, Sanofi, Sunovion, Teva and Valeant. R.W.L. reports grants from CIHR, Janssen, Lundbeck, Pfizer, Asia-Pacific Economic Cooperation, VGH-UBCH Foundation, BC Leading Edge Endowment Fun and St. Jude Medical; personal fees from Allergan, Akili, CME Institute, CANMAT, Lundbeck, Otsuka, Medscape, Hansoh, Janssen, Lundbeck, Pfizer; and other from Asia-Pacific Economic Cooperation and Mind Mental Health Technologies. M.G.K., K.K., T.C., G.S., H.Q. and H.W. have nothing to disclose.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This study and the CANMAT trial were funded by the Canadian Institutes of Health Research.
