Abstract
This paper examines the current literature on matters specific to women patients with bipolar disorder, emphasizing gender differences in aspects of epidemiology, phenomenology and treatment. It also focuses upon sex differences in psychiatric and physical comorbidity. In approaching the pharmacological treatment of women with bipolar disorder, a number of gender considerations need to be taken into account, including differences in pharmacokinetics and pharmacodynamics of mood-altering drugs. Furthermore, the current literature on the impact of the female reproductive cycle on bipolar disorder is reviewed and special considerations necessary when managing women through pregnancy and the postpartum period are emphasized.
Epidemiological and phenomenological considerations
Epidemiological characteristics
The lifetime prevalence of bipolar I disorder has been found to be around 1% for both men and women [1]. Bipolar II disorder has usually been reported as being more common in women [2–4] with lifetime prevalence rates ranging from 5% to 10% [5], [6]. However, a recent Australian study by Mitchell et al. [7] found the 12-month prevalence of bipolar disorder (I and II combined) to be equal in men and women at 0.5%. Baldessarini et al. found a gender difference in the relationship between onset of symptoms and commencement of maintenance treatment. Men with bipolar I started such treatment after 6 years, while women with bipolar II disorder had a mean latency of 11 years [4].
Gender differences in clinical presentation of bipolar disorder
Some authorities have proposed that women have a ‘depressive diathesis’ that affects the expression of their bipolar disorder [8], [9]. Such a diathesis may explain the observation that women diagnosed with bipolar disorder report more episodes of depression than men, while conversely men report more manic episodes [8], [10]. This is reflected in admission rates, with men more likely to be admitted during a manic episode and women while depressed [8], [10]. Depressive episodes in women have been found to be of longer duration and to be more treatment refractory [11]. Although studies focusing onmixed episodes have had significant methodological limitations (e.g. changes in the definition), it appears that women experience more ‘mixed’ episodes than men [12]. It is also apparent that women are more likely than men to have a depressive episode precede a hypomanic/manic episode. Men with bipolar disorder appear to present more commonly with mania followed by depression [13]. Gender differences in the symptomatology of mania has also been reported by some authors, with men more likely to present with hyperactivity, risk-taking behaviour and grandiosity and women with racing thoughts and distractibility [14].
Psychiatric comorbidity
About 65% of patients with bipolar disorder have a comorbid psychiatric or physical condition [15]. Strakowski et al. [16], examining rates of comorbidity in mania at first hospitalization, found that woman were 2.7 times more likely than men to have a comorbid diagnosis.
The Epidemiological Catchment Area (ECA) study found lifetime prevalence rates of 46% for alcohol abuse/dependence (general population rate 14%) and 41% for substance abuse/dependence (general population rate 6%) in subjects with bipolar I disorder [14], [18]. Kraeplin [19] and Sonne et al. [20] have both reported that men with bipolar disorder had a greater prevalence of alcoholism than women. However, Frye et al. [21] found that women with bipolar disorder had a much greater risk of alcoholism than women in the general population, and that this relative risk was greater than the relative risk for men with bipolar disorder compared to males in the general population. More than 40% of people with bipolar disorder have a comorbid anxiety disorder, with women having higher rates (particularly of panic disorder and social phobia) than men with bipolar disorder [6], [16], [22].
Suicide and suicide attempts
Individuals with bipolar disorder are usually considered to have a risk of 10–19% of completed suicide, a rate about 15 times that of the general population [23], [24]. A recent study by Dalton et al. [25], however, found that the lifetime risk of suicide in patients with bipolar disorder (types I and II considered together) may be even higher, at 26%. No gender difference has been found in terms of completed suicide rates for bipolar disorder, unlike the general population in which suicide rates of males are three to four times those of women. A study by Bottlender et al. [17] examined suicidality as well as completed suicide in bipolar depressed and unipolar depressed inpatients. They found that female gender was associated with a lower probability of suicidal tendencies at the time of admission.
Differences in the course of bipolar disorder in women
Rapid-cycling pattern
Several studies have now shown that women are more likely than men to experience a rapid-cycling pattern during the course of their bipolar illness [8], [12], [26], [27]. The explanation for this sex difference is not clear. Three hypotheses to account for a greater likelihood of rapid cycling in females include: gonadal steroid effects, hypothyroidism and the greater use of antidepressant medication in women [8], [12], [27].
Mixed episodes
Although comparison of studies is confounded by differing definitions of mixed episodes, one study which reviewed pooled data from 13 investigations found that women had an increased propensity to such episodes (1.9:1) [9], [28]. Why women may be more susceptible to mixed episodes has not been fully established, with many differing theories; however, the main speculations concern the hypothalamic–pituitary–adrenal and thyroid axes [8], [12], [29].
Diagnosis and prognosis
Bipolar disorder in women appears to remain unrecognized and untreated for a longer period of time than in men, with a mean delay in treatment of 11 years versus 6 years [4], [11], [30]. It may be that the more frequent depressive presentations inwomenwith bipolar disorder are responsible. Routine inquiry about hypomanic or manic episodes in all women presenting with depressive symptoms is therefore recommended. With respect to outcome, one study estimated that for a woman diagnosed at the age of 25 years, the condition would shorten her lifespan by 9 years and lead to a loss of 14 years of effective functioning [13]. There have been no such published actual data or estimations for males.
Impact of the reproductive cycle on the course of bipolar disorder
Menstrual cycle
It is known that gonadal steroids can modulate the central activity of serotonin, noradrenaline and γ-aminobutyric acid (GABA). It has been postulated that the fluctuations in oestrogen and progesterone which occur during the reproductive cycle in women play a role in some women with bipolar disorder in exacerbating symptoms [81]. This mechanism is seen in women with premenstrual dysphoric disorder. The over-representation of women in rapid-cycling bipolar disorder has led to the hypothesis that alterations in female reproductive hormones may be in part responsible [82]. This is supported by studies that have found an increase in suicide rates, increased severity of suicidal intent and increased rates of hospitalization in women with bipolar disorder during the premenstrual and menstrual phases of the menstrual cycle [83]. Two studies have found that about 65% of womenwith bipolar disorder report regular exacerbations of mood symptoms during their menstrual cycle [82], [84]. Other studies, however, have not found a relationship between the menstrual phase and mood in women with rapid-cycling bipolar disorder [85].
Oral contraceptives
There is a lack of studies of the effect of exogenous oestrogens and progesterone on the course of bipolar disorder [8]. One recent prospective study of mood changes with the menstrual cycle in a ‘normal’ population reported an incidental finding that women taking oral contraceptives did not have the significant mood changes across the cycle seen in women who were not on such agents [82]. Further studies are needed to clarify this potentially important observation.
Pregnancy
In the past, pregnancy was thought to be a time during which women are less likely to experience episodes of affective illness [86]. For example, a retrospective study by Grof et al. [87] found that women with lithiummresponsive bipolar I disorder had fewer episodes during pregnancy compared to other 9-month periods before or after delivery. However, the sample in this study appears to have been biased, as all women had become pregnant before starting lithium treatment [88]. Another study by Viguera et al. [89] found that for women discontinuing lithium maintenance treatment, the rates of recurrence in the first 40 weeks were similar in pregnant (52%) and non-pregnant women (58%). Studies by Blehar et al. [84] and Freeman et al. [90] also found that 45–50% of women with bipolar disorder reported an exacerbation of their symptoms during pregnancy.
Post-partum
It has long been recognized that the post-partum period represents a time of particular risk for women for the development of an affective disorder or episode of psychosis, particularly women with pre-existing bipolar disorder. Rates of recurrence in women with bipolar disorder in the immediate post-partum period have been reported to be between 25% and 40% [87], [91], and relapse within 3–6 months post-partum in women with bipolar disorder has been variously reported to be between 67% and 82% [84], [89], [90]. A study by Viguera et al. [89] found that in women who remained stable in the first 40 weeks after discontinuation of lithium, the rates of recurrence increased 2.9 times in the post-partum period compared to non-pregnant women during weeks 41–64 (70% vs 24%). A further study suggested that mixed states are more common than pure mania or depressive episodes during this time [92].
The rate of post-partum psychosis increases from 10% to 20% in women with bipolar disorder [86], [93]. Postpartum psychosis presents with a rapid onset of symptoms, including auditory, visual and tactile hallucinations, often within 48–72 hours of delivery. It is not fully understood why post-partum relapse is so common in women with bipolar disorder. The association with the sudden decline in oestrogen and progesterone levels has led to speculation about the possible role of these steroids in treatment. Several studies have reported response to administration of oestrogen in postnatal depression. In both of these studies, though, the women were also on antidepressant treatment [94], [95].
A family history of puerperal psychosis appears to be a strong risk factor for women with bipolar disorder to develop a post-partum psychotic episode [96]. Several studies have confirmed that once a woman with bipolar disorder has experienced a post-partum affective episode, her risk for a subsequent episode in the post-partum period in the future is greatly increased. The range of increased risk has been estimated to range from 50% to 90% [88],[97–99].
Perimenopause and postmenopause
There is little information on the impact of menopause on the course of bipolar disorder [100]. Freeman et al. [90] reported on a study of the impact of reproductive events on the course of bipolar disorder, with 22 of the 50 women being either perimenopausal or menopausal. Three women, all of whom had bipolar I disorder, reported onset during or after the perimenopause. Twelve out of the 22 women reported a worsening of mood (increased depressive symptoms) and 10women reported no change after the onset of menopause; none of the women reported an improvement. Other studies have also reported worsening of mood symptoms, mostly depression, in postmenopausal women with bipolar disorder [84].
Biological treatment of bipolar disorder in women: special considerations
Pharmacokinetic and pharmacodynamic differences
Studies have identified some pharmacokinetic and pharmacodynamic differences between men and women, which may impact upon the pharmacological treatment of bipolar disorder [12]. Findings have been mixed with the results being confounded by small sample size and a lack of control on other variables (age, weight, dietary factors hormonal status and concomitant medications). The differences that have been found may be less important for psychotropic medications with a wider therapeutic index.
Pharmacokinetic differences
Women have lower hepatic metabolic rates than men [12]. They can also experience fluctuations in this rate related to the cyclic changes in reproductive hormones. The peak hepatic metabolic rate appears to occur mid menstrual cycle. This may cause lower plasma medication levels in the luteal phase of the cycle, with a potential to contribute to premenstrual worsening of bipolar disorder symptoms. A slower rate of metabolism in the follicular phase of the menstrual cycle could lead to higher circulating drug levels and an increased risk of side effects [31]. Women also have reduced absorption rates that may affect serum drug concentrations. This is thought to be because of the decreased secretion of gastric acid and progesterone in the luteal phase slowed gastric emptying. The menstrual cycle also affects this gastric motility as dilution via fluid retention results in lower plasma levels [12]. The slower renal elimination in females has the potential to prolong the half-life of lithium in women and raise plasma levels. Women may therefore require a lower dose compared to men [12], [32].
The normal increase in glomerular filtration rate and effective renal plasma flow during pregnancy [33] necessitates careful monitoring of lithium levels and may lead to the requirement for increased dosage. The sudden reduction in maternal blood volume, which occurs post-partum, can lead to lithium toxicity unless levels are checked, and dosage reduced accordingly. Oral contraceptives reduce the activity of some hepatic cytochrome P450 enzymes, which may lead to higher serum concentrations of some antipsychotics used in bipolar disorder, such as olanzapine. It has been speculated that young women may have higher activity levels of the enzymes CYP3A4 and CYP2C19, which may lead to lower plasma concentrations of medications such as sertraline, carbamazepine and citalopram compared to men [34]. However, overall there is a lack of data to support gender differences in genetic polymorphisms of the CYP enzymes.
Pharmacodynamic differences between men and women
Recently, attention has focused on possible gender differences in response to antidepressants. The studies have only focused on unipolar depression and results are contradictory. Specific studies are needed to explore this issue in bipolar disorder [12],[35–37]. It is unclear if there are substantive gender differences in the likelihood of adverse effects.
Electroconvulsive therapy
Women generally have lower seizure thresholds than men [35], [38]. It has been postulated that women may also experience less cognitive impairment than men [39]. According to one study, however, women may be at a higher risk of relapse after electroconvulsive therapy (ECT) [40]. As these studies have been fewin number and have focused on unipolar depression rather than bipolar depression, further focused studies in bipolar disorder are required.
Gender considerations in using medications
In general, guidelines that have been developed for the management of bipolar disorder [24], [41], [42] do not distinguish between men and women; however, some agents do raise specific gender issues. The teratogenic effects of the mood stabilizers are considered separately.
Mood stabilizers
Sodium valproate is a widely used mood stabilizer. There has been controversy whether valproate is associated with increased rates of endocrine abnormalities in women such as hyperandrogenism, hyperinsulinaemia and polycystic ovary syndrome (PCOS). This debate followed reports of a high-point prevalence of polycystic ovaries (PCO) in women who were prescribed valproate for epilepsy [43], [44]. Isojarvi et al. hypothesized that the weight gain associated with valproate treatment is progressive, and associated with hyperinsulinemia and low serum levels of insulin-like growth-factor-binding protein 1, which may lead to hyperandrogenism and PCO. Several studies have found higher rates of menstrual abnormalities in women receiving valproate for bipolar disorder (and epilepsy) compared to women not taking this anticonvulsant [45]. Specific side-effects of sodium valproate which may be of particular concern to women include fluid retention, weight gain and alopecia. Like lithium, valproate can be taken with oral contraceptives as it does not interfere with their efficacy, despite strong protein-binding affinity [46].
Possible gender differences in the side-effects of lithium have not been well-studied, apart from the greater vulnerability to hypothyroidism in women. Other adverse effects such as weight gain and hair loss appear clinically to be more of concern to women, but it is not clear if women are more vulnerable to these.
If a woman is taking carbamazepine there are some important issues that need to be considered. Carbamazepine can induce the metabolism of oral contraceptives through induction of cytochrome P450 enzymes, particularly CYP3A4. It is vital therefore, that all women being prescribed carbamazepine who rely on an oral contraceptive are warned of the potential reduction in its effectiveness. They need to be on either a higher dosages of the pill, or use barrier or other contraceptives [8]. Lamotrigine is effective for bipolar depression both in acute treatment [47] and in prophylaxis [48], [49]; however, gender differences in rates of response and side-effects have yet to be studied.
Antidepressants
A sex difference in response rates of unipolar depression has been postulated but whether such a difference exists in bipolar depression is yet to be established [24], [37]. The safe use of antidepressants in bipolar disorder has been debated at length in the literature. As women are at higher risk of developing a rapid-cycling course, it has been proposed that use of antidepressants should be minimized in women [27].
Antipsychotics
‘Typical’ antipsychotics (e.g. haloperidol, chlorpromazine) and the ‘atypical’ agent risperidone are all associated with menstrual dysfunction, because of hyperprolactinemia. This increase in prolactin levels may lead to anovulation, sexual dysfunction and enhanced risk of osteoporosis. Menstrual dysfunction may also occur with other atypical antipsychotics such as quetiapine, clozapine and ziprasidone, but at a much lower rate (<1%) [50]. Some of the atypical antipsychotics, particularly olanzapine and clozapine, can produce significant weight gain, hyperlipidemia and diabetes [51] – a concern for male and female patients alike. Clinicians should inform their patients of this risk, record baseline weight and give advice on how this can be minimized with good diet control and exercise, especially as such weight gain may be more of a concern for females.
Hormonal therapies
Evidence for the role of hormonal therapy per se in the treatment of women with bipolar disorder is limited. Exogenous oestrogen has been linked with the induction of mania and rapid cycling [52–54]. Other studies, however, have reported mood stabilization with the combination of oestrogen and progesterone [55]. The period of greatest risk of relapse for women with bipolar disorder is the post-partum period. This is a period of marked hormonal change, with a sudden decline in oestrogen and progesterone levels. There have been case reports of women with post-partum depression and low oestrogen levels responding to treatment with sublingual oestradiol [49], but further studies need to be performed [8]. Progesterone has been proposed as a possible treatment of post-partum mania, but with little success [56].
There have been several case reports of the benefit of exogenous steroids in postmenopausal women with treatment refractory bipolar disorder who had experienced mood episodes related to their menstrual cycles [55]. However, there has also been one case report of a treatment-resistant postmenopausal woman who developed rapid-cycling illness after treatment with high-dose conjugated oestrogen [57].
Novel therapeutic options
Omega-3 fatty acids have been increasingly studied in the treatment of bipolar disorder, depression and schizophrenia. The studies so far are limited and as yet gender differences have not been reported [58], [59]. A recent small study by Wisner et al. [60] reported response to verapamil in women with acute manic and mixed episodes. Although the numbers were limited, the authors also found low toxicity during pregnancy and in lactation which may suggest its role as a useful alternative in this group if findings can be replicated in future studies.
Teratogenic effects of medications used in bipolar disorder
Mood stabilizers
Managing a woman with bipolar disorder through pregnancy and the post-partum period presents many challenges to the clinician. The fact that most mood stabilizing agents have significant teratogenic effects is one major reason for these difficulties. These risks always need to be discussed with women as an ongoing process of informed consent.
The use of lithium in the first trimester has serious effects on the developing fetus and exposure at this time should be avoided. Specifically, there is an associated increased risk of Ebstein's anomaly. Recent studies have suggested lower rates than previously thought, namely, of one in 2000 (0.05%); however, this remains a 10-fold elevation of the risk in the general population [61]. The most common toxicity effect in newborns exposed to lithium during late pregnancy is the ‘floppy baby’ syndrome. This is characterized by cyanosis and hypotonicity. Lithium has also been reported to cause neonatal hypothyroidism and nephrogenic diabetes insipidus [62].
Sodium valproate and carbamazepine are both human teratogens and are associated with an increased risk of neural tube defects such as spina bifida (0.5–1% and 3–5%, respectively) [63], [64]. Combining anticonvulsants increases the risk, so such combinations should be avoided. As well as these serious neural tube defects, these anticonvulsants may cause other teratogenic effects including anencephaly, craniofacial defects, growth retardation, cardiac abnormalities and intrauterine growth retardation. Valproate has been associated with possible mental retardation as part of the ‘fetal valproate syndrome’. Carbamazepine has been associated with transient hepatic toxicity hepatitis and direct hyperbilirubinemia [62]. These complications may even occur after exposure, late in pregnancy [65]. It is important to note, however, that women in these studies were usually taking valproate for treatment of epilepsy and as such may have been on higher dosages than those used in bipolar disorder.
The use of lamotrigine during the first trimester has been investigated in a prospective naturalistic study. Risk of major malformations was found to be low at 1.8%, a level little different from the baseline population rate of 1.0% [50], [66]. However, the sample size was small and further studies are required to replicate these results. Two studies have reported a decrease in concentration of lamotrigine as clearance rates increase in late pregnancy.
Antidepressants
Tricyclic antidepressants (TCAs) have not been associated with an increased risk of major congenital abnormalities; however, exposure in the third trimester has been reported to cause anticholinergic effects in the neonate after delivery [68]. In terms of the safety of the selective serotonin re-uptake inhibitors (SSRIs) in pregnancy, a study byKulin et al. [69] examining 267 pregnantwomen exposed to sertraline, paroxetine or fluvoxamine did not find increased teratogenic risk. Three studies have specifically investigated the teratogenic effect of fluoxetine [70] and reported an increase in minor, but not major, congenital malformations. One study reported a non-significant increase in miscarriage rates in women treated with fluoxetine. However, these studies, as discussed by Leibenluft [71], did not separate medication effects from illness effects, which may in themselves increase miscarriage rates. Fluoxetine has been reported in other studies [72] to cause an increase inminor anomalies and to be associated with neonatal difficulties (e.g. respiratory difficulties, jitteriness). A recent study found that neonates exposed to SSRIs (citalopram or fluoxetine 20–40 mg day−1) during late pregnancy were at increased risk of serotonergic central nervous system adverse effects [72]. Other uncontrolled studies have found that SSRIs and the serotonin noradrenaline re-uptake inhibitors (SNRIs) may be used as monotherapy in pregnant women with bipolar II disorder for whom mood stabilizers have either been ineffective, or for whom there is potential teratogenic risk [73].
Medication and breast-feeding
Mood stabilizers
Use ofmood stabilizers during lactation is again a difficult clinical area as carbamazepine, valproate and lithium have all been associated with serious effects in the infant. However, we now know that the post-partum period is the time of maximum lifetime risk of relapse for a woman with bipolar disorder, especially if she ceased the mood stabilizer before becoming pregnant. Rather than being able to state definite preferences, the choice of mood stabilizer should be guided by the woman's past history of medication response, her risk of relapse without a mood stabilizer and her plans in regards to breast-feeding, as well as the clinical state of the infant [64], [74].
The amount of lithium that passes into the infant's system through maternal breast milk has been reported to be 10–50% of maternal lithium serum levels. After the first week this declines to around 33% [75], [76]. Renal clearance in the infant is decreased until at least 5 months of age; therefore, it has been recommended that use of lithium should be avoided until this time [77]. Lithium has been associated with thyroid dysfunction, poor muscle tone and cyanosis in the infant. A review by Chaudron and Jefferson [74] reported on 11 cases of women using lithium while breast-feeding. Two of these infants had evidence of lithium toxicity, as seen clinically by restlessness, muscle twitching, cyanosis, hypotonia, hypothermia and abnormal electrocardiogram (ECG). Eight infants had detectable serum lithium levels. However, for the woman who is prone to relapses without lithium and is insistent upon breast-feeding, the risks of becoming ill may outweigh the risks to the infant. In these cases, close monitoring of the infant by a paediatrician and monitoring of its serum lithium levels may minimize some of the potential risks [77].
Both valproate and carbamazepine have been considered to be compatible with breast-feeding by the American Academy of Paediatrics [64]. However, as both have been associated with hepatic dysfunction, it is recommended that liver enzymes, bilirubin and the white blood count should be monitored in such infants [77], [78]. The relative dose (per cent ratio of infant to maternal concentrations) of valproate in a nursing infant has ranged from 1.5% to 40% [79]. There has been one case report of valproate being associated with neonatal anaemia and thrombocytopenia [80]. Wisner and Perel reported that the relative dose of carbamazepine was 15–65% [79]. However, these fell to insignificant levels after 3–4 weeks post-partum. There is limited data on the safety of lamotrigine in breast-feeding infants; more studies are needed before this medication can be considered to be safe during breast-feeding.
Antidepressants
A recent review [77] examined all case reports and studies on the outcome of use of TCAs, SSRIs, SNRIs and other antidepressants in breast-feeding mothers. In general, the TCAs appeared to be safe. However, of concern was one case report of respiratory depression in one of two cases using doxepin, which resolved 24 hours after cessation of the medication. Although this is only one case report, it may be preferable to avoid doxepin in the post-partum period. No relationship was found between adverse effects and maternal dose of doxepin, infant serum level of doxepin or infant age. No other adverse effects have been reported with the other TCAs.
For those infants exposed to SSRIs, adverse effects were reported in 10 of 190 infants on fluoxetine. In six infants, adverse effects were non-specific and unconfirmed, three infants had colic and one infant had a transient seizure-like activity at 3 weeks of age and episodes of unresponsiveness at 4 months. No adverse effects were reported with the other SSRIs, though numbers in some of the studies were very small and were only based on case reports. More studies are clearly needed. No adverse effects were reported in the infants exposed to the other antidepressants.
In determining the treatment of women with bipolar depression in the post-partum period, ECT should be given serious consideration. The lack of adverse effects seen with the SSRIs sertraline and paroxetine would also make these suitable first-line treatments. The level of antidepressant (albeit already low) that passes through into breast milk is minimized if the medication can be taken immediately after breast-feeding and the mother attempts not to breast-feed for 7–11 hours afterwards, using formula in the interim.
Antipsychotics
Post-partum psychosis or mania will almost invariably require the use of antipsychotics. However, ECT is also indicated and may be the treatment of first choice in many cases. Burt et al. [77] reviewed 10 studies or case reports of infants exposed to antipsychotics during breast-feeding. Of the total of 34 infants in these studies, 25 infants had no adverse effects. Of those exposed to chlorpromazine, four of the 14 infants had adverse effects including lethargy and declining scores on developmental testing. These four infants had also received combination therapy with other antipsychotics. Monotherapy would therefore appear to be advisable. Of the 11 infants exposed to haloperidol, three infants showed declining scores on developmental testing and again these had also been exposed to combination treatment. The small numbers in the olanzapine group and lack of data on other atypical antipsychotics makes it difficult to comment on this group as a whole. Overall, the clinical condition of the mother may be more important. If antipsychotics need to be used in a mother committed to breast-feeding, the infant should be closely monitored for side-effects such as somnolence, tremor and rigidity.
Practical considerations in managing women through pregnancy
Overview of the issues
A collaborative approach is needed when managing women with bipolar disorder during this period. Involvement of the spouse or partner helps when difficult decisions about treatment need to be made. Important areas that need to be discussed with the couple include family planning, risks of exposure for a fetus to medication, the treatment choices that are available [88], [101] and the genetic risks of bipolar disorder.
The dilemma all clinicians face is the task of minimizing potential risks to the fetus (by choosing whether or not to continue psychotropic medications), while at the same time optimizing the mental health of the mother and not exposing her to risk of relapse by ceasing medication prematurely. Rather than following set guidelines, several authors [62], [88] have proposed an individualized approach, which base decisions on treatment on the severity of illness, prior frequency of episodes and any past history of relapse in pregnancy and the post-partum period. It is now known that abrupt discontinuation of medication should be avoided because of the increased risk of sudden relapse [88]. Of all the mood stabilizers, lithium is preferred over valproate and carbamazepine during the second and third trimester. In view of the toxicity of all mood stabilizers in the first trimester, none are considered safe and should be avoided. A planned pregnancy would allow a slow withdrawal of such medication before conception with reinstatement, if indicated, during the second trimester [27]. All women on anticonvulsants should be encouraged to take folate supplements because of the high risk of neural tube defects. As these anomalies occur early in pregnancy, women need to be already taking the folate to experience a protective benefit [50], [66], [67].
Developing an individualized management plan
Ideally, conception should be planned to occur when the woman is euthymic. As well as discussing use of psychotropic medications, planning should involve discussion on all aspects of the women's health and wellbeing to before conception, that is, balanced nutrition, exercise and weight control and ceasing smoking, drug and/or alcohol use [62]. Unplanned pregnancies are more likely to lead to abrupt cessation of ongoing mood stabilizing medication on discovery of the pregnancy. Earlier recommendations [102] discouraged use of mood stabilizers at any time during pregnancy; this view is no longer supported by the literature. The risk of relapse for women with bipolar disorder who have a lifetime history of more than four episodes, and who rapidly stop lithium (<2 weeks) once they fall pregnant, has been reported to be significantly higher than those with fewer lifetime episodes and who discontinue lithium more slowly (66% vs 38%) [101]. In developing an individualized approach, Viguera et al. propose that all women with bipolar disorder are considered ‘high risk’ in terms of obstetric care, and as such require close monitoring during pregnancy. A woman's previous course and specific illness characteristics can assist the clinician to formulate with her a specific management plan. See 27], [64], [88], [101].
A suggested approach for the use of mood stabilizers in planning toward pregnancy for a woman with pre-existing bipolar disorder
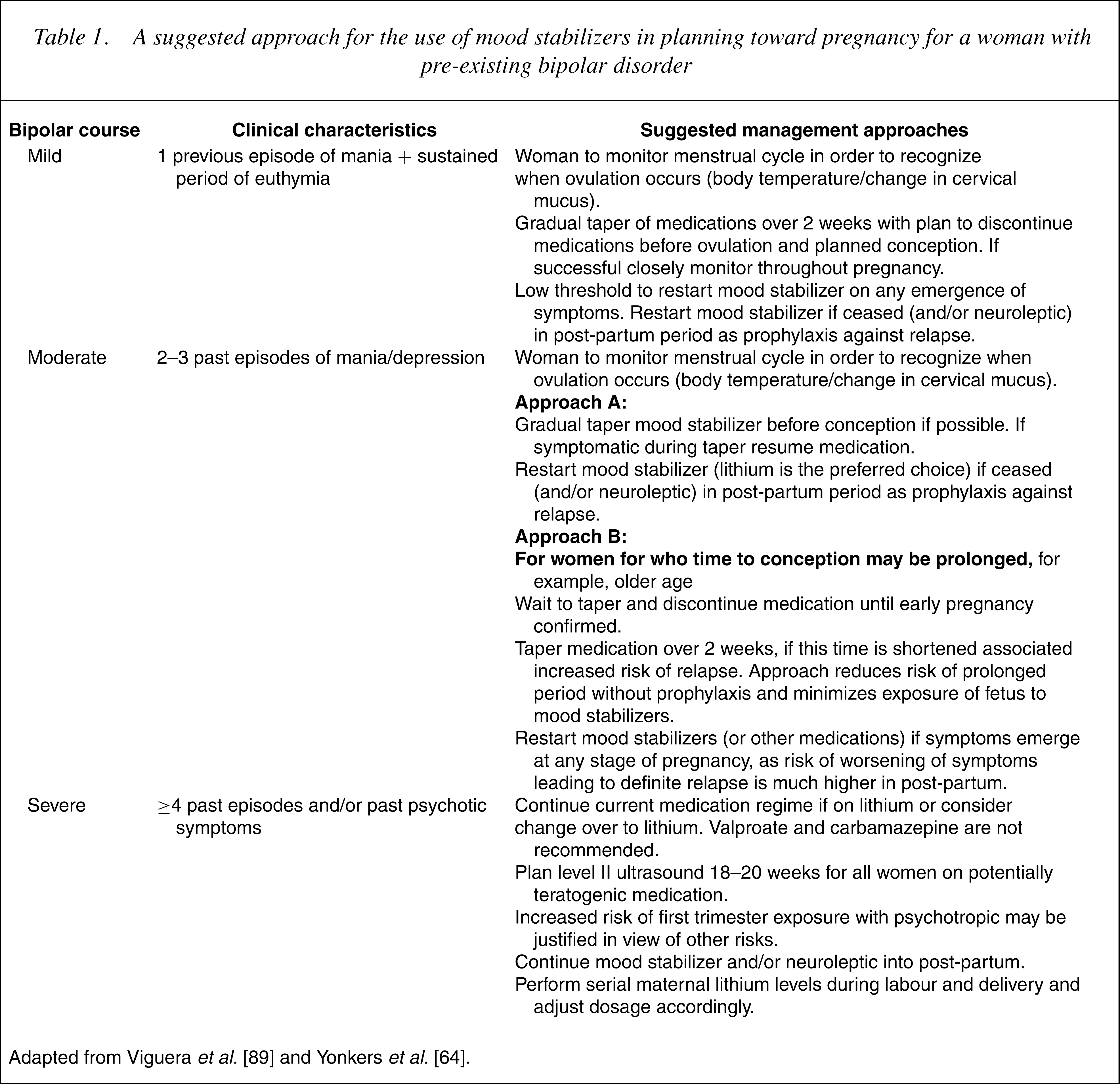
Practical considerations in managing women in the post-partum period
It is vital that women with bipolar disorder are fully aware of both the risks they face in the post-partum period and the importance of continuing or restarting prophylactic medication. A retrospective study by Cohen et al. [103] followed 27 women with bipolar disorder through their pregnancy and post-partum period. They reported that the relative risk of relapse for women who did not receive preventive treatment was 8.6 times that of women who received prophylaxis with a mood stabilizer. Several studies have found that the use of lithium reduces the risk of relapse fivefold if re-instituted shortly before delivery (at about week 36), or within 48 hours of delivery and is continued into the post-partum period [88], [103], [104]. As neonatal toxicity can occur, careful monitoring of maternal lithium levels needs to occur during and immediately after delivery.
Some studies have postulated that the disruption of the sleep–wake cycle that occurs after birth plays an important role in the onset of relapse at this time. One study found both fathers as well as post-partum mothers with bipolar disorder having an increased risk of relapse of bipolar disorder at this time [105]. Leibenluft et al. found that decreased total time asleep as well as early waking were associated with relapse of manic symptoms [106]. Planning for extra support during this period, especially during the night, to allow the mother some periods of uninterrupted sleep is an important part of the post-partum management plan. If the mother breast-feeds while taking medication regular monitoring of the infant's growth, behavioural and physical assessments has been recommended, aiming to use the lowest dose of medication possible in the mother and to limit the number of medications used [64].
Summary
As more research is undertaken in this area, it is becoming increasingly clear that gender differences have important clinical applications in bipolar disorder. There are still many uncertainties in knowledge that need to be clarified, in particular: the impact of the menopause on bipolar disorder; the effect of hormone replacement therapy (HRT) and oral contraceptives; the relationship of gender and side-effects; and the safety during pregnancy and lactation of many of the newer medications now being used in the treatment of bipolar disorder. Management of women with bipolar disorder through pregnancy remains a challenging task for clinicians. It is hoped that consideration of an individualized and collaborative approach to management will assist clinicians in this task.
Footnotes
Acknowledgements
The research activities of Philip Mitchell are supported by an Australian National Health and Medical Research Council Program grant (no. 22708).
