Abstract
Objective:
Activation encompasses energy and activity and is a central feature of bipolar disorder. However, the impact of activation on treatment response of bipolar depression requires further exploration. The aims of this study were to assess the association of decreased activation and sustained remission in bipolar depression and test for factors that could affect this association.
Methods:
We assessed participants with Diagnostic and Statistical Manual of Mental Disorders (4th ed) bipolar depression (n = 303) included in a comparative effectiveness study of lithium- and quetiapine-based treatments (the Bipolar CHOICE study). Activation was evaluated using items from the Bipolar Inventory of Symptoms Scale. The selection of these items was based on a dimension of energy and interest symptoms associated with poorer treatment response in major depression.
Results:
Decreased activation was associated with lower remission rates in the raw analyses and in a logistic regression model adjusted for baseline severity and subsyndromal manic symptoms (odds ratio = 0.899; p = 0.015). The manic features also predicted lower remission (odds ratio = 0.934; p < 0.001). Remission rates were similar in the two treatment groups.
Conclusion:
Decreased activation and subsyndromal manic symptoms predict lower remission rates in bipolar depression. Patients with these features may require specific treatment approaches, but new studies are necessary to identify treatments that could improve outcomes in this population.
Introduction
Energy and activity are key symptoms in bipolar disorder (BD), which are increased during manic and decreased during depressive episodes (Faurholt-Jepsen et al., 2016). Several studies identifying a hyperactivity factor during mania concluded that increased energy is not only important, but a core feature of manic states (Cheniaux et al., 2014). As a consequence of these studies, the Diagnostic and Statistical Manual of Mental Disorders (5th ed.; DSM-5), unlike previous editions, requires elevated energy or activity as a prerequisite for a diagnosis of a manic (or hypomanic) episode. While decreased energy or activity is not necessary for the diagnosis of a depressive episode, it may be a core feature (American Psychiatric Association, 2013).
Scott et al. (2017) recently proposed the concept of activation to describe the phenomena implied by the DSM terms of activity and energy. These authors performed a systematic review on activation in BD and found that increased activation is the most frequently observed factor during mania, psychomotor retardation is more frequent in bipolar depression than in major depressive disorder (MDD) and individuals with BD have decreased activity levels during depressive and euthymic periods (Scott et al., 2017). Activation symptoms are present in the prodromal phase of BD (Egeland et al., 2000; Skjelstad et al., 2010), and change in energy is among the first recognized symptoms of recurrence both for manic and depressive episodes (Goossens et al., 2010; Houston et al., 2007).
Interest in activation symptoms in mood disorders is not a recent development. Psychomotor retardation is a central feature in historical descriptions of melancholia (Parker, 2007). Kraepelin (1921) described manic-depressive insanity in terms of changes in activity as well as mood and thinking. Studies used objective assessments of motor activity in BD more than 40 years ago (Kupfer et al., 1974; Post et al., 1977).
Interestingly, some studies indicate that there is a relationship between activation symptoms of mood disorders and energy metabolism. Brain circuits that regulate mood may be intertwined with those that regulate homeostasis (Liu et al., 2014). Another study found that mitochondrial function was impaired in depression and that the decline in mitochondrial respiratory activity was correlated with depressive symptoms of loss of energy, fatigue and difficulties with concentration (Karabatsiakis et al., 2014). Resting energy expenditure of individuals with BD is increased during manic episodes (Caliyurt and Altiay, 2009) and decreased during maintenance treatment (Soreca et al., 2007). While there have not been any studies on bipolar depression, total energy expenditure is reduced in unipolar depressive episodes (Wielopolski et al., 2015).
Additional studies confirm the relevance of activation symptoms in MDD. Mean levels of activity, assessed by actigraphy, are decreased in MDD patients compared to controls (Krane-Gartiser et al., 2015). An instrument assessing psychomotor retardation during depressive episodes was associated with psychotic symptoms increased severity (Caldieraro et al., 2013) and higher inflammatory marker levels (Spanemberg et al., 2014). A large study reported that the only depressive symptoms associated with C-reactive protein levels were fatigue, low energy, restless sleep and feeling depressed (White et al., 2017). In patients treated for MDD, lack of energy is a common residual symptom inclusive in those who achieve the criteria for remission (Nierenberg et al., 2010; Nil et al., 2016). Most importantly, fatigue and lower activity levels are associated with lower response to antidepressant treatment (Ferguson et al., 2014). Uher et al. reported that a dimension of interest–activity symptoms (i.e. low interest, reduced activity, indecisiveness and lack of enjoyment) predicted treatment resistance in two different large antidepressant trials (Uher et al., 2012).
Despite the relevance of activation symptoms in BD, the association of decreased activation with treatment response in bipolar depression has been insufficiently explored in previous studies. The aim of this study is to assess whether symptoms of decreased activation at baseline can predict remission rates in patients with bipolar depression who participated in the Bipolar Clinical Health Outcomes Initiative in Comparative Effectiveness (Bipolar CHOICE) study (Nierenberg et al., 2016). We also tested for other factors that could moderate this association.
Methods
Bipolar CHOICE was an 11-site, 6-month randomized comparative effectiveness study comparing lithium (a classic mood stabilizer) to quetiapine (a second-generation antipsychotic) with adjunctive personalized treatments (APTs; evidence-based, guideline-informed treatment based on illness course, treatment history and current symptomatology) in adult outpatients with BD-I or BD-II. The quetiapine + APT group could receive all available treatments except for lithium or other antipsychotics, and the lithium + APT group could receive all available treatments except for antipsychotics. Patients were evaluated at baseline and eight follow-up assessments (weeks 2, 4, 6, 8, 12, 16, 20 and 24). A detailed description of the study protocol can be found elsewhere (Nierenberg et al., 2014). The study protocol was approved by the Institutional Review Board (IRB) at each site, and participants provided written informed consent before starting any study-related procedure. The authors assert that all procedures contributing to this work comply with the ethical standards of the relevant national and institutional committees on human experimentation and with the Helsinki Declaration of 1975, as revised in 2008. The Bipolar CHOICE study was registered on ClinicalTrials.gov (NCT01331304).
Participants
Inclusion and exclusion criteria were minimal in the Bipolar CHOICE study to obtain a more diverse and generalizable sample. Eligible patients diagnosed with BD-I or BD-II at any mood state entered the study with at least mild BD symptoms (Clinical Global Impressions–Severity–Bipolar Version [CGI-S-BP] overall illness severity score ⩾3). Potential participants were excluded from the study if they had any contraindication to lithium or quetiapine (e.g. pregnancy, prior hypersensitivity, severe renal disease, lack of treatment response after an adequate trial); were currently in crisis such that hospitalization or more acute care was necessary; were currently taking lithium or quetiapine or were unable to comply with study requirements. For this secondary analysis, the CHOICE sample was restricted to individuals who were depressed at baseline.
Assessments
Diagnosis and symptom severity
Lifetime and current diagnoses according to the Diagnostic and Statistical Manual of Mental Disorders (4th ed., Text Revision; DSM-IV-TR), including BD and other psychiatric comorbidities, were established at the screening visit with the electronic Mini-International Neuropsychiatric Interview (eMINI-PLUS), an electronic extended version of a validated structured diagnostic interview (Sheehan et al., 1998).
Mood symptom severity was assessed with the Bipolar Inventory of Symptoms Scale (BISS; Bowden et al., 2007) and the CGI-S-BP (Spearing et al., 1997). The BISS assesses individual mood symptoms in a structured interview that yields an overall severity score and multiple subscores, including mania and depression. Participants presenting with manic/hypomanic or mixed episodes were excluded in this analysis to focus on bipolar depressive episodes. Therefore, manic symptoms, when present, were in the subsyndromal level. The CGI-S-BP assesses the severity of BD based on the clinician’s view of the patient’s global functioning and provides three scores: overall illness severity, severity of depression and severity of mania (Spearing et al., 1997).
Sustained remission
The treatment outcome measure used for this study was sustained remission, defined by CGI-S-BP severity scores ⩽2 for at least 8 weeks. To prevent the risk of false-positive results caused by multiple comparisons, this outcome variable was selected a priori and other analyses using different definitions of treatment response/remission were not performed. This definition of sustained remission was adopted because it is consistent with DSM-IV criteria for remission, and it has been used in previous publications on the Bipolar CHOICE cohort (Deckersbach et al., 2016; Reilly-Harrington et al., 2016; Sylvia et al., 2017) and previous studies examining treatments in depression and BD (Keller et al., 1993; Perlis et al., 2006).
Decreased activation
To evaluate decreased activation, we used selected BISS items to create an adapted version of the original ‘interest–activity’ dimension identified in patients with MDD by Uher et al. (2008). This dimension predicted response to antidepressant treatment in separate datasets from two independent antidepressant clinical trials (Uher et al., 2008, 2012).
The selected BISS items and the corresponding symptom in the original ‘interest–activity’ dimension were as follows: BISS 8—Low energy (energy/fatigue); BISS 10—Loss of interest/anhedonia (interest/enjoyment); BISS 11—Social withdrawal (Interest in people); BISS 12—Reduced sex drive (interest in sex) and BISS 21—Impaired concentration (concentration/decisions). We could not identify a BISS item that corresponded to the symptom of work activities in the original ‘interest–activity’ dimension. Moreover, we assessed the symptoms based only on a clinician-rated scale (i.e. BISS), while the original dimension includes both clinician- and patient-rated scales.
Statistical analyses
Data were analyzed using SPSS 20.0 (Chicago, IL). Univariate analyses were performed using t-tests for continuous variables with a normal distribution and Mann-Whitney U-tests for continuous variables with a non-normal distribution. Categorical variables were compared using chi-square (χ2) tests with exact tests when indicated. We used a two-tailed significance threshold of p < 0.05. Comparisons of individual-item baseline scores between remitters and non-remitters included a Bonferroni correction, and p values < 0.0011 were considered statistically significant in these specific analyses.
To test the baseline total score of the decreased activation items as a predictor of sustained remission, we performed a binary logistic model controlling for baseline severity (CGI-S-BP overall illness) and subsyndromal manic symptoms (BISS mania). A second model was performed to test baseline depression severity (BISS depression) as a predictor of sustained remission and used the same covariates. These analyses were repeated using models with additional terms defined a priori for bipolar subtype (I vs II), sex, age, treatment group (quetiapine vs lithium) and comorbidity with anxiety disorders.
Results
Out of the 303 patients who presented with a depressive episode at enrollment, 58 (19.1%) achieved sustained remission during the study follow-up. The difference in the remission rates of individuals treated with lithium + APT (21.2%) and quetiapine + APT (17.2%) was not statistically significant (p = 0.372). Baseline characteristics of remitters and non-remitters are presented in Table 1. There were no statistically significant differences in general demographic characteristics, except for the education level. The proportion of patients diagnosed with BD-I was higher in non-remitters (69%.0) than in remitters (46.6%; p = 0.001). Non-remission was also associated with higher rates of comorbidity with panic disorder, agoraphobia, social phobia and generalized anxiety disorder.
Baseline demographic and clinical characteristics of the sample.
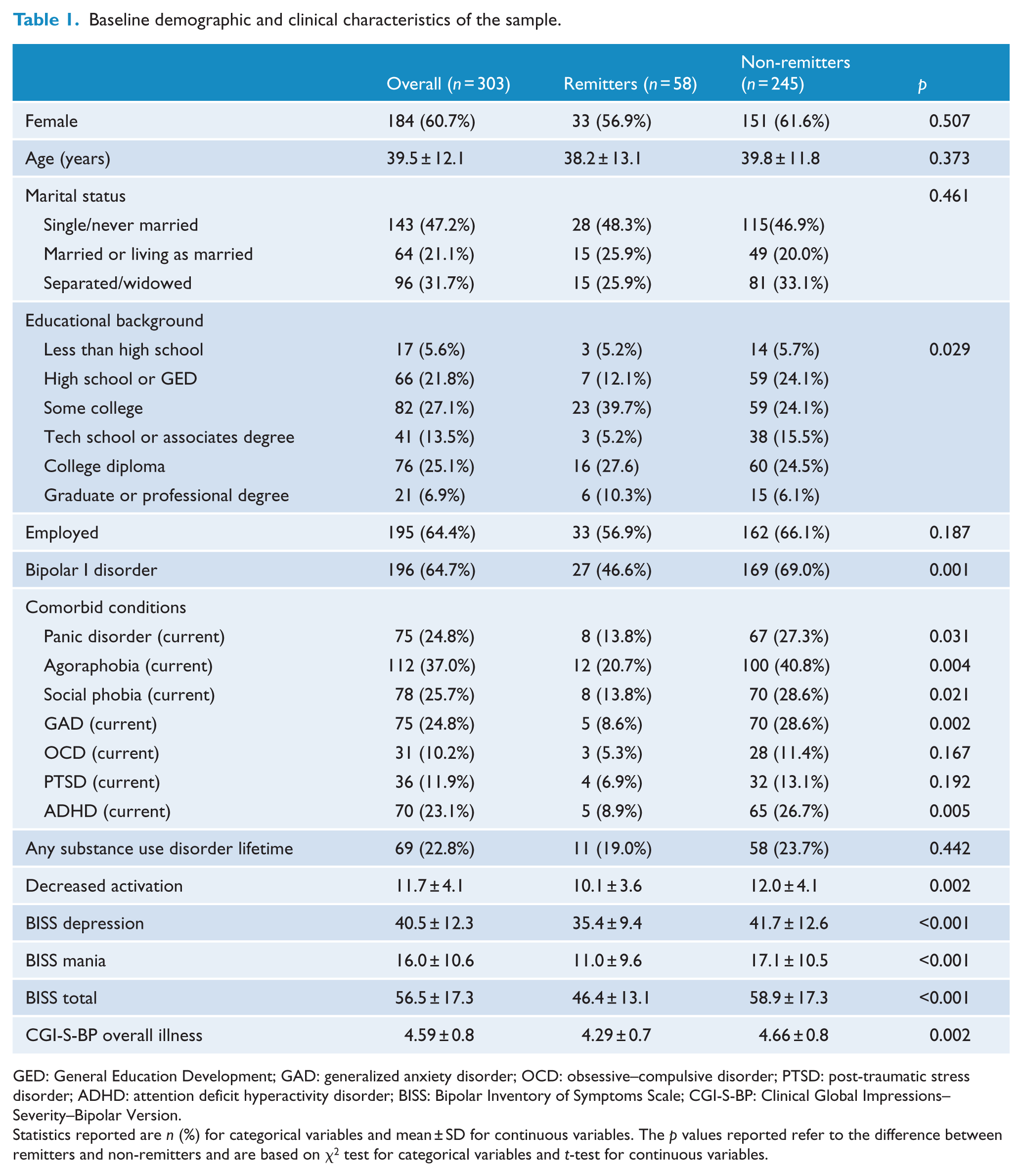
GED: General Education Development; GAD: generalized anxiety disorder; OCD: obsessive–compulsive disorder; PTSD: post-traumatic stress disorder; ADHD: attention deficit hyperactivity disorder; BISS: Bipolar Inventory of Symptoms Scale; CGI-S-BP: Clinical Global Impressions–Severity–Bipolar Version.
Statistics reported are n (%) for categorical variables and mean ± SD for continuous variables. The p values reported refer to the difference between remitters and non-remitters and are based on χ2 test for categorical variables and t-test for continuous variables.
In addition to the severity of the decreased activation symptoms, we also assessed the severity of overall depression, subsyndromal manic symptoms and overall illness severity (total BISS and CGI-S-BP). Higher baseline scores in all these measures were associated with non-remission (see Table 1). As an exploratory step, we compared baseline scores of each of the 45 BISS items between remitters and non-remitters. After a correction for multiple comparisons, seven individual items associated with remission. One of these items was from the depression subscale: suicidality (p < 0.001). The other six items were from the mania subscale: reported irritability (p < 0.001), pressured speech (p < 0.001), distractibility (p = 0.001), persecutory ideas (p < 0.001), delusions (p < 0.001) and appearance (p < 0.001).
Higher baseline scores on the decreased activation symptoms also predicted lower chance of remission (odds ratio (OR) = 0.899; p = 0.015) in a logistic regression adjusted for overall baseline severity (CGI-S-BP) and baseline score in the mania subscale of the BISS (Table 2). The mania subscale was included in this model because most of the items associated with the remission rates assess manic symptoms at the individual item level. In a model controlling for the same variables, baseline overall depression severity (BISS depression) did not predict remission (Table 2). Furthermore, adjustment for sex, age, treatment group, bipolar subtype (I vs II) and comorbidity with anxiety disorders did not impact the association between decreased activation and sustained remission. Similar results were observed when the control for baseline manic symptoms was performed using the score in the mania domain of the BISS (Thompson et al., 2010), which does not include the psychotic symptoms present in the mania subscale of the BISS.
Predictors of sustained remission.
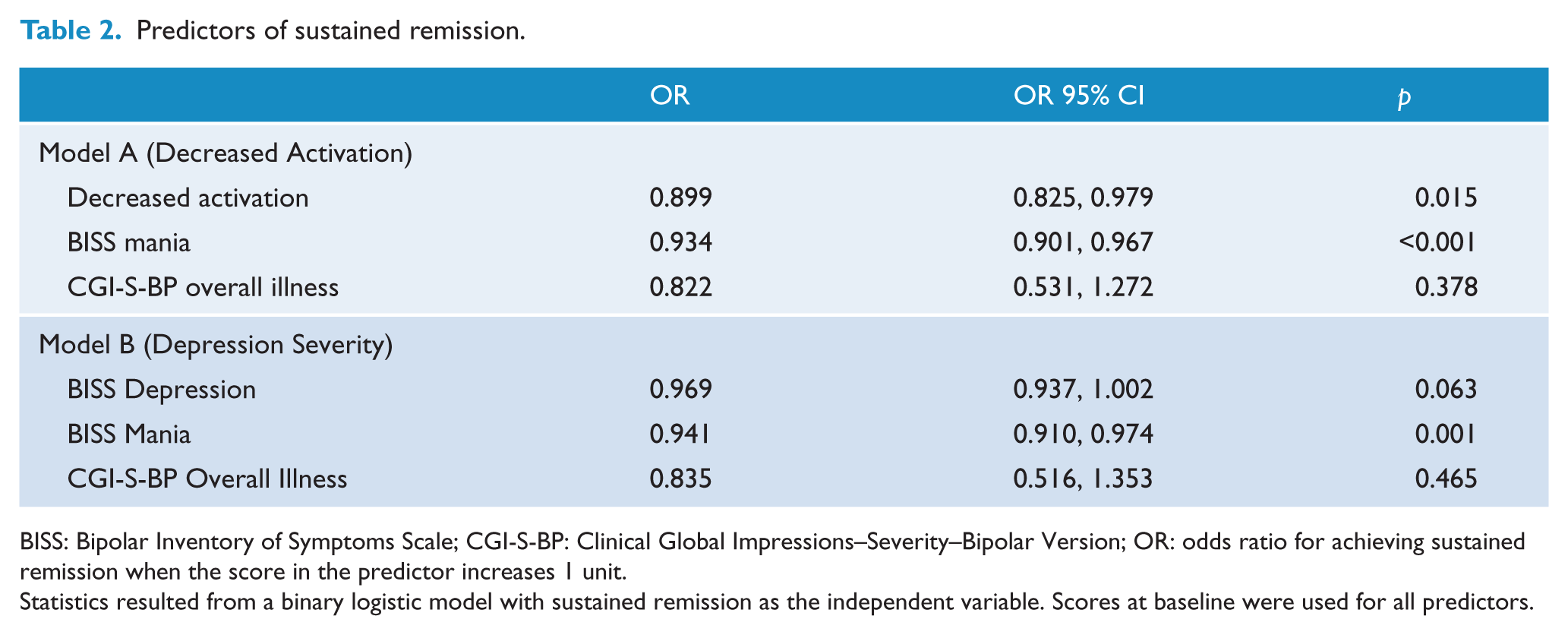
BISS: Bipolar Inventory of Symptoms Scale; CGI-S-BP: Clinical Global Impressions–Severity–Bipolar Version; OR: odds ratio for achieving sustained remission when the score in the predictor increases 1 unit.
Statistics resulted from a binary logistic model with sustained remission as the independent variable. Scores at baseline were used for all predictors.
Baseline severity of manic symptoms was negatively associated with sustained remission in both of these models (also shown in Table 2), confirming that manic symptoms were significant predictors of non-remission in bipolar depression. Finally, the remission rates were similar in the two treatment groups of the study in a model controlling for manic symptoms (OR = 1.165; 95% confidence interval (CI): [0.645, 2.104]; p = 0.614). This result indicates that the severity of subsyndromal manic symptoms was not associated with a distinct efficacy of one treatment over the other.
Discussion
In this secondary analysis of a large effectiveness treatment trial in BD, we found that decreased activation is a predictor of lower remission rates in bipolar depressive individuals receiving treatments based on a classic mood stabilizer (lithium) or an US Food and Drug Administration (FDA)-approved treatment for bipolar depression (quetiapine). We also found that subsyndromal manic symptoms are a significant predictor of non-response to these treatments. In both cases, the observed associations persisted after adjusting for overall illness severity.
Activation symptoms are a central feature in the phenomenology of BD (Scott et al., 2017) and present more frequently in bipolar depression than in MDD (Motovsky and Pecenak, 2013). However, their value as predictors of treatment response in bipolar depression has been understudied. Our finding that decreased activation at baseline is associated with a lower probability of achieving remission is in accordance with results observed in unipolar depression (Uher et al., 2012). In line with the relevance of activation for the treatment outcomes of bipolar depression, a recent study found that early improvement in the symptom ‘loss of energy’ was associated with durable recovery, and early improvement in psychomotor retardation was associated with a subsequent affective switch (Mizushima et al., 2017). The relevance of our results with activation symptoms was reinforced by the finding that overall depression severity was not a predictor of remission in a similar model controlling for baseline overall depression severity and subsyndromal manic symptoms. The DSM-5 recognized the relevance of activation during mania, by requiring the presence of increased energy for the diagnosis of these episodes. Our results indicate that activation is also central to bipolar depressive episodes.
The presence of subsyndromal manic features during a depressive episode is frequent in both MDD and BD (McIntyre et al., 2015b). We found a strong association of these symptoms with decreased remission rates. This result is consistent with findings from previous studies indicating that these features are associated with a more severe and more complex clinical state (Goldberg et al., 2009) as well as greater treatment resistance (Rosenblat and McIntyre, 2017). We also found that the presence of subsyndromal manic symptoms was not associated with a difference in the efficacy of a lithium- or quetiapine-based treatment. To date, no studies have examined the efficacy of classic mood stabilizers for bipolar depressive episodes with mixed features. Previous studies reported the efficacy of ziprasidone (Patkar et al., 2012), lurasidone (McIntyre et al., 2015a), olanzapine (Tohen et al., 2014) and olanzapine-fluoxetine combination (Benazzi et al., 2009) in the treatment of bipolar depressive episodes with mixed features. However, the previous studies, similar to the present one, were mostly secondary analyses and did not used the DSM-5 definition of mixed features, indicating the need for future treatment trials on this population. Our finding suggests that lithium should also be considered for these future trials.
The overall remission rates observed in the Bipolar CHOICE study were similar to those reported in other trials on BD (Nierenberg et al., 2013; Sachs et al., 2007). The presence of comorbidities, particularly with anxiety disorders, was also more frequent in non-remitters. This finding is in accordance with previous studies that report that anxiety disorders are frequently comorbid and associated with a more severe course in BD (Perlis et al., 2006).
The results of this study should be interpreted considering some limitations. This is a secondary analysis, and the original trial was not designed to assess activation. Patients were included using the DSM-IV criteria, so the assessment of subsyndromal manic symptoms was not based on the DSM-5 mixed-features specifier. Our selection of symptoms to assess activation was based on a dimension previously studied by Uher et al. (2012) in MDD, but it is not equivalent to that dimension. We used only a clinician-rated scale, while the original dimension included both clinician- and self-rated scales.
In sum, we found that decreased activation and subsyndromal manic symptoms are associated with reduced remission in patients with bipolar depression. This effect was similar in patients receiving a lithium- or a quetiapine-based treatment. These results are clinically relevant, as they can help clinicians to make a more realistic prognosis and consider alternative approaches for patients presenting with these features. In terms of research, our results suggest that future studies should explore new treatments for this population. For example, it would be important to study whether the use of medications that have a significant effect on energy symptoms, such as stimulants, would increase remission rates in bipolar depression with decreased activation. For bipolar depression with mixed features, future trials should examine the efficacy of classical mood stabilizers as well as second-generation antipsychotics.
Footnotes
Declaration of Conflicting Interests
Dr Caldieraro reports no competing interests. Ms Walsh reports no competing interests. Dr Deckersbach’s research has been funded by NIH, NIMH, NARSAD, TSA, IOCDF, Tufts University, DBDAT, Otsuka Pharmaceuticals and Cogito, Inc. He has received honoraria, consultation fees and/or royalties from the MGH Psychiatry Academy, BrainCells Inc., Clintara, LLC., Systems Research and Applications Corporation, Boston University, the Catalan Agency for Health Technology Assessment and Research, the National Association of Social Workers Massachusetts, the Massachusetts Medical Society, Tufts University, NIDA, NIMH and Oxford University Press. He has also participated in research funded by DARPA, NIH, NIMH, NIA, AHRQ, PCORI, Janssen Pharmaceuticals, The Forest Research Institute, Shire Development Inc., Medtronic, Cyberonics, Northstar, Takeda and Sunovion. Dr Bobo has received research support from NIMH, AHRQ and the Mayo Foundation for Medical Education and Research. Dr Gao have been on a speakers bureau and an advisory board of Sunovion and received grant supports from AstraZeneca, Brain and Behavior Research Foundation and Cleveland Foundation. Dr Ketter has the following financial interests/arrangements or affiliations that could be perceived as real or apparent conflicts of interest: Grant/Research Support from the AstraZeneca Pharmaceuticals LP, Cephalon Inc., Eli Lilly and Company, Pfizer Inc. and Sunovion Pharmaceuticals; Consultant Fees from Allergan, Inc., Avanir Pharmaceuticals, Bristol-Myers Squibb Company, Cephalon Inc., Forest Pharmaceuticals, Janssen Pharmaceutical Products, LP, Merck & Co., Inc., Sunovion Pharmaceuticals, Teva Pharmaceuticals; Lecture Honoraria from Abbott Laboratories, Inc., AstraZeneca Pharmaceuticals LP, GlaxoSmithKline and Otsuka Pharmaceuticals and Publication Royalties from American Psychiatric Publishing, Inc. In addition, Dr Ketter’s spouse is an employee of and holds stock in Janssen Pharmaceuticals. Dr Shelton has received grant funding from Alkermes, Inc.; Assurex Health; Avanir Pharmaceuticals; Cerecor, Inc.; Janssen Pharmaceutica; Novartis, Inc.; Otsuka Pharmaceuticals; Nestle’ Health and Takeda Pharmaceuticals. He has been a consultant for Allergan; Cerecor, Inc.; Clintara LLC; Janssen Pharmaceutica; Medtronic, Inc.; MSI Methylation Sciences, Inc.; Nestle’ Health; Pfizer, Inc. and Takeda Pharmaceuticals. Dr Reilly-Harrington receives royalties from Oxford University Press, the American Psychological Association and New Harbinger. She serves as a consultant for United Biosource Corporation and was a shareholder in Concordant Rater Systems. Dr Tohen has been a consultant for AstraZeneca, Abbott, BMS, Lilly, GSK, J&J, Otsuka, Roche, Lundbeck, Elan, Allergan, Alkermes, Merck, Minerva, Neuroscience, Pamlab, Alexza, Forest, Teva, Sunovion, Gedeon Richter and Wyeth. He was a full-time employee at Lilly (1997 to 2008). His spouse is a former employee at Lilly (1998–2013). Dr Calabrese has received federal funding from the Department of Defense, Health Resources Services Administration and National Institute of Mental Health as well as grant support from Abbott Laboratories; AstraZeneca; Bristol-Myers Squibb Company; Cephalon, Inc. (now Teva Pharmaceutical Industries Ltd.); Dainippon Sumitomo Pharma Co., Ltd.; GlaxoSmithKline; Janssen Pharmaceuticals, Inc.; Eli Lilly and Company; Intra-Cellular Therapies, Inc.; Pfizer, Inc; H. Lundbeck A/S; Sunovion Pharmaceuticals Inc. and Takeda Pharmaceutical Company Limited. Dr Calabrese has served as a consultant/advisory board member/speaker for Abbott Laboratories; Allergan; AstraZeneca; Bristol-Myers Squibb Company; Cephalon, Inc. (now Teva Pharmaceutical Industries Ltd.); Dainippon Sumitomo Pharma Co., Ltd.; GlaxoSmithKline; Janssen Pharmaceuticals, Inc.; H. Lundbeck A/S; Merck & Co., Inc.; Otsuka Pharmaceutical Co., Ltd.; Pfizer, Inc; Repligen Corporation; Servier; Sunovion Pharmaceuticals Inc.; Solvay Pharmaceuticals, Inc. and Takeda Pharmaceutical Company Limited. Dr Thase has been an advisor/consultant: Alkermes; Allergan; AstraZeneca; Bristol-Myers Squibb Company; Cerecor, Inc.; Eli Lilly & Co.; Fabre-Kramer Pharmaceuticals, Inc.; Forest Laboratories; Gerson Lehrman Group; GlaxoSmithKline; Guidepoint Global; H. Lundbeck A/S; MedAvante, Inc.; Merck and Co. Inc. (formerly Schering Plough and Organon); Moksha8; Naurex, Inc.; Neuronetics, Inc.; Novartis; Ortho-McNeil Pharmaceuticals (Johnson & Johnson; Janssen); Otsuka; Pamlab, L.L.C. (Nestle); Pfizer (formerly Wyeth-Ayerst Pharmaceuticals); Shire US Inc.; Sunovion Pharmaceuticals, Inc.; Takeda and Trius Therapeutical, Inc. Dr. Thase receives grant funding from the Agency for Healthcare Research and Quality; Alkermes; AssureRx; Avanir; Forest Pharmaceuticals; Janssen; National Institute of Mental Health and Otsuka Pharmaceuticals. He has equity holdings in MedAvante, Inc. and receives royalty income from American Psychiatric Foundation, Inc., Guilford Publications, Herald House, Oxford University Press and W.W. Norton & Company. His wife is employed as the Group Scientific Director for Peloton Advantage which does business with Pfizer. Dr Kocsis has received research grants and contracts from AHRQ, NIMH, Pritzker Consortium, Qatar National Research Fund and Rockefeller Treatment Development Fund. He holds an Elan Patent (No. 8,853,279), entitled ‘Method for Determining Sensitivity or Resistance to Compounds That Activate the Brain Serotonin System’. Dr Sylvia was a shareholder in Concordant Rater Systems and serves as a consultant for United Biosource Corporation and Clintara. She receives royalties from New Harbinger. Dr Nierenberg is a consultant for Abbott Laboratories, Alkermes, American Psychiatric Association, Appliance Computing Inc. (Mindsite), Basliea, Brain Cells, Inc., Brandeis University, Bristol-Myers Squibb, Clintara, Corcept, Dey Pharmaceuticals, Dainippon Sumitomo (now Sunovion), Eli Lilly and Company, EpiQ, L.P./Mylan Inc., Forest, Genaissance, Genentech, GlaxoSmithKline, Healthcare Global Village, Hoffman LaRoche, Infomedic, Intra-Cellular Therapies, Lundbeck, Janssen Pharmaceutica, Jazz Pharmaceuticals, Medavante, Merck, Methylation Sciences, NeuroRx, Naurex, Novartis, Pamlab, Parexel, Pfizer, PGx Health, Otsuka, Ridge Diagnostics Shire, Schering-Plough, Somerset, Sunovion, Takeda Pharmaceuticals, Targacept and Teva; consulted through the MGH Clinical Trials Network and Institute (CTNI) for AstraZeneca, Brain Cells, Inc, Dainippon Sumitomo/Sepracor, Johnson and Johnson, Labopharm, Merck, Methylation Science, Novartis, PGx Health, Shire, Schering-Plough, Targacept and Takeda/Lundbeck Pharmaceuticals. He is a stakeholder in Appliance Computing, Inc. (MindSite), Brain Cells, Inc. and Medavante. He receives research support from American Foundation for Suicide Prevention, AHRQ, Brain and Behavior Research Foundation, Bristol-Myers Squibb, Cederroth, Cephalon, Cyberonics, Elan, Eli Lilly, Forest, GlaxoSmithKline, Intra-Cellular Therapies, Janssen Pharmaceutica, Lichtwer Pharma, Marriott Foundation, Mylan, NIMH, Pamlab, PCORI, Pfizer Pharmaceuticals, Shire, Stanley Foundation, Takeda and Wyeth-Ayerst. Honoraria include Belvoir Publishing, University of Texas Southwestern Dallas, Brandeis University, Bristol-Myers Squibb, Hillside Hospital, American Drug Utilization Review, American Society for Clinical Psychopharmacology, Baystate Medical Center, Columbia University, CRICO, Dartmouth Medical School, Health New England, Harold Grinspoon Charitable Foundation, IMEDEX, International Society for Bipolar Disorder, Israel Society for Biological Psychiatry, Johns Hopkins University, MJ Consulting, New York State, Medscape, MBL Publishing, MGH Psychiatry Academy, National Association of Continuing Education, Physicians Postgraduate Press, SUNY Buffalo, University of Wisconsin, University of Pisa, University of Michigan, University of Miami, University of Wisconsin at Madison, APSARD, ISBD, SciMed, Slack Publishing and Wolters Klower Publishing, ASCP, NCDEU, Rush Medical College, Yale University School of Medicine, NNDC, Nova Southeastern University, NAMI, Institute of Medicine, CME Institute, ISCTM, World Congress on Brain Behavior and Emotion, Congress of the Hellenic Society for Basic and Clinical Pharmacology and ADAA. He has copyright joint ownership with MGH for Structured Clinical Interview for MADRS and Clinical Positive Affect Scale.
Funding
The Bipolar CHOICE study was funded by the Agency for Healthcare Research and Quality (AHRQ; 1R01HS019371-01). Marco Antonio Caldieraro was supported by Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES), program 227—Pesquisa Pós-doutoral no Exterior (grant 88881.120434/2016-01).
