Abstract
Serotonergic neurotransmitter systems have been implicated in the aetiogenesis of obsessional symptoms since the 1960s, although still today a coherent neurochemical/ neuroendocrine explanatory model for serotonergic dysfunction in obsessive-compulsive disorder (OCD) has not been accepted [1]. This is despite the now well-established and validated role of SRIs (serotonin re-uptake inhibitors) [2] – including the tricyclic antidepressant clomipramine and the selective serotonin re-uptake inhibitors – as first-line agents for the pharmacological treatment of OCD, attested to in efficacy by numerous controlled trials, including large multicentre collaborative studies [3]. SRIs are, however, found to be effective as monotherapy in only 50–60% of OCD patients [4]. In this context, there is growing evidence to support treatment augmentation in SRI-resistant OCD patients with antipsychotics [5–7] which are likely to afford clinical improvement in around 50% of resistant patients [8].
The literature on antipsychotic augmentation of SRI treatment in OCD also serves potentially to validate subtypes based upon differences in treatment response and other variables. Studies of combination SRI/dopamine antagonist treatments more robustly benefiting specific subtypes of OCD, have focused most extensively on OCD with comorbid tic-disorder. Such interest has been founded on the established phenomenological, genetic, neurochemical and neuroanatomic overlap between OCD and Tourette's syndrome, and preclinical evidence pointing to interactions between 5-HT and dopamine systems in the brain [6].
This article reviews antipsychotic augmentation studies of both conventional and newer antipsychotic agents in OCD, and elucidates some of the subgroup associations which have been informed by this literature.
Methods
A computerized literature search (MEDLINE: 1966 to December 2003, EMBASE: 1982 to December 2003) was performed to locate relevant literature. Search terms included ‘obsessive-compulsive’, ‘antipsychotic’ and ‘subtypes’; no restrictions were imposed on searches. Additionally relevant literature was retrieved from reference lists of scrutinized articles.
Results
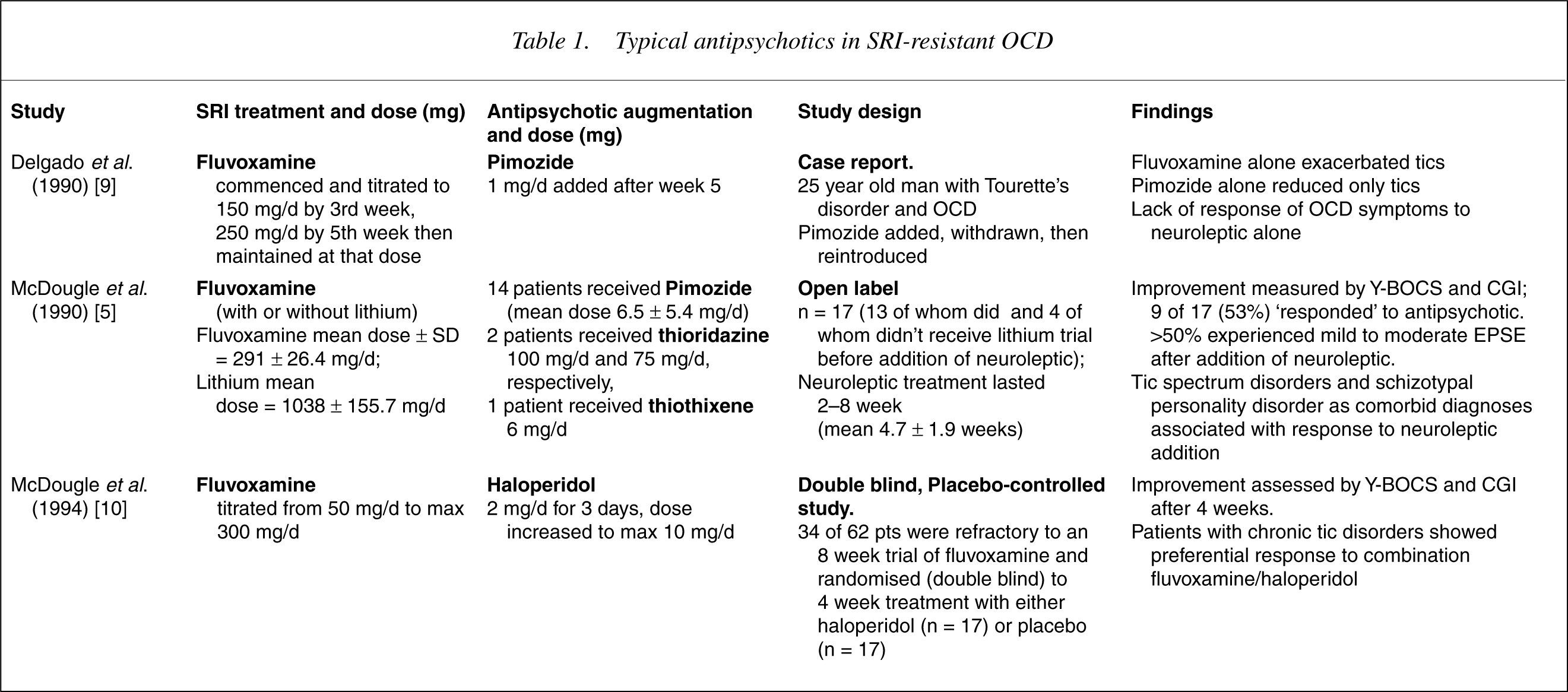
Literature examining the role of typical antipsychotics in OCD comprises several studies of varying methodologies, including a case report, an open label study of pimozide, a placebo-controlled study of haloperidol, and a related retrospective case-controlled study of SRI treatment effects in relation to comorbidity of tic disorder (see Table 1 and below). Subsequent investigations into atypical antipsychotic augmentation of SRIs have developed from those initial findings. These more recent studies (see Table 2 and below) comprise: case reports; 11 open-label studies (4 of risperidone, 6 of olanzapine, 1 of quetiapine); one double blinded randomized controlled trial of risperidone; one single blinded randomized controlled trial of quetiapine; and one uncontrolled retrospective trial of quetiapine. The majority of the identified studies recruited subjects from specialized outpatient clinic settings, although there was sometimes a mixture of inpatients and outpatients within studies, notably in the earlier typical antipsychotic studies.
Typical antipsychotics in SRI-resistant OCD
Atypical antipsychotics in SRI-resistant OCD


Typical antipsychotics (refer Table 1)
A case report of OCD with tic disorder in which small doses of the dopamine antagonist pimozide were added to an SRI, with subsequent discontinuation of both sequentially [9], showed that pimozide alone reduced only tics, whereas the combination of SRI and pimozide seemed necessary for improvement of OCD symptoms. This study highlights the notion of a dopaminedependent subtype of OCD with unresponsiveness to SRIs alone, but benefit from dopamine antagonism. Another early open label study examined the efficacy of the addition of low-dose pimozide to SRIs in refractory patients [5]. This study demonstrated a preferential augmentation response in subjects with schizotypal personality disorder and tic spectrum disorders as comorbid diagnoses.
A placebo-controlled investigation of haloperidol in 34 SRI-refractory patients [10] found superiority of fluvoxamine-haloperidol over fluvoxamine-placebo in reducing the severity of obsessive compulsive symptoms as measured by the Yale-Brown Obsessive Compulsive Scale (Y-BOCS), with overall response rates of 65% at 4 weeks. Again, this study supports the hypothesis that OCD patients with a comorbid chronic tic disorder constitute a subtype requiring combined SRI/antipsychotic therapy. A related retrospective case-controlled analysis by Mc Dougle et al. [11] found that fluvoxamine monotherapy was less effective in OCD patients with, than in those without, comorbid chronic tics.
Atypical antipsychotics
Studies examining atypical antipsychotic augmentation of SRIs in OCD are presented in 7], the response was not confined to patients who had a tic disorder or schizotypal personality disorder. In one of the open trials of olanzapine [12], comorbid schizotypal personality disorder was significantly associated with response whereas a comorbid tic disorder was not. The quetiapine studies did not differentiate any particular subtype of patient being more or less likely to respond.
Discussion
The studies reviewed here show that typical antipsychotics have generally been effective for augmentation of SRI-resistant OCD accompanied by tics, but have not been shown to augment SRI action in the absence of tics. Typical antipsychotics have also been found to be effective when used to augment SRI treatment in OCD with schizotypal personality disorder. However, studies of the typical agents are few in number and limited in design (viz case report, open label study). The only double-blind placebo-controlled data (for haloperidol) is still limited by small sample size (only two patients out of 17 with comorbid schizotypal personality disorder).
There is more substantial evidence supporting a role for atypical antipsychotics as augmentation, but the published studies should be interpreted with some caution. Obvious limitations in appraising this literature stem from paucity of controlled studies. The majority of the studies are open-label series with small samples (largest sample 23 patients), and improvements that have been demonstrated only in the short term (maximum periods of review being 3 months). Also, different studies use different cutoff scores for response rates (e.g. 50% decrease in Y-BOCS in the Weiss et al. study [12], compared to 35% in the study of McDougle et al. [7]). Differences in the criteria for treatment resistance exist also between the studies, such that inclusion criteria in some studies was a failure of response to at least 3 adequate SRI treatments, whereas in many of the other trials subjects had failed only one SRI trial. Thus, comparison across studies or pooling of data is precluded.
Clinical considerations
Is monotherapy with antipsychotics of any value in OCD?
We are aware of no published studies, either openlabel or controlled, of typical or atypical antipsychotic monotherapy demonstrating efficacy in treating OCD. It is unlikely even that clozapine, which McDougle et al. examined in an open-label investigation of 20 treatment resistant OCD patients for 10 weeks [24], produces significant alteration of OCD symptoms in refractory patients. This appears to be the case even in ‘psychotic’ OCD where insight is lacking. Indeed, there is emerging evidence that atypical antipsychotics used alone can exacerbate or cause de novo OC symptoms [25–27].
Maintenance of augmentation therapy
While there are few data on the efficacy of antipsychotic augmentation over the long-term, there is initial evidence (from retrospective data) that in patients who respond to antipsychotic augmentation, this strategy should be maintained over prolonged periods to prevent relapse [28].
Dosing
McDonough and Kennedy in their review of pharmacological management of OCD [29], suggested augmentation dose ranges for antipsychotics in keeping with those for psychosis (e.g. risperidone, 2–6 mg daily; olanzapine 5–20 mg daily), but they note that response often tends to occur at lower doses, and that a 4 week therapeutic trial at the maximum tolerated dose is required.
Are there concerns in combining SRIs and antipsychotics?
Generally the combination is well tolerated [29]. Caution is required with dosing of risperidone, whose breakdown by the cytochrome P450 enzymes is potentially inhibited by SRIs, leading to elevation of plasma risperidone levels [30]. Thus, added vigilance for sideeffects such as postural hypotension and extrapyramidal side-effects is required. Clinically significant interactions include the potentially toxic interaction between fluvoxamine and clozapine (serum levels of clozapine are raised by fluvoxamine) [31], and the combined effect on decreased platelet aggregation of both SRIs and antipsychotics.
Mechanism of action of antipsychotic augmentation of SRIs in OCD
Pharmacokinetic effects
Alterations of the blood levels of SRIs mediated by antipsychotics has been suggested as a possible contributory mechanism in the effect of augmentation. Against this, it has been noted that quetiapine, unlike risperidone, is a only a weak inhibitor of the cytochrome P450 isoenzymes [32] with a clinically irrelevant effect in vivo. Thus, pharmacokinetic interactions are unlikely to account fully for the clinical effect of augmentation, at least with quetiapine and SRIs [22].
Pharmacodynamic effects
Evidence for the pharmacodynamic mechanisms of action of the atypical agents in OCD has been gathered empirically by examining group and specific agent effects, and relating these to known receptor binding profiles of agents.
As a group, the efficacy of atypical antipsychotics as an adjunct to SRIs in refractory OCD may be due to direct dopamine – D2 blockade [32] separate from or together with antagonism of 5-HT2 receptors. It has been hypothesized that patients with SRI-refractory OCD have additional abnormalities in dopaminergic function that require augmentation with dopamine-blocking agents [33]. For example, risperidone's effect at low dose may be due to the antagonism of dopamine receptors, and that effect may be dose dependent [34]; the deleterious effect sometimes observed with high dose risperidone or clozapine may be related to decreased serotonergic neurotransmission [35].
The fact that in some instances atypical antipsychotic augmentation strategies have been effective in patients refractory to a combination of SRI and typical neuroleptics might indeed be explained by the strong antagonistic effects on serotonin receptor subtypes (5-HT2A, 5-HT2C, 5-HT1A, 5-HT1D and 5-HT7) by the atypical agent. These receptors are down-regulated by chronic SRI use, and consequently the blockade of serotonin receptors by atypical antipsychotics may potentiate the action of the SRI [21]. Yet, alone, other drugs with relatively potent 5-HT2 antagonism such as trazodone hydrochloride, have not proven useful in the treatment of OCD [36].
Subtypes of OCD
Can these concepts and findings inform and expand our understanding of meaningful OCD subtypes? The heterogeneity in response of OCD patients to modern treatments has supported the hypothesis of OCD being an heterogeneous condition comprising a number of subtypes, some of which are beginning to be more clearly defined [37]. Lochner and Stein, in their review of the heterogeneity of OCD [38], highlight some current approaches to the conceptualization of OCD subtypes. Subgroups on the basis of OCD symptoms, age of onset (earlier age aggregating with familial forms), presence of tic disorders and comorbid psychiatric disorders have been suggested, and there are potential treatment implications [39]. For example, it is known that OCD with comorbid tics is characterized by earlier onset, a particular range of OCD symptoms, and worse response to SRIs [38]. There has also been interest over the past decade in the concept of ‘psychotic OCD’, or, according to DSM-IV ‘OCD without insight’, as a meaningful subtype with putative poorer response to treatment [40], [41]. Further study of clinical characteristics such as treatment response and illness trajectory and outcome will likely help further delineate meaningful subtypes.
Conclusions
There is growing evidence confirming the efficacy of antipsychotic augmentation in SRI-refractory OCD. While the published literature is supportive, further controlled studies are warranted, specifically examining the efficacy of the range of atypical agents that could be used in augmentation, the doses and durations of treatments, and predictors of response.
Despite the inherent plausibility and face validity of some of the pharmacodynamic theorizations of how antipsychotics influence dopaminergic and serotonergic mediation in OCD, these conceptualizations and hypotheses remain broad, integrative schemata which are still speculative. The complexities of the exact mechanisms of action remain undetermined and additional research is warranted.
In the future we can expect further advances in the characterization of OCD subtypes on the basis of psychobiology and phenomenology. Specific subtypes of OCD based on treatment response are beginning to be elucidated, and provide a framework from which further to conceptualize, integrate, and delineate what we understand of the variations in symptomatology, neurobiology and treatment response in OCD.
