Abstract
Previous neuropsychological investigations of first episode schizophrenia (FES) have provided clear evidence that cognitive deficits are present at onset [1–4] and remain relatively stable during the early course of the disorder [5], [6]. The majority of FES studies also suggest that the profile of cognitive functioning is similar to that of chronic schizophrenia patients, with generalized impairment across the majority of cognitive domains overlaid with more selective impairments in memory and learning, executive functioning, attention and speed of information processing [1], [4],[7–9].
However, the question of whether schizophrenia can be differentiated from other psychotic disorders on the basis of neuropsychological test performance remains inconclusive. Few studies have addressed this issue and most have focused exclusively on comparisons between schizophrenia and psychotic affective disorders. Of these, the majority suggest that psychotic affective disorder patients show similar impairment to schizophrenia patients on a broad range of cognitive tasks, irrespective of illness duration [10–14]. These findings are also consistent with research indicating that schizophrenia and psychotic affective disorders share similar genetic vulnerability and clinical syndrome patterns [15–17]. Other studies, however, have found different patterns of cognitive functioning among diagnostic groups [18–20]. For instance, Motjabai et al. [19] found that FES patients showed significantly poorer attention, concentration and mental tracking skills than those with psychotic affective disorders, whereas Verdoux et al. [20] reported that individuals with schizophrenia performed significantly more poorly on tasks of memory functioning than those with bipolar affective disorder, non-schizophrenic psychotic disorder or major depression.
This disparity in findings is problematic and reflects wide variations in study design and methodology. For instance, some studies assessed stabilized community outpatients two years after their first episode [19], whereas others assessed acutely ill inpatients [10]. Some used comprehensive assessment batteries [10], [19] whereas others have focused on specific areas of functioning such as executive and memory abilities [12], [13], [20]. Medication status of groups also varied considerably, from those predominantly prescribed conventional antipsychotics [10] to those receiving a heterogenous mix of pharmacological treatments [11], [13]. Moreover, to our knowledge Verdoux et al. [20] are the only group to date to examine the cognitive functioning of other psychotic disorders in addition to schizophrenia and affective psychoses, although their sample comprised a mixture of firstepisode and chronic patients.
This report attempts to overcome some of the design limitations of previous studies by using a comprehensive battery of tests to compare the cognitive profiles of three first-episode psychosis (FEP) diagnostic groups against that of a healthy control group. Two important features of the present study were the young age of the clinical sample and the fact that the majority were receiving atypical antipsychotic medication. There have been few neuropsychological studies of adolescents and young people with psychosis and the majority have focused on schizophrenia alone. Results of these suggest that the cognitive profile of adolescent-onset schizophrenia is comparable to that of adult-onset schizophrenia [21], [22]. However, other studies have indicated that earlier age of onset may be associated with more severe cognitive deficits [23] and poorer outcome [24]. More importantly, assessing young people at the earliest stages of psychotic illness increases the probability of identifying primary cognitive deficits, while minimizing the confounding effect of factors such as duration of illness and adverse life events. Second, to our knowledge only one recent study has investigated the neuropsychological functioning of FEP patients receiving atypical antipsychotic medications [25], despite the clinical advantage of these drugs over conventional antipsychotics due to their improved cognitive side-effect profile [26]. Clinical trials with mainly chronic, treatment-resistant samples have also highlighted the superior effects of atypical antipsychotics on cognitive functioning [27], [28].
Our aims in this study are to determine whether the pattern and extent of neuropsychological deficits varied as a result of diagnostic group membership and to determine the contribution of demographic and clinical factors to neuropsychological performance among FEP patients. On the basis of previous research [1], [8], [18] we predicted that individuals with psychosis would perform more poorly in the areas of attention, memory, executive function and speeded processing than healthy agematched controls, but that the schizophrenia group would show significantly more impaired performance on these measures than either of the other two psychotic groups. We also hypothesized that younger age of onset and higher levels of antipsychotic medication would be associated with poorer cognitive functioning in FEP patients.
Materials and method
Subjects
Subjects were participants in the Western Sydney First Episode Psychosis Project. A full description of recruitment and methodology is reported in Harris et al. [29]. A total of 105 FEP patients consented to take part in the present study, but 11 were subsequently excluded following evidence of previously undisclosed head injury or neurological disease (n = 4), lack of clear evidence of psychosis (n = 2), or inability to co-operate with the assessment process (n = 5). A further 11 patients did not meet criteria for inclusion in the diagnostic groups outlined below.
The final sample of 83 FEP patients consisted of 59 community outpatients and 24 voluntary inpatients, who were subsequently assigned to one of three diagnostic groups according to DSM-IV and ICD-10 criteria: schizophrenia (schizophrenia, n = 41; schizophreniform disorder, n = 11); affective disorders (bipolar affective disorder, n = 12; mania with psychosis, n = 1; major depression with psychotic features, n = 5) and substance-induced psychosis (n = 13).
At the time of assessment, 88% (n = 73) were receiving atypical antipsychotic medication, with a mean chlorpromazine-equivalent dose of 258.0 mg/day (SD = 194.0 mg/day) [30]. Two people were prescribed conventional antipsychotics (mean = 75 mg/day, SD = 35.4 mg/ day, in addition to a mood stabilizer) and eight were not receiving any antipsychotic medication. In all, 14 people were prescribed mood stabilizers and 12 were receiving antidepressant medication. A minority of the group were prescribed benzodiazepines (n = 6) or anticholinergic medication (n = 3).
Healthy control subjects were recruited from local high schools, vocational education centres and a first-year undergraduate psychology course. In addition to the exclusion criteria applicable to FEP subjects, control subjects were excluded if they reported any personal or family history of mental illness. After complete description of the study to all subjects, written informed consent was obtained. Additional parental/ guardian written consent was obtained for individuals under 18 years. Table 1 presents the demographic details of the four groups.
Demographic and clinical characteristics of first episode psychosis patients and healthy controls
Psychiatric assessment
The presence and severity of psychotic symptoms was evaluated by the Positive and Negative Syndrome Scale (PANSS) [31] which provides scores on three subscales: positive symptoms, negative symptoms and general psychopathology. The extent of mood disturbance was evaluated with the Calgary Depression Rating Scale [32] and the Young Mania Rating Scale [33]. A more detailed description of the psychiatric measures and assessment process can be found in Harris et al. [29].
Neuropsychological assessment
A comprehensive neuropsychological battery of tests was administered, from which selected measures from individual tests were combined to form 10 broad cognitive domains (Table 2). Decisions about grouping individual measures into composite functional domains were based on a priori assessment of content validity of individual tests [34] and with reference to previous studies using similar procedures [1], [9], [10], [35]. Due to the age range of participants in the study, child (< 16 years, 11 months) and adult (< 16 years, 11 months) versions of individual tests within the neuropsychological battery were administered as appropriate.
Neuropsychological test battery with tests grouped by domain
In order to calculate composite scores for each cognitive domain, the healthy control group was divided into two age brackets (< 16: 11 years, and < 16: 11 years) and mean raw scores for individual tests were calculated for both age groups. On tests where higher scores indicated impairment, scores were transformed (reverse-scored) so that high scores always indicated better functioning. Composite scores for each cognitive domain were calculated for FEP patients by converting individual raw scores to standardized z-scores based on the ageappropriate control group mean scores. Z-scores were then summed and averaged across tests in each domain to provide a single score.
To limit the confounding effects of acute symptomatology and conceptual disorganization on test performance, assessments were commenced when FEP patients had achieved stabilization of acute symptoms, as indicated by a score of less than 5 on the ‘conceptual disorganization’ item of the PANSS positive subscale. In addition, in the case of voluntary inpatients, assessments were conducted as close to discharge as possible. In order to ensure reliability of associations between levels of symptomatology and cognitive functioning, the neuropsychological battery was generally administered within 1–2 days of symptom ratings, with a maximum time interval of 14 days. Tests were administered by Masters level neuropsychologists and psychologists and given in the same order to all subjects. The battery of tests was typically completed in one session of 3–4 h duration, with rest breaks given as needed. All tests were administered and scored according to published standardized instructions.
Statistical analyses
Initial screening of data indicated violations of multivariate normality assumptions on three cognitive domains (attention/vigilance, verbal acquisition and verbal recall) due to the presence of univariate outliers and negatively skewed distributions of sampling means. Normalization of these measures was achieved via logarithmic transformation prior to further analysis. Missing values occurred in only seven out of 114 cases and no more than two missing values were recorded for any one individual. As multivariate analysis deletes cases with missing values, which potentially would affect the representativeness of the sample, missing values were replaced with the age-appropriate (< 16 years, 11 months or < 16 years, 11 months) group mean.
Multivariate analysis of variance (MANOVA) was used as the primary test of group differences, with diagnostic group (schizophrenia, affective disorders, substance-induced psychosis, controls) as the betweengroup factor and the 10 cognitive domains as dependent variables. Pillai's criterion was used as the significance test, due to the unequal size of the groups. To control for the effect of potentially confounding variables, multivariate analysis of covariance (MANCOVA) was employed where indicated. Univariate analyses of variance were performed for demographic variables and for individual summary measures (Bonferroni-adjusted for inflated type 1 error rate), followed by Bonferroni-adjusted post-hoc comparisons where significant.
To evaluate whether the profiles of the FEP groups differed in areas of selective impairment, difference scores were calculated by subtracting the mean of each cognitive domain from the mean of the remaining nine domains. Mean profile contrasts were performed (GLM procedure, simple contrast method) to test whether diagnostic group profiles deviated from flatness relative to the healthy control group (where the expected value of the mean contrast is zero). Bonferroni adjustment was used to control for inflated type 1 error rate (critical p = 0.005).
Step-wise multiple regressions were additionally performed for cognitive domains on which significant between-group differences emerged, to determine the relative contribution of demographic and clinical factors. Age, premorbid IQ, Calgary Depression Scale score, Young Mania Rating Scale score, positive, negative and general scores of the Positive and Negative Syndrome Scale (PANSS) and chlorpromazine-equivalent medication dosage were entered as independent variables, with cognitive domain as the dependent variable. All statistical tests were carried out using the Statistical Package for the Social Sciences (SPSS), Version 10.0.
Results
Demographic and clinical characteristics of the FEP and healthy control groups are provided in Table 1. After Bonferroni correction for inflated type 1 error rate (p = 0.016) there were no significant betweengroup differences in years of education or estimated premorbid intellectual functioning. However, groups differed in age at time of assessment, with the healthy control group demonstrating a younger mean age than either the schizophrenia or substance-induced psychoses groups. Age was therefore entered as a covariate in subsequent analyses to control for possible confounding effects.
Figure 1 shows the mean z-score profile of cognitive functioning for the three FEP groups relative to the healthy control group. By definition, the healthy control group mean is represented by the zero line with a standard deviation of one for all cognitive domains. Group means and standard deviations for individual tests comprising the composite domains are provided in Table 3. Overall MANCOVA controlling for age revealed a significant difference in neuropsychological performance among groups, Pillai's criterion, F(30,306) = 3.16, p < 0.001, and subsequent univariate analyses of covariance (Bonferroni corrected, p = 0.005), indicated significant between-group differences in all cognitive domains except for working memory and visuospatial ability (Table 3).
Cognitive domain and individual test mean and standard deviation scores for first-episode psychosis and healthy control groups
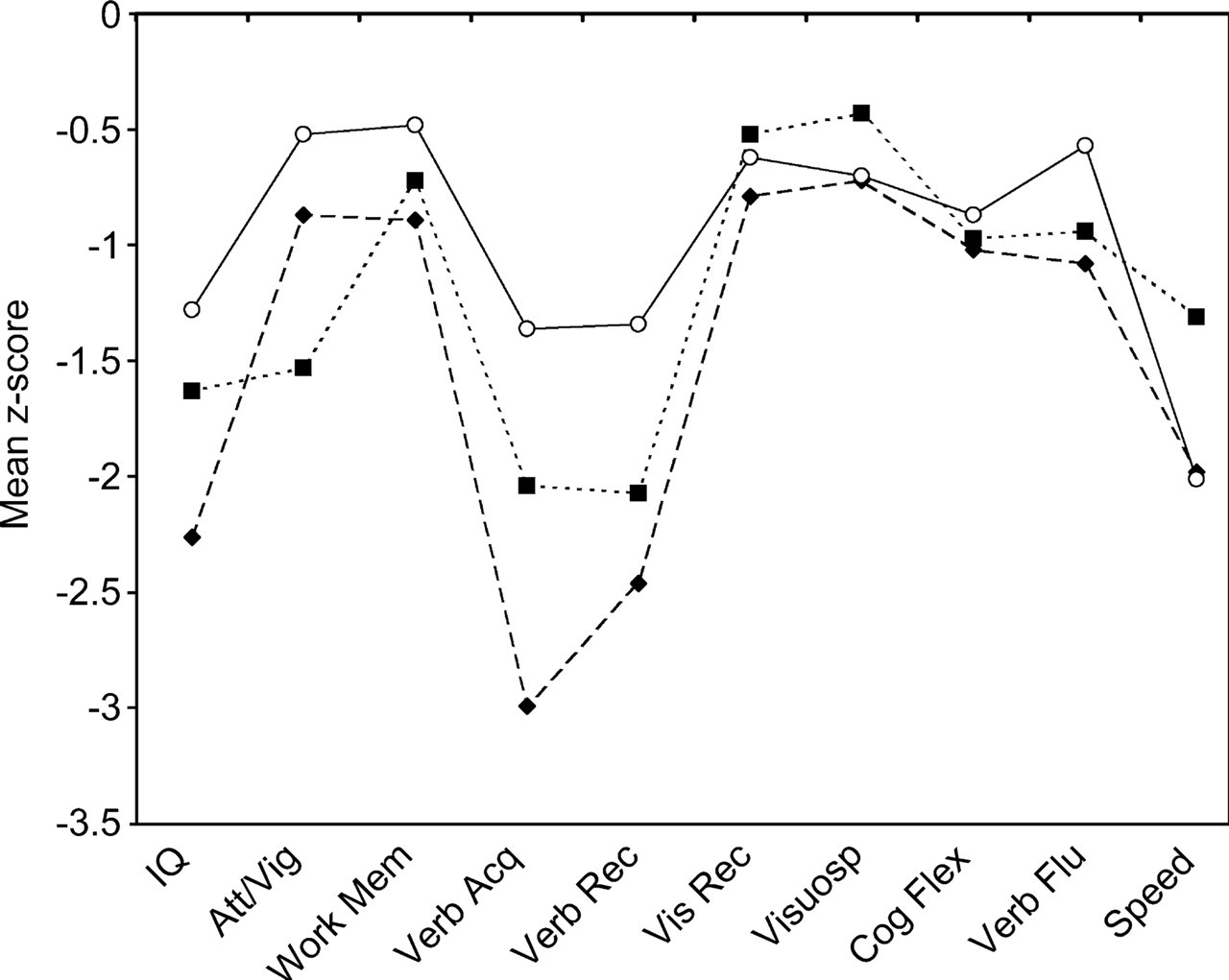
Mean z-score cognitive domain profiles of the three first episode psychosis diagnostic groups compared to control group (represented by zero line, with standard deviation of 1).▪, substance-induced psychosis (n = 13); ♦, schizophrenia (n = 52); ○, affective disorders (n = 18).
Bonferroni-adjusted post-hoc comparisons revealed that these differences mainly lay between the healthy control group and the three FEP groups. All three psychotic groups performed significantly more poorly than healthy controls in four cognitive domains: current IQ (schizophrenia, p < 0.001; affective disorders, p = 0.034; substance-induced psychosis, p = 0.036); verbal acquisition (schizophrenia, p < 0.001; affective disorders, p = 0.012; substance-induced psychosis, p = 0.001); verbal recall (schizophrenia, p < 0.001; affective disorders, p = 0.028; substance-induced psychosis, p < 0.001); and speeded processing (schizophrenia, p < 0.001; affective disorders, p < 0.001; substance-induced psychosis, p = 0.010). In addition the schizophrenia group demonstrated significantly poorer performance than healthy controls on a further four domains: visual recall (p = 0.001); cognitive flexibility (p < 0.001); attention/vigilance (p = 0.018) and verbal fluency (p < 0.001); whereas the substanceinduced psychosis group performed more poorly than healthy controls on three of these domains: cognitive flexibility (p = 0.024); attention/ vigilance (p = 0.006); and verbal fluency (p = 0.034).
Visual examination of the neuropsychological profiles of the three FEP groups (Fig. 1) suggested that the schizophrenia group performed more poorly on the majority of cognitive domains compared to the other two FEP groups. However, post-hoc analyses (Bonferroniadjusted) revealed that these differences were significant only on measures of verbal learning and memory, with the schizophrenia group performing significantly more poorly than the affective disorders group on verbal acquisition (p = 0.001) and verbal recall (p = 0.015). Between-group differences on the remaining cognitive domains failed to reach significance.
After Bonferroni adjustment (critical p = 0.005), results of planned mean profile contrasts indicated significant deviations from flatness in profile among the diagnostic groups relative to the healthy controls on seven out of 10 cognitive domains: current IQ, F(3,110) = 5.22, p = 0.00; attention/vigilance, F(3,110) = 5.10, p = 0.001; verbal acquisition, F(3,110) = 13.96, p < 0.001; verbal recall, F(3,110) = 5.63, p = 0.00; visual recall, F(3,110) = 5.38, p = 0.002; visuospatial ability, F(3,110) = 9.41, p < 0.001; speeded processing, F(3,110) = 4.60, p = 0.004. Relative to their mean performance on other domains, the schizophrenia group showed specific weaknesses in current IQ (p < 0.001), verbal learning (p < 0.001) and verbal recall (p < 0.001) and relative strengths in attention/vigilance (p = 0.004), visual recall (p < 0.001) and visuospatial ability (p < 0.001). The affective disorders group demonstrated specific weakness in speeded processing only (p = 0.001), whereas the substance-induced group demonstrated no specific areas of weakness, but a relative strength in visuospatial ability (p = 0.001).
Mean effect sizes were calculated for the three diagnostic groups (in z-score units, reflecting the number of standard deviations below the healthy control group mean). The schizophrenia group demonstrated the most impaired performance, with an overall profile mean that fell 1.6 standard deviations below that of healthy controls. In comparison, the substance-induced psychosis and affective disorders groups, respectively, demonstrated overall profile means of 1.3 and 0.9 standard deviations below that of the healthy control group.
Results from the regression analyses are presented in Table 4. Variables that were excluded in the equations (i.e. change in explained variance [R2] failed to reach significance at the 5% level) are not reported. Premorbid IQ was the strongest predictor of all cognitive domains examined (excluding visual recall, where the PANSS negative score entered the equation first), explaining between 6% and 31% of the variance. PANSS negative was also an independent predictor of current IQ (8%), verbal acquisition (7%), verbal recall (7%) and visual recall (11%). Chlorpromazine-equivalent dosage was an independent predictor of verbal acquisition (6%), verbal recall (6%), cognitive flexibility (6%) and speeded processing (5%).
Stepwise regression analyses for predicting cognitive domain scores†
Discussion
Few studies have examined the neuropsychology of broadly defined first episode psychosis. This study examined the extent and nature of cognitive deficits in a large sample of young individuals in the earliest stages of psychotic illness. Our results provide evidence that although young people with psychosis exhibit significant cognitive deficits at onset of illness, individuals with schizophrenia demonstrate significantly greater impairment than other diagnostic groups across a broad range of cognitive domains when compared to healthy controls. Our findings also suggest that while the pattern of deficits is similar across diagnostic categories, areas of selective deficit may differentiate schizophrenia from psychotic affective disorders. Comparison of FEP diagnostic groups revealed significantly greater deficits on tasks of verbal learning and memory among individuals with schizophrenia compared to those with affective disorders, which is consistent with previous findings [20].
In the present study, individuals with schizophrenia under-performed healthy controls by an average of 1.6 standard deviations, and showed significantly impaired performance on measures of global intellectual functioning, attention/vigilance, verbal learning and memory, visual memory, speeded processing, cognitive flexibility and verbal fluency. In addition, measures of intellectual ability, verbal learning and verbal memory were identified as areas of specific weakness. This pattern of generalized impairment with specific weakness in verbal learning and memory is consistent with findings from previous FES studies [1],[8–10] and has also been documented in previous studies of adolescent-onset schizophrenia [22], [36], [37]. The finding of a specific weakness in overall intellectual functioning (WAIS-III or WISC-III full scale IQ score) relative to the healthy control group should be interpreted with caution, given that our healthy controls scored above the average range on this measure (see Table 4). This above-average performance by the control group was limited to measures of intellectual functioning only, and is common to previous studies [1], [3]. In fact, comparison of intellectual functioning scores for the schizophrenia group revealed a similar level of performance to that reported in previous FEP and chronic studies [3], [11], [25].
The performance of the affective disorders group in the present study is comparable to results of previous first episode studies of affective psychoses [10], [20]. However, the extent of impairment on speeded processing was quite surprising, given the short duration of illness and young age of the present sample. These findings do not appear to be confounded by medication effects, given that the majority of subjects were receiving atypical antipsychotic medications which not only have a more favourable side-effect profile than conventional neuroleptic medications but also have been associated with improved performance on tasks of reaction time [26], [38]. Similarly, although lithium has been associated with cognitive slowing [39] only five individuals in the affective disorders group were prescribed lithium and subsequent analysis confirmed no differences in speeded processing between those prescribed lithium and the remainder (data not shown). Additionally, no significant associations were observed between speeded processing and levels of symptomatology when these were examined for the affective disorders group alone, suggesting that the selective deficit observed in speeded processing was independent of symptom effects. This, together with the observation that cognitive slowing was not exclusive to the affective disorders group suggests that impaired speed of processing may be a significant, if non-specific, feature of the cognitive profile evident at onset of psychosis. Whether the cognitive profile of the present affective disorders group remains stable, or demonstrates improvement in line with periods of remission as previously observed [19], remains an issue to be explored in our longitudinal follow-up study.
The overall cognitive profile of the substance-induced psychosis group showed more resemblance to that of the schizophrenia group than the affective disorders group, demonstrating significant impairment on seven out of 10 cognitive domains compared to healthy controls. In general, their performance lay between that of the schizophrenia and affective disorders groups, suggesting that the observed differences may be more quantitative than qualitative in nature and would appear to support the hypothesis of a common underlying aetiology among the psychoses expressed along a continuum of illness severity [15]. However, it should also be noted that previous research indicates that approximately one-quarter of those initially diagnosed with non-specific psychoses receive a diagnosis of schizophrenia at follow-up [40], and only 45% of those with drug-induced psychoses continue to receive the same diagnosis at 24-month follow-up [41]. It is therefore feasible that in this study, the similarity in performance observed between the schizophrenia and substance-induced psychosis groups reflects some degree of diagnostic overlap and instability, with a percentage of subjects in the latter group likely to progress to a diagnosis of schizophrenia as their illness evolves.
The present finding that estimated premorbid intellectual functioning was the strongest predictor of neuropsychological performance in FEP patients on the majority of cognitive domains was not surprising and supports the hypothesis that higher premorbid intelligence may be a protective factor against more severe cognitive impairment [42]. Clinical factors such as negative symptomatology and medication dosage (chlorpromazine-equivalent levels) also contributed to performance on different domains of cognition. Negative symptoms were more predictive of cognitive impairments on tasks of learning and memory, whereas higher levels of medication were predictive of greater impairments on tasks of learning, executive functioning and speeded processing. These clinical factors may reflect the severity of psychotic illness, insofar as higher levels of negative symptomatology and/or medication dosage may be indicative of more severe and intractable illness. Positive symptomatology may not have been a significant contributing factor due to our assessment criteria of low levels of thought disorder (represented by a score of less than 5 on the ‘conceptual disorganization’ item of the PANSS positive subscale). In addition, our finding that levels of symptomatology and medication were stronger predictors of cognitive impairment than measures of depression and mania suggests that cognitive deficits are more strongly influenced by severity of psychotic illness rather than factors such as mood disturbance. This is in contrast to findings from previous studies where depression was found to be a more significant contributor to cognitive deficits than psychotic symptomatology [43], [44].
The results from the present study should be interpreted with several limitations in mind. First, it is possible that the smaller group sizes in the affective and substance-induced psychosis groups limited the statistical power of our analyses in observing between-group differences in the FEP groups. However, it should also be noted that both the size of individual groups and overall sample size in this study compares favourably with other neuropsychological studies of psychosis and similarities in neuropsychological functioning across psychotic disorders has been previously reported [10], [11]. Second, it is well-recognized that the interpretation of cognitive processes underlying neuropsychological performance should be treated with caution, given that many individual neuropsychological tests involve more than one cognitive process [4], [35]. Although the construction of the cognitive domains in the present study was guided by previous research and a priori theoretical and clinical considerations, we acknowledge previous concerns that the use of summary scores may obscure the true nature of the cognitive processes involved, due to the different cognitive demands and psychometric properties of individual tests within each domain [4]. On the other hand, we would suggest that using scores from more than one measure may be a more valid assessment of an area of functioning as it reduces the possibility of administration error or individual task properties confounding results. We would also argue that the comprehensiveness of the assessment battery employed in the present study allows for a more complete understanding of the neuropsychological profile of FEP.
In summary, results from the present study support previous evidence that tasks of verbal learning and memory distinguish schizophrenia and psychotic affective disorders even in the earliest stages of illness and that the two disorders can be differentiated on the basis of areas of selective deficit [10], [19], [20]. However, the present results also support the hypothesis of a continuum of illness severity, insofar as significant cognitive deficits are evident in all diagnostic groups at onset of psychosis, but appear most severe among those with schizophrenia [15], [21], [22], [36]. More generally, the extent of cognitive deficits observed in our young sample reinforces the need for early detection and intervention strategies, such as cognitive rehabilitation programmes, for individuals experiencing a first episode of psychosis. Finally, the significant impact of negative symptomatology on cognitive functioning also reinforces the need for the development of more effective pharmacotherapy for negative symptoms.
Footnotes
Acknowledgements
The authors thank Wayne Reid and Susanne Meares for their contributions to the development and implementation of this study and Mark Kneebone, Martyn Patfield, Megan Chambers and Susan Coleman for their assistance and cooperation.
This research was supported by a grant from the Centre for Mental Health (NSW Health, Australia).
