Abstract
Carbon steel is a candidate material for the disposal container of high-level nuclear waste in a deep geological repository in China, which will be subject to general corrosion and localised attack. In this work, the effects of Cl−, SO4 2– and CO3 2− concentrations on the general corrosion and crevice corrosion of carbon steel were studied by immersion tests and electrochemical measurements. The results show that Cl− promotes general corrosion, while SO4 2− and CO3 2− inhibit general corrosion. In a single ion solution, SO4 2− is the most obvious in promoting the crevice corrosion, which is different from the result of carbon steel in groundwater solution. Cl−, rather than SO4 2−, dominates the crevice corrosion process of carbon steel in groundwater solution.
This paper is part of a supplement on the 6th International Workshop on Long-Term Prediction of Corrosion Damage in Nuclear Waste Systems
Introduction
A clean and efficient energy, nuclear power has been widely used around the world, but the high-level radioactive waste (HLW) has become a major risk. The generally accepted strategy for dealing with HLW is deep underground burial (about 500 m down) in stable geological formations [1–4]. As early as 1985, China has also chosen the deep geological disposal as the main direction to address the high-level radioactive waste issue. Lots of preliminary works have been carried out ever since. The major activities of HLW program in China are as follows: (1) Siting and Site characterisation since 1986; (2) Backfill material study: Bentonite from Inner Mongolia; (3) Migration behaviour study of radionuclide; (4) Performance assessment; (5) Natural analogue study. According to China's relevant policies, three-step long-term plan for geological disposal of HLW in China: from 2006 to 2020, laboratory studies and site selection for HLW repository; during 2021 to 2040, underground in situ tests; from 2041 to 2050, repository construction.
The metal container is the first barrier to protect high-level radioactive waste against the leakage. At present, packaging material selection for geological disposal is different in variety countries because of different geological conditions. In short, current candidates for waste container material of the canister are carbon steel, cast iron, titanium, stainless steel, Hastelloy alloy, copper and copper alloys [5]. Owing to carbon steel has the characteristics of easy processing, low manufacturing cost, and can effectively contain nuclear waste for a long time, many countries (such as South Korea, Switzerland, Spain, etc.) have chosen carbon steel as the preferred material. In order to save costs, the United States put forward the second design scheme for the Yucca Mountain Repository, which used carbon steel A588 as outer packing container material [6].
According to the planned concept of China's HLW disposal program, it can be known that carbon steel is a candidate container material for the disposal of high-level nuclear waste in a deep geological repository in China. Carbon steel canisters in underground repository will be subject to general corrosion and localised attack during long-term geological disposal. Hornus and Rodr'guez [7] think that localised corrosion, such as crevice or pitting corrosion, is one of the most important degradation processes that will limit the lifetime of containers. Crevices will occur when HLW containers are in contact with supporting structures or with undersurface deposits, corrosion products, passive film destroyed, etc [7–9]. The corrosion degree will depend on the concentration of the groundwater in contact with the canister surface and, especially, the anions, such as Cl−, SO4 2− and CO3 2−. In this work, the effects of Cl−, SO4 2− and CO3 2− concentrations on the corrosion behaviour of carbon steel were studied by immersion test and electrochemical measurement.
Experimental
Carbon steel was used here and its chemical composition (wt-%) was: C 0.36, Si 0.08, Mn 0.41, P 0.36, S 0.009, N 0.05. Before immersion test, the sample surface was mechanically ground by silicon carbide paper progressively up to 150 grit. After drying in hot air, the sample was weighed and then stored in a desiccator. The crevice corrosion based on ASTM G48-11 was carried out on an artificial crevice assembly [10]. The crevice was composed of polytetrafluoroethylene (PTFE) and metal, as shown in Figure 1. To ensure reproducibility, at least three groups of parallel samples were run for each experiment.
The crevice sample.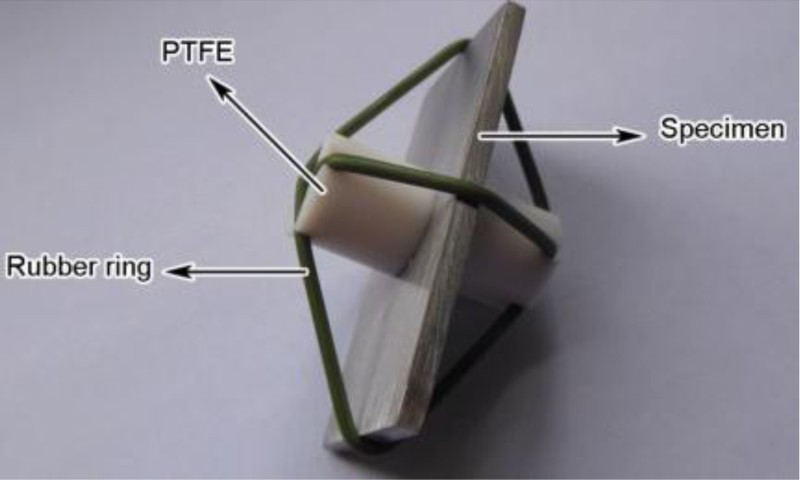
Chemical composition of the simulated groundwater (mg L−1).

Polarisation curves were measured using a CS350 electrochemical workstation. The electrochemical cell consisted of three electrodes: a working electrode (WE), a reference electrode (RE, Ag/AgCl), and a platinum sheet 20 mm × 20 mm was used as the counter electrode (CE). All the electrochemical tested specimens were enclosed with epoxy resin, leaving a working area of 1.0 cm2 and the test solution was 200 mL. Before the potentiodynamic polarisation curve measurement, the samples were immersed into the solution for 30 min to stabilise. The polarisation potential was swept from −0.25 to −0.4 V vs. the open circuit potential (OCP) at a scan rate of 0.5 mV s−1. All of the potentials reported here were measured with respect to the value of the Ag/AgCl (3 M KCl).
To remove the corrosion products, the specimens were pickled in 10% hydrochloric acid (HCl) inhibited with 10 gL−1 of hexamethylenetetramine (urotropine), rinsed subsequently in deionised water and acetone. After the experiments, the surface morphologies of samples were observed by Zeiss Evo 18 type of scanning electron microscope (SEM). The crevice corrosion depth was analysed by Dektak 150 Surface Profiler.
Results and discussion
Potentiodynamic polarisation studies
To clarify the effects of different anions on the corrosion behaviour of carbon steel in the groundwater solution, potentiodynamic polarisation curves of carbon steel were measured, as shown in Figure 2. All tests exhibit active corrosion behaviour, such that the anodic current density increased continuously with increasing potential. This behaviour typically indicates that no passive film formed on the specimen surface [11]. The fitting results are listed in Table 2. Compared with four polarisation curves and the results of Table 2, it can be observed that when Cl− was added to the solution, the corrosion rate experienced a slight increase. When SO4 2− or CO3 2− was added to the solution, the corrosion rate experienced a slight decrease. The results indicate that Cl− promoted the general corrosion of carbon steel, while the presence of SO4 2− or CO3 2− exhibited a suppressive effect.
Potentiodynamic polarisation curves of carbon steel in the solutions with different anion concentrations. Electrochemical test results of steels immersed in the solutions with different anion concentrations.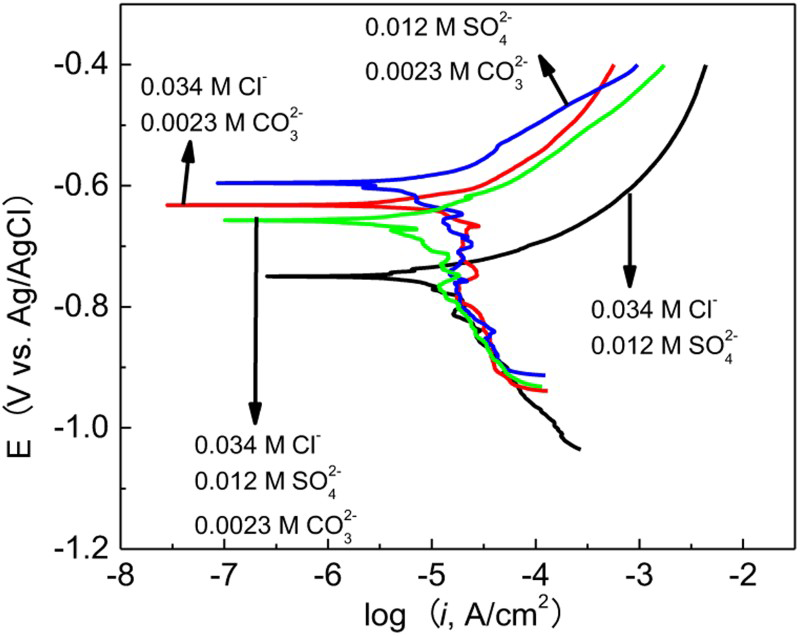

The crevice corrosion in a single anion solution
After the crevice corrosion, the corrosion products were removed, the steel surfaces were characterised using SEM and the surface profiler. Figures 3–5 show that the specimens exposed to different solutions have different surface appearances. The crevice corrosion attack occurred near the crevice mouth and inside the crevice of the carbon steel. It can be observed that the crevice corrosion of steel immersed in a single SO4 2− solution was the most serious, followed by that in a single Cl− solution. Carbon steel underwent slight corrosion on the surface inside the crevice in a single CO3 2− solution.
Surface morphologies of samples after removing the corrosion product film in single Cl− solutions with different concentration. Surface morphologies of samples after removing the corrosion product film in single SO4 2− solutions with different concentration. Surface morphologies of sample after removing the corrosion product film in single CO3 2− solutions with different concentration.
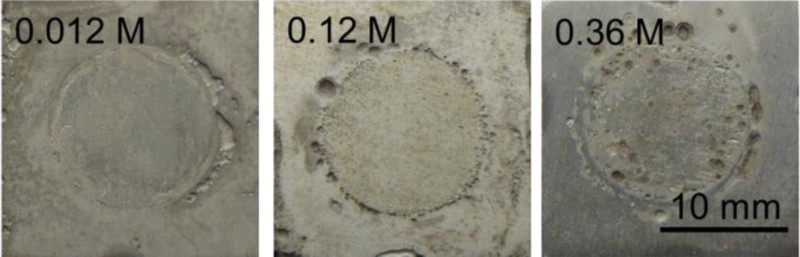

Figure 6 shows the crevice corrosion depth after the immersion tests in different single anion solutions. With the increase of Cl− concentration, the crevice corrosion depth exhibited an increased trend. The crevice depths of steel immersed in single SO4 2− solutions were higher than that in single Cl− solutions, but when the concentration of SO2− 4 was higher, the crevice depth decreased. The increase of CO3 2− concentration led to the decrease of crevice corrosion depth, which indicates that CO3 2− had an inhibiting influence on the crevice corrosion.
Crevice corrosion depths of carbon steels in different single anion solutions.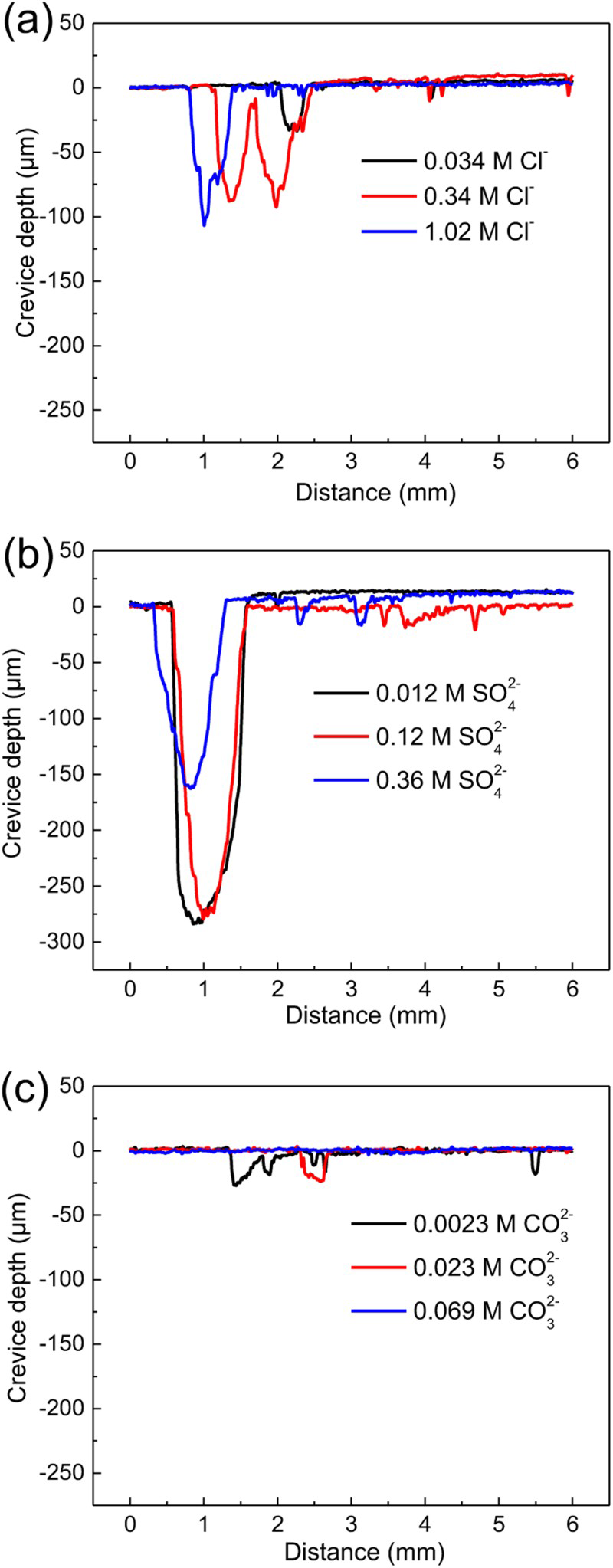
The crevice corrosion in the groundwater solution containing different concentrations of Cl−, SO4 2− and CO2− 3
Influence of Cl−
Figures 7 and 8 show SEM morphologies and crevice corrosion depth of samples after crevice corrosion immersion tests in the groundwater solution containing different Cl− concentrations, respectively. The corrosion attack was different inside the crevice, as shown in Figure 7. With the increase of Cl− concentration, the crevice corrosion depth decreased initially then increased gradually. When the Cl− concentration was higher than 3.4 × 10−2 M, the sample occurred serious crevice corrosion.
Surface morphologies inside the crevice after removing the corrosion product film in groundwater solution containing different Cl− concentrations. Crevice corrosion depth of steel in the groundwater solution as a function of Cl− concentration.
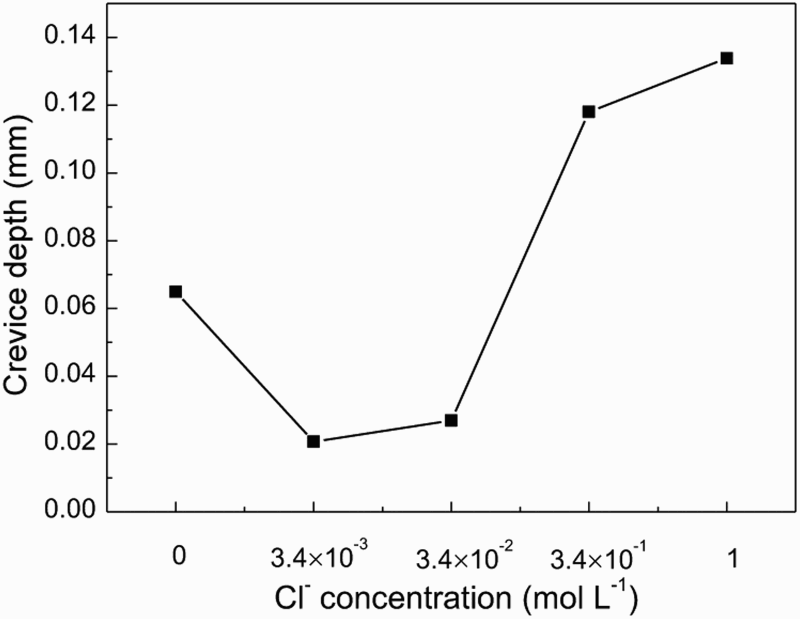
Crevice corrosion of carbon steel usually occurs through the following processes: first, the anodic dissolution of Fe and the transfer of Cl− into the crevice, and then the hydrolysis of metal chloride leading to the reduction of pH inside the crevice, finally the accelerated dissolution inside the crevice, which is a process of cycle and autocatalysis [12,13]. When there was no Cl−, SO4 2− was the main anion inside the crevice to promote the crevice corrosion of carbon steel. When Cl− was introduced, it would migrate into the crevice, leading to the reduction of SO4 2− concentration inside the crevice. It was demonstrated that there was a certain relationship between the concentration of Cl− and the pH of inside the crevice, i.e. pH = 3.0-0.172[Cl−] [14]. So with the increased of the Cl−concentration, the pH value decreased inside the crevice resulting in the more serious crevice corrosion.
Influence of SO4 2−
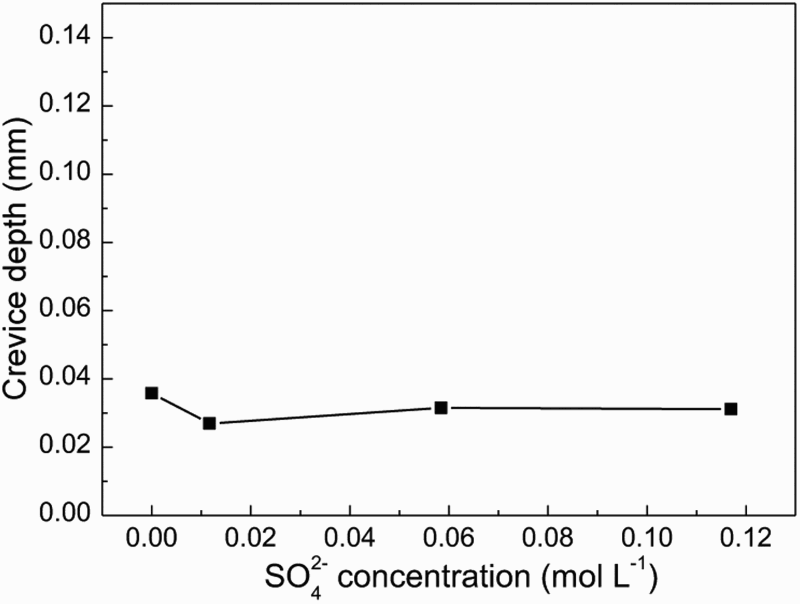
Figures 9 and 10 show SEM morphologies and crevice corrosion depth of samples after the crevice corrosion immersion tests in groundwater solution containing different SO2− 4 concentrations, respectively. The crevice corrosion depth changed little with SO2− 4concentration, which indicated that SO2− 4 had little effect on the crevice corrosion. This was because Ca2+ concentration was high in the groundwater solution, CaSO4 precipitated easily in the solution. The higher the concentration of SO4 2−, the higher the tendency of precipitation film formed. The precipitation reaction of SO4 2− occurred mainly outside the crevice, it was difficult to play a role on corrosion inside the crevice with high concentration of SO4 2−. So the variation of SO4 2− concentration in the groundwater solution had little effect on the crevice corrosion of carbon steel.
Surface morphologies inside the crevice after removing the corrosion product film in the groundwater solution containing different SO4 2− concentration. The crevice corrosion depth of steel in the groundwater solution as a function of SO4 2− concentration.
Influence of CO3 2−
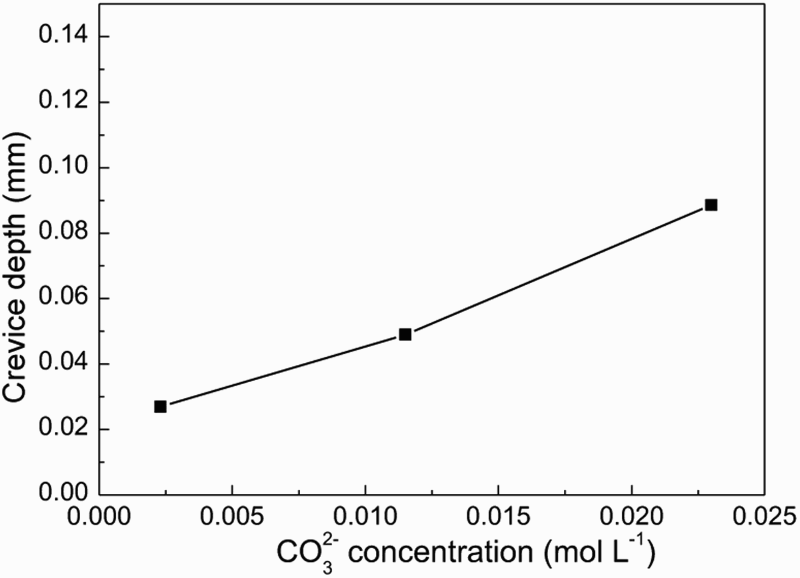
Figures 11 and 12 show SEM morphologies and crevice corrosion depth of samples after the crevice corrosion immersion tests in the groundwater solution containing different CO3 2− concentration, respectively. With the increase of the CO3 2− concentration, the crevice corrosion depth increased slightly. CO3 2− exhibited higher influence on crevice corrosion than that of SO4 2−, and lower effect than that of Cl−. CO3 2− could promote crevice corrosion of steel might be due to damage local passivating film inside the crevice, thus localised corrosion occurred in some areas. However, on the other hand, CO3 2− could increase pH value inside the crevice and inhibit crevice corrosion. So the degree of crevice corrosion induced by CO3 2− was lower than that of Cl−.
Surface morphologies inside the crevice after removing the corrosion product film in the groundwater solution containing different CO3 2− concentration. The crevice corrosion depth of steel in the groundwater solution as a function of CO3 2− concentration.
Conclusions
The corrosion behaviour of carbon steel was studied in a solution simulated to the groundwater in the northwest part of china. The effects of anions on the corrosion behaviour were evaluated. The following results have been obtained:
Cl− promoted general corrosion, while SO4 2− and CO3 2− inhibited general corrosion; In a single anion solution, the crevice corrosion of carbon steel was the most sensitive to SO4 2−; however, Cl− played a dominated role in the crevice corrosion process of carbon steel in the simulated groundwater solution.
Footnotes
Disclosure statement
No potential conflict of interest was reported by the authors.
