Abstract
Stability of organic solar cells requires development before their commercialisation is possible. This review will give a brief overview of organic solar cells and their stability, before focussing on the photochemical stability of the active layer. The photo-oxidation of the donor polymers will be looked at first which has been studied quite extensively and then fullerene electron acceptors, such as widely used phenyl-C61-butyric acid methyl ester, which has been considerably less studied. It has been shown that oxidation of the fullerene cage on phenyl-C61-butyric acid methyl ester results in oxides with a deeper lowest unoccupied molecular orbital (LUMO) level than the fresh electron acceptor. These oxides act as electron traps, leading to deterioration of the blend photoconductivity. The significance of fullerene photo-oxidation on device stability has been indirectly shown via research on: photoconductivity; organic solar cells made with an oxidised fullerene derivative and organic field effect transistors. Techniques that could be developed to increase photochemical stability of fullerene electron acceptor resistance to photo-oxidation include: reducing its LUMO level; increasing its crystallinity or aggregation and changing its chemical structure. Improving the photochemical stability of organic solar cells would move us one step closer to a more accessible solar power.
This review was submitted as part of the 2016 Materials Literature Review Prize of the Institute of Materials, Minerals and Mining run by the Editorial Board of MST. Sponsorship of the prize by TWI Ltd is gratefully acknowledged.
Introduction to organic solar cells
With the global energy demand growing, fossil fuel reserves depleting and the increasing need to tackle climate change, there is an ever growing need to develop alternative clean energies. Solar energy is one such technology and is by far the largest exploitable resource, providing the Earth more energy in 1 h than all of the energy currently used by mankind in an entire year. 1 This energy can be harnessed and converted directly into electricity by a group of devices called solar cells. An integrated group of solar cells orientated in one plane constitutes a solar panel or module. Current commercially available solar modules are mainly based on crystalline silicon, an inorganic semiconductor, which, despite a steady (and recently sharp) price reduction of modules, are still not competitive against fossil fuels.2,3 This promotes the development of a cheaper solar technology to increase its commercialisation and bridge the potential energy gap.
Organic solar cells, architectures shown in Fig. 1, are one such candidate with potential to be more versatile and cheaper than their silicon counterparts for the following reasons: (1) The semiconducting layers of organic solar cells are composed of organic polymers or small molecules, deposited in thin films (100–200 nm), which with development could be cheaper than silicon, which is an expensive raw material to purify to get it to the necessary quality for semiconductors. This means that organic solar cells will make solar more accessible both on a large scale for use in business when the transition from fossil fuels to clean energy is made easier, and for deployment in developing countries, where electricity has yet to be introduced or where the supply is unreliable. (2) The chemical structures of these photo-active materials can be varied so that their properties can be tuned, including the wavelengths at which they absorb thus giving them the capability to maximise their absorption by matching the solar spectrum or making them semi-transparent – a property that silicon solar cells do not possess. (3) The polymers and some small molecules are soluble in organic solvents, enabling them to be produced with low temperature solution processing such as roll-to-roll processing, vastly reducing the price and making the manufacture of organic solar cells far easier than that of silicon systems. (4) These coating methods allow for organic solar cells to be fabricated onto a vast range of substrates, including flexible plastic, making them light weight and highly mechanically flexible. These properties increase the versatility of organic solar cells and could lead to novel applications such as Building Integrated Photovoltaics where photovoltaics replace conventional building materials in the roof or facade; power generating windows; or solar textiles. These factors mean that organic solar cells have the potential to be a more cost-effective and versatile method than silicon solar cells to harness the power of the Sun.
Typical organic solar cell architectures: a conventional structure and b inverted structure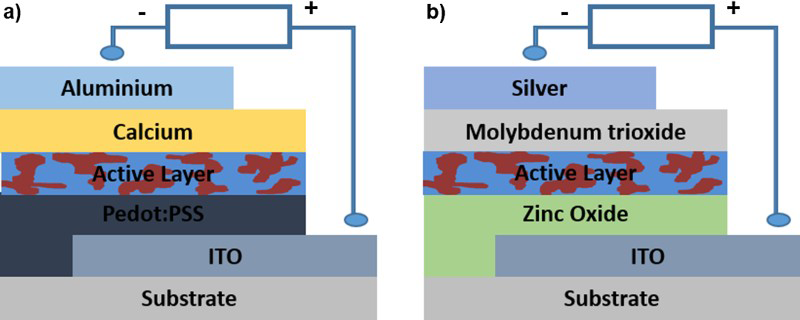
Comparison on silicon and organic solar cells in terms of efficiency, lifetime and upscaled power density

Although all three areas need improvement to make organic solar cells commercially viable, it can be argued that their stability is the most pressing of these issues. This is due to the fact that the testing of stability is not so established and has only recently had some testing protocols being outlined by the community. 13 Before these guidelines were established, much of the research was carried out under different testing conditions which sometimes did not simulate real-life weathering conditions. For example, the two values stated earlier for the best organic solar cell lifetimes were obtained without lifelike weathering conditions such as high humidity taken into account and the LED lamp source is very far from the solar spectrum, with especially low levels of UV radiation. As stability seems to be the area where the most development is required, this will be the focus of this review.
Introduction to the stability of organic solar cells
The limited device stability of organic solar cells is one of the main issues that needs addressing before commercialisation is possible.14,15 This is a complex issue that has numerous stress factors: oxygen, light, humidity, thermal stress and physical stress. These stress factors either cause the device materials to undergo a chemical reaction and/or change their morphology, classified as photochemical or morphological degradation, respectively.
These active layers are made up of a blend of a donor polymer and an electron acceptor which form different domains, maximising the interface between the two materials. The most commonly used electron acceptor for organic solar cells is phenyl-C61-butyric acid methyl ester that will be referred to as PCBM herein. This PCBM contains a fullerene (C60) ball; however, other fullerenes can be used and will be referred to in this literature review stating the size of the fullerene ball; for example, PC70BM that contains the C70 fullerene. Commonly used donor polymers are shown in Fig. 2 and electron acceptors in Fig. 3.
Chemical structure of typical donor polymers. From left to right, Top: poly(p-phenylene vinylene) with a high glass transition temperature (‘High Tg PPV’); poly[2-methoxy-5-(3′,7′-dimethyloctyloxy)-1,4-phenylenevinylene] (MDMO-PPV); poly({4,8-bis[(2-ethylhexyl)oxy]benzo[1,2-b:4,5-b′]dithiophene-2,6-diyl}{3-fluoro-2-[(2-ethylhexyl)carbonyl]thieno[3,4-b]thiophenediyl} (MEH-PPV). Middle: Poly[2-methoxy-5-(2-ethylhexyloxy)-1,4-phenylenevinylene] (PTB7), diketopyrrolopyrrole-thieno[3,2-b]thiophene thiophene (DPP-TT-T); poly(9,9dioctyl)fluorine (PFO); polythiophene. Bottom: poly(3-hexylthiophene-2,5-diyl) (P3HT); poly[N-9′-heptadecanyl-2,7-carbazole-alt-5,5-(4′,7′-di-2-thienyl-2′,1′,3′-benzothiadiazole)] (PCDTBT); poly(2,5-bis(3-hexadecylthiophen-2-yl)thieno[3,2-b]thiophene) (PBTTT); poly[[5-(2-ethylhexyl)-5,6-dihydro-4,6-dioxo-4H-thieno[3,4-c]pyrrole-1,3-diyl][4,8-bis[(2-ethylhexyl)oxy]benzo[1,2-b:4,5-b′]dithiophene-2,6-diyl]] (PBDTTPD) Chemical structures of fullerenes and typical fullerene electron acceptors. From left to right, Top: C60 fullerene; C70 fullerene; phenyl-C61-butyric acid methyl ester (PCBM); phenyl-C71-butyric acid methyl ester (PC70BM). Bottom: 1′,4′-dihydro-naphtho[2′,3′:1,2][5,6]fullerene-C60 (ICMA); bis(1-[3-(methoxycarbonyl)propyl]-1-phenyl)-[6,6]C62 (BisPCBM); 1′,1′′,4′,4′′-tetrahydro-di[1,4]methanonaphthaleno[1,2:2′,3′,56,60:2′′,3′′][5,6]fullerene-C60 (ICBA); indene-C60-tris-adduct (ICTA)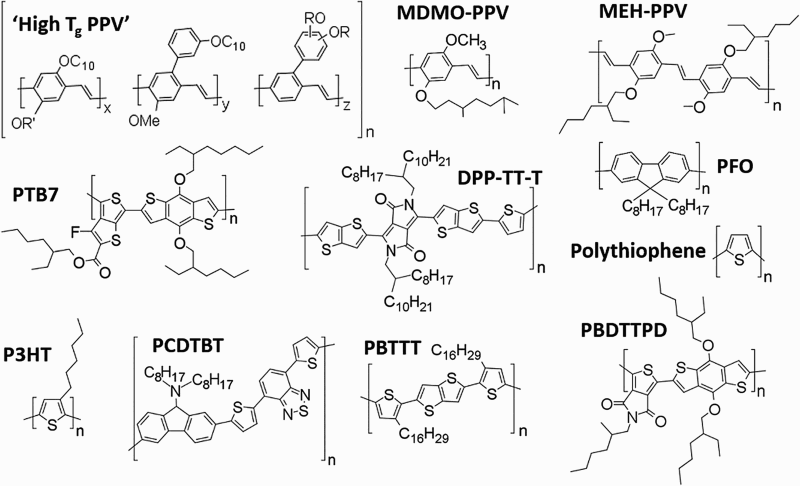
The stability of organic solar cells can be further broken down into problems associated with each component layer in the device structure (Fig. 4). The photo-active layer is prone to photochemical and morphological degradation. The device reaches high temperatures (∼85°C) after long exposure to the Sun that can cause the change of nanomorphology of the active layer, affecting charge generation, recombination and transport.13,16,17 An example of this is shown in Fig. 5, where two polymers poly[2-methoxy-5-(3′,7′-dimethyloctyloxy)-1,4-phenylenevinylene] (MDMO-PPV) and poly(p-phenylene vinylene) with a high glass transition temperature (‘High Tg PPV’) are annealed at 110°C for up to 16 h, yielding the formation of large PCBM-clusters for MDMO-PPV but a more stable morphology for ‘High Tg PPV’, indicating that this morphological stability is polymer dependent.
18
The organic semiconducting materials can also undergo photo-oxidation under ambient conditions.19,20 There are two pathways for the photodegradation that studies focus on: the generation of singlet oxygen (high energy quantum state of oxygen) or superoxide radical anions (O−2) (a negative ion with an unpaired valence, or outer orbital, electron which makes it highly chemically reactive), both of which change the electrical and optical properties of the active layer materials, creating charge traps and will be described further in the next section.
19
This review will focus on the photochemical stability of the active layer materials but first will be a short summary of other degradation mechanisms within organic solar cells.
A cross-sectional schematic of a conventional organic solar cell illustrating some of the degradation processes Morphology changes in the active layer of: top MDMO-PPV:PCBM 1:4 and bottom ‘High Tg PPV’:PCBM 1:4, as a result of annealing at 110°C
18
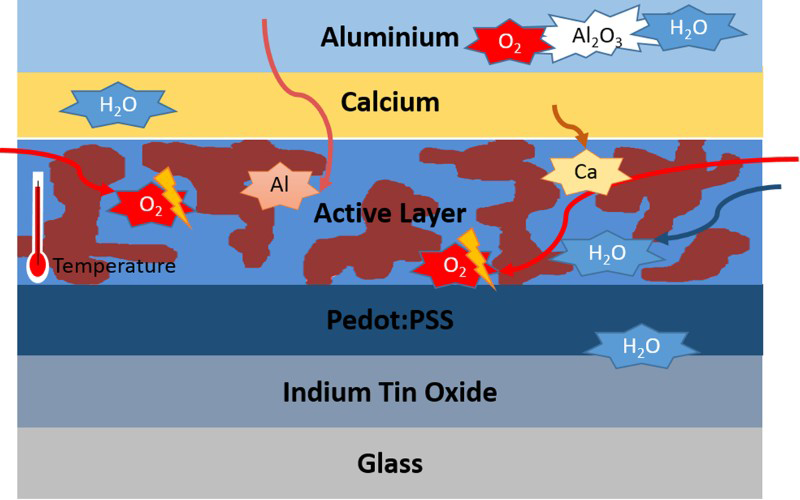

In a conventional organic solar cell device, the aluminium electrode, calcium interlayer and other low work function metals can easily be oxidised in the presence of oxygen and water to form electrically insulating metal oxides at the metal/organic interface.19,21 Furthermore, aluminium can diffuse through the device structure, explained by a phenomenon known as interlayer diffusion, which then when are oxidised, further deteriorates device performance.19,22 The indium in the indium tin oxide electrode has also been shown to diffuse all the way through the device structure, although this has not yet been correlated to the degradation of device performance. 22 The hole-transport layer PEDOT:PSS is also highly susceptible to water and can corrode the anode (indium tin oxide) layer due to its acidic and water absorption properties.19,21 These degradations at the cathode, anode and hole-transporting layer hinder charge collection, resulting in poorer device performance. As demonstrated, each layer of organic solar cells has its own stability issues associated with it, but the photochemical stability of the photo-active layer seems to be the most complex. Further complication is added when the interfacial properties are considered as these affect the processing of devices. For example, benchmark organic solar cells’ stability under thermal stress was substantially increased in cells deposited on a zinc oxide substrate in an inverted structure compared to those using a PEDOT:PSS substrate in conventional structure cells correlated to PCBM nucleation. 23 It should also be noted that encapsulation can be used to hinder the diffusion of oxygen and water through the device; however, a perfect encapsulation has not yet been developed. The photo-oxidation of fullerene electron acceptors will be the focus of this review but first, a comparison of polymer photochemical stability will be undertaken and note taken on what can be applied to the electron acceptors.
Photochemical stability of electron donors (polymers)
Photochemical degradation in the presence of light and oxygen has been observed for different benchmark organic solar cell polymers, with the degradation rates varying significantly, and will be explored in this section. The cause of the organic solar cell polymer photochemical degradation is not fully understood. The literature suggests a number of possible causes or factors electron donor stability can be related to including: chemical structure; formation of singlet oxygen and superoxide radical anions (O−2).19,24,25 The latter two mechanisms will be described below before the review of polymer photo-oxidation is continued.
The generation of singlet oxygen, a high-energy quantum state of molecular oxygen, is one of the mechanisms for photo-oxidation of organic solar cell polymers.
20
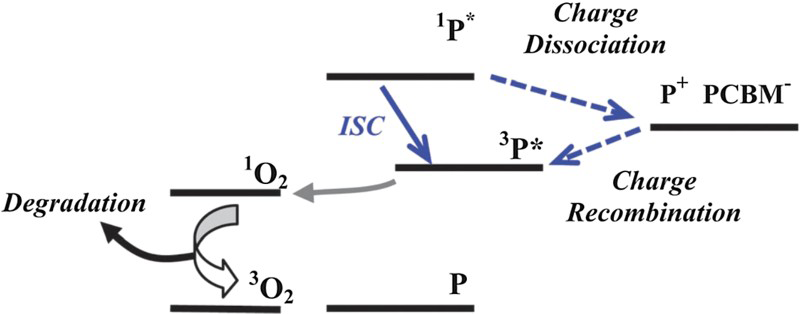
Figure 6 illustrates the triplet-mediated photo-oxidation pathways which can result in singlet oxygen. The blue solid arrow shows the triplet formation in a neat polymer film via intersystem crossing from the polymer singlet state (1P*) to the lower energy triplet state (3P*). The dashed blue arrows show the triplet formation via charge recombination of dissociated charges (P+ and PCBM−) in a blended polymer:PCBM film. Depending on the energy and lifetime of the polymer triplet state, oxygen quenching may occur. This is the transfer of excitons from the polymer triplet state to the singlet state of the oxygen molecule, reducing the number of triplet state electrons that will decay via fluorescence to the ground state (P). Singlet oxygen is highly reactive and so can attack the polymer, degrading it.
Triplet formation mechanism in neat polymer films (blue solid arrow) and in blend films (blue dashed arrow) which can lead to the generation of singlet oxygen and polymer photo-oxidation
20
The generation of superoxide anions (O−2), an oxygen molecule with an unpaired valence electron that makes it highly chemically reactive, is another mechanism for polymer photo-oxidation of organic solar cells.
25
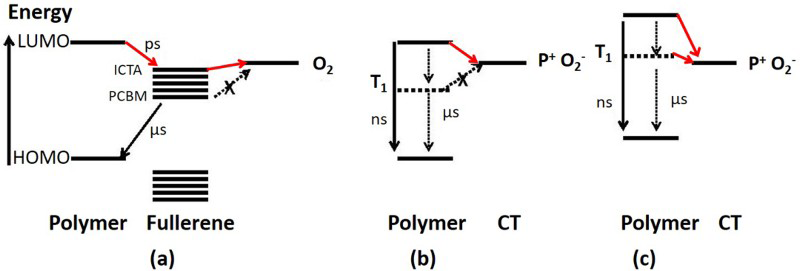
The higher the lowest unoccupied molecular orbital (LUMO) level of the fullerene, the more likely photogenerated electrons on the fullerene will transfer to oxygen to form a superoxide anion which will degrade the polymer. Figure 7a illustrates the generation of superoxide anions from polymer:fullerene blends in the presence of oxygen. Microsecond charge recombination from the fullerene LUMO to the polymer highest occupied molecular orbital (HOMO) competes with electron transfer to oxygen (electron affinity of 3.75 eV). The discrete acceptor energy levels represent increasingly electronegative fullerene derivatives, from ICTA (LUMO = 3.77 eV) to PCBM (LUMO = 4.07 eV). Figure 7b depicts nanosecond recombination of the singlet exciton competing with electron transfer from the polymer to oxygen to form a polymer-oxygen charge transfer (CT) state complex. Figure 7c shows how polymers with significant intersystem crossing yields and high LUMO levels, triplet excitons on the polymer may also undergo CT to oxygen and form substantial yield of superoxide anions.
Proposed mechanisms for superoxide anion generation from a polymer:fullerene blends b, a pure polymer film and c, a pure polymer film with significant intersystem crossing yields and high LUMO level
25
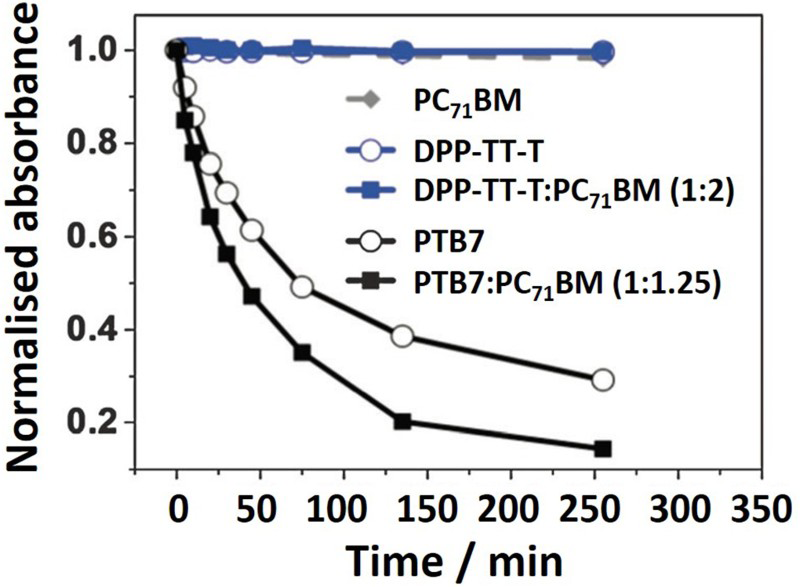
Poly({4,8-bis[(2-ethylhexyl)oxy]benzo[1,2-b:4,5-b′]dithiophene-2,6-diyl}{3-fluoro-2-[(2-ethylhexyl)carbonyl]thieno[3,4-b]thiophenediyl}) (PTB7) completely photobleaches (loss of fluorescence) in 250 h, whereas there is little or no change in the UV–Vis absorbance of diketopyrrolopyrrole-thieno[3,2-b]thiophene thiophene (DPP-TT-T) (Fig. 8).
20
The relative instability of PTB7 is due to the generation of singlet oxygen from triplet excitons, as described above. However, DPP-TT-T is actually unstable when in a device, suggesting that photobleaching may not be a direct indicator of the level of photo-oxidation. A small number of traps could form, degrading the device performance and yet not be detectable through UV–Vis absorbance measurements. There are other methods of investigating the photo-oxidation of organic solar cell polymers which will now be discussed.
Decrease in the peak ground state absorbance of PTB7 and DPP-TT-T films, both neat and blended with PC71BM, during photo-oxidation under pure oxygen and 80 mW cm−2 illumination
20
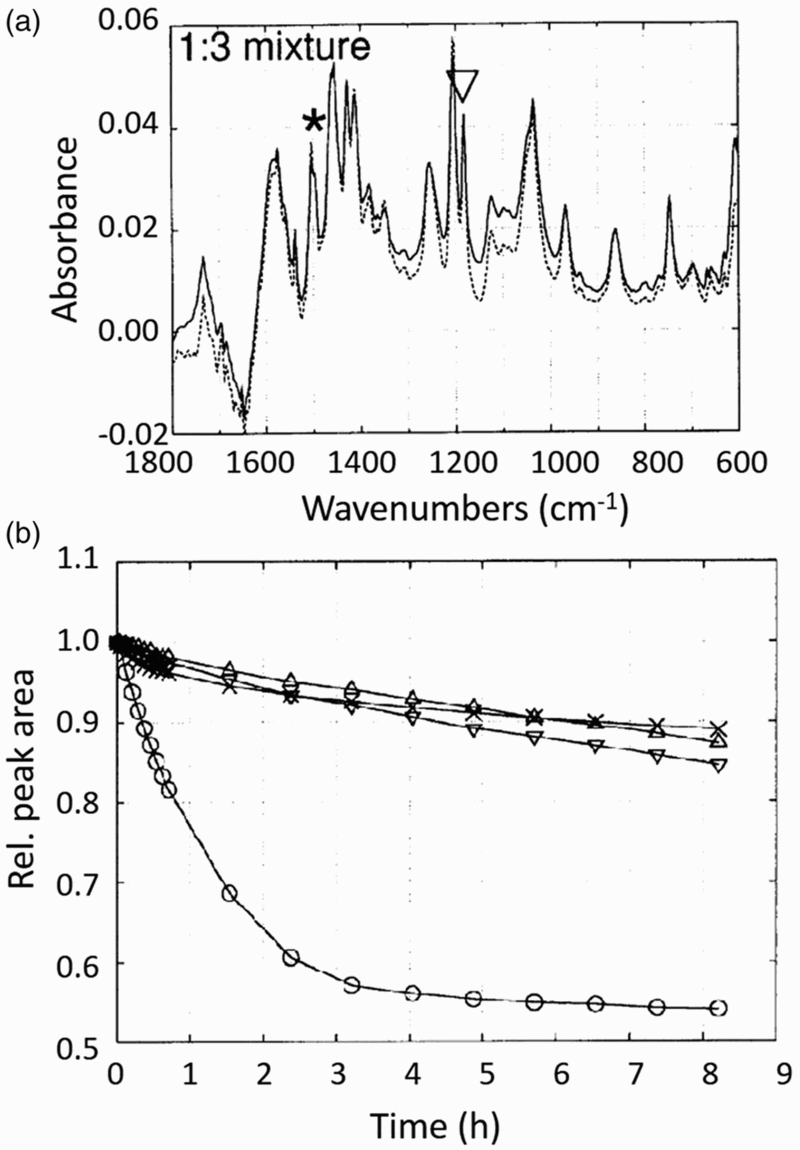
A blend of a polymer with fullerene or PCBM has a lower rate of photo-oxidation than the neat polymer as seen via Fourier Transform Infrared spectroscopy (FTIR) and current voltage measurements on devices.26–28 FTIR can detect changes in the chemical structure of a material by probing the molecular vibration of the chemical bonds and comparing the peak heights of the resultant spectra after different amounts of exposure to the degradation conditions (Fig. 9). The photo-oxidation is shown by the loss in the specific absorbance bands at 1182 cm−1 (C60) and 1506 cm−1 (MDMO-PPV). The rate of degradation of the MDMO-PPV is reduced greatly with the addition of the fullerene which can be seen when comparing the loss of the specific absorbance bands for the fullerene, polymer and blend as a function of degradation time. By adding the fullerene to the polymer, the polymer triplet state is quenched and therefore singlet oxygen formation is avoided, decreasing the photo-oxidation of the polymer.
28
This occurs due to the lower LUMO of the fullerene which means it can quench the singlet state of MDMO-PPV which then prevents the formation of the triplet state. Blending a fullerene acceptor with a polymer decreases the rate of photo-oxidation of the polymer, increasing its photochemical stability.
Photochemical degradation measured via FTIR of: (+) MDMO-PPV 1506 cm−1, (X) fullerene 1182 cm−1, (□) blend 1506 cm−1, (*) blend 1182 cm−1
27
There have been some studies on how the crystallisation of a polymer affects its triplet exciton dynamics. It has been reported that the conjugation length (length of polymer measured along backbone) of α-oligothiophene polymers influences the triplet photophysics.29,30 Furthermore, thermally annealing poly(9,9dioctyl)fluorine (PFO) films significantly lower yield of triplet excitons and there was an observed presence of triplet states in polythiophene films when amorphous but not when crystalline.31,32 Furthermore, a study of broad range of polymers showed that the more crystalline a polymer is, the shorter its triplet lifetime. 33 This is due to the yield of oxygen quenching of polymer triplet states increasing strongly with the reduction in crystallinity of the polymer. This quenching increases the stability of the polymer as charges recombining to triplet excitons result in a significant loss of photocurrent generation, reducing organic solar cell performance.
The structure of a polymer is another factor that can affect its photochemical stability. For example, introducing a silicon atom to bridge solubilising alkyl side-chains in a polymer backbone has shown to improve the photochemical stability of the resultant polymer. This is because the silicon atom blocks the unpaired electron from moving towards the C5 ring, forming a reactive macroradical. The photochemical stability of over 25 different π-conjugated polymers was observed in a study and five crucial trends in structure that affected polymer photostability were identified. 24 First, exocyclic double bonds in the main backbone – such as in poly[2-methoxy-5-(2-ethylhexyloxy)-1,4-phenylenevinylene] (MEH-PPV) and MDMO-PPV – lead to poor stability. Second, moieties that contain a quaternary site – part of the molecule with one centralised positive atom bound to four side-chains – are very unstable as these sites are easily oxidised, for example fluorene or cyclopentadithiophene. Stability is also restricted by the presence of readily cleavable bonds – such as C–N or C–O. Side-chains are also an important factor in conjugated polymer stability as removing them generally improves stability and keeping their number low as possible – no matter of their chemical nature – is a good rule to follow. Finally, good photochemical stability is generally displayed by aromatic polycyclic units – hydrocarbons composed of numerous rings in which the electrons are delocalised. These rules could be implemented to attempt to find a photochemical polymer.
There are a number of techniques that can be used to measure the levels of photo-oxidation in polymers. First, as seen above, FTIR is a useful technique that can observe the addition of C=O to the rings (at 1782 cm−1) on the fullerene, causing them to be strained, or C–O stretches (at 1182 cm−1). However, a drawback to this technique as a polymer may have a similar oxidation product to the fullerene and hence have similar peaks on an FTIR spectrum, making them difficult to distinguish. However, there is another characteristic fullerene ball peak (centred at 526 cm−1) which would rapidly decrease with oxidation and would be a good indicator of photo-oxidation.27,28 Another technique that could be used is mass spectrometry which again would measure the chemical composition of a material, but this time would give a more quantitative measurement of photo-oxidation. 34 Ultraviolet photoelectron spectroscopy (UPS) and inverse photoemission spectroscopy (IPES) could be used to measure the photo-oxidation of organic solar cell semiconducting materials from an electronic point of view. UPS can be used to measure the HOMO level and IPES to measure the LUMO level of a material and could be employed to compare materials before and after photo-oxidation.35,36 Finally, hole-only (and electron only) devices can be used to measure the charge trap density in a polymer (or in an acceptor), with charge traps decreasing the organic solar cell performance.37,38 Employing these techniques could improve the understanding of photo-oxidation of polymers which will assist in the development of more stable polymers.
The photo-oxidation of organic solar cell polymers has been widely studied and it has been suggested that it results from two main mechanisms, the generation of singlet oxygen and superoxide radical anions. Additionally, there have been a number of polymer characteristics correlated to its susceptibility to photo-oxidation, such as its structure, crystallinity and ability to inhibit either of the two suggested degradation pathways. Interestingly, it was also observed that blending a polymer with a fullerene electron acceptor reduces its rate of photo-oxidation. Techniques such as FTIR, UPS, IPES, hole-only and electron only devices could be employed to investigate this photo-oxidation further and more stable polymers developed. There has been quite an extensive study of photo-oxidation of organic solar cell polymers but less on fullerene electron acceptors which will be described in the next section.
Photochemical stability of electron acceptors (fullerenes)
Fullerene electron acceptors are widely used in organic electronics and PCBM is the most commonly employed electron acceptor of organic solar cells. This is down to the low lying excited states of the fullerene anion, promoting fast charge separation, and its high solubility in organic solvents, making solvent processing possible. 39 The majority of studies use PCBM as an acceptor when testing new polymers, architectures or upscaling methods. However, despite being well established, the photo-oxidation of fullerene electron acceptors is far less studied than that of polymers.
It has been shown via FTIR that PCBM degrades under harsh photo-oxidation conditions (an oxygen environment with argon laser light resembling part of the solar spectrum that resonates the polymer band gap or UV radiation in ambient conditions), in agreement with the previous section.27,28 The reduction of this degradation kinetic of the polymer is attributed to the scavenging effect of the fullerene whereby it chemically traps radicals that would otherwise react with the polymer.40,41 It has been shown that this oxidation of the fullerene cage leads to the deterioration of the photoconductivity of the blend film in which it resides.
42
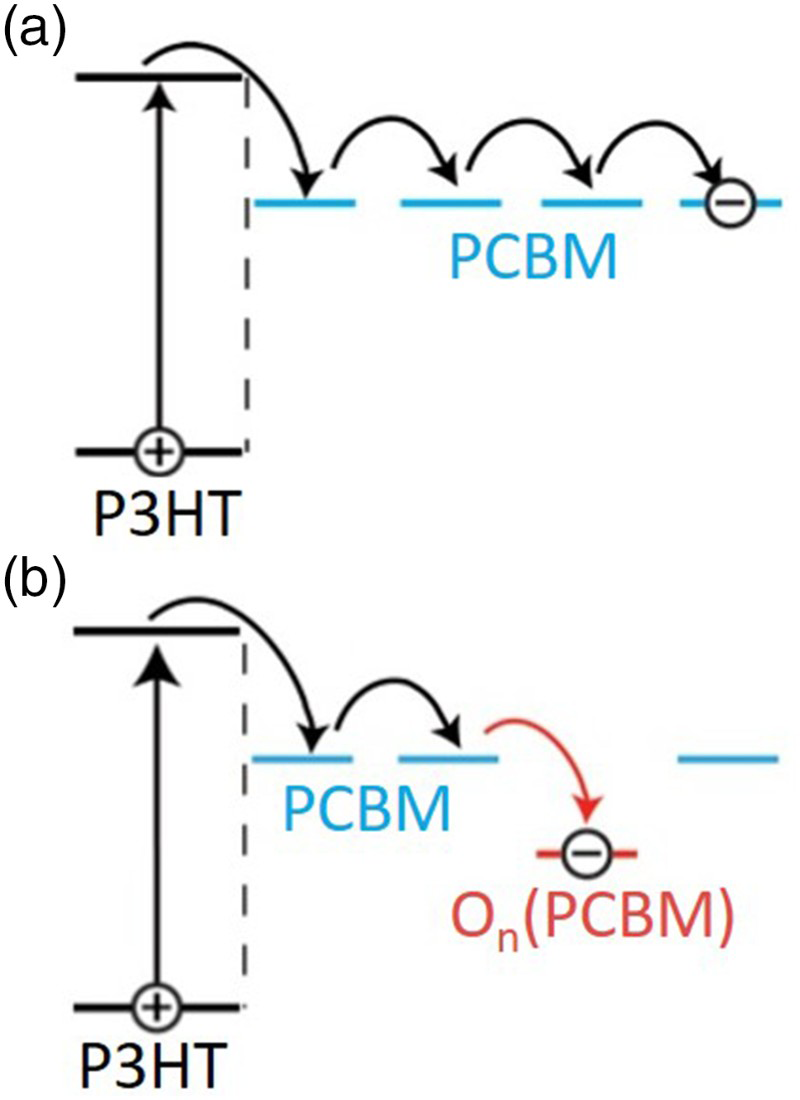
The resultant PCBM oxides have a deeper LUMO level than fresh PCBM and therefore act as electron traps in the PCBM domains Fig. 10.
42
Illustration of a photogeneration in a P3HT:PCBM film and b how the deeper LUMO level of oxidised PCBM introduces trap states that would inhibit mobility in PCBM domains
42
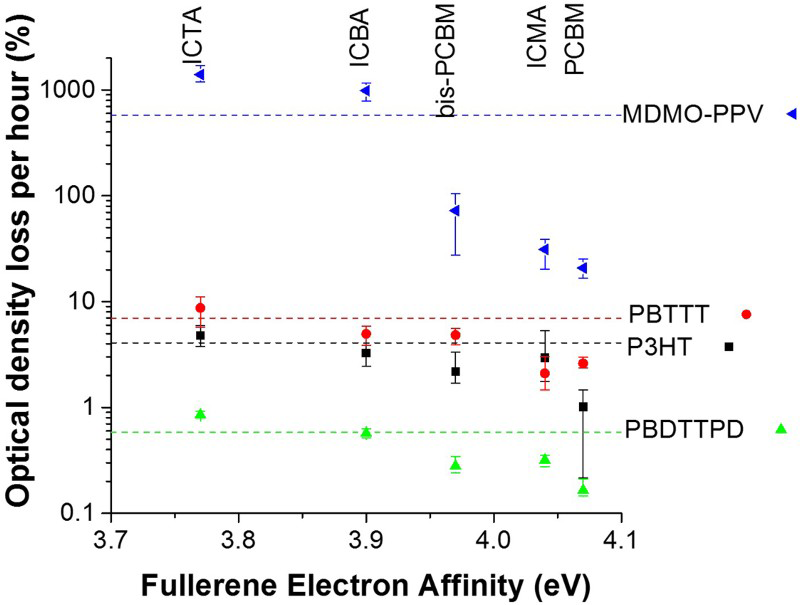
Not only does the addition of fullerene to the polymer slow its rate of photo-oxidation, the electron affinity (the change in energy of a neutral atom when an electron is added to form a negative ion) of the fullerene acceptor further affects the rate of photo-oxidation of a polymer:fullerene blend (Fig. 11).
25
Polymers blended with a fullerene with a higher electron affinity, or a lower LUMO level, had increased photochemical stability (Fig. 9). This is consistent with the generation of highly reactive superoxide (O2−) radical anions which then degrade the polymer (Fig. 7). PCBM and other fullerene acceptors with higher LUMO levels were studied but none with lower LUMO levels so presumably developing a fullerene acceptor with a lower LUMO level would increase the photochemical stability of the polymer. The study also did not test how the LUMO level affected the stability of the fullerene acceptor itself or devices. It has been seen that a lower LUMO level of a fullerene acceptor in a blend with a polymer can increase photochemical stability, but there is still scope for further investigation into how it affects the fullerene and device stability (Fig. 11).
The rate of photobleaching of organic solar cell polymers as a function of fullerene electron affinity. Dashed lines denote the photobleaching rate of pure polymer films. The error bars show the minimum and maximum measured values for different equivalent samples ± a standard error in the fitted rate
25
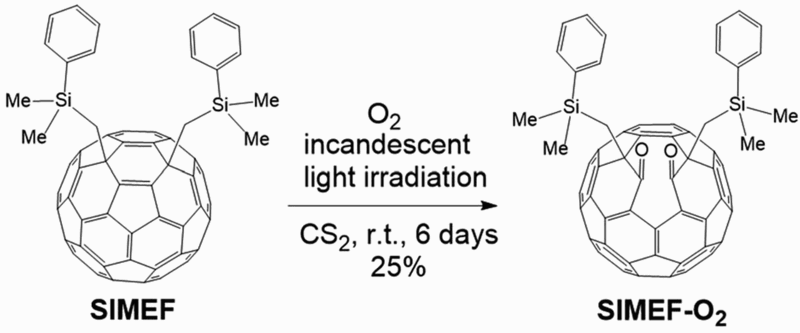
The photo-oxidation of a fullerene acceptor, 1,4-bis(dimethylphenylsilylmethyl)[60]fullerene (SIMEF), has also been shown to react with molecular oxygen when in an oxygen-saturated environment, under a 60 W incandescent lamp to give SIMEF-O2 and other more oxygen-heavy products (Fig. 12).
43
Moreover, organic solar cells were fabricated with various amounts of SIMEF-O2 added to the fresh SIMEF and device performance swiftly decreased as the amount of SIMEF-O2 was increased (with a slight recovery when the composition became 100% SIMEF-O2). It should be emphasised that even an addition of 1 SIMEF-O2 wt-% was enough to almost half the efficiency of an organic solar cell (3.2–1.9%). This suggests that even if a minute amount of a fullerene acceptor is oxidised, then the organic solar cell efficiency can be drastically reduced.
Chemical structures of fullerene acceptor SIMEF and when it is oxidised as SIMEF-O2
43
As seen in the previous section, polymer crystallisation affects its photo-oxidisation. A similar effect could be possible for fullerene electron acceptors where the crystallinity or aggregation level could affect photo-oxidation, as it would be physically more difficult for the oxygen to penetrate through the film, enhancing absorption and charge transport. However, if the domain size is too large, the efficiency of CT may be reduced, then domain size will have to be optimised in order to balance these effects. It has been seen that fullerene solution after light exposure forms fullerene epoxides, where oxygen binds onto the fullerene ball, straining the ring to which it binds, making it highly reactive. 44 This subsequently leads to the fullerenes aggregating, coulombically bound, in the solution. If these epoxides could be mixed with PCBM to form aggregates, this may reduce the rate of PCBM photo-oxidation as it will be harder for the oxygen to penetrate through as the aggregated areas will be physically denser. The photo-oxidation of fullerene could be altered by its aggregation or crystallinity and methods such as the addition of fullerene epoxides could be utilised to control these properties.
Fullerene derivatives are also used as electron transport layers in organic field effect transistors.45–47 It is well known that the performance of n-type organic field effect transistors is far inferior to that of the p-type. 48 This is due to the susceptibility of charge trapping in n-type transistors when exposed to environmental oxidants (oxygen and water), reducing electron carrier mobility.49,50 The air stability of fullerene electron transport layers in n-type organic field effect transistors was found to depend upon the LUMO level of the fullerene and energetic disorder (distribution of the energy levels) in the semiconducting layer. 51 There have been attempts to address this problem one of which is changing the structure of the electron transport material such that it has less of a tendency to react with oxygen or water.50,52 For example, the addition of a solubilising group to a fullerene backbone saw the creation of fullerene monoadducts with long-term stability in air. 52 Additionally, it is thought that lowering the LUMO level of the acceptor below the trap energy of environmental oxidants would make it stable against charge trapping.49–51,53 Considerable research has been undertaken by the organic field effect transistor community on the photo-oxidation of fullerene acceptors, highlighting that it is indeed a significant problem. Hence, photo-oxidation of fullerene acceptors is an issue that should be scrutinised within the organic solar cell community and some of the methods to reduce transfer from organic field effect transistors.
Although fullerene-based electron acceptors, mainly PCBM, have been in organic solar cells for over 20 years, there has been little research on their photochemical stability in the presence of light and oxygen. Studies undertaken by the organic field effect community suggest that the photo-oxidation of fullerene is a significant problem and methods to reduce it have been investigated, including methods to increase the aggregation of the fullerene acceptors or reducing their LUMO level. However, if this proves unsuccessful, the use of non-fullerene acceptors, commonly used for organic light emitting diodes, can be employed. One such non-fullerene acceptor is perylene which has demonstrated an organic solar cell efficiency of 6.3% and not only could improve stability but is also a cheaper acceptor material than PCBM.54–56 The photo-oxidation of fullerene acceptors clearly requires further investigation and this may lead to shift in the focus of organic solar cell research.
Conclusion
Stability is a key area of development for organic solar cells before commercialisation is possible, so research has increased in this area. However, there have been few studies looking at the photo-oxidation of the electron acceptor materials, such as widely used phenyl-C61-butyric acid methyl ester. Several papers (on photoconductivity; devices made with an oxidised fullerene derivative; and organic field effect transistors) indirectly showed the significance of fullerene photo-oxidation on device stability. Some methods of probing photo-oxidation can be transferred from those used on polymers, including: FTIR; UPS; IPES; and electron only devices. Some techniques have been suggested to increase fullerene acceptor resistance to photo-oxidation including: reducing its LUMO; increasing its crystallinity or aggregation; and changing its chemical structure. By applying techniques and developing an electron acceptor with more resistance to photo-oxidation, more photochemically stable organic solar cell devices could be developed, and hence, a more accessible solar energy would be one step closer.
Footnotes
Acknowledgements
The author would like to thank her supervisor W.C. Tsoi for his advice and guidance when writing this literature review. Informative discussions with K.H. Lee were also much appreciated. The Materials and Manufacturing Academy (M2A) kindly provided publication costs. This review was undertaken during a PhD sponsored by Ser Solar and the National Research Network.
