Abstract
As a result of the increasing in the world population, the development of technology to meet increasing energy needs and to find other energy resources is one of the biggest tests for society. Triphenylamine and its derivatives attract the attention of researchers due to their many applications such as in solar cells, electronics, and medicine. The nitro group, an electron-accepting chromophore, is a good candidate for D–π–A sensitizers for dye-sensitized solar cells. In this study, the power conversion efficiency of an anchor nitro group in dye-sensitized solar cells and the synthesis of new compounds containing a donor triphenylamine unit, functionalized with a nitro group as an anchor group and possessing a π-conjugated structure are investigated. The structures of the compounds are determined by FTIR, UV-Vis spectrometry, NMR, and MS. Considering the photovoltaic performance of the produced dye-sensitized solar cell devices, the anchor group, and its acceptor effect, it is observed that the efficiency increases, in the order,

Introduction
Global warming has become one of the most important factors threatening the world. Fossil fuels, which are mainly used for energy requirements, take first place among the causes of global warming. Intensive consumption of fossil fuels causes depletion of the world’s fossil fuel reserves, environmental pollution, and contributes to the greenhouse effect. Various renewable energy sources are used as alternatives and serve as solutions to these problems. Of all the forms of renewable energy, solar energy is the most popular. Solar cells attract significant attention as one of the most convenient ways to convert solar energy into electricity. 1 Traditional costly and less efficient silicon-based solar cells are being replaced by dye-sensitized solar cells (DSSCs).
In DSSCs, ruthenium-containing dyes show superior photovoltaic performance compared to other metal complexes. 2 However, the large-scale use of ruthenium-based paints in DSSC applications is limited. As ruthenium has high production costs and is toxic, it causes environmental pollution. Therefore, many metal-free organic dyes are being tested as effective dye-sensitizing solar cells. 3 Among metal-free organic dyes, triphenylamine (TPA) dyes are promising candidates as high-throughput DSSCs. 4
TPA dyes are widely used as sensitizers in DSSCs due to their strong electron-donating ability. They exhibit high molar extinction coefficients due to intermolecular π–π* transitions. TPA dyes are widely used as sensitizers because they are strong electron donors, and their derivatives occupy an important place in many industrial applications. For example, they are widely used in organic photovoltaic functional materials, 1 and as block structures for conductive polymers 5 and high-spin polyradicals.6,7 TPA derivatives have also been used in pharmacology and in biosensor construction. Furthermore, as an organic semiconductor, TPA is used as a hole-transfer material for optoelectronic applications. In addition to its very good pore-carrying properties, it also prevents aggregation due to its non-planar structure. 1
In this study, we describe the synthesis and solar cell applications of organic electronic materials with D–π–A properties. The constructed products possess a nitro group–containing moiety, as an anchor group, high absorption bands in the visible region, and demonstrate attached to the TPA structure of their UV spectrum.
Results and discussion
Characterization
The new ligands

Synthesis of
Both compounds exhibit good solubility in common polar organic solvents such as ethanol, methanol, and chloroform. The UV-Vis spectra of the ligands (Figure 1) showed two types of transitions, the first occurring in the 270–320 nm range, which is due to transitions containing molecular orbitals that can be assigned to the π–π* transition. The second type of transition occurs in the 320–450 nm range, which can be assigned to the n–π* transition, in which the bands become wider.

Absorption spectra of ligands
In the mass spectra, the molecular ion peaks of
Compounds
For DSSC fabrication, transparent TiO2 paste was deposited on the conductive side of an FTO (fluorine-doped tin oxide) substrate using Doctor Blade’s method. The photoanode was prepared by immersing the coated FTO substrate in a solution containing 0.3 mM
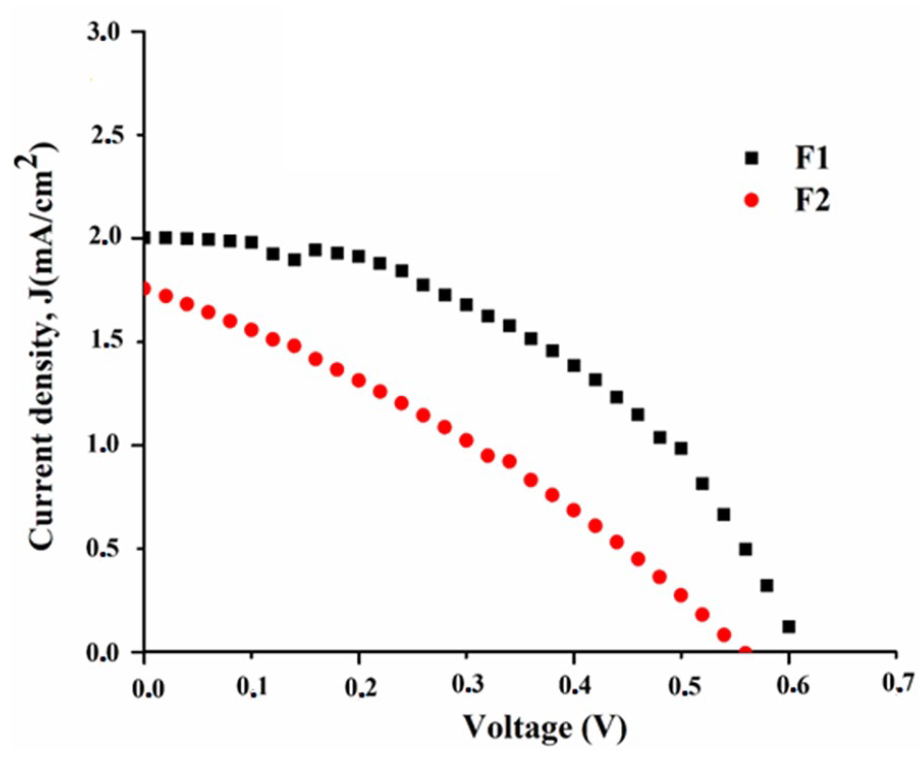
Current density–voltage curves of illuminated DSSCs using dyes
The current–voltage (
Photovoltaic parameters for the DSSCs sensitized from

The effect of the electron-withdrawing chlorine atom close to the anchor NO2 group has a negative impact on achieving a better PCE value. It can be seen from the obtained PCE values that the electron density on the anchor NO2 in sensitizer
Cong et al.
9
synthesized a functionalized dye (

Structures of compounds
Conclusion
In this study, sensitizers
Experimental
Materials
All chemicals were reagent grade from Merck and Fluka. Solvents were distilled from appropriate drying agents prior to use. Commercially available reagents were used without further purification unless otherwise stated. Flash column chromatography was carried out using silica gel 60 (0.04–0.063 mm) from Merck. 4-(Diphenylamino)benzaldehyde was prepared according to the Vilsmeier–Haack reaction, which is widely used to obtain aryl aldehydes. 11
Equipment
FTIR spectra were recorded on a Spectrum One Perkin Elmer FTIR spectrophotometer using attenuated total reflectance (ATR). The mass spectra were acquired on an Agilent 6530 + Agilent high-performance liquid chromatography (HPLC) liquid chromatography–mass spectrometry (LC-MS) Quadrupole time-of-flight (QTOF) instrument, and gas chromatography–mass spectrometry (GC-MS) mass spectra were recorded at the Yıldız Technical University Science and Technology Application and Research Center (BITUAM), Turkey. 1H NMR spectra were recorded in CDCl3 solutions on a Varian 500 MHz spectrometer.
Synthesis
Synthesis of 4-{[(4-Nitrophenyl)imino]methyl}-N ,N -diphenylaniline (F1 )
An ethanol solution of 4-nitroaniline (0.1 g, 0.73 mmol) was added dropwise to a solution of triphenylamine aldehyde (0.2 g, 0.73 mmol) in ethanol (10 mL). The resulting mixture was refluxed for 18 h in the presence of piperidine (0.045 mL, 0.45 mmol) as the catalyst. The solvent was evaporated, and the residue was purified by column chromatography (
Synthesis of 4-{[(2-chloro-4-nitrophenyl)imino]methyl}-N ,N -diphenylaniline (F2 )
An ethanol solution of 2-chloro-4-nitroaniline (0.126 g, 0.73 mmol) was added dropwise to a solution of triphenylamine aldehyde (0.2 g, 0.73 mmol) in ethanol (10.0 mL). The resulting mixture was refluxed for 18 h in the presence of piperidine (0.045 mL, 0.45 mmol) as the catalyst. The solvent was evaporated, and the residue was purified by gradient column chromatography (
Fabrication and characterization of the DSSCs
For DSSC fabrication, two transparent TiO2 pastes were deposited using Doctor Blade’s method on the conductive side of an FTO substrate, as reported earlier.
12
In order to prepare the dye-sensitized (
The electrical performance of the solar cell is determined by the short-circuit current (

where the

Supplemental Material
sj-pdf-1-chl-10.1177_17475198231168948 – Supplemental material for Investigation of the effect of a nitro group as an anchor group in dye-sensitized solar cells
Supplemental material, sj-pdf-1-chl-10.1177_17475198231168948 for Investigation of the effect of a nitro group as an anchor group in dye-sensitized solar cells by Fatma Aytan K1l1çarslan and İbrahim Erden in Journal of Chemical Research
Footnotes
Author contributions
All authors contributed to the study conception and design. Synthesis, purification, and characterization studies of substances were performed by Associate Professor F.A.K. Examination of the applicability of the solar cell was performed by Professor Dr İ.E. The first draft of the article was written by F.A.K., and all authors have made the necessary corrections and comments. All authors have read and approved the final article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the Yıldız Technical University Scientific Research Projects Coordination Department (project no. FBA-2021-4051).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
