Abstract
Tic disorders are common in children and adolescents, and the point prevalence rates in epidemiological studies range from 4.8% to 6.6% of 7–15-year-old children [1–3]. They are associated with significant morbidity, and adversely affect the young person's academic achievement, social relationships within their peer group and family life [4], [5]. Frequently, tic disorders are comorbid with obsessive–compulsive disorder (OCD), attention-deficit–hyperactivity disorder (ADHD) and/or depressive disorders [6], [7], and hence need to be managed synergistically with them [8–10]. Typical and atypical dopamine D2 receptor [11] antagonists, such as haloperidol, pimozide and risperidone in Australia and tiapride in China, are frequently used treatments for tic disorders. Clinical effectiveness, however, remains variable [12–15], with approximately 40% of patients remaining symptomatic [16].
Currently the noradrenergic system is underresearched as the target for medication intervention in tic disorders, despite its key role in modulating the dopaminergic system directly [17], [18]. Clonidine, whose main site of action is the locus ceruleus, is an α2-adrenoceptor agonist that decreases the functional activity of the noradrenergic system [19]. It has demonstrated beneficial effects on tic disorders, particularly Tourette disorder [20–24]. The clonidine adhesive patch is a transdermal therapeutic system (TTS) that releases clonidine at a relatively constant rate for 7 days without peak or valley plasma concentration changes [25], [26]. Because of its ease of use (self-administered patch, needing to be changed only at weekly intervals, with easy disposal, and a rapid onset of action), patient compliance rates are high [27]. To date there are no extant controlled trials of the clonidine adhesive patch in tic disorders. The aim of the present study was therefore to evaluate the therapeutic effectiveness and adverse effects profile of the clonidine adhesive patch using a rigorous controlled study design.
Methods
Participants
A total of 437 participants who met the inclusion and exclusion criteria were recruited from eight clinical academic child psychiatry centres across China (transient tic disorder, 5%; chronic motor or vocal tic disorder, 40%; or Tourette disorder, 55%). There were 326 participants in the active treatment group, 111 in the clinical control group, and six did not complete the study (1.37% of the sample studied): two actively withdrew their informed consent to participate in the study, three did not attend for subsequent follow up as required by the study's protocol, and one experienced a seizure that was recorded as an adverse event.
Inclusion criteria were that participants met Chinese Classification of Mental Disorders–third edition (CCMD-3) [28] criteria for transient tic disorder, chronic motor or vocal tic disorder and/or Tourette disorder, were aged 6–18 years, were clonidine medication naïve, had a mainstream educational background and had a guardian that was able to complete informed consent forms on their behalf. The CCMD-3 is comparable to both DSM-IV and ICD-10 for tic disorders, anxiety disorders, mood disorders, psychotic disorders and disruptive behaviour disorders. It differs from both in its conceptualization of neurasthenia and specific culture-bound syndromes and disorders. The term ‘mainstream educational background’ refers to children without intellectual disability (mental retardation in China) who are educated in mainstream schools rather than special schools for the intellectually disabled. Children with an intellectual disability were excluded from the sample studied. Participants were excluded from the study if they had bodyweight ≤20 kg, they were diagnosed with one or more comorbid CCMD-3 psychiatric disorders, they had one or more comorbid medical disorders, they were known to be hypersensitive to the clonidine adhesive patch or to have medication allergies for more than two medicines, they were using antipsychotic/antidepressant/mood stabilizing and/or anti-epileptic medication, they were using medication for a tic disorder within 4 weeks of starting the trial, they had taken part in a medication trial within the 3 months before the start of the trial and/or they had no guardian or could not take medicine according to the trial dosing regimen. The CCMD-3 was used to determine the presence of comorbid disorders and 1503 cases were screened in order to identify the 437 patients included in the sample studied. This is feasible in China because each child mental health centre manages approximately 10 000 cases per year.
Measures
Yale Global Tic Severity Scale
This scale is a semi-structured clinician-rated instrument of motor and vocal tic severity [29]. It consists of two domains, motor and vocal tics, assessed on frequency, intensity, complexity and interference. It has robust psychometric properties as a measure of change in tic severity and has been used in a number of studies internationally [30], [31]. Therapeutic effect is judged by the change in total Yale Global Tic Severity Scale (YGTSS) score from initial assessment to that at the end of week 4. Clinical recovery is defined as ≥80% decrease in the total YGTSS score; obvious improvement as ≥50% and <80% decrease in the total YGTSS score; improvement as ≥30% and <50% decrease in the total YGTSS score; and ineffective as <30% decrease in total YGTSS score.
Clinical Global Impression scale
The Clinical Global Impression scale (CGI) consists of 0–7 points [32]. It is used to evaluate the severity of clinical symptoms and measure change in them over the course of the study. It has robust psychometric properties as a measure of change in clinical symptoms and has been used in a number of studies internationally [32].
Procedure
Layering segmenting randomization of 800 random numbers was used. Participants received treatment for 4 weeks, randomly assigned to the clonidine adhesive patch treatment group or the placebo control group, according to the randomized number; the rate of the participants who used clonidine to those who used placebo was 3:1. Participants were classified into three groups according to their bodyweight: low (20 kg < bodyweight ≤ 40 kg); middle (40 kg < bodyweight ≤ 60 kg) and high (bodyweight >60 kg). The low-bodyweight group received a 1.0 mg clonidine adhesive patch, the middle-bodyweight group a 1.5 mg clonidine adhesive patch and the high-bodyweight group a 2.0 mg clonidine adhesive patch. The patch would be pasted on the left or right infrascapular region each week in turn. The previous patch could not be removed unless the next one had been used for at least 1 day. Participants whose YGTSS score decreased <30% and CGI-I score was ≥4 by the end of week 3, were withdrawn from the trial and offered free alternative medication treatment, tiapride. This practice is consistent with that in China currently. Most clinicians and their patients will accept a 3 week trial of treatment after which ineffectiveness or partial effectiveness will usually lead to a change in treatment strategy.
Clinical evaluation occurred at screening, 0 days, 7 days, 14 days, 21 days and 28 days after clonidine adhesive patch or placebo treatment. At each visit the YGTSS and CGI were completed by the study physician along with measurement of vital signs (blood pressure and pulse rate) and examination of the condition of the skin where each patch was pasted. Further, an electrocardiogram (ECG) was completed at each visit. There was harmonization of methods across the centres, training was standardized and the proportion of active: placebo was similar across the centres. IRB approval and informed consent was obtained from the participants’ legal guardians. Each stage of the assessment, treatment and evaluation processes was conducted by separate staff blind to the other stages.
Statistical analysis
According to the intention-to-treat principle, we defined the following sets: (i) full analysis set (FAS) for all the participants who had been grouped randomly into the test group and the control group, treated by clonidine adhesive patch or placebo, and followed up at least once (each last observation was carried forward for missing values of each major measure index); (ii) per protocol set (PP) for those participants who were recruited into the study, received clonidine adhesive patch or placebo, were followed up at least once but who were withdrawn from the trial after 3 weeks of treatment; and (iii) a safety set (SS) for all the participants who had been treated at least once after randomization.
The statistical program SAS6.12 (SAS Institute, Cary, NC, USA) was used. The variables are described as mean ±SD. Where appropriate, t-tests, analysis of variance (ANOVA) and non-parametric tests were used to evaluate the progress each group made after treatment. Enumeration data were analysed by frequency analysis (constituent ratio). Non-parametric tests were used to analyse the change each group had made after the intervention.
The reason and exact time of dropout cases were recorded in detail. χ2 tests (or exact probability method) were used to analyse the different total rate of dropout and rate of dropout due to adverse events between the two groups.
The YGTSS total score was the primary index of therapeutic effect. The different rate of decreased scores between the two groups was analysed using centre effect-concerned ANOVA while the different therapeutic response between the two groups was compared on repeated measures ANCOVA, covarying for initial YGTSS, age and gender.
Adverse events were described in detail. χ2 tests (exact probability) were used to analyse the different rates of adverse events between the two groups.
Results
Baseline comparison
The two groups did not differ in age, gender, nationality, educational background, blood pressure, heart rate or bodyweight. Further, the two groups did not differ in severity of tics (YGTSS) or clinical symptoms (Table 1).
Baseline subject comparison
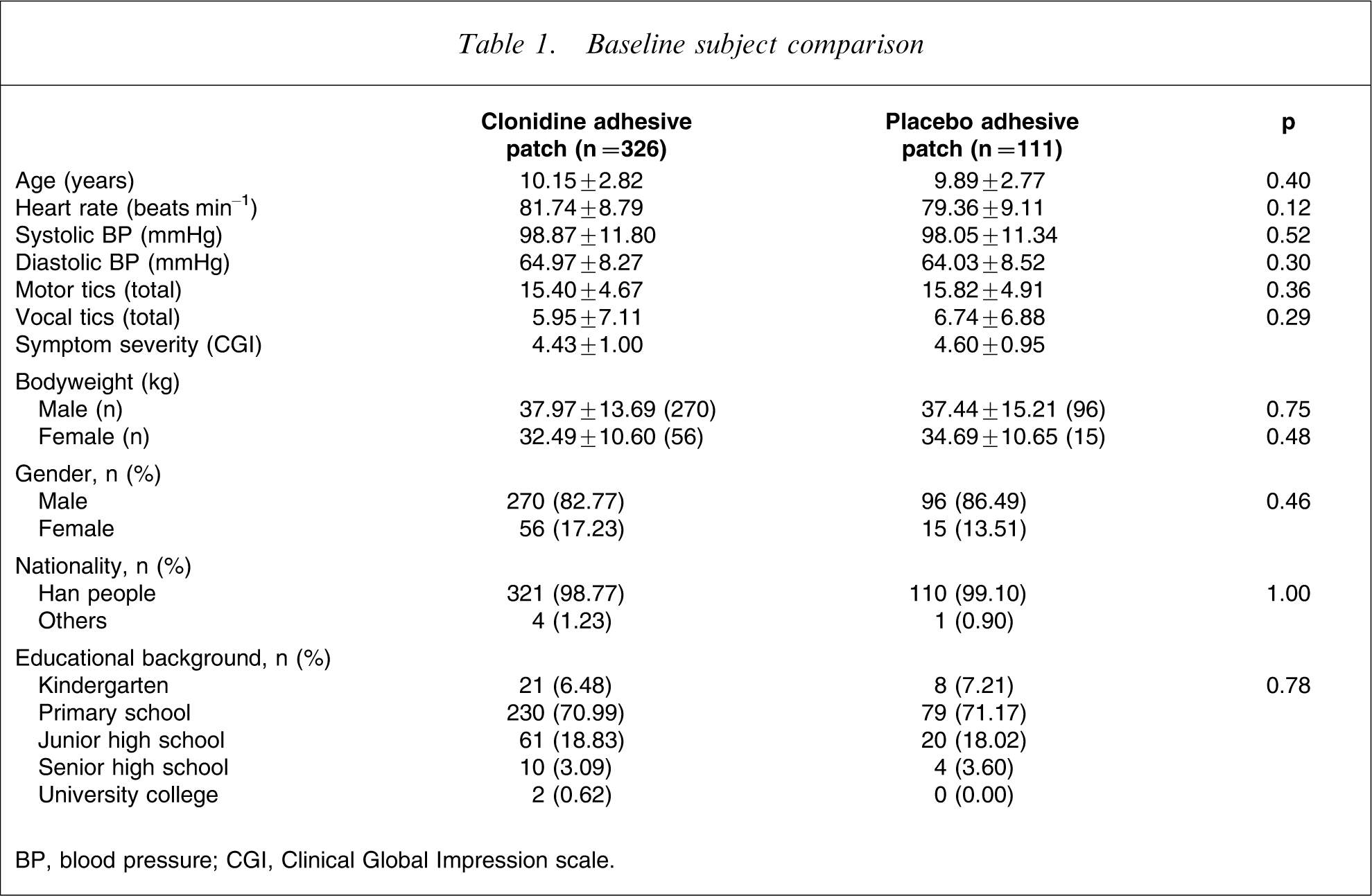
BP, blood pressure; CGI, Clinical Global Impression scale.
YGTSS comparison
There were 437 patients in the FAS and in the SS: 326 were in the test group and 111 in the control group. There were 432 patients in the PP: 321 in the test group and 111 in the control group.
Total score of YGTSS in the FAS
After 4 weeks of treatment, the YGTSS total score in the test group decreased from 21.35±8.67 to 9.83±7.77 (difference, −11.53±8.22), while in the control group it decreased from 22.56±8.79 to 11.84±8.01 (difference, −10.72±7.50).A repeated measures ANCOVA indicated that the test group had a significantly lower total YGTSS score than the control group after week 4 (Table 2).
Corrected mean YGTSS total score
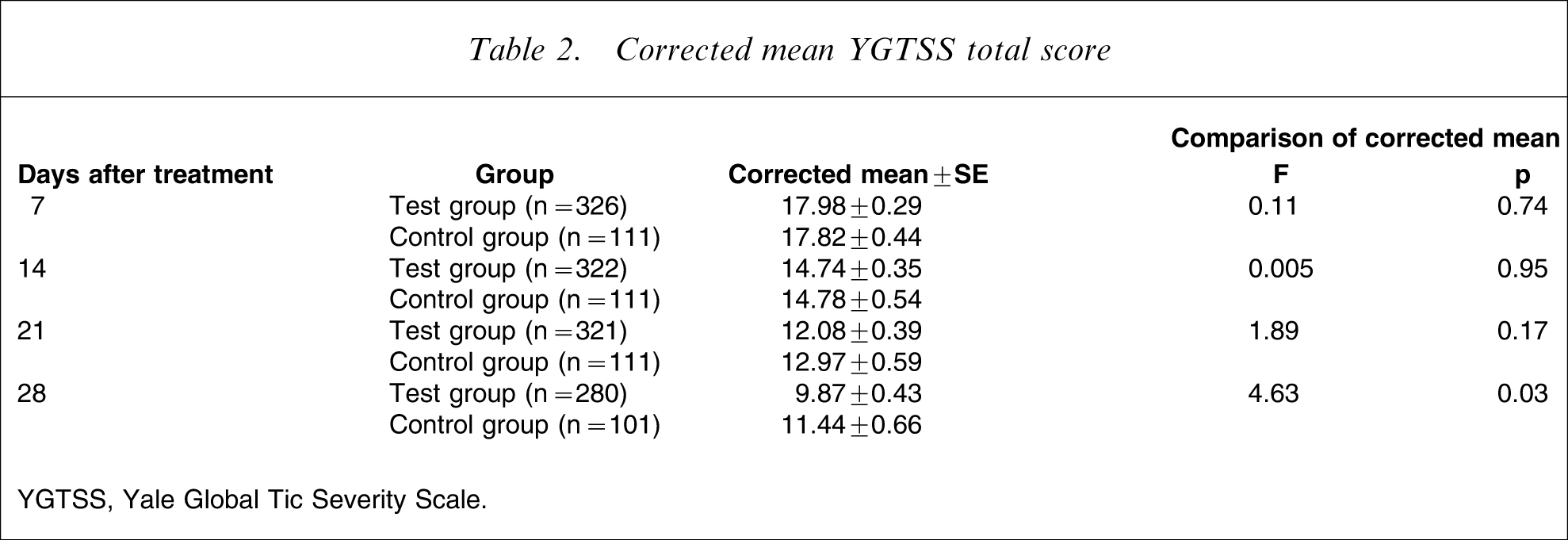
YGTSS, Yale Global Tic Severity Scale.
Rate of decreased total score of YGTSS
A repeated-measures ANCOVA showed that the test group had a significantly lower rate of decreased total YGTSS score than the control group after week 4 (Table 3).
Decreased total raw YGTSS score
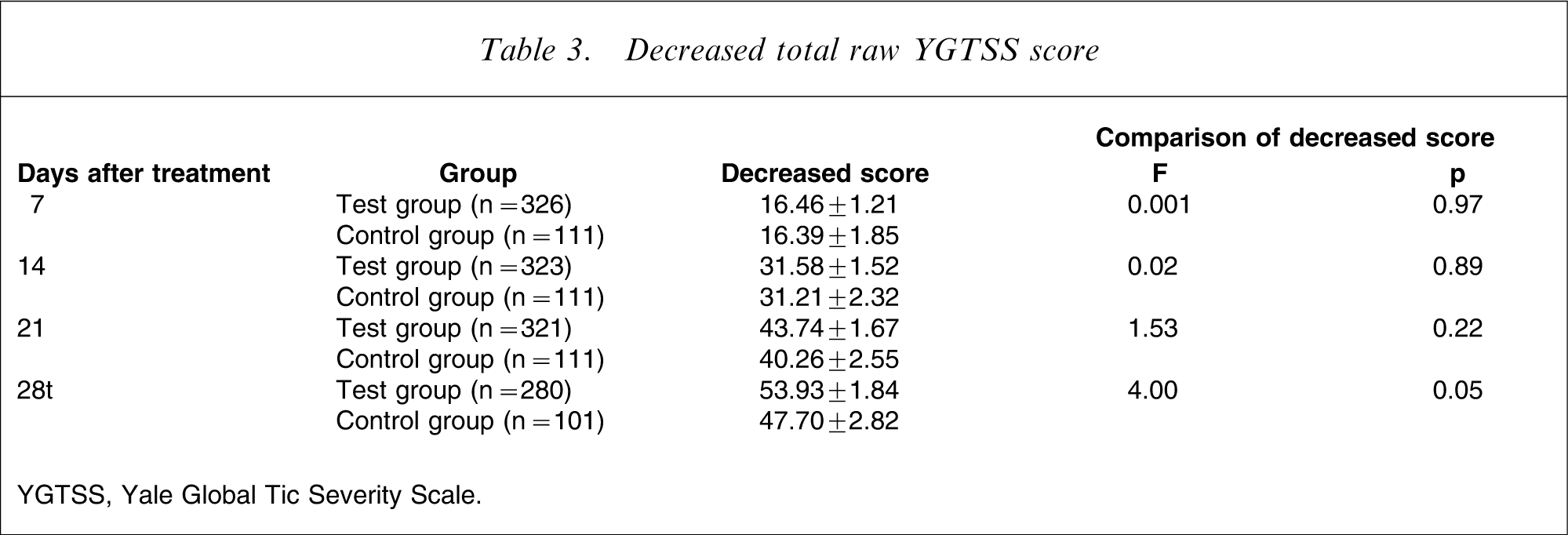
YGTSS, Yale Global Tic Severity Scale.
Therapeutic response
After 4 weeks of treatment, 19.31% of participants (62/321) in the test group were clinical recovered, 49.53% (153/321) were obviously improved, 11.21% (36/321) were improved and 19.94% (64/321) were in the ineffective group. In the control group, 11.71% of participants (13/111) were clinically recovered, 35.14% (39/111) were obviously improved, 35.14% (39/111) were improved, and 18.02% (20/111) were in the ineffective group. The difference between the two groups was significant (χ2=9.15, p = 0.003). The total response rate was 68.85% in the test group and 46.85% in the control group, which was again statistically significant (χ2=16.98, p < 0.0001).
Safety assessment
There were 437 participants assessed: 326 in the test group and 111 in the control group. In the test group three participants (0.92%) took medicine for 1 week, two (0.62%) for 2 weeks, 41 (12.58%) for 3 weeks and 280 (85.89%) for 4 weeks. In the control group, 10 subjects (9.01%) took medicine for 3 weeks and 101 (90.99%) for 4 weeks.
Adverse reactions
Eight participants (3.08%) in the test group experienced adverse reactions: three (0.92%) experienced rashes on the skin where the patch was pasted, two (0.62%) had abnormal ECG, one participant (0.31%) had somnolence, one (0.31%) had lightheadedness and one (0.31%) had insomnia. Eight patients had adverse reactions (7.21%) also occurred in the control group, including six cases of rash (5.41%), one of nausea and one of dry mouth, lightheadedness, dizziness and somnolence. The severity of adverse reaction ranged from mild to moderate and the two groups did not differ significantly. In particular, the two abnormal ECG findings were (i) atrial premature beat and bigeminal rhythm; and (ii) mildly raised ST wave and myocardial ischemia, both of which returned to normal at the following visit. The abnormal ECG findings did not lead to withdrawal from the study.
Vital signs
After 4 weeks of treatment the heart rate of the test subjects increased slightly (mean 80.80 beat min−1 before treatment to mean 81.84 beat min−1 after treatment), blood pressure decreased a little (systolic pressure from 98.87 mmHg to 97.60 mmHg; diastolic pressure from 64.97 mmHg to 64.01 mmHg) and bodyweight increased slightly (from 37.98 kg to 38.00 kg in male participants). All these changes were statistically significant. In contrast, the male control subjects had only slightly increased bodyweight.
Discussion
These data are the first to conclusively demonstrate, through a rigorous controlled trial, the effectiveness and safety of the clonidine adhesive patch for the treatment of tic disorders in children and adolescents. Importantly, the clonidine and the placebo adhesive patch groups did not differ on key demographic, physiological and clinical severity measures; the clonidine adhesive patch group had a significantly decreased frequency and severity of motor and vocal tics and a better therapeutic response compared to the placebo adhesive patch group. The findings are consistent with those of Gaffney et al. and Leckman et al., who studied the effectiveness of oral clonidine on tic disorders [19], [20]. Further, the response rate of risperidone, a widely used medication for tic disorder treatment, is reported as being between 60% and 90% [14], [16], [19]. The present finding of a 69% response rate for the clonidine adhesive patch is commensurate.
The relatively high placebo response rate of 47% is consistent with previous controlled studies in the field [19], [20]. Most likely it is associated with the known exacerbation of tics with increased stress and their remission with decreased stress and the myriad psychological and social factors that may mediate such changes in perceived and experienced stress.
No serious adverse events occurred during the present trial. Further, the rate of adverse events in the clonidine adhesive patch group was low and the two groups did not differ. Hence, the clonidine adhesive patch appears safe and well-tolerated in children and adolescents with tic disorders. The minor increase in heart rate, decrease in blood pressure and increase in bodyweight in the male participants were inconsequential and expected, given the known pharmacodynamics of clonidine.
The effectiveness of clonidine in tic disorders is consistent with emerging neurodevelopmental models. Baym et al. recently reported functional magnetic resonance imaging event-related data that support hyperactivation of both (i) direct basal ganglia dopaminergic pathways and (ii) compensatory subthalamic and prefrontal cortical pathways in Tourette disorder [33]. Noradrenaline is known to modulate dopamine activity in all of these brain regions in order to optimize prefrontal cortically linked neural networks [34]. Hence, medication that alters the functional activity of either/both of these neurotransmitter systems can correct putative imbalance(s) in neural pathways involving them [35].
In conclusion, the present study provides conclusive evidence that the clonidine adhesive patch is effective, safe and well-tolerated in the treatment of tic disorders in children and adolescents. Future systematic research will need to study its effect on tic disorders in the presence of key comorbid conditions (e.g. OCD, ADHD and/or depressive disorders) and its effect on the treatment of these comorbid conditions.
Footnotes
Acknowledgements
The authors would like to acknowledge the hard work of Fan Juan, Zhao Zhi-Min, Tang Hui-Qin, Liu Yi, Qian Yun, Zhou Lin-Lin, Wang Cun-Mei, Zhang Hao, Yu Shun-Ying, Xu Yun from Shanghai Mental Health Center; Zhu Yan, Geng Yao-Guo, Su Qiao-Rong, Huang Guang-Fei, Li Fei from Mental Health Research Institute of Central South University; Chen Yi-Xin, Yu Lin, Zou Bing, Zhang Jiu-Ping, Luo Shuo-Jun, Jiao Gong-Kai, Hang Yue-Yue from Nanjing Brain Hospital; Wang Lei, Liu Li from Harbin Maternity and Child Care Hospital; Zhong You-Quan, Tao Xuan-Hua, Wu Ju, Zhou Wen-Zhi from Chengdu Children's Hospital; Zhang Feng-Ling from Shanghai Children's Hospital; Qiao Jing, Yang Li-Fang, Liu Chun-Feng, Lei Hong-Tao, Wang Ke-Sheng, Gao Xiao-Min, Tang Hua, Guo Xiang-Yang, Gao Ying from Shaanxi Province People's Hospital; and Zhang Xiao-Yan, Wu Bao-Zheng, Wang Hui-Qin, Liu Zheng-Yin from Shanxi Province Children's Hospital. This research was funded by grants from Shanxi Ruifulai Pharmaceutical, Taiyuan, Shanxi Province, China. The research number is: L02689-91. Professor Yasong Du has received research funding from Xi'an-Janssen Pharmaceutical, Eli Lilly, and Shanxi Ruifulai Pharmaceutical.
