Abstract
Schizophrenia is a serious disease characterized by impairment in the perception or expression of reality, leading to occupational and social dysfunction [1]. Symptoms of schizophrenia have been categorized as ‘positive symptoms’, which typically include delusions, hallucinations and thought disorder, and ‘negative symptoms’, which describe inappropriate or non-present emotion, poverty of speech, and lack of motivation.
The use of antipsychotic medication is now universal in the first-line treatment of schizophrenia. The newer atypical antipsychotic drugs are increasingly being preferred over older typical antipsychotic medication due to their favourable adverse drug reaction (ADR) profile [2]. Olanzapine, an atypical antipsychotic introduced in 1996 [3], is now commonly used as a first-line drug in the treatment of acute symptoms of the disease, and also for long-term maintenance therapy. Recent studies have shown that this drug is likely to induce weight gain and presents an increased risk for obesity-related diseases, even metabolic syndrome [2]. Indeed, in the USA, olanzapine comes with a specific warning for increased risk of diabetes mellitus [4].
Amisulpride, a substituted benzamide derivative, is a selective dopamine antagonist that is reported to work against negative symptoms of schizophrenia [5], in addition to positive symptoms, and was approved for marketing in India on 1 August 2006 [6]. Although trials have been reported from Western countries regarding the effectiveness of amisulpride [7,8], studies with this drug are still limited in India. This prompted us to conduct a randomized controlled clinical trial to assess the effectiveness and safety of amisulpride in the Indian scenario in comparison to the standard atypical antipsychotic olanzapine.
Methods
The study was designed as a prospective, single-blind, parallel-group, randomized controlled trial of amisulpride versus olanzapine in schizophrenia patients attending the psychiatry outpatient department (OPD) of Bangur Institute of Neuroscience and Psychiatry (BINP), Kolkata, India. The study protocol conformed to the Declaration of Helsinki (as revised in Edinburgh 2000) and was approved by the Ethics Committee of Institute of Postgraduate Medical Education and Research, Kolkata, India, of which BINP is a part.
The sample size involved 40 evaluable patients in each group. This was estimated to detect a difference, between groups, of 6 in Brief Psychiatric Rating Scale (BPRS) score, with 80% power and 5% probability of Type I error.
Patients diagnosed on DSM-IV criteria [9], 18–65 years of age, were recruited between June 2007 and June 2008, subject to written informed consent from their legally acceptable representatives. Subjects were screened by psychiatry residents and the diagnosis was confirmed by the senior psychiatrist among the authors (MG). The screening included routine blood tests and a standard 12-lead resting electrocardiogram (ECG). Women who were pregnant or breast-feeding, subjects with severe symptoms that precluded an antipsychotic-free run-in period of even 5 days, and those having neurological disease with potential symptomatology resembling schizophrenia symptoms, were excluded. Also excluded were patients with significant disease of vital organs, adrenal or pituitary glands, those receiving medication known to prolong QT interval on ECG or capable of otherwise interacting with study medication, those with history of alcohol or substance abuse and those without sufficiently reliable caregivers at home.
Randomization was done in blocks of 10 by computer-generated random number list. For the individual subject, the treatment duration was 12 weeks following randomization. Follow-up visits were scheduled at 4 weeks and 8 weeks, with provision for additional interim follow ups, if needed, for assessing adverse events.
Each recruited subject received either amisulpride (100–800 mg total daily dose) administered as a single oral daily dose (or in two equal divided doses daily if the total dose exceeded 200 mg) or olanzapine (10–20 mg total daily dose) administered once daily orally. The starting dose was decided by the concerned clinician, depending on the severity of illness. Uptitration, if needed, was permitted only once during the course of the study, within the maximum limits stated here. The medication was supplied to the family members of the patients in airtight screw-cap containers, in 4 week instalments, at baseline, first follow-up and second follow-up visits, with careful instructions to ensure adherence to treatment. The compliance was assessed by the traditional pill count method. Laboratory tests and ECG were repeated at the end of 12 weeks of treatment.
There was no provision for continued supply of study medication to subjects who reported good response to study drugs at the end of 12 weeks of treatment and wished to continue treatment with the same. Appropriate alternatives, however, were prescribed from the Psychiatry OPD as per the discretion of the consultant who followed up the patient after the trial period.
Effectiveness assessment
The primary effectiveness variable for this study was symptom relief as assessed on BPRS score [10]. Secondary efficacy variables were extent of change in negative symptoms assessed on Scale for Assessment of Positive Symptoms (SAPS) [11], extent of change in positive symptoms assessed on Scale for Assessment of Negative Symptoms (SANS) [11] and Clinical Global Impression (CGI) rating by the physician.
Safety assessment
Adverse events recorded included treatment-emergent events reported spontaneously by subjects at any time during the 12 week treatment, serious events reported for up to 4 weeks after completing the scheduled course of study medication, those elicited as clinical signs by the investigators during the scheduled visits and adverse laboratory test results. Causality analysis of adverse events was done using World Health Organization–Uppsala Monitoring Centre standardized case causality assessment criteria [12]. Events with less than ‘possible’ causality rating were ignored.
Data analysis
Effectiveness analysis was on an intention-to-treat basis for subjects reporting for at least one post-baseline visit. All subjects randomized were considered for adverse event analysis. Missing values were dealt with using the last observation carried forward strategy.
Null hypothesis was that amisulpride was not inferior to olanzapine in the treatment of schizophrenia. Primary effectiveness variable and other numerical variables were compared between groups on Student's unpaired t-test. Within-group comparison was on repeated measures analysis of variance followed by Tukey's multiple comparison test for post-hoc testing between two individual time points. Laboratory data were compared between baseline and end-of-study visits using Student's paired t-test. Categorical data were compared between groups using Fisher's exact test or χ2 test as appropriate. All analyses were two tailed and p < 0.05 was taken to be statistically significant.
Analysis was done using SPSS version 11.5 (SPSS, Chicago, IL, USA) and STATISTICA version 6.0 (StatSoft, Tulsa, OK, USA).
Results
We screened 124 subjects for this study, of whom 80 (64.52%) were recruited, and 40 randomly assigned to each group. Seventy-seven subjects (96.25% of recruited) were analysable: 39 in the amisulpride and 38 in the olanzapine groups. The flow of patients is given in Figure 1.
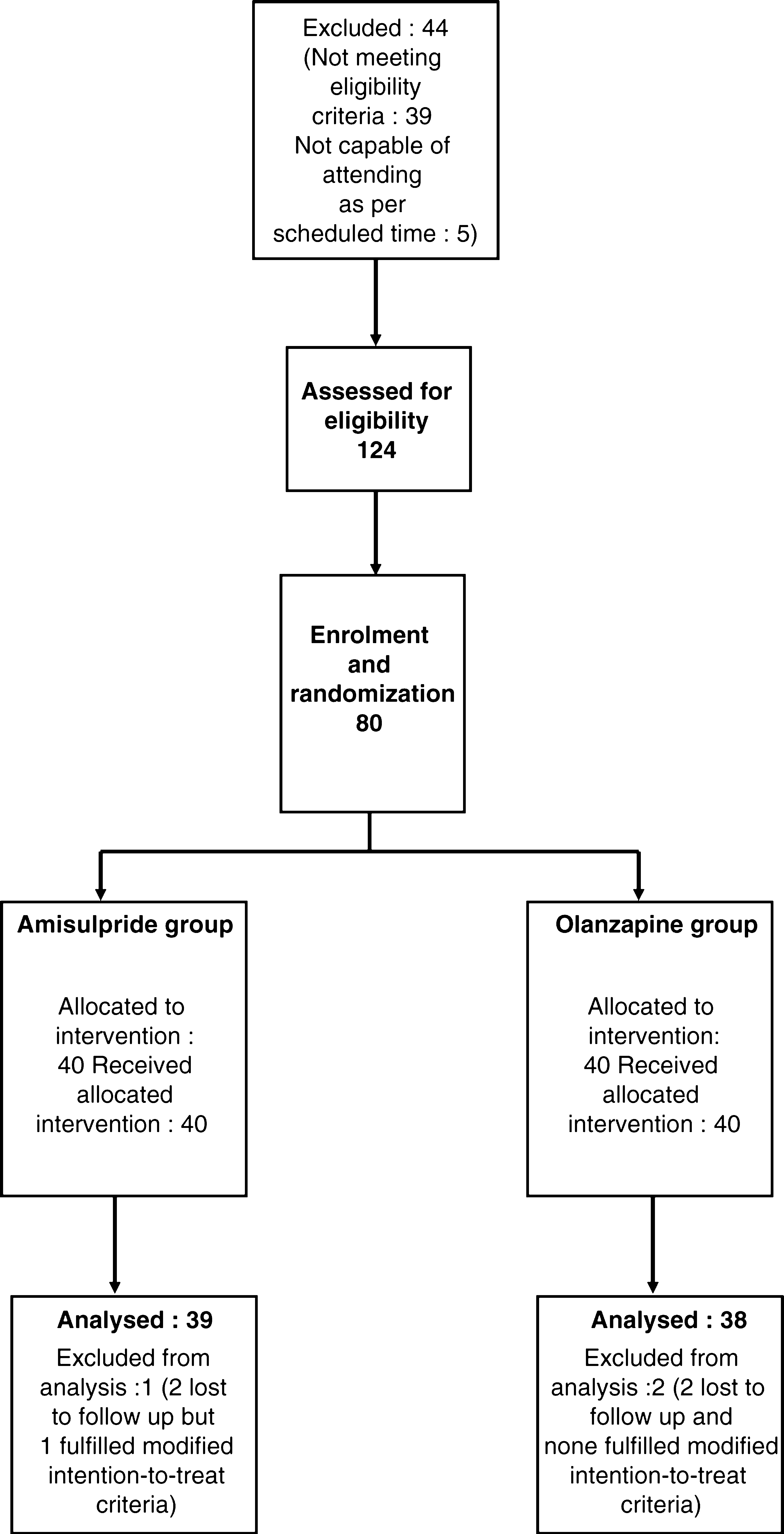
Flowchart of the study population.
The groups were comparable at baseline with respect to demography, duration of illness, and symptom scores. The mean ±SD age was 31.8 ± 7.5 years and 31.4 ± 7.7 years in the amisulpride and olanzapine groups, respectively, indicating a preponderance of young adult subjects. Men comprised 53.85% (amisulpride) and 52.63% (olanzapine) of the sample in the two groups. The mean duration of schizophrenia in patients receiving amisulpride and olanzapine was 25.1 ± 8.8 months and 25.8 ± 9.5 months, respectively.
Effectiveness parameters
The BPRS score was the primary effectiveness parameter, and serial changes over the 12 week treatment period are listed in Table 1. There was steady and statistically significant decline in both groups, with the net reduction in the olanzapine group (mean difference between study end and baseline = 28, 95% confidence interval (CI) = 25.27–30.78) being greater than in the amisulpride group (mean difference = 25, 95%CI = 21.73–27.15). The difference between groups was statistically significant from 4 weeks onwards and remained so at study end.
Brief Psychiatric Rating Scale score changes
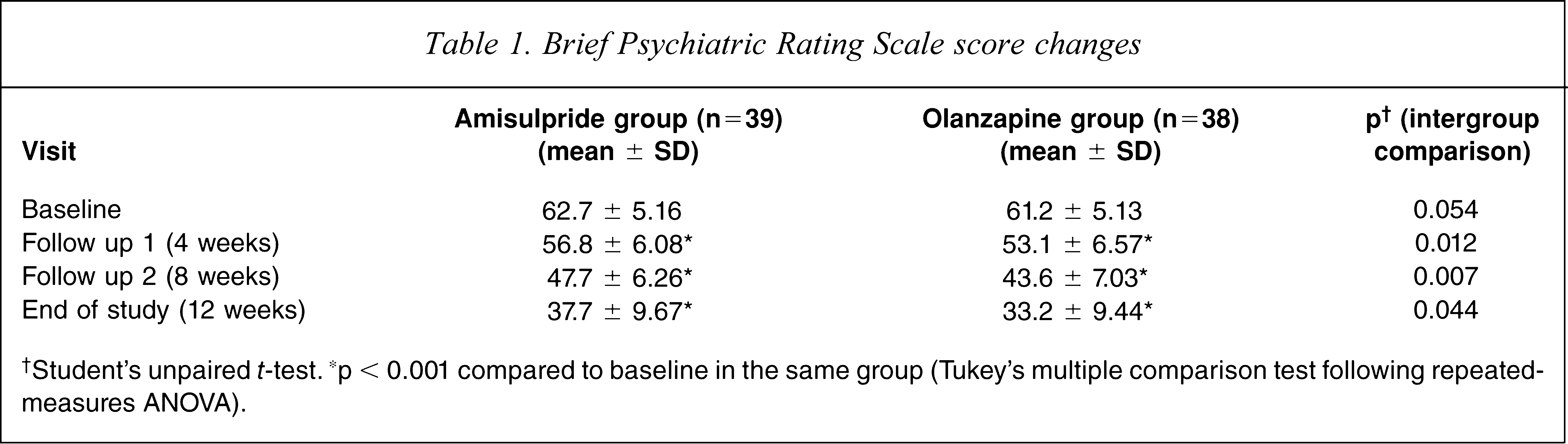
†Student's unpaired t-test. ∗p < 0.001 compared to baseline in the same group (Tukey's multiple comparison test following repeated-measures ANOVA).
Changes in SAPS and SANS scores over the 12 weeks are listed in Table 2. With amisulpride, the mean reduction from baseline to study end was 21.3 (95%CI=16.54–24.13) in SAPS score and 25.7 (95%CI=22.22–29.21) in SANS score. With olanzapine, the corresponding reductions were 31.4 (95%CI=27.14–35.70) in SAPS score and 26.3 (95%CI = 23.6–28.97) in SANS score. Individually, both parameters showed marked improvement in each of the study groups and the improvement was statistically significant from the first follow up at the end of 4 weeks of treatment. Compared between groups, the scores remained comparable from baseline to study end, although the trend was towards greater reduction in SAPS score in the olanzapine arm.
Positive and negative symptom score changes

SANS, Scale for Assessment of Negative Symptoms; SAPS, Scale for Assessment of Positive Symptoms. †Student's unpaired t-test. ∗p < 0.001 compared to baseline in the same group (Tukey's multiple comparison test following repeated-measures ANOVA).
The CGI rating was administered by the physician during the follow-up and end-of-study visits and the summary counts are given in Table 3. Although there was a statistically better outcome in the olanzapine arm at the first follow-up visit, the groups became comparable at the subsequent follow-up visit and remained so at study end.
Changes in physician-rated CGI score
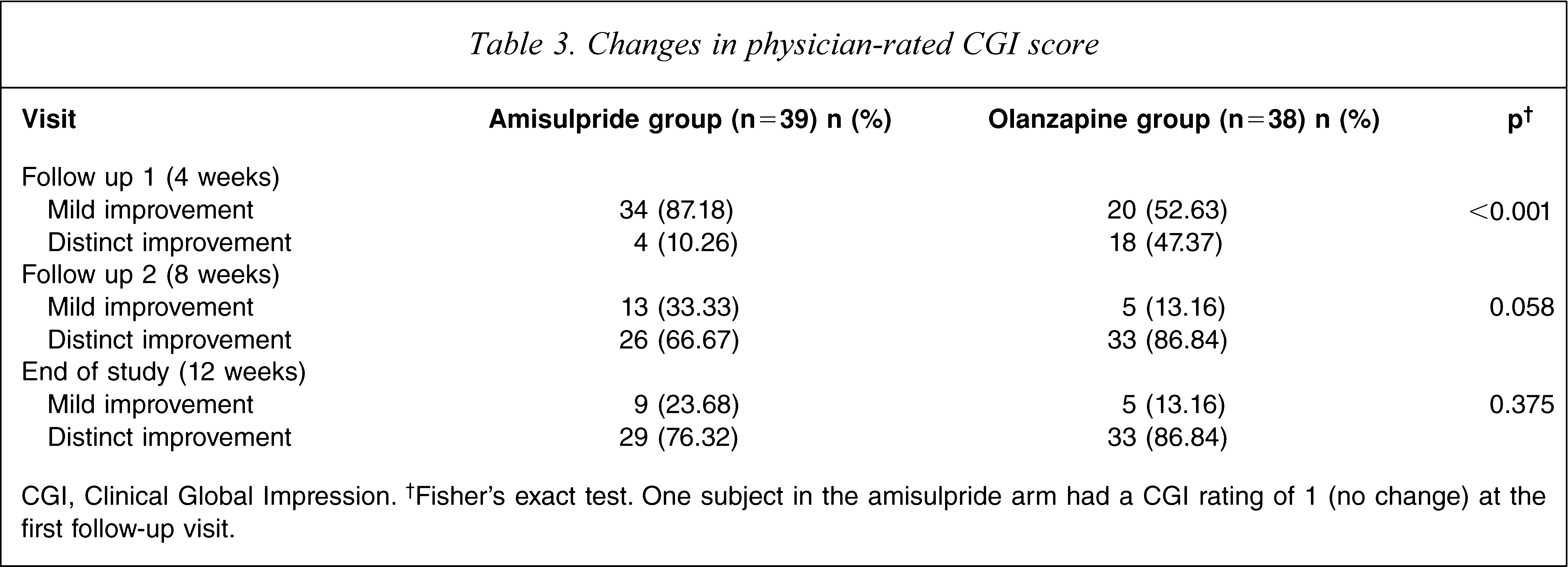
CGI, Clinical Global Impression. †Fisher's exact test. One subject in the amisulpride arm had a CGI rating of 1 (no change) at the first follow-up visit.
Safety parameters
Mean heart rate, systolic and diastolic blood pressures did not show any significant changes and values remained in the normal range throughout in both groups.
Practically all patients in the present study, except one in the amisul-pride group, had at least 0.5 kg weight gain from baseline to study end. The average gain in olanzapine-treated patients was significantly higher than their amisulpride-treated counterparts (4.7 ± 1.18 kg vs 3.5 ± 1.42 kg, p < 0.001). The difference was also statistically significant when considering the number of subjects having >10% gain over their basal bodyweight (12 vs 4, p = 0.026).
No statistically significant difference was recorded between baseline and end-of-study values of routine biochemistry parameters, and individually the mean values were also within clinically normal range. No clinically significant ECG changes were observed for either drug.
Overall, 76 treatment-emergent adverse events were recorded from 46 subjects during the study: 27 subjects (67.5% of the 40 randomized) in the amisulpride arm and 19 (47.5%) in the olanzapine arm reported at least one event. This difference was not statistically significant (p = 0.113). The suspected ADR profiles are listed in Table 4. The incidence of tremor and insomnia was greater with amisulpride, while weight gain and sedation occurred more frequently with olanzapine. The events were mild or moderate in severity. There were no hospitalizations or other serious adverse events.
Individual suspected adverse drug reactions
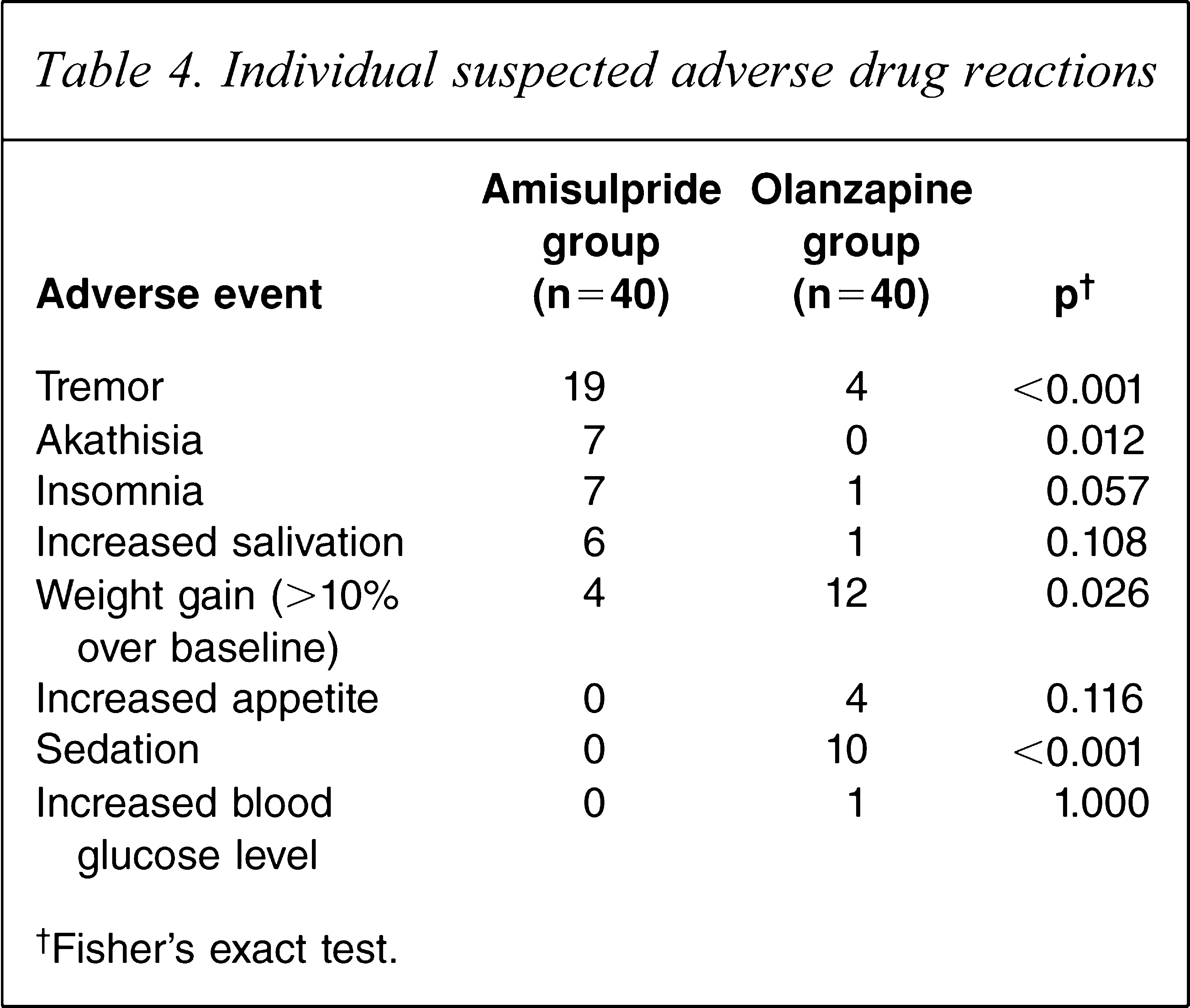
†Fisher's exact test.
Discussion
Both amisulpride and olanzapine were effective in combating the various symptoms of schizophrenia over the 12 week treatment period, as evident from the declining BPRS, SAPS and SANS scores. The overall decline in BPRS score was higher in the olanzapine arm, and the difference between groups was statistically significant at study end. Although the SAPS score remained comparable between the two groups throughout the study period, olanzapine showed a distinct trend towards greater reduction. The reduction in SANS score was comparable. Physician CGI rating, although initially favouring olanzapine, became comparable at study end. These suggest that although amisulpride was comparable to olanzapine in some measures, it could not match the improvement in BPRS and SAPS scores.
Three large randomized control trials comparing amisul-pride and olanzapine in schizophrenia have been published to date [7,8,13]. The present results are in agreement with those of a French study that compared olanzapine with amisulpride in patients experiencing predominantly negative symptoms [7]. Here, olanzapine showed significantly greater improvement than placebo and amisulpride in the positive and negative symptom scales total scores. The neurological tolerance of olanzapine, amisulpride and placebo were comparable. In the European Schizophrenia Outpatient Health Outcomes study, >10 970 patients from 10 European countries, who were initiating or changing antipsychotic medication for the treatment of schizophrenia, were studied for 3 years [13]. The patients treated with olanzapine and clozapine tended to have somewhat better outcomes than patients treated with other atypical or typical antipsychotics. That study also showed that olanzapine reduced the positive and negative symptoms of schizophrenia better than amisulpride.
In a large British study by Mortimer et al. comparing the two drugs for a period of 6 months, the primary efficacy variable was change from baseline in the BPRS score, assessed through a non-inferiority analysis [8]. The evolution of positive and negative symptomatology, depression, social functioning and quality of life were assessed. Safety evaluation included adverse events reported, neurological status and bodyweight. The improvement of BPRS score was 32.7% in the amisulpride group and 33.0% in the olanzapine group, thus, the efficacy of amisulpride was not inferior to that of olanzapine.
Tolerability of both drugs was satisfactory in the present study, matching the experience in other studies. Although ADRs were frequent in both arms, their intensity and frequency did not force withdrawal in any case. The spectrum encountered with amisulpride is similar to the known ADR profile of the drug to date [14].
The present study had certain limitations. The sample size was calculated on the basis of a relatively large effect size with respect to BPRS score. A larger sample may have shown a clear statistical difference between groups in other measures such as SAPS score and CGI rating. The study was required to be single blind because we lacked the resources required for a truly double-blind study. The medication was obtained in their commercial packs and then repackaged in a coded fashion; they were similar but not identical in appearance. Also, the dosing frequency of amisulpride was twice daily, when the total daily dose exceeded 200 mg Antipsychotic drugs are known to increase serum prolactin [15] but we could not measure prolactin levels because of logistic constraints. Finally, the treatment period in this study was relatively short (12 weeks), and hence provided no scope for assessing comparative effectiveness of long-term therapy or the detection of delayed adverse events.
In conclusion, in Indian schizophrenia patients, amisul-pride, although comparable to olanzapine with respect to improvement of negative symptoms, did not match the improvement seen with the latter drug in BPRS and SAPS scores. Although overall tolerability was satisfactory for both drugs, some differences were encountered in the ADR profiles. Therefore, when it is a question of choosing between the two drugs, amisulpride should be the secondary choice, unless it is important to avoid known problems with olanzapine such as weight gain and sedation, or unless the patient presents with predominantly negative symptoms. The fact that amisulpride brands currently marketed in India are costlier than olanzapine adds support to this conclusion.
Footnotes
Acknowledgements
We are grateful to Dr Dipankar Kanjhi and Dr. Sanchari Roy, Residents, Department of Psychiatry, Bangur Institute of Neuroscience and Psychiatry, Kolkata, India, for their assistance in screening subjects for recruitment. The study was undertaken as an academic project. It was not sponsored by any pharmaceutical company or contract research organization. None of the investigators had any conflict of interest in the conduct of this trial and did not receive any remuneration for the project. The investigators, however, approached Sun Pharmaceutical Industries, Mumbai, India for donation of the study drugs and gratefully acknowledge this donation. This company markets both olanzapine and amisulpride in India, but played no role in the design and conduct of the trial or in the analysis of the results.
