Abstract
It has long been recognized that people with schizophrenia experience a range of cognitive deficits particularly in memory, attention, verbal fluency and executive functions. These deficits in cognitive functioning are of the order of one to two standard deviations below the mean of normal healthy individuals [1–4]. Positive symptoms of schizophrenia have been shown to be unrelated to cognitive deficits [4–6], but significant relationships have been found between negative symptoms and deficits in memory, verbal fluency, executive functions and processing speed [4], [7].
Previous research has demonstrated an important association between cognitive deficits and functional abilities in people with schizophrenia. Attention, verbal learning and fluency are related to successful performance of social skills [8], verbal memory has been found to predict independence and community involvement [9–11], and executive functioning is strongly related to work performance and activities of daily living [11–13]. These findings have led to major initiatives to improve cognition in schizophrenia, in the hope that this may lead to improved vocational outcomes [14], [15].
The aim of mental health clinicians working with people with schizophrenia is not just to improve symptoms and functional outcome, but also to improve their overall quality of life (QOL). It can be assumed that there will be some relationship between symptoms, cognition and QOL, but the extent to which each of these dimensions contributes to QOL is unclear. Defining this relationship has proven challenging, and the present study was designed to address this issue.
Defining and measuring QOL are not easy. The World Health Organization (WHO) defines QOL as the individual's perception of their position in life in the context of the culture and value systems in which they live and in relation to their goals, expectations, standards and concerns [16]. QOL includes physical and psychological health, level of independence, and social relationships.
QOL can be conceptualized as consisting of both subjective and objective dimensions, and assessment of each dimension presents challenges in people with schizophrenia. Schizophrenia is characterized by impairments in judgement, insight and mood, and the presence of psychotic symptoms such as hallucinations and delusions, which may affect the person's subjective assessment of their situation. Objective QOL includes everyday functioning and activities, and people with schizophrenia are not always accurate in rating their community functioning [17]. A number of instruments have been developed to measure QOL. Some, such as the WHO scale for measuring QOL (WHOQOL-Bref) are designed to allow comparisons between groups of people who may differ in culture or economic status, or in the type of disability they have. The WHOQOL-Bref has been used in schizophrenia research [18], [19], although there are few comparisons between people with schizophrenia and those with other conditions [20]. The Short-Form 36-item Health Survey (SF-36) and the EuroQnol-5 Dimension (EQ-5D) are also generic QOL instruments shown to have good validity in schizophrenia [21], [22], but these instruments are not sensitive to the specific problems experienced by people with schizophrenia.
The Quality of Life Scale (QLS), the instrument selected for the present study, is a clinician-rated instrument that measures objective QOL, with a focus on deficit symptoms [23]. The QLS is widely used in schizophrenia research [24] and a shortened version has been developed for outcome research [25]. It has four subscales: interpersonal relationships and social network; instrumental role functioning; intrapsychic foundations; and common objects and activities. The QLS has been shown to have a moderate relationship with subjective measures of QOL in schizophrenia, although the QLS score was more strongly influenced by negative symptoms and occupation status [26]. More recently, objective measures of functioning have been developed that directly test a subject's ability to perform a range of tasks, such as the University of California San Diego (UCSD) Performance-Based Skill Assessment [27]. But the ability to perform a task does not mean that the person necessarily chooses to use this ability in their day-to-day life, so there may not always be a direct relationship between laboratory-assessed functional abilities and QOL.
Reviewing previous research on the association between symptoms, cognition, functional outcome and QOL, it is apparent that the types of measures used to assess QOL may influence the results. Several studies have demonstrated a relationship between negative symptoms of schizophrenia and QOL [4], [5] but it has been noted that these studies tended to use the QLS, which includes deficit symptoms but does not address subjective well-being [28]. Norman et al. found that both positive and negative symptoms and level of functioning were related to QOL, measured with the QLS [28]. Only positive symptoms were related to scores on a General Well-being Scale, a self-rated index of subjective well-being. Positive symptoms are also thought to have a detrimental impact on functional abilities, such as productivity in the workplace, social skills and concentration [29].
More recently, Hofer et al., using the WHOQOL-Bref, found that both positive and negative symptoms were associated with QOL, and cognitive symptoms influenced occupational functioning [30]. George et al. also found that QOL was negatively affected by cognitive deficits in 120 people with chronic schizophrenia [7]. In contrast, in a study of 42 outpatients with schizophrenia, Heslegrave et al. found that cognitive deficits were largely unrelated to QOL [4]. Only attention span and memory were related to QOL, and these were the least impaired cognitive functions. Heslegrave et al., however, used the Sickness Impact profile, a health-related QOL measure, which may be applicable only to a narrow component of QOL [4].
Herman used the WHOQOL-Bref scale and an extensive neuropsychological battery to examine the relationship between cognitive deficits and QOL [31]. Executive Functioning was found to be the strongest predictor of QOL, over general intelligence and memory. Galletly et al. and Alptekin et al. also found a moderate relationship between QOL and cognitive functioning, such that more severe cognitive deficits were associated with a poorer QOL [32], [33]. Addington and Addington reported that in both first-episode and multi-episode schizophrenia patients, cognitive impairment was associated with a lower total QLS score and with poorer performance on a videotaped vignette test of interpersonal problem solving, but there was no relationship between cognition and scores on a self-report instrument assessing social functioning [34]. It appears therefore that in that study assessments rated by clinician interview correlated with cognition while assessments rated by the patients themselves did not show this relationship. Perlick et al. found that symptom severity accounted for 16% of the variance in QLS score, while cognition explained only 8% of the variance [24].
The aim of the present study was to investigate the relationships between QOL, cognitive function and symptoms in people with schizophrenia. We selected the QLS because this is the most widely used QOL instrument in schizophrenia research and the use of the QLS allows our findings to be compared with those of previous studies. Based on the literature reviewed here, we hypothesized that poorer QOL would be related to poorer cognitive functioning and to more severe positive and negative symptomatology. We anticipated that cognitive dysfunction would be related to more severe negative symptoms, but not to severity of positive symptoms. Further, we expected that cognitive deficits and symptomatology would each make a unique contribution to the variance in QOL scores, after controlling for age, gender, and substance (drug and/or alcohol) abuse.
Exploratory analyses were also conducted to examine the relationships between specific aspects of QOL and the different domains of cognitive function, positive symptoms, negative symptoms, and general psychopathology, and to determine whether age, gender, or substance abuse contributed to the variance explained in QOL.
Methods
Participants
Fifty-seven outpatients fulfilling the DSM-IV-TR [35] criteria for schizophrenia and living in the community were recruited from the Northern Mental Health Service, Adelaide. The sample was self-selected and represents all eligible outpatients who agreed to participate during the time available for data collection. Participants were >18 years of age, maintained on a stable dose of antipsychotic medication, fluent in English and able to participate in cognitive assessment. The ethics committee of the North West Area Health Service, Adelaide, approved the study and all participants gave written informed consent. Ethics approval did not allow the collection of information from non-participants to determine the representativeness of the sample. Exclusion criteria included presence of another Axis I diagnosis, intellectual disability, a history of significant head injury, neurological condition, or substance abuse that was deemed severe enough to have had an independent effect on cognitive functioning. There were no payments to participants. The final sample consisted of 43 men and 14 women, with an age range of 21–55 years (mean age = 36.07 years, SD = 9.09).
Measures and procedure
Administration of all study measures was undertaken by a research nurse trained in the use of these instruments. The research nurse was blind to the study hypotheses. Positive and Negative Syndrome Scale (PANSS) training was undertaken using standardized training DVDs in addition to individual supervised training with a consultant psychiatrist (CG). At baseline, diagnoses were confirmed, and comorbid Axis I diagnoses excluded, using the Mini-International Neuropsychiatric Interview [36]. On confirmation of eligibility for the study, participants completed the PANSS [37]. QOL was measured with the Quality of Life Scale (QLS) [23]. A QLS Total score can be calculated by adding scale scores, with higher scores reflecting better QOL.
After a rest break, neuropsychological functioning was assessed with the Brief Assessment of Cognition in Schizophrenia (BACS) [38]. This clinician-administered assessment takes 30–40 min to complete and is designed to be used by a variety of testers including nurse clinicians and other mental health workers. The BACS testing was performed by the research nurse who had undertaken specific training provided by the research team who designed the BACS [39], with supervision provided by a consultant psychiatrist (CG) trained in neuropsychological testing procedures. The BACS includes measures of verbal memory (learning a list of 15 words), working memory (a digit sequencing task), motor function (placing 100 plastic discs into a container), attention and speed of information processing (symbol coding), verbal fluency (FAS and category instance generation) and executive functioning (Tower of London task). A BACS Total score provides a measure for overall cognitive performance. The control data used to compare the performance of our subjects with a normal control sample were collected by Keefe et al. [39] and is described in their paper reporting the reliability and validity of the BACS [39]. The mean age of the control sample (n = 50) was 34.8±11.4 years and there were 19 women and 38 men.
Statistical analysis
All results are presented as mean±SD. Two-tailed independent samples t-tests were used to compare participants’ scores on the component measures of the BACS with standard scores [39]. Effect sizes were calculated and interpreted according to the guidelines specified by Cohen [40]. Pearson's product–moment correlations were used to examine the primary associations between aspects of cognitive function and both QOL and symptomatology. The threshold for statistical significance was set at p = 0.05. It is acknowledged that this is a conservative threshold to apply for this modest sample size, but it was decided that a conservative statistical approach was the most ethically appropriate in this vulnerable sample. A Bonferroni correction to p = 0.007 was applied where multiple comparisons were undertaken. Hierarchical multiple regression analysis and simultaneous multiple regression analysis were used to develop more specific models of the contributions of cognitive deficits and symptomatology to QOL, while accounting for the impact of demographic and additional psychopathological variables. All analyses were conducted using the SPSS version 13.0 [41].
Results
Symptomatology and QOL
The sample had relatively low mean levels of PANSS positive symptoms (18.25±6.61), negative symptoms (20.82±7.32), and general psychopathology (36.56±21.37), as would be expected in outpatients maintained on stable doses of antipsychotic medication. QLS scores demonstrated moderately impaired Interpersonal Relations (20.65±10.95), Instrumental Role Functioning (10.60±5.22), Intrapsychic Foundations (19.23±8.37), Common Objectives/Activities (6.37±2.74), and moderate impairment in overall QOL, as reflected by QLS Total scores (56.82±23.19). Sixteen subjects (25.8%) abused alcohol and 18 subjects (29%) abused other substances.
BACS scores (Table 1) showed that participants had significantly poorer working memory (t(105) = 1.80, p < 0.05, d = 0.35) and executive functioning (t(105) = 2.26, p < 0.05, d = 0.44) than healthy controls.
BACS subtest scores (mean±SD)
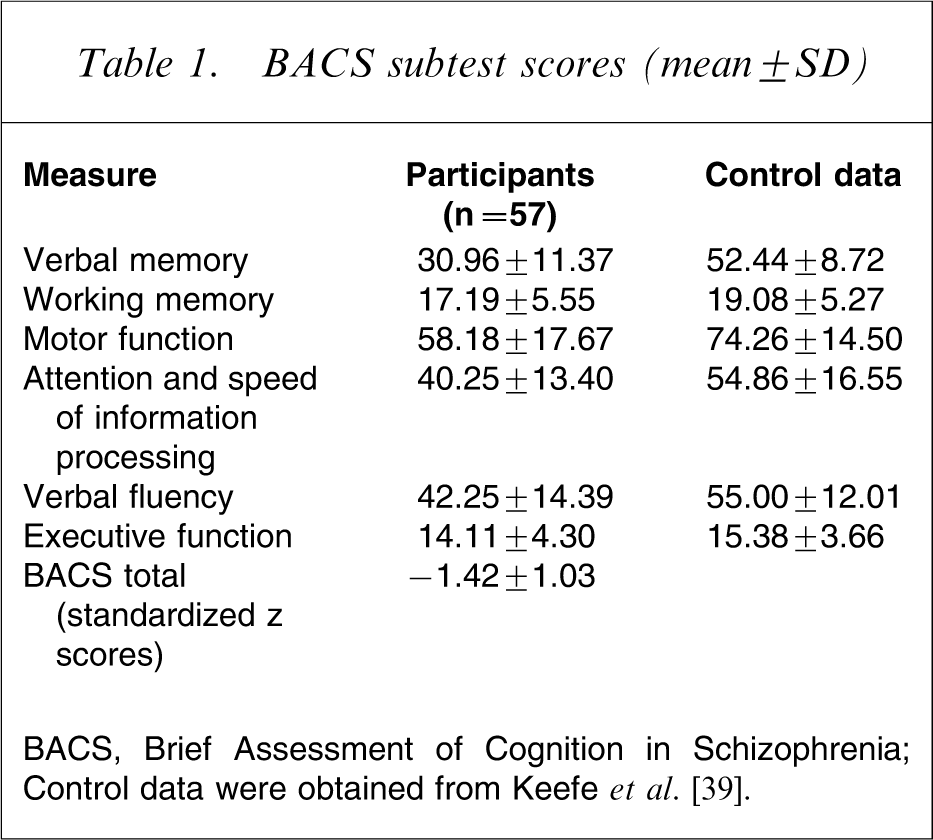
BACS, Brief Assessment of Cognition in Schizophrenia.
Control data were obtained from Keefe et al. [39].
Cognitive functioning and symptom severity
It was hypothesized that the severity of negative symptoms, but not positive symptoms, would be associated with poorer cognitive functioning. Using the Total BACS score and PANSS negative symptom and positive symptom scores, respectively, poorer cognitive functioning was found to have a moderate association with increased negative symptoms (r= − 0.30, p < 0.05). There was also a small relationship between cognitive functioning and positive symptoms, but the relationship did not reach statistical significance in this sample (r = − 0.16, p > 0.05) [40].
QOL and cognitive functioning
It was hypothesized that poorer QOL would be related to poorer cognitive functioning. Using Total QLS and BACS scores, poorer cognitive functioning was associated with poorer QOL (r = 0.44, p < 0.01).
QOL and symptom severity
Using Total QLS and PANSS scores, greater symptom severity was found to be related to poorer QOL (r = − 0.71, p < 0.05). On closer examination, Total QLS scores were lower among participants experiencing more positive symptoms of schizophrenia (r = − 0.51, p < 0.01), and also among those experiencing more negative symptoms (r = − 0.64, p < 0.01).
Prediction of QOL using total scores for cognitive function and symptom severity
A hierarchical multiple regression analysis was conducted to examine the unique contribution of cognitive functioning and symptomatology to QOL, while controlling for demographic and clinical variables. The model summary after step 1 demonstrated that age, gender, substance dependence and substance abuse did not significantly predict QLS Total scores, Adjusted R2= − 0.06, F(4,52) = 0.18, p > 0.05. These variables in combination explained only 1.4% of the variance in Total QLS. The addition of BACS Total and PANSS Total scores at step 2 produced a model that significantly accounted for an additional 57.0% of the variance in QLS Total scores, (adjusted R2=0.53, F(6,50) = 11.61, p < 0.001). The results also demonstrated that BACS Total score and PANSS Total score were both significant independent predictors of QLS Total scores (Table 2).
Hierarchical regression analysis for variables predicting QLS Total (n = 56 complete cases)
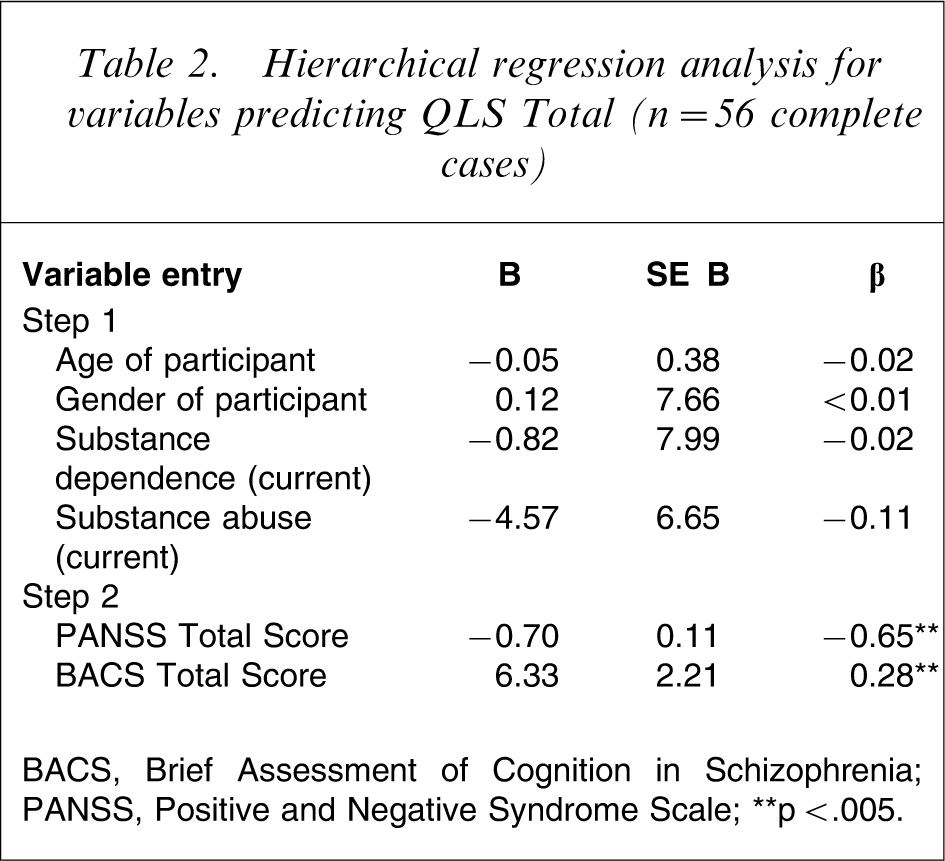
BACS, Brief Assessment of Cognition in Schizophrenia; PANSS, Positive and Negative Syndrome Scale.
∗∗p<.005.
Unique relationships between individual cognitive domains and domains of QOL
After applying a Bonferroni correction for multiple comparisons, 11 significant correlations were found between measures of specific components of cognitive functioning and aspects of QOL as measured by the QLS subscales (Table 3).
Correlations between domains of cognitive functioning, and quality of life
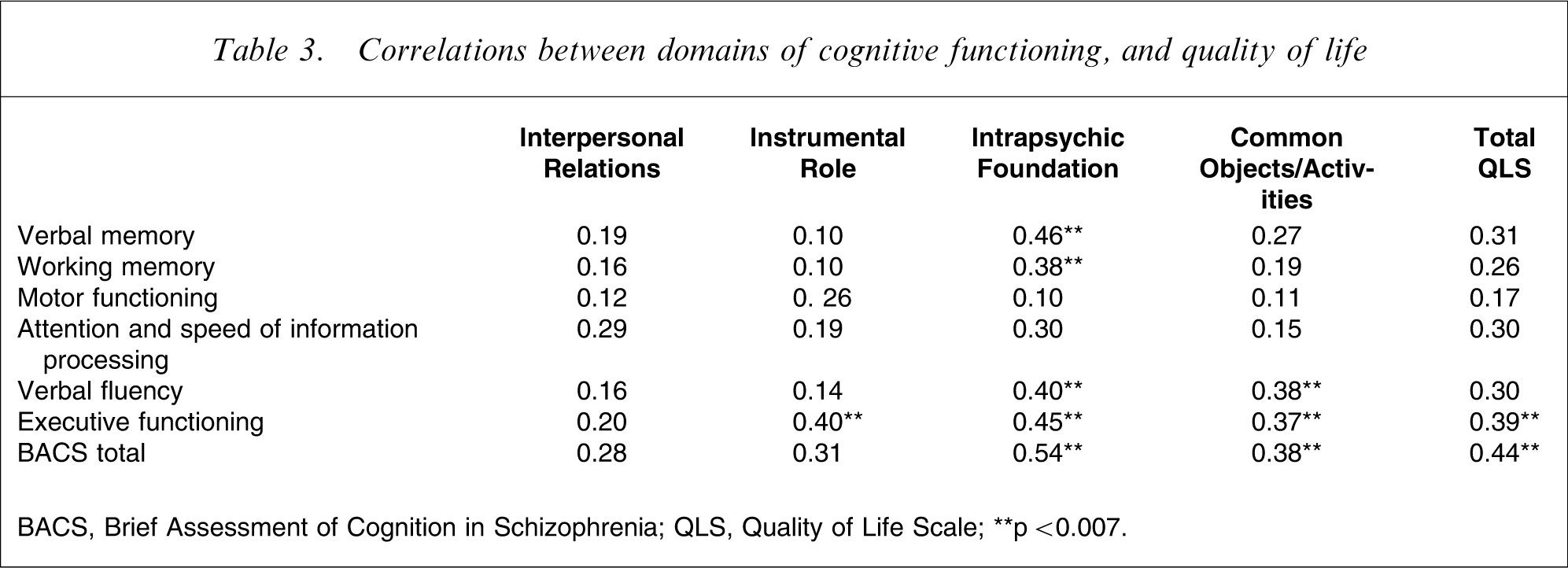
BACS, Brief Assessment of Cognition in Schizophrenia; QLS, Quality of Life Scale.
∗∗p < 0.007.
To determine the unique contributions of verbal memory, working memory, motor function, attention and speed of information processing, verbal fluency and executive functioning to overall QOL, a simultaneous multiple regression analysis was performed. Results demonstrated that the overall model approached statistical significance (adjusted R2=0.11, F(6,50) = 2.20, p = 0.06), but that executive function was the only cognitive subtest to approach significance as an independent contributor to Total QLS scores (β = 0.23, p = 0.10).
Unique relationships between symptom types and domains of QOL
A series of Pearson's correlations, entering all PANSS and QLS scores, was conducted to explore whether different symptom types (i.e. positive and negative symptoms, and general psychopathology) were associated with specific aspects of QOL. Statistically significant moderate − strong relationships were found between all symptom scores and QLS subscale scores (Table 4).
Correlations between symptom type, and quality of life domain
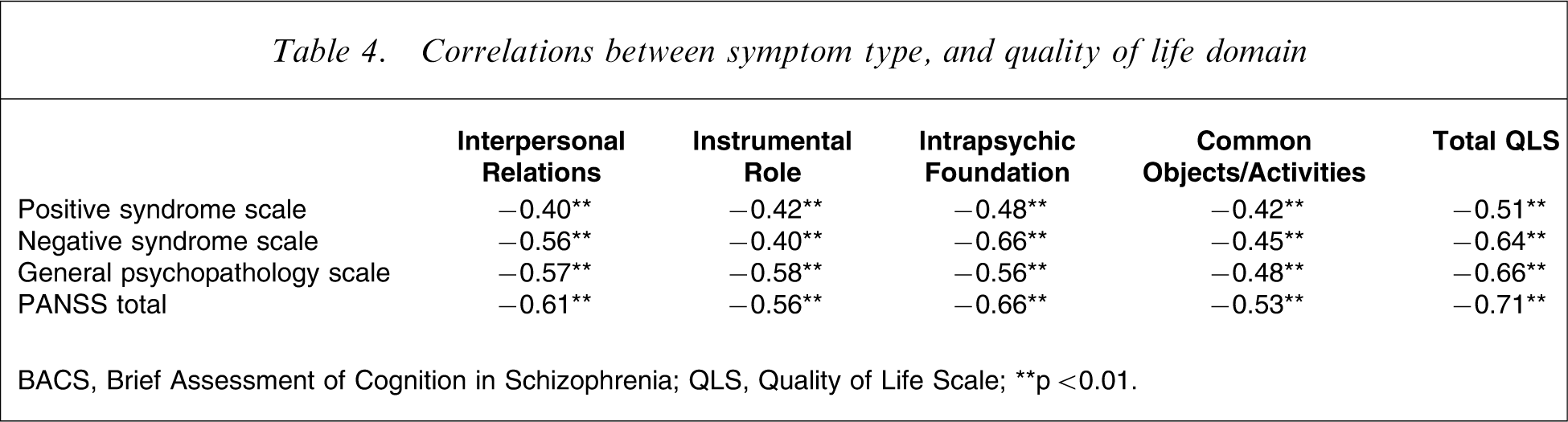
BACS, Brief Assessment of Cognition in Schizophrenia; QLS, Quality of Life Scale.
∗∗p < 0.01.
To determine the unique contribution of positive symptoms, negative symptoms and general psychopathology to overall QOL, a simultaneous multiple regression analysis was performed. Results demonstrated that PANSS positive and negative scores explained a significant proportion of the variance in QLS Total (adjusted R2=0.50, F(3,53) = 19.79, p < 0.001). Negative symptoms and general psychopathology significantly predicted QLS Total scores (Table 5).
Simultaneous regression analysis for symptomatic variables predicting QLS Total scores
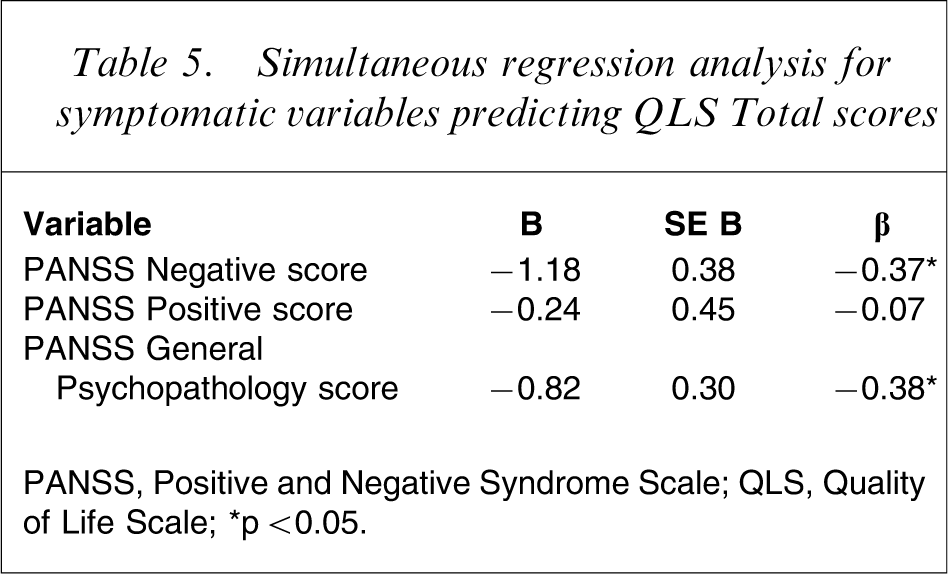
PANSS, Positive and Negative Syndrome Scale; QLS, Quality of Life Scale.
∗p < 0.05.
Discussion
The aim of the present study was to explore the relationships between cognitive functioning, symptoms and QOL in patients with schizophrenia living in the community. The participants demonstrated significant deficits in working memory and executive functioning compared to a healthy standardization sample. They also had lower scores for motor functioning, verbal fluency, attention, speed of information processing and verbal memory than the healthy standardization sample, but these differences did not reach significance. The degree of cognitive impairment found among participants in the present sample is consistent with the cognitive deficits found in previous studies of individuals with schizophrenia [2–4], [12]. The mean Total QLS scores for the present sample were comparable with those reported by Addington and Addington, who reported a mean score of 54.8±21.2. for first-episode subjects attending a first-episode psychosis programme and 67.7±19.4 for multi-episode schizophrenia subjects attending an outpatient clinic [34]. Perlick et al. reported a lower mean QLS score of 45.1±17.0 for an older sample of US veterans with schizophrenia [24].
The present study supported previous research demonstrating a significant positive correlation between cognitive functioning and QOL, such that better cognitive functioning was related to better QOL [7], [31], [33]. Poorer cognitive functioning was also found to be moderately and significantly related to more severe negative symptoms of schizophrenia while a smaller and statistically non-significant relationship was found between cognitive functioning and positive symptoms, lending partial support to the hypothesis that only negative symptoms would be associated with cognitive deficits. Given that negative symptoms and cognitive deficits have a degree of independence from the positive symptoms of schizophrenia, they should therefore be addressed as separate targets of treatment. The Measurement and Treatment Research to Improve Cognition in Schizophrenia (MATRICS) initiative specifically addresses cognition in schizophrenia [15], and it can be anticipated that enhancement of cognition will have benefits in terms of both QOL and vocational outcome.
As shown previously, QOL was associated with symptomatology [4–6]. Both negative and positive symptoms of schizophrenia were found to be strongly associated with overall QOL, such that more severe symptoms were related to poorer QOL. Given that the QLS does include measures of deficit symptoms, an association between negative symptoms and QLS score would be expected. The association between more severe positive symptoms and poorer QOL was anticipated, in that positive symptoms such as hallucinations and delusions are very difficult to tolerate as part of day-to-day living.
In examining the unique relationships between domains of cognitive functioning and QOL, it was found that impaired executive functioning had the most pronounced effect, being significantly associated with all areas of QOL except Interpersonal Relationships, again supporting findings of previous studies [11], [12]. The present study further found that poor verbal memory, working memory, and verbal fluency correlated significantly with the Intrapsychic Foundation domain of the QLS. Because the Intrapsychic Foundation subscale reflects some of the negative symptoms of schizophrenia, these findings may also reflect the association between cognitive deficits and negative symptoms.
Although many domains of cognitive functioning appear to have strong associations with QOL, it must be noted that separate components of cognitive functioning were not found to make significant independent contributions to the variance explained in total QOL scores. It appears, therefore, that cognitive functioning is only a significant predictor of QOL when considered in total, and in conjunction with the symptoms of schizophrenia. Conversely, the positive and negative symptoms of schizophrenia, in combination with general psychopathology, contributed a significant proportion to the variance explained in QOL scores. Further examination showed that while all symptom types correlated significantly with all QOL domains, only negative symptoms and general psychopathology emerged as significant predictors of QOL. We can conclude, based on these findings, that clinical measures addressing global cognitive functioning, negative symptoms, and non-psychotic symptoms such as anxiety and depression are most likely to be helpful. More broadly, these interventions may include addressing drug and alcohol abuse; lifestyle programmes addressing fitness, healthy eating and general well-being; anxiety management and other cognitive behavioural therapies; and increased engagement in social activities. As yet there has been very little research into the effects of such interventions on QOL in schizophrenia.
There were some limitations to the present study. The sample was small and self-selected, limiting statistical power. In combination with the correlational design of the study, this reduced our ability to reach strong conclusions about the relationships between the variables in question and imposed limits on the generalizability of the findings. There are challenges associated with the conduct of research in this population, including instability of housing and living arrangements, erratic contact with outpatient services and the obvious contribution of symptomatology to the willingness of individuals with schizophrenia to participate in research. These factors limited our ability both to improve design and increase the size of the sample. There is no doubt that research in larger samples, and of longitudinal design would be a welcome addition to the literature, but we believe that cross-sectional studies such as the present study, in the context of the existing literature, assist in building understanding of these complex relationships.
Considering the present study in the broader context of research into cognition in schizophrenia, the present results lend some support to the hope that improving cognition might be associated with improved QOL. Cognitive remediation therapies are showing some benefits and could be included more extensively within standard rehabilitation programmes [42], [43]. While clinician-delivered cognitive remediation is time-intensive and therefore relatively expensive, computer-delivered remediation programmes have also been shown to be beneficial and are considerably more cost-effective [13], [44]. McGurk et al., in a meta-analysis of cognitive remediation in schizophrenia, found that cognitive remediation therapy resulted in significant improvements with a mean effect size of 0.41 for global cognitive performance, 0.52 for verbal working memory, 0.41 for attention and vigilance and 0.47 for reasoning and problem solving [45]. The effect sizes were smaller for less proximal outcome measures, with 0.36 for improvement in psychosocial functioning, and 0.28 for symptomatic improvement. McGurk et al. note the lack of long-term follow up in most of these studies [45]. Although overall there was some improvement in psychosocial functioning, many studies did not include measures of psychosocial outcome.
Cognitive deficits and symptomatology contributed 57% to the variance explained in QOL scores in the present sample, after controlling for other demographic and clinical variables. It can be speculated that the remaining variance in QOL may be related to other factors such as the secondary consequences of chronic mental illness. People with schizophrenia have high rates of unemployment, often rely on government benefits, and therefore generally live in poor social circumstances [46]. They tend to be socially isolated, and their physical health is often worse than the general community [47]. Stigma and discrimination can further contribute to social adversity [48]. Although research (including the present study) generally focuses on psychopathological and neurocognitive variables, a broader research approach with increased attention to social and psychological factors may enable us to identify other targets for intervention that may improve QOL in this especially disadvantaged population.
Footnotes
Acknowledgements
The authors wish to acknowledge the contributions of Ms Beverly Hisee, Dr Peter Tyllis, Dr Darryl Watson and the Northern Mental Health Research Unit.
