Abstract
Background
Previous voxel- or surface-based morphometric analysis studies have revealed alterations in cortical structure in patients with chronic migraine, yet with inconsistent results. The discrepancies may be derived partly from the sample heterogeneity. Employing both methods in a clinically homogenous group may provide a clearer view.
Methods
Structural MRI data from 30 prevention-naïve patients with chronic migraine without medication overuse headache or a history of major depression and 30 healthy controls were analyzed. Vertex-wise (surface-based) or voxel-wise (voxel-based) linear models were applied, after controlling for age and gender, to investigate between-group differences. Averaged cortical thicknesses and volumes from regions showing group differences were correlated with parameters related to clinical profiles.
Results
Surface-based morphometry showed significantly thinner cortices in the bilateral insular cortex, caudal middle frontal gyrus, precentral gyrus, and parietal lobes in patients with chronic migraine relative to healthy controls. Additionally, the number of migraine days in the month preceding MRI examination was correlated negatively with right insular cortical thickness. Voxel-based morphometry (VBM) did not show any group differences or clinical correlations.
Conclusion
Patients with chronic migraine without medication overuse headache, major depression, or prior preventive treatment had reduced cortical thickness in regions within the pain-processing network. Compared to voxel-based morphometry, surface-based morphometry analysis may be more sensitive to subtle structural differences between healthy controls and patients with chronic migraine.
Introduction
Migraine is a leading cause of disability worldwide, with a prevalence of 10–15% (1). Epidemiological studies have shown that about 2% of the general population suffers from chronic migraine (CM) (2), with a 3% (2–4%) annual conversion rate from episodic migraine (EM) to CM (3).
The pathophysiology of migraine is far from being understood. Using structural neuroimaging methods; for example, surface-based morphometry (SBM) or voxel-based morphometry (VBM), researchers have found that, in EM, specific regions within the pain matrix exhibit alterations in cortical thickness or volume. These regions include the somatosensory cortex (4–6), motor cortex (7,8), frontal/prefrontal cortices (7–12), visual cortex (7,13), insula (6,11), and anterior/middle cingulate cortex (6,8,11), among others. However, the direction of change was inconsistent across these studies. For example, although some studies reported a thicker cortex in EM over somatosensory (4–6), visual (7,13), or frontal regions (5,7) compared with healthy controls (HCs), other studies reported no differences (14–16) or thinner cortex (6,7,17). Both SBM and VBM results have been conflicting. Differences in methodologies and clinical characteristics of the patients may partly explain the discrepancies.
Compared to EM, CM is characterized by an increased disease burden and could thus be associated with more widespread brain structural changes. Few studies have addressed this important issue with SBM (18) or VBM (19–21), with the latter yielding inconsistent results. The only study (18) to investigate cortical thickness in CM did not find any significant differences relative to HCs; however, one third (10/30) of their study patients were comorbid with medication overuse headache (MOH) (22). As CM with and without MOH to some extent exhibit different morphological alterations (19), combining the two groups may obscure subtle changes in either group (23).
Although both SBM and VBM provide measures of brain structure, the two methods provide complementary rather than redundant information (5,6,16,24). As depicted in Figure 1, VBM measures gray matter (GM) volume, while SBM is capable of probing various aspects of cortical shape including cortical thickness, degree of gyral convolution (i.e. the gyrification index), and sulcal depth, among others. Thus, combining SBM and VBM may provide further insight into how brain structures are affected in CM.

Basic workflow of VBM and SBM. After segmentation, VBM measures GM volume, while SBM can determine various aspects of cortical shape, including cortical thickness, degree of gyral convolution (the gyrification index), and sulcal depth, after applying different strategies in registration to the templates.
In the present study we aimed to evaluate brain structural changes using both SBM and VBM in a clinically homogeneous group of CM patients who were without MOH or major depression and naïve to preventive medications. We examined two hypotheses. Our first hypothesis was that these patients would show alterations in SBM or VBM metrics in pain-related brain regions. Our second hypothesis was that some of these alterations would correlate with clinical parameters, such as disease duration, CM severity, or headache frequency.
Methods
Subjects
Patients with CM were recruited from the Department of Neurology at Taipei Veterans General Hospital. At their first visit, they completed a structured headache questionnaire consisting of headache profiles and assessment of disease severity (Migraine Disability Assessment [MIDAS]) and screening for anxiety and depression (Hospital Anxiety and Depression Scale [HADS]). Information on other common pain syndromes (e.g. widespread pain index as in fibromyalgia, arthralgia, toothache, and back pain), as well as the duration and severity of the pain, were collected. The inclusion criteria for CM patients were: Aged 18–65 years; fulfilling the ICHD-3 criteria for CM (22); and migraine onset before the age of 50 years. The exclusion criteria were: Headache type other than CM; comorbid MOH as defined by the ICHD-3 criteria (22); history of major depression; use of migraine prophylaxis before enrolment; CM onset after the age of 60 years; comorbidity with other pain syndrome for more than 3 months, being pregnant or nursing; having a history of renal or hepatic disorder, or other severe systemic disease; and having a condition incompatible with MRI, such as metallic or electric implants or claustrophobia. After the initial visit, eligible patients who met the ICHD-3 criteria for CM were asked to keep headache diaries during a 4-week prospective baseline period. Subjects with ≥15 monthly headache days (MHD) and ≥8 monthly migraine days (MMD) within this period then underwent brain MRI examination, with structural, functional, and spectroscopic measurements. Age and gender-matched HCs without a history of migraine were also enrolled. Infrequent tension-type headache (TTH) (<1/month) (22) was allowed in the HC group, in line with the inclusion criteria for HC in the field of migraine research (5,11,14,16). Apart from a diagnosis of CM in the patient group, all participants had normal physical and neurological examinations.
Prior to entering the study, all participants were informed about the study orally and in writing, and gave informed written consent to participate. The institutional review board of Taipei Veterans General Hospital approved the protocol (VGHIRB: 2012-04-046B).
MRI acquisition
The study was conducted on a 3-T magnetic resonance scanner (Trio, Siemens Medical Solutions, Erlangen, Germany) located at National Yang-Ming University. Images were acquired with a 32-channel head coil array. The participants’ heads were immobilized in the scanner by foam pads. The structural scan consisted of a high-resolution three-dimensional MPRAGE (Magnetization Prepared Rapid Acquisition Gradient Echo) sequence with the following parameters: Repetition time, 2530 ms; echo time, 3.03 ms; flip angle, 7 degrees; field of view, 224 × 256 × 192; and voxel size, 1 × 1 × 1 mm.
SBM
The SBM analysis was carried out with the Computational Anatomy Toolbox (CAT12, http://www.neuro.uni-jena.de/cat/), an extension for the Statistical Parametric Mapping 12 (SPM12; https://www.fil.ion.ucl.ac.uk/spm/software/spm12/) running in the MATLAB environment (R2017b; MathWorks, Natick, MA). The analysis was semi-automated by applying default parameters for all processing steps, as described by Niddam et al. (25). The high-resolution T1-images were initially segmented into different tissue types, including GM, white matter (WM), and cerebrospinal fluid (CSF). The pial surface and the WM surface (GM/WM boundary) were extracted, and an outer hull layer that tightly wraps the pial surface was reconstructed (Figure 1). Cortical thickness was estimated by calculating the distance between the pial and WM surfaces. Two additional geometric cortical measures (e.g. sulcal depth and degree of gyrification [gyrification index =
VBM
The VBM analysis was conducted in SPM 12, and followed the procedures described by Niddam et al. (29). In short, the high-resolution T1-images were segmented into different tissue types, including GM, WM, and CSF. An optimized GM template in standard space was created with the DARTEL toolbox. Registration parameters and deformation maps generated during this step were then used for spatial warping of each individual’s GM images. During this step, the GM volume was preserved to ensure that GM volumes would be compared rather than GM concentrations. Voxels with a GM concentration less than 0.2 were excluded from the analysis to avoid potential boundary effects between GM and WM, as well as to preserve homogeneity. Finally, a 4‐mm3 FWHM Gaussian kernel was used for spatial smoothing of the GM images. The smoothed images were used for between-group analyses.
Statistical analysis of structural images
Between-group differences in cortical thickness, gyrification index, and sulcal depth (by SBM) and GM volume (by VBM) were assessed with two-sample t-tests controlling for age and gender. Total intracranial volume (TIV) was also controlled for in the VBM models as recommended (29). Statistical maps were thresholded first at a voxel-level at
To label significant clusters with the corresponding anatomical structures, Montreal Neurological Institute (MNI) coordinates were first transformed into Talairach space using an MNI to Talairach Converter (https://bioimagesuiteweb.github.io/webapp/mni2tal.html), and then entered into the Talairach Client (http://www.talairach.org/client.html).
Given that previous conflicting results on brain structural changes in patients with migraine may have been due to different approaches, two additional analyses were performed. Firstly, threshold-free cluster enhancement (TFCE), a relatively liberal statistical method, was performed with a cluster-wise threshold at
Statistical analysis of clinical parameters
Statistical analyses of clinical variables were conducted in SPSS software (version 21 for Windows; IBM Inc., Armonk, NY). Differences between groups were examined with independent t-tests for continuous data and with either the chi-squared test or Fisher’s exact test for nominal data, where appropriate. Statistical significance was defined as
Results
Demographics and baseline clinical characteristics
Thirty patients with CM and 30 HCs were recruited for this study from June 2012 to December 2014. All participants were Taiwanese (East Asians). The demographic characteristics of the participants in each group are summarized in Table 1. The CM patient and HC groups were gender-matched (
Demographics of healthy controls and patients with chronic migraine.

Note: Data are shown as mean (SD).
CM: chronic migraine; CSF: cerebrospinal fluid; GM: gray matter; HADS-A/D/T: Hospital Anxiety and Depression Score, anxiety/depression/total score; HC: healthy control; M0: the month preceding MRI scan; MIDAS: migraine disability assessment scale; NSAID: non-steroidal anti-inflammatory drugs; WM: white matter.
SBM analysis
Comparisons of surface metrics between the CM and HC groups revealed significant between-group regional differences in cortical thickness (Figure 2), but not in sulcal depth or gyrification index. Compared to HCs, reduced cortical thickness was found in the CM group in clusters located in bilateral precentral gyri, caudal middle frontal gyri (MFG), insula, and parietal lobes (Tables 2 and 3). No clusters of increased cortical thickness were observed in the CM group relative to HCs. Correlation analyses of mean cortical thicknesses (extracted from significant clusters) with headache parameters in CM patients revealed a negative correlation between mean cortical thickness within the right insula and MMD (n = 30,
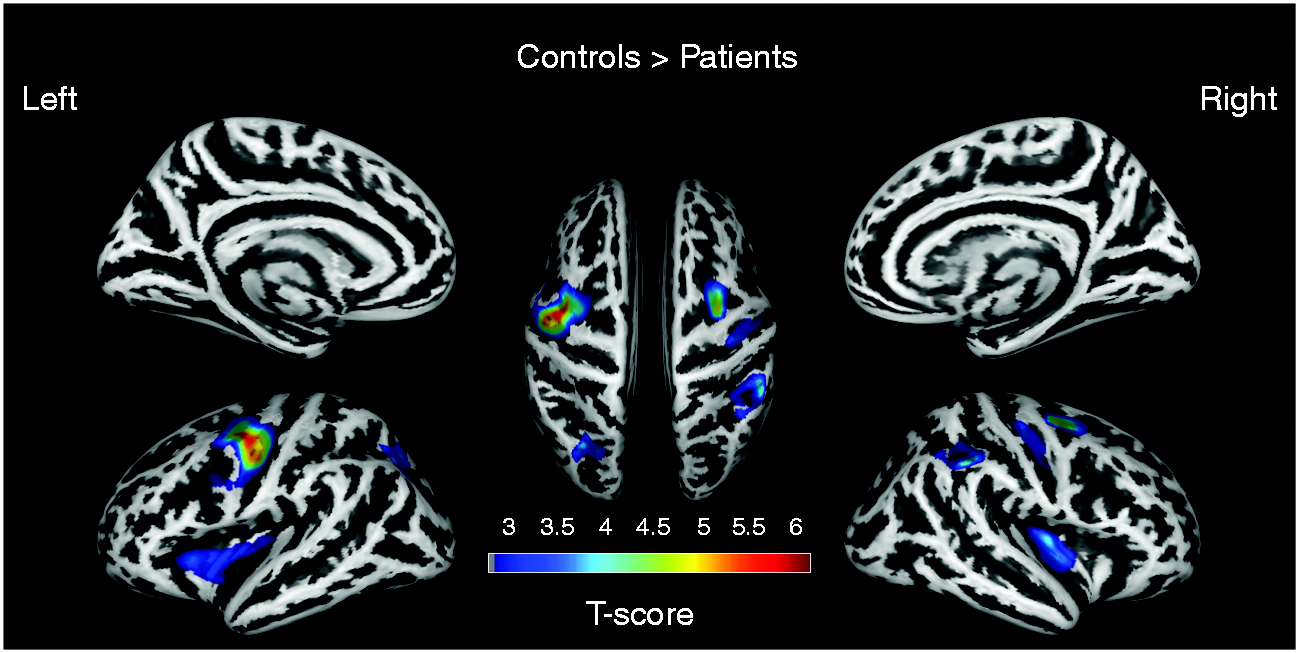
Cortical thickness abnormalities in patients with CM. Reduced cortical thickness in CM patients was observed in the bilateral precentral gyri, bilateral middle frontal gyri, bilateral insula, and bilateral parietal lobes (corrected
Significant clusters exhibiting reduced cortical thickness in patients with CM compared with HCs.

Note:
BA: Brodmann area; FDR: false discovery rate; G: gyrus; L: lobe/lobule; LT: left; MNI: Montreal Neurological Institute; RT: right.
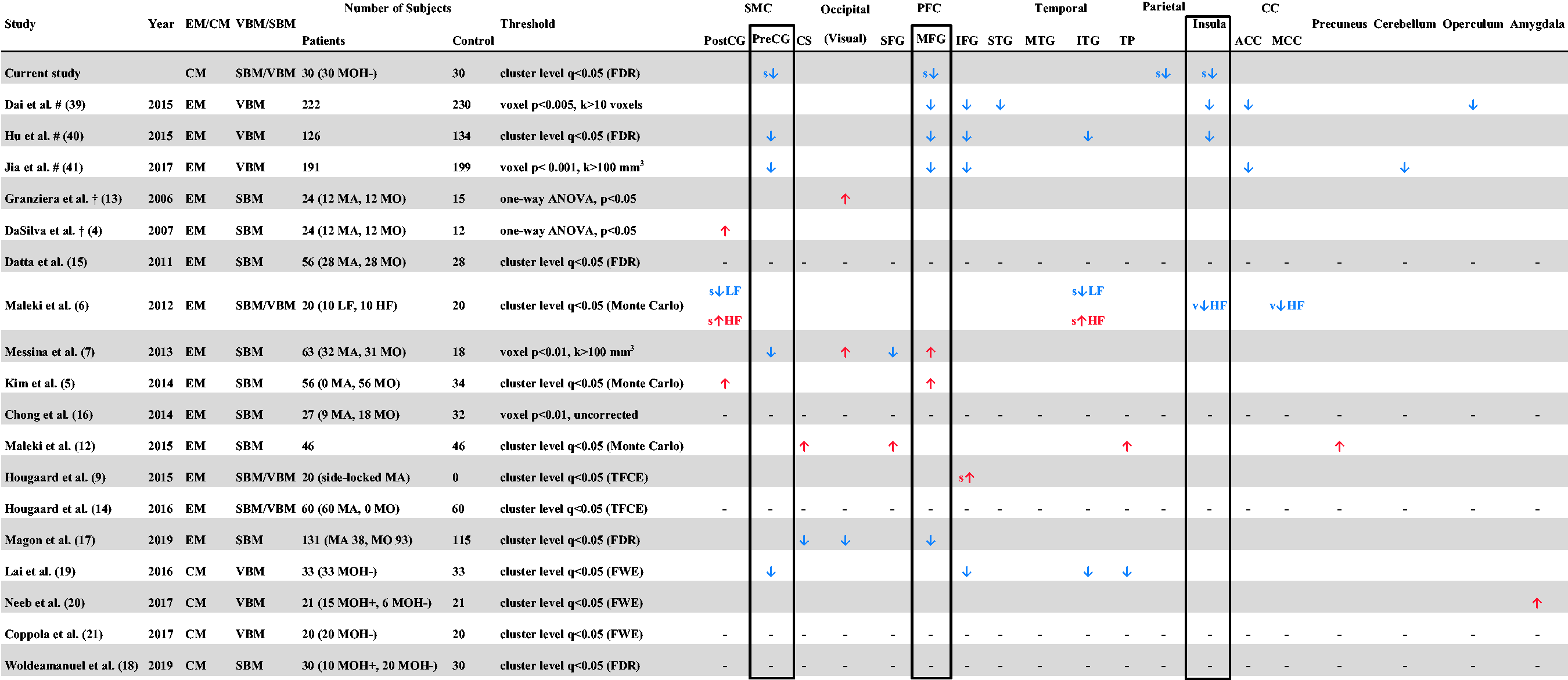
Summary of findings from voxel- or surface-based morphometric studies.

CM: chronic migraine; FDR: false discovery rate; GM: gray matter; HC: healthy control; ROI: region-of-interest; SBM: surface-based morphometry; TFCE: threshold-free cluster enhancement; VBM: voxel-based morphometry.

Association between mean cortical thickness and baseline migraine frequency. A negative correlation was observed between the mean cortical thickness of the right insula and monthly migraine frequency at baseline (n = 30,
VBM analysis
Voxel-wise whole brain comparisons showed no significant differences in GM volume between the CM and HC groups (Table 3). No clusters passed the more liberal TFCE threshold. Finally, between-group comparisons of mean GM volumes in regions with corresponding significant clusters in the SBM analysis also failed to reveal any significant differences.
Analysis in women only
To further increase the specificity of the study cohort, an additional analysis restricted to women was performed (CM: n = 23; HC: n = 22). They were age-matched (
Discussion
In the present study, we investigated cortical morphological changes using both SBM and VBM in patients with CM not comorbid with MOH or major depression, and not on any preventive medications. Furthermore, all subjects included in the study had a relatively homogenous demographic background (i.e. all Taiwanese). In contrast to our negative VBM results, SBM showed regional cortical thinning in CM patients, compared to HCs, in the bilateral insular cortex, caudal MFG, precentral gyrus, and parietal lobes. These regions are considered parts of the pain matrix and are involved in pain perception, evaluation, and modulation (31,32). In addition, we observed a correlation between the cortical thickness of the right insula and clinical severity.
SBM might be more sensitive than VBM
Although VBM and SBM adopt similar preprocessing steps (e.g. GM/WM segmentation), the two methods use different strategies in aligning each individual’s image to the template (Figure 1). VBM applies voxel- (volume-) based registration to compensate for individual variability, which is relatively prone to registration artifacts induced by global volume change (15). The highly variable folding pattern across individuals may further reduce the accuracy of alignment in VBM (33). SBM, in contrast, adopts an alternative approach for registration by matching the gyral and sulcal geometry to an inflated spherical atlas (5,15,24). Such an approach greatly reduces the potential misalignment caused by complex folding pattern, as well as global volume change. Moreover, SBM is capable of measuring different important cortical features, such as cortical thickness, sulcal depth, gyrification index, and surface area among others, while VBM only provides a measure of GM volume, which may depend on some of the surface derived characteristics (5).
In this study, between-group differences were found with SBM (cortical thickness), but not VBM (GM volume). A similar result was reported recently in a study applying both methods in patients with EM (9). Both studies thus lend support to the above-mentioned notion that SBM might be more accurate or sensitive than VBM in detecting morphological changes within the brain. Of note, in our SBM analysis, only cortical thickness, but not gyrification index or sulcal depth, showed between-group differences. The absence of changes in the latter features may suggest that no overt atrophic changes occur in patients with migraine, as alteration in gyrification index or sulcal depth are frequently seen in neurodevelopmental (34) or neurodegenerative diseases (35).
The observation that cortical thinning was not associated with a corresponding reduction in GM volume also deserves further discussion. The superior spatial registration used in SBM may in part explain this difference. In addition, although GM volume is assumed to be influenced by both surface area and thickness, there are indications that GM volume depends more on the former than the latter (24). It can, however, not be ruled out that an enlarged surface area developed in patients with CM to compensate for the reduction in cortical thickness, and resulted in a preserved GM volume. Nevertheless, further studies are needed to clarify this point.
Comparison to previous morphological studies in CM and EM
Our results in CM patients are partly in accordance with previous studies of CM, as well as those of EM (see summary in Table 4). For example, in another recent VBM study of CM, no between-group differences were observed (21). Nevertheless, two other VBM studies of CM demonstrated regional differences in the precentral gyrus, prefrontal cortex, temporal lobe, and amygdala (19,20), and one SBM study failed to show between-group differences (18). The causes of the discrepancies may partly be attributed to differences in comorbidities, disease burden, MRI parameters, and analytic approaches (36). For instance, CM with MOH is known to differ from CM without MOH with respect to cortical alterations (19). Of note, 33–71% of the CM patients were comorbid with MOH in the aforementioned previous studies (18,20). Regarding disease burden, the mean MIDAS score in our patient cohort (45.6 ± 50.7) was lower compared to the others (ranging from 46.2–67.3) (18,20,37,38), which could be explained again by the absence of MOH (37) or comorbid depression (38) as both were associated with higher MIDAS scores. On the other hand, the difference in the inclusion criteria for HC (i.e. the allowance of infrequent TTH (5,11,14,16) vs. completely headache-free (18–21)) may also partly contribute to the inconsistent results.
Summary of brain morphological studies in patients with migraine.

Note: Most commonly involved regions, including those found in the current study, are marked in rectangles.
ANOVA: analysis of variance; A/MCC: anterior/middle cingulate cortex; CM: chronic migraine; CS: central sulcus; EM: episodic migraine; FDR: false discovery rate; FWE: family-wise error; HF: high-frequency migraine; k: number of voxel; LF: low-frequency migraine; MA: migraine with aura; MO: migraine without aura; MOH: medication-overuse headache; PFC: prefrontal cortex; Post/PreCG: post-/pre-central gyrus; s: SBM-based analysis; SBM: surface-based morphometry; SMC: sensorimotor cortex; S/M/IFG: superior/middle/inferior frontal gyrus; S/M/ITG: superior/middle/inferior temporal gyrus; TFCE: threshold free cluster enhancement; TP: temporal pole; v: VBM-based analysis; VBM: voxel-based morphometry; – : no between-group differences; #: meta-analysis, †: region-of-interest (ROI)-based analysis.
Interestingly, the CM results reported here are comparable to those derived from SBM studies, as well as from meta-analyses involving VBM studies, in EM (39–41) (see summary in Table 4). This finding may imply that SBM is capable of detecting subtle structural differences that are only apparent with VBM using a much larger study cohort.
Comparison to previous morphological studies in other chronic pain syndromes
It should be emphasized that the regions demonstrating between-group differences in Table 4 are not specific to EM/CM. Previous studies on non-migraine headache disorders and other chronic pain syndromes also showed alterations in these regions; that is, the pain matrix (42,43). The observation that nearly all studies investigating brain alterations in patients with various types of chronic pain disorders yield comparable results suggests that these changes are highly correlated with the presence of chronic pain and may either result from repeated input or an inherited susceptibility to develop chronic pain (42). As insular, premotor cortex, and caudal MFG were affected in this study, it could be that the affective-motivation and motor components (43) were more selectively involved in our patient population. Nevertheless, further large-scale studies or meta-analyses using SBM may help delineate the extent of involvement of the pain matrix in patients with CM.
Clinical implication from correlation analysis
With respect to the observations of cortical thinning, it is important to clarify whether such changes are causal to or consequent of chronic pain. The observation that reduced cortical thickness within the right insular cortex correlated with baseline migraine frequency suggests that the alteration within this region may result from the impact of repetitive high-frequency attacks. This supposition is supported by a recent study showing decreased GM volume in the insular cortex in high-frequency EM patients (8–14 days/month) with respect to both HC and low-frequency EM patients (1–2 days/month) (6). The fact that effective treatment of some forms of chronic pain results in a reversal of localized GM reductions (e.g. within the insular cortex, anterior cingulate cortex, and dorsolateral prefrontal cortex) further strengthens this viewpoint (44).
It is worth noting that most of the regions showing between-group differences did not correlate with clinical parameters. A recent SBM study in a large cohort of patients with EM also showed similar findings (17). This could mean that the observed alterations may be inherent to the migraine brain, and might potentially be a predisposing factor. This interpretation is supported by a recent SBM study in twins with migraine with aura, demonstrating morphological changes as being a trait rather than a consequence of the disease (45).
Limitations
Our study has limitations. First, the number of subjects was relatively small and hence may not fully reflect group differences. Further large-scale studies may be needed to demonstrate cortical thickness alterations not detected here. Second, because we recruited a relatively homogenous group of patients with CM, our results may not be generalizable to the broader CM population, of whom 50% have comorbid MOH and 60% of whom have major depression. Third, owing to the limitation of the software (CAT12), surface area was not calculated. The speculation that patients with CM had enlarged surface area to compensate for cortical thinning requires further studies. Fourth, since this is a cross-sectional study, it is difficult to determine whether clusters showing between-group differences, yet without clinical correlation, are predisposing factors for the development of CM. Further studies using a longitudinal follow-up design are needed. Finally, given the inherent limitation, SBM is not capable of analyzing subcortical regions (46,47), including the hypothalamus and the brainstem, both of which have been recognized recently to play crucial roles in migraine pathogenesis (48–51). The dynamic alterations of functional connectivity between the hypothalamus and some specific nuclei within the brainstem (e.g. the dorsal pons, and the spinal trigeminal nucleus) have been shown to be responsible for the cyclic nature of migraine attacks (49,50). Also, the hyper-responsiveness to painful trigeminal stimulation in the anterior part of the hypothalamus is involved in migraine chronification (51). All these findings were demonstrated by a newly developed high-resolution functional MRI paradigm focusing on brainstem structure (52). In the coming era of ultra-high field MRI, combined analysis of both high-resolution structural and functional images may provide more information regarding the regions involved in the pathogenesis of CM.
Conclusions
In summary, our study showed that in patients with CM, specific regions located within pain-processing networks had reduced cortical thickness compared to HCs. SBM appears to be more sensitive than VBM for studying subtle structural differences in cortical regions. Further longitudinal studies aimed at examining cortical thickness within these regions after successful treatment are warranted.
Clinical implications
Patients with chronic migraine without MOH, major depression or prior prophylactic treatment had reduced cortical thickness within regions related to pain process, which could be a consequence of or a predisposing factor to chronic pain. Surface-based morphometry analysis appears to be more sensitive than using voxel-based morphometry for studying subtle structural differences in cortical regions.
Supplemental Material
sj-pdf-1-cep-10.1177_0333102420920005 - Supplemental material for Cortical morphological changes in chronic migraine in a Taiwanese cohort: Surface- and voxel-based analyses
Supplemental material, sj-pdf-1-cep-10.1177_0333102420920005 for Cortical morphological changes in chronic migraine in a Taiwanese cohort: Surface- and voxel-based analyses by Kuan-Lin Lai, David M Niddam, Jong-Ling Fuh, Wei-Ta Chen, Jaw-Ching Wu and Shuu-Jiun Wang in Cephalalgia
Supplemental Material
sj-pdf-2-cep-10.1177_0333102420920005 - Supplemental material for Cortical morphological changes in chronic migraine in a Taiwanese cohort: Surface- and voxel-based analyses
Supplemental material, sj-pdf-2-cep-10.1177_0333102420920005 for Cortical morphological changes in chronic migraine in a Taiwanese cohort: Surface- and voxel-based analyses by Kuan-Lin Lai, David M Niddam, Jong-Ling Fuh, Wei-Ta Chen, Jaw-Ching Wu and Shuu-Jiun Wang in Cephalalgia
Supplemental Material
sj-pdf-3-cep-10.1177_0333102420920005 - Supplemental material for Cortical morphological changes in chronic migraine in a Taiwanese cohort: Surface- and voxel-based analyses
Supplemental material, sj-pdf-3-cep-10.1177_0333102420920005 for Cortical morphological changes in chronic migraine in a Taiwanese cohort: Surface- and voxel-based analyses by Kuan-Lin Lai, David M Niddam, Jong-Ling Fuh, Wei-Ta Chen, Jaw-Ching Wu and Shuu-Jiun Wang in Cephalalgia
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Brain Research Center, National Yang-Ming University from The Featured Areas Research Center Program within the framework of the Higher Education Sprout Project by the Ministry of Education (MOE) in Taiwan; Ministry of Science and Technology of Taiwan [MOST 108-2321-B-010-014 -MY2 and 108-2321-B-010 001 and 108-2314-B-010-023-MY3 and 107-2314-B-010-018-MY3]; Ministry of Health and Welfare, Taiwan [MOHW 103-TDU-B-211-113-003, MOHW 104-TDU-B-211-113-003, MOHW 105-TDU-B-211-113-003, MOHW 106-TDU-B-211-113001, MOHW 107- TDU-B-211-123001, MOHW 108- TDU-B-211-133001]. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.