Abstract
Objective:
Gender dysphoria (GD) is characterized by distress due to inconsistency between gender identity and biological sex. Individuals with GD often desire to be the other gender, which is called transgender. Although altered brain volumes in transgender people, particularly transgender women, have been reported, the particular brain regions have been inconsistent among studies. This study aimed to investigate neuroanatomical differences in transgender men without physical interventions.
Method:
T1-weighted magnetic resonance images (MRIs) were acquired in 21 transgender men and 21 cisgender women matched for biological sex and age. Whole-brain comparisons using voxel-based morphometry (VBM) were performed to identify gray matter volume (GMV) differences between transgender men and cisgender women.
Results:
Transgender men showed greater GMV in the right posterior cingulate gyrus (
Conclusion:
These findings suggest that transgender men have altered brain structure. We suggest that larger posterior midline structures may contribute to sensitivity to self-referential processing through altered visual perception in transgender people.
Introduction
Gender dysphoria (GD) refers to the distress that may accompany the inconsistency between one’s experienced or expressed gender and one’s assigned biological sex, according to the criteria in the
To reveal the causal factor of GD, differences in cerebral morphology between transgender people and cisgender people whose gender identity matches their assigned sex at birth have been investigated using the voxel-based morphometry (VBM) technique (Hoekzema et al., 2015; Luders et al., 2009; Savic and Arver, 2011; Simon et al., 2013; Spizzirri et al., 2018). These studies have found that transgender people had increased or decreased gray matter volume (GMV), e.g., in the insula, hippocampus, precuneus and posterior cingulate, compared with cisgender people; however, the particular regions and the directionality of the associations have been inconsistent among studies. Some studies investigated the differences in cerebral structures between transgender women and cisgender people, consisting of both cisgender men and cisgender women (Luders et al., 2009; Savic and Arver, 2011; Spizzirri et al., 2018), while other studies investigated the differences in cerebral structures among transgender women, transgender men, cisgender men and cisgender women (Hoekzema et al., 2015; Simon et al., 2013). Furthermore, hormone-treated or heterosexual transgender people were included in some studies (Hoekzema et al., 2015; Simon et al., 2013). After reducing heterogeneity in study outcomes by focusing on participants, particularly transgender women and cisgender people (cisgender men and cisgender women), the detected regions and the directionality (decreased or increased) remained inconsistent among studies.
The reason for inconsistent outcomes among previous studies may be due to heterogeneity in transgender people among studies. It is known that transgender women show two broad developmental trajectories: early-onset transgender woman, which begins in childhood and persists into adolescence and adulthood, and late-onset transgender woman, which emerges around puberty or much later in life (American Psychiatric Association, 2013). Early-onset transgender women are sexually attracted to men (androphilic) and seek out sex reassignment therapies at a relatively earlier age. Meanwhile, late-onset transgender women show greater fluctuation in the degree of GD: they frequently engage in transvestic behavior with sexual excitement, and most of them are considerably gynephilic and cohabit or get married with cisgender woman. These variations in age at onset, sexual orientation and behaviors of seeking treatment in transgender women could result in heterogeneity in brain morphology among transgender people.
In contrast, transgender men have a more homogeneous etiology and clinical course than transgender women. Most transgender men show early onset, which begins in childhood and persists into adolescence and adulthood. Transgender men are generally attracted to women (gynephilic) and seek out sex reassignment therapies at an earlier age (American Psychiatric Association, 2013). It is well known that early-onset disorders, such as autism spectrum disorder (Ecker, 2012; Rojas et al., 2006) and schizophrenia (Jiang et al., 2018; Vita et al., 2012), display more prominent brain differences than late-onset disorders, such as major depressive disorder (Bora et al., 2012; Du et al., 2012), and schizophrenia patients with early onset exhibit more decreased GMV than those with late onset (Fraguas et al., 2016; Tordesillas-Gutierrez et al., 2015). Schizophrenia occurs in individuals with GD at rates higher than in the general population (Rajkumar, 2014), suggesting that cerebral alteration at early stages of life may contribute to relationships between experiencing GD in early stages of life and psychiatric conditions such as schizophrenia. Therefore, we hypothesized that individuals with GD, particularly transgender men, would show aberrant brain structure compared with cisgender women based on neurodevelopmental processes.
In this study, we investigated GMV differences between transgender men and cisgender women using voxel-based morphometry (VBM) analysis because participants in previous studies were limited to transgender women and cisgender men. Furthermore, the transgender people included in the current study consisted of untreated and homosexual transgender people, i.e., androphilic transgender men who have never undergone cross-sex hormone treatment or sex reassignment surgery. Therefore, our participants consisted of a more homogeneous sample of transgender men.
Methods
Participants
All transgender men who complained of GD were recruited at the Department of Psychiatry, Gifu University Hospital during 2013 and 2018. Transgender men who had never visited psychiatric clinics/hospitals were also recruited through advertisements via our departmental website and direct announcements to local transgender community organizations during the period. All transgender men were diagnosed with GD according to the
Both transgender and cisgender people were interviewed to exclude those with a history of any of the following: endocrine disorder, hormonal treatment (e.g. cross-hormone therapy and oral contraceptive), surgery for sexual organs (including sex reassignment surgery), severe medical conditions that could potentially affect the central nervous system, severe neurological diseases, intellectual disability, autism spectrum disorder or disorders of sex development.
The Mini-International Neuropsychiatric Interview (M.I.N.I.) (Sheehan et al., 1998) was applied to exclude severe psychiatric conditions, such as psychotic disorders, substance abuse and major depressive disorder. Individuals who had any medication with psychotropic effects (antipsychotics, benzodiazepines, antidepressants or lithium) were also excluded. Serum sex hormone levels (estradiol and free testosterone) were assessed at the date of magnetic resonance imaging (MRI) scanning. This study was performed according to the World Medical Association’s Declaration of Helsinki and was approved by the Research Ethical Committee of Gifu University. Written informed consent was obtained from all subjects after the procedures were fully explained.
Magnetic resonance imaging acquisition
MRI scans using a 3-Tesla scanner (Achieva 3.0 Tesla TX, Philips, Amsterdam, the Netherlands) equipped with an 8-channel head coil were performed at Chubu Medical Center (Gifu, Japan). Whole-brain anatomical images were obtained by T1-weighted images (T1 W, 3D, TFE) with the following sequence parameters: repetition time (TR) =8.0 ms, echo time (TE) = 4.6 ms, field of view = 256 mm, acquisition matrix = 256 × 256, flip angle = 8°, voxel size = 1 mm × 1 mm × 1 mm, yielding 200 contiguous sagittal slices. Routine fluid-attenuated inversion recovery (FLAIR) images were acquired to rule out neurological mass lesions. Each scan was visually checked for image artifacts and gross neurological abnormalities by a psychiatrist (TF) and expert radiological technologists. No images were excluded for the presence of artifacts and/or anomalies.
Statistical analysis
All statistical analyses, except for the VBM analyses, were performed with R version 3.6.2 software (www.r-project.org; The R Foundation for Statistical Computing, Vienna, Austria). Demographic variables such as age and education years as well as serum hormonal levels were fitted to a normal distribution using the Kolmogorov-Smirnov test (
For VBM preprocessing, we used the SPM12 software package (www.fil.ion.ucl.ac.uk/spm/; Welcome Department of Imaging Neuroscience, London, UK) running under MATLAB R2018b (MathWorks Inc., Natick, MA, USA). Each scan was manually reoriented along the anterior commissure-posterior commissure line, and the anterior commissure was set as the origin of spatial coordinates on the view tool equipped with SPM12.
We applied the default preprocessing parameters in SPM12 to the following procedure. T1 images were segmented into five tissue types (GM, white matter, cerebrospinal fluid, bone and soft tissue). All segmented images were visually inspected. Diffeomorphic Anatomical Registration through Exponentiated Lie algebra (DARTEL) (Ashburner, 2007) registration using the template created by all T1 images was carried out and normalized into the standard Montreal Neurological Institute (MNI) atlas space with 1.5 mm × 1.5 mm × 1.5 mm resolution. Finally, the resulting GM images were also modulated and smoothed with a Gaussian kernel of 8 mm FWHM.
Comprehensive exploratory whole-brain research was conducted using SPM12 statistical tools. We conducted a two-sample
After exploring GM regions with a voxel-level height threshold of
Results
Demographic and clinical measures
Through the inclusion and exclusion criteria as mentioned in the methods section, 42 individuals were finally registered in the current study, which were composed of 21 transgender men and 21 cisgender women. The demographic and clinical characteristics of our participants are summarized in Table 1. The serum free testosterone levels were lower in transgender men than in cisgender women (
Demographic and clinical data.
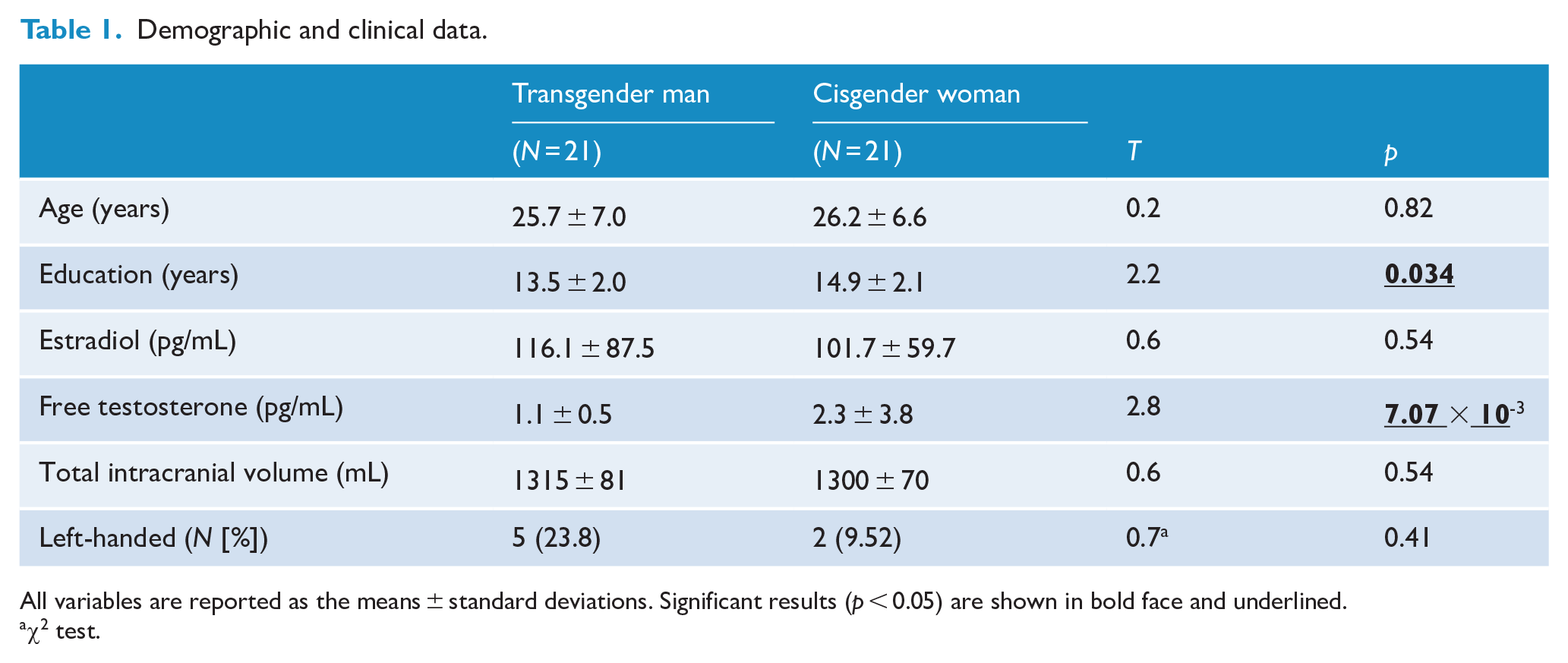
All variables are reported as the means ± standard deviations. Significant results (
Regions with altered GMV between transgender men and cisgender women
We investigated GMV differences in the whole-brain analysis between transgender men and cisgender women (Figure 1). Compared with cisgender women, transgender men had significantly increased GMV in the right posterior cingulate gyrus (
Differences in gray matter volume (GMV) between transgender men and cisgender women.
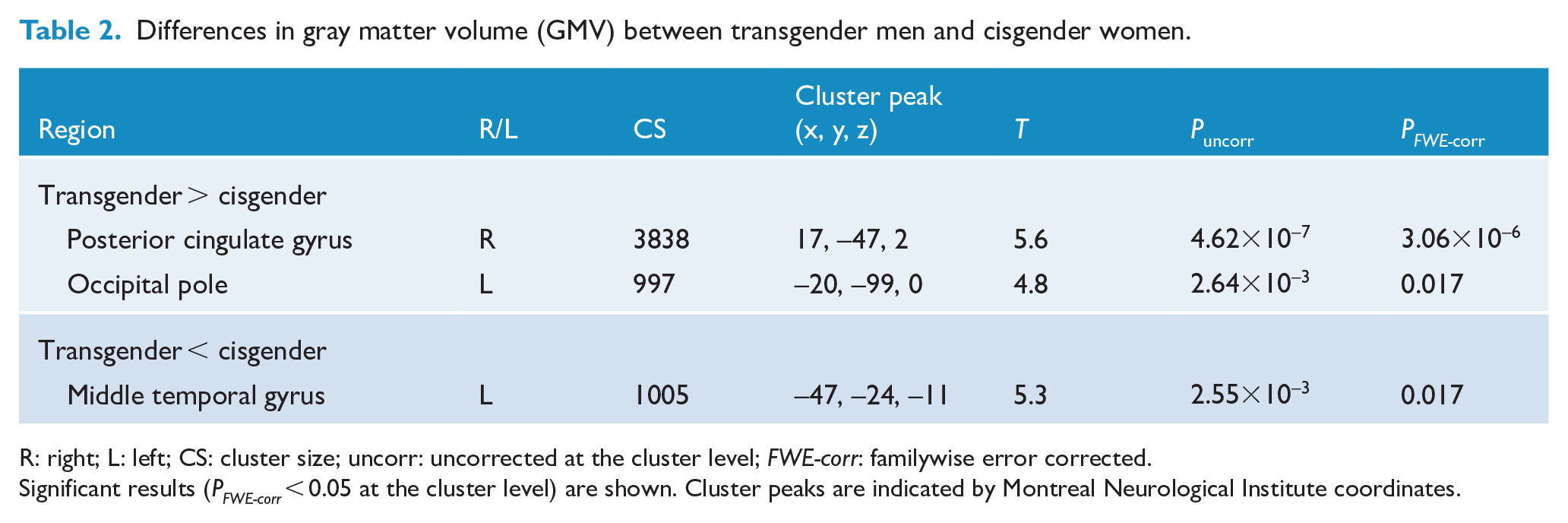
R: right; L: left; CS: cluster size; uncorr: uncorrected at the cluster level;
Significant results (
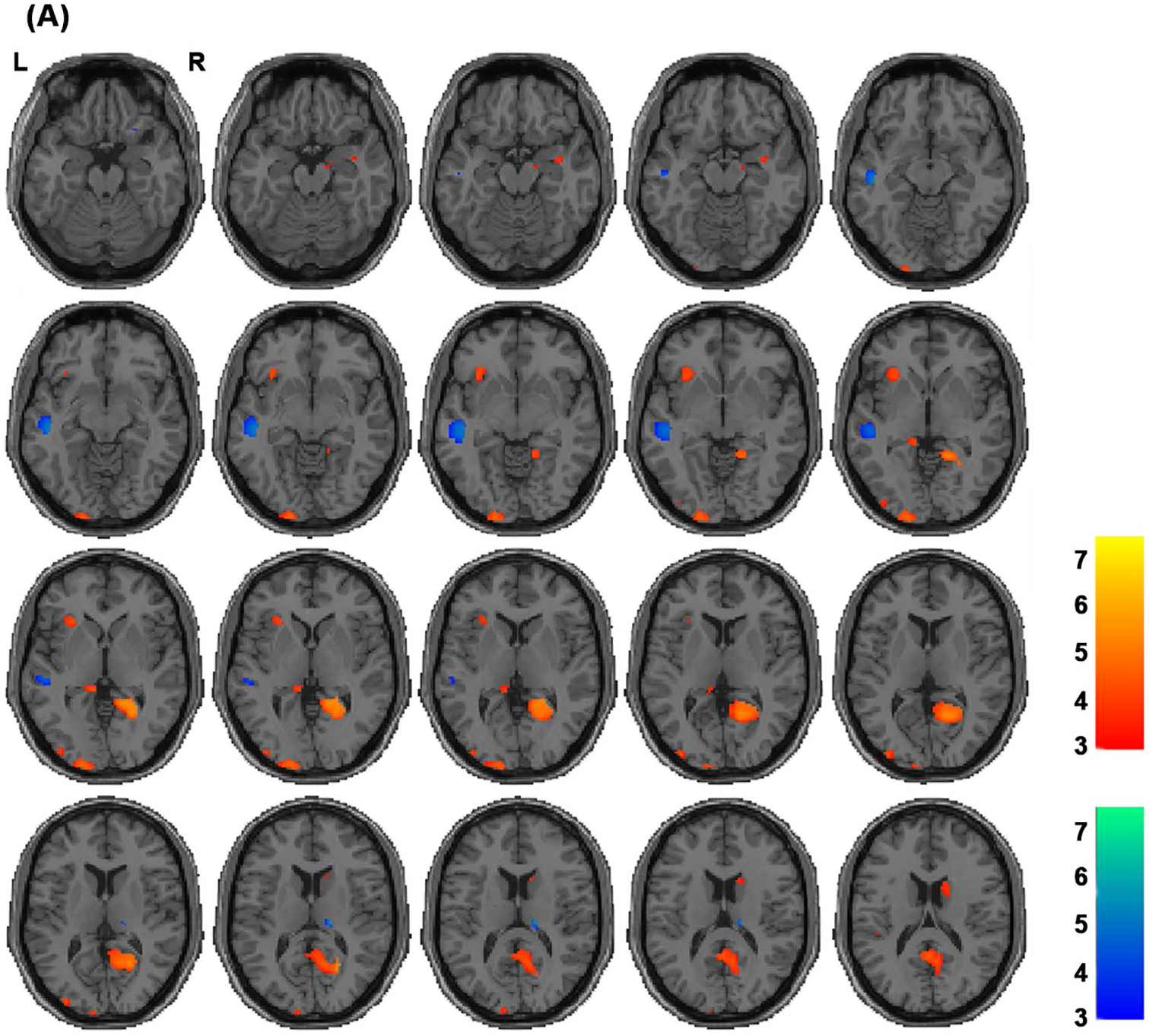
Altered gray matter volume (GMV) regions between transgender men and cisgender women. Red areas indicate regions with increased GMV in transgender men compared with cisgender women, whereas blue areas indicate regions with decreased GMV in transgender men compared with cisgender women. Each color bar shows
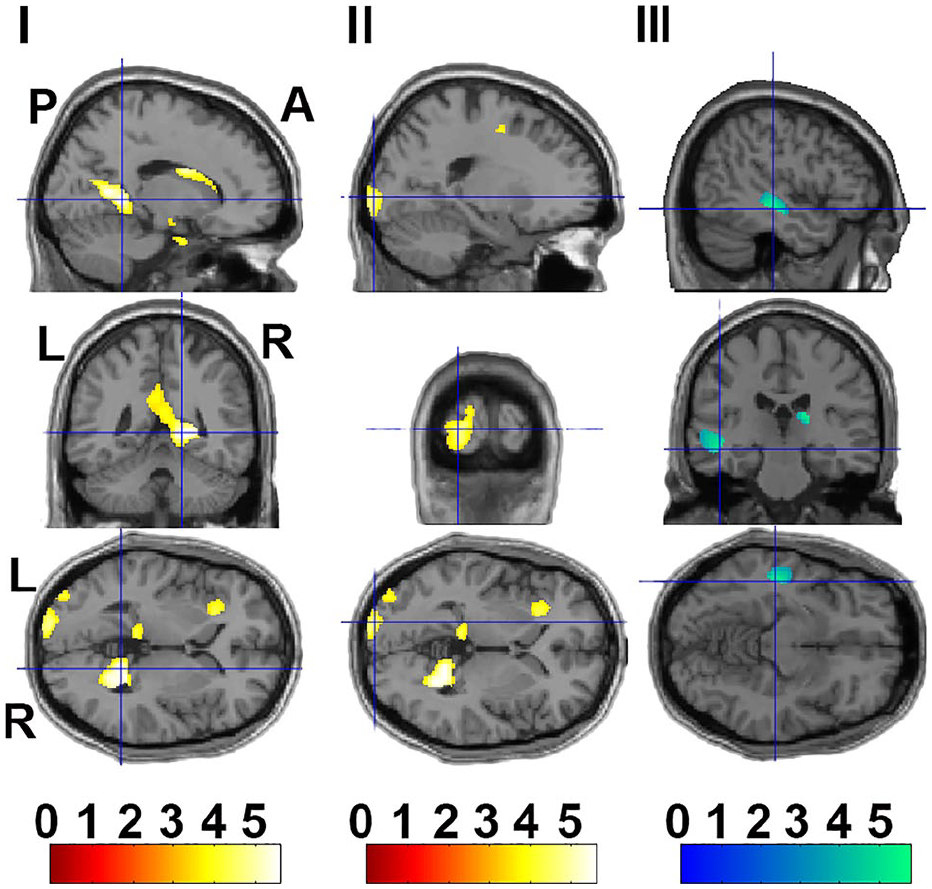
Cluster’s peak voxel regions with significant differences in GMV between transgender men and cisgender women. Anatomical localizations of three cluster peaks are indicated by cross-hairlines (
Discussion
This study is the first to investigate differences in GMV between transgender men and cisgender women using the VBM technique based on our hypothesis that transgender men have more prominently altered brain volumes than cisgender women, i.e. those sharing the same biological sex. We revealed that the transgender men, compared with the cisgender women, had regions with increased GMV (right posterior cingulate gyrus and left occipital pole) and decreased GMV (left middle temporal gyrus). These findings suggested that individuals with GD, transgender men, display altered brain structure compared with cisgender women.
Consistent with previous studies (Luders et al., 2009; Manzouri et al., 2017; Savic and Arver, 2011; Zubiaurre-Elorza et al., 2013) mainly focusing on GMV differences between transgender women (biologically male) and cisgender men, we found that transgender men (biologically female), compared with cisgender women, had an increased right posterior cingulate gyrus extending to the right lingual gyrus beyond the biological sex. The posterior cingulate gyrus and its surrounding regions are called cerebral midline structures, which involve self-referential processing in emotional, facial and social domains (Northoff and Bermpohl, 2004; Northoff et al., 2006). Larger thickness and hypoactivity of cerebral midline regions during the resting state in transgender men have been hypothesized to be biological markers for dysphoric own-body perception (Manzouri et al., 2017). Imbalance between internal self-perception and perception of the external world is related to alteration in self-related processing (Northoff and Panksepp, 2008). Therefore, we suggest that larger posterior midline structures may contribute to sensitivity to self-referential processing, including visual perception of their unexpected body in transgender people.
Furthermore, we detected that transgender men, compared with cisgender women, had increased left occipital pole and decreased left middle temporal gyrus. However, these regions were not significant after correcting for serum sex hormone levels, especially testosterone levels. Although serum testosterone or estradiol levels were not significantly correlated with any GMV in our participants (
There are some limitations that should be considered in this study. First, as the sample sizes in our study were relatively small (
Conclusion
We examined differences in GMV between transgender men and cisgender women, i.e., biologically female individuals. Compared with cisgender women, transgender men had increased GMV in the right posterior cingulate gyrus and the left occipital pole and decreased GMV in the left middle temporal gyrus. Particularly, the alteration in the posterior cingulate gyrus was not affected by serum sex hormone levels, and the posterior cingulate gyrus was associated with transgender men as well as transgender women. Further study is required to explore functional differences in the posterior cingulate between GD individuals and cisgender individuals.
Footnotes
Acknowledgements
We would like to thank Prof. Jun Shinoda and the technical staff at Chubu Medical Center for acquiring magnetic resonance imaging data. We would also like to thank gid. jp and GID proud for supporting recruitment of transsexual participants.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
