Abstract
Objectives:
Differentiating schizophrenia from mania in acutely psychotic patients can be difficult, but is important in determining immediate and subsequent management. Such differentiation is generally addressed by clinical interviews, but an observational approach may assist. This paper therefore describes the development of a relevant observational measure.
Methods:
We developed a provisional list of 49 items (weighting features with suggested specificity to schizophrenia and mania) for independent completion by two nurses and judged its ability to predict diagnosis provided by consultant psychiatrists.
Results:
Eighty-seven psychotic patients were recruited, and 173 completed data sets were analysed. We refined the item set to two sets of 10 items that best-differentiated schizophrenia from mania and vice versa. Optimal differentiation was achieved with a score of at least 7 on both the schizophrenia and mania item sets. Difference scores (i.e. schizophrenia items affirmed minus mania items affirmed) were also generated, with a difference score of +1 (i.e. one or more schizophrenia items being affirmed than mania items) showing optimal differentiation (sensitivity 0.67, specificity 0.82) between the two conditions. Evaluating all potential difference scores, we demonstrated that, as difference scores increased, diagnostic accuracy in identifying each condition was very high.
Conclusion:
Analyses allow the properties of an observational measure (the 20-item Sydney Psychosis Observation Tool) to be described. While a single cut-off difference score was derived with acceptable discriminatory ability, we also established the capacity of varying difference scores to assign both schizophrenia and mania diagnoses with high accuracy.
Introduction
When an individual is admitted to an acute psychiatric unit in a psychotic state, common differential diagnoses include schizophrenia, mania and organic states – with the last commonly involving illicit drug use. Organic psychoses can usually be identified based on the presenting history, associated features (e.g. abnormal vital signs, altered levels of consciousness, seizures) and basic investigations (e.g. blood tests, urine drug screen). Differentiating schizophrenia from mania, however, is more complex given the absence of biomarkers and the fact that many patients can exhibit prototypical symptoms of both diagnoses. We briefly review relevant literature on this distinction before detailing the development of a diagnostic measure that employs observational ratings.
The distinction between schizophrenia and mania originated with Emil Kraepelin’s (1899) separation of ‘dementia praecox’ (schizophrenia) from ‘manic-depressive illness’ (bipolar disorder) in his classificatory scheme (and leading to the so-called ‘Kraepelinian dichotomy’). However, since it was first proposed, this division has been contested due to the frequency with which some patients show features of both conditions. This led Kasanin (1933) to propose a third category, ‘schizoaffective disorder’, to encompass such patients, which is now a DSM-5-TR diagnostic category. More recently, the original dichotomy has been challenged by evidence of shared genetic underpinnings (Greene, 2007). The distinction nevertheless has remained influential due to distinct differences in clinical management both during acute and ongoing management phases. Thus, an important clinical priority is to diagnose each condition accurately.
While patients with schizophrenia and mania exhibit psychosis, each condition may be more likely to present with certain features (Kendler, 1986). In relation to hallucinations, their expressions in mania have been characterised as infrequent, incomplete, and closely related to mood, whereas those in schizophrenia have been characterised as non-sensical and more likely to take the form of hearing one’s thoughts spoken aloud or in third person (Schneider, 1959). For delusions, those in manic states have been characterised as grandiose and having a basis in reality, whereas those in patients with schizophrenia may be more likely to be fantastic, bizarre and/or involve themes of altered bodily agency, ideas of reference, and thought broadcasting and manipulation (Dikeos et al., 2006; Kendler, 1986; Lake, 2008; Solovay et al., 1987). With respect to thought disorder, those with mania often evidence distractibility and tangentiality, while in those with schizophrenia, it may be more likely to involve non-sequiturs, incoherence, and disorganisation (Kendler, 1986; Solovay et al., 1987).
Patients with schizophrenia and mania may also differ in other domains. The affect in patients during a manic episode is often unrestrained, elevated, cheerful, irritable or involve atypical increases of these emotions (Dikeos et al., 2006; Kendler, 1986; Lake, 2008; Murray et al., 2005). By contrast, the affect in patients with schizophrenia is often blunted, reflecting the negative symptoms associated with the condition (Kendler, 1986; Walker et al., 1988). Differences in psychomotor activity may also be evident. Patients in manic states are more likely to show agitation and high energy levels, evidenced by restlessness, talkativeness and gregariousness (Klein, 1982). By contrast, patients with schizophrenia are more likely to show anhedonia, apathy and catatonia (Kendler, 1986).
As already noted, differentiating between the two conditions is important due to differences in prognosis and management (e.g. preferentially prescribing or co-prescribing a mood stabiliser if the patient is in a manic state and subsequent management). However, few tools exist to aid clinicians in differentiating the two conditions. The Schedule for Affective Disorders and Schizophrenia (SADS) (Endicott and Spitzer, 1978) collects clinical feature details and encompasses other mental disorders aside from bipolar disorder and schizophrenia (e.g. depression, anxiety, obsessive-compulsive disorder) but is of considerable length. The Brief Psychiatric Rating Scale (BPRS; Overall and Gorham, 1962) is of a more convenient length, but has a convoluted scoring system, and generates subtyping of mania/schizophrenia presentations which are too specific for simple diagnostic purposes. The computer-based OPCRIT criteria system (McGuffin et al., 1991) covers a wide range of affective and psychotic symptomology, produces diagnoses according to multiple definitions and has reasonable concordance with clinical diagnoses (Brittain et al., 2013). Such scales require a clinical interview and prioritise symptoms as against observable behaviour or signs. There is one nurse rating scale – the Nurses’ Observational Scale for Inpatient Evaluation (NOSIE; Honigfeld et al., 1966), but this measures only six constructs in patients with schizophrenia – being designed to measure patient status and change over time rather than being a diagnostic scale. Nevertheless, the NOSIE-30 does have considerable advantages, particularly with regard to its non-interview format and focus on nurse observations.
Perhaps due to the challenges in differentiating schizophrenia and mania, particularly in a busy psychiatric ward, a frequent default may be a diagnosis of schizoaffective disorder. This, however, may sometimes simply reflect the practical difficulties of assessment, rather than valid categorisation. Diagnostic assessments generally weight the interview of the patient by a clinician and involve the patient being interviewed when seated, and risk constraining the patient’s spontaneous verbal responses and behaviours. However, when psychotic patients are observed ‘in vivo’ or ‘free ranging’ (i.e. in the ward, gardens, and airing courts), they may show diagnosis-specific behaviours or signs that may not be evidenced in the interview. In such contexts, nurses are front-line observers. Such a reality encouraged us to develop a nurse-rated observational measure (though this measure need not necessarily be limited to nursing staff) that would help differentiate patients with schizophrenia and mania.
Methods
Study design
The study was undertaken across three acute psychiatric wards at Prince of Wales Hospital in Sydney, Australia, with recruitment proceeding from January 2021 to August 2022. These wards routinely admit individuals with acute psychoses. Inclusion criteria for study subjects required them to be (i) currently hospitalised with a psychotic illness (i.e. bipolar mania, schizophrenia or some unspecified functional psychosis), (ii) not experiencing a psychosis that was drug-induced and (iii) 18 years of age or older. Exclusion criteria were (i) having another primary psychiatric disorder, (ii) having a bipolar disorder but currently experiencing a depressive episode, and (iii) having any distinct comorbid medical condition (e.g. hyperthyroidism) that might confound diagnostic differentiation.
As all patients were experiencing acute psychosis, it was not possible to obtain informed consent from them. As such, the study was designed such that all the necessary requirements to obtain waivers of consent (as outlined in Section 2.3.10 of the Australian National Health and Medical Research Council’s National Statement on Ethical Conduct in Human Research) were met. These procedures, and all other elements of this study, were approved by the Human Research Ethics Committee of the South Eastern Sydney Local Health District (SESLHD; project 2020/STE05396).
Procedures
Study registrars (trainee psychiatrists) were responsible for recruiting eligible subjects. Registrars identified patients who had been admitted to the ward and met the study criteria. The registrars then identified two nurses responsible for the care of each patient (and who had observed them for a sufficient period) and invited them to complete a nurses’ questionnaire (which rated different aspects of the patient’s behaviour; see section ‘Measures’). These two nurses completed their questionnaires independently of each other (i.e. each nurse was unaware of the other’s scores), thus allowing inter-rater reliability to be quantified. The registrar also contacted the patient’s consultant psychiatrist, who was blinded to the nurses’ ratings, to provide a diagnosis for the patient using one of five categories across the schizophrenia-mania spectrum (see section ‘Measures’).
Measures
Nurse questionnaire
We generated a test measure comprising 49 descriptors of candidate condition-specific signs. These were compiled from an extensive literature review of symptoms and signs typifying mania and schizophrenia (with a focus on those that differentiated the two conditions as considered in the Introduction) and from the clinical experience of the researchers. The total list of items is provided in Supplementary Table 1. Nurses were required to rate each item descriptor as ‘not at all relevant’, ‘evident to some degree’ or ‘distinctly evident’. Informal reports from several rating nurses suggested that they found it difficult to judge the severity of any features present. Thus, in all analyses reported below, the comparison data comprised the amalgamated latter two rating options versus the first, so that ratings effectively indicated whether an item was ‘present’ or ‘absent’. The age and gender of each patient were also recorded.
Psychiatrist questionnaire
The patients’ consultant psychiatrist could choose from one of five options in providing their diagnosis of study patients. Allowed options included ‘definitely schizophrenia’, ‘most likely schizophrenia’, ‘definitely mania’, ‘most likely mania’, and ‘diagnosis unclear – features of schizophrenia and mania both present’ (while the latter option sought to capture those with a schizoaffective disorder, it clearly also captured diagnostic uncertainty). In our principal analyses, and in light of the relatively small sample size, those assigned to the first and second groups were combined to form a schizophrenia (SZ) group, while the third and fourth formed a mania (MN) group. We also positioned those in the ‘diagnosis unclear’ group as potentially having a schizoaffective (SA) condition. Comparisons between these groups formed the basis of the analyses.
Analyses
While the questionnaire contained 49 items, we judged that our measure would need to have distinctly fewer items if it were to be acceptable to those employing it for clinical or research purposes. Odds ratio analyses were used to identify items that most discriminated the SZ and MN patients in terms of their being affirmed as present. After selecting a reduced item set for the final measure (by deleting non-differentiating or minimally differentiating items), receiver operating characteristic (ROC) analyses were then undertaken to assess the optimum cut-off scores for three candidate measures of diagnostic differentiation: a schizophrenia score, a mania score, and an overall difference score (i.e. number of schizophrenia items affirmed minus the number of mania items affirmed). Specifically, these cut-offs were determined by maximising the product of sensitivity and specificity (Liu, 2012). Further analyses also considered the probability of individuals having an SZ or MN diagnosis given their score, and how SA patients were classified. All analyses were conducted in R (version 4.2.1) using the pROC package (Robin et al., 2011).
Results
Sample characteristics
The registrars recruited 87 patients: 40 with schizophrenia (as diagnosed by their consultant psychiatrist), 34 with mania and 13 with an ‘unclear’ diagnosis potentially indicating a schizoaffective disorder (but also diagnostic uncertainty). The mean age of subjects was 42.1 (SD = 12.9) years, and 58 (66.7%) were male. Each participant (bar one patient with mania) had two nurse ratings, yielding a total of 173 responses. Of these, 80 referred to a patient diagnosed as having definite or probable schizophrenia (48 and 32, respectively), 67 with definite or probable mania (51 and 16, respectively) and 26 with a potential schizoaffective disorder.
Item refinement
We first sought to identify the most differentiating items. Analyses (as indicated by the odds ratio statistics reported in Table 1) established that 29 of the 50 items significantly differentiated the SZ and MN groups. Items that failed to significantly differentiate the SZ and MN groups included ones assessing poor hygiene and grooming, frequent changing of clothes, speech content unconnected to surroundings, persistent anger and hostility, the compilation of lots of lists/notes, disorganised speech, distractibility, irritability, feeling entitled, a tangential speech pattern, being ‘bright-eyed and bushy-tailed’, wanting to spend lots of money, strange mannerisms such as posturing or grimacing, dressing in bright clothes, seeming paranoid thinking, hanging lots of material on bedroom walls, evidencing peculiar use of words and neologisms, having differing ideas and statements intruding into speech, evidencing unusual ideas that did not make sense, and the patient linking ideas or sentences by rhyme or other non-logical connections.
Final list of 20 discriminating items and their statistical characteristics.
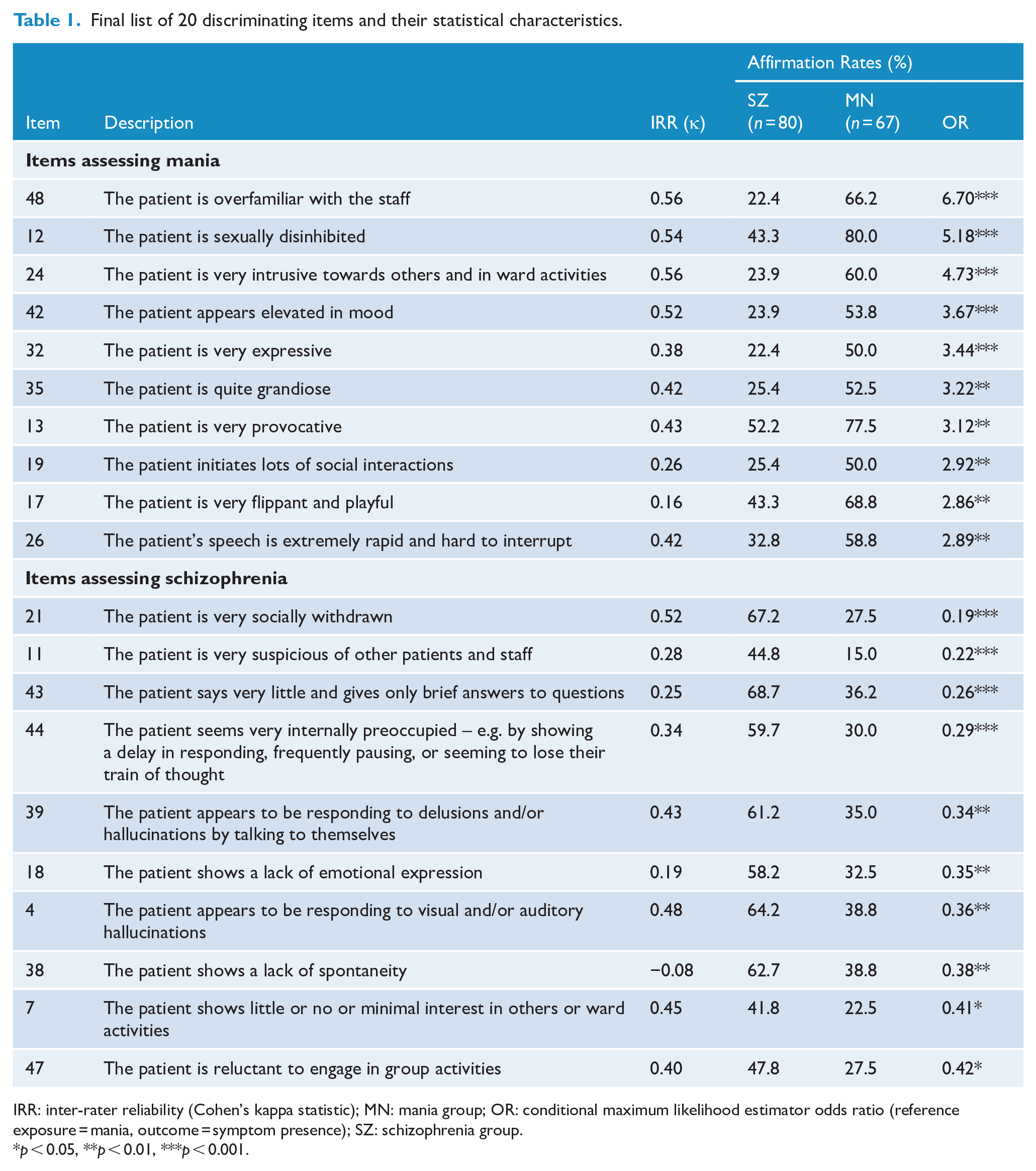
IRR: inter-rater reliability (Cohen’s kappa statistic); MN: mania group; OR: conditional maximum likelihood estimator odds ratio (reference exposure = mania, outcome = symptom presence); SZ: schizophrenia group.
p < 0.05, **p < 0.01, ***p < 0.001.
In seeking to develop a relatively brief measure that would advance its application, we set a target of 20 items (comprising 10 SZ and 10 MN items). We therefore reduced the 29-item set to two sets of 10 items that displayed the greatest difference in affirmation rates across the SZ and MN groups. These items are listed in Table 1, together with their frequency of affirmation in the two groups. Table 1 also reports the level of agreement between the two nurses in rating each item, with Cohen’s κ’s ranging from -0.08 to 0.56 (mean = 0.31).
ROC analyses and test performance
Our first two ROC analyses sought to determine how many of the items indexing schizophrenia and mania were required to be affirmed to achieve optimal differentiation between the two diagnostic groups. The ROC analyses established a cut-off of seven or more items for both item sets, with the sensitivity and specificity being 0.59 and 0.79 for the SZ set, respectively, and 0.66 and 0.76, respectively, for the MN set. For individuals affirming 7 SZ items or above, 77.0% of those were assigned as SZ, while the equivalent figure for MN (i.e. proportion of those who scored 7 or more MN items who had been assigned as MN) was 69.8%. Meanwhile, for those scoring less than 7 on each scale, the proportion of individuals not assigned as SZ/MN was 61.6% and 72.6%, respectively. These data are displayed in Table 2 along with the overall accuracy estimates.
Classificatory performance of the three ROC analyses according to their optimum cut-offs.
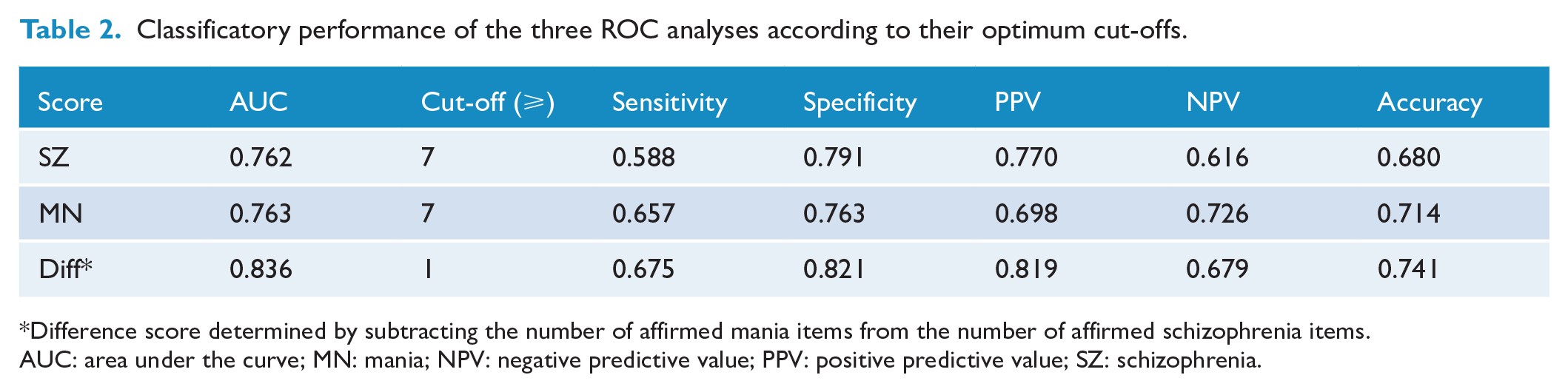
Difference score determined by subtracting the number of affirmed mania items from the number of affirmed schizophrenia items.
AUC: area under the curve; MN: mania; NPV: negative predictive value; PPV: positive predictive value; SZ: schizophrenia.
A third ROC analysis was undertaken to determine the optimal ‘difference’ score between the two 10-item sets considered in combination. Overall, as quantified by the area under the curve (AUC), the difference score performed better as a classifier (AUC = 0.84) than the SZ (AUC = 0.76) or MN scores (AUC = 0.76) alone. In this analysis, a score greater than or equal to +1 (i.e. a schizophrenia diagnosis is indicated if one or more SZ items than MN items are affirmed, otherwise a mania diagnosis is given) was identified as being optimal (sensitivity 0.67, specificity 0.82) in distinguishing between the two principal groups. Specifically, 81.9% of those assigned as having SZ by the difference score cut-off had been designated as SZ, while 67.9% assigned as having MN had been designated as MN. Figure 1 provides a graphical representation of all three ROC analyses.
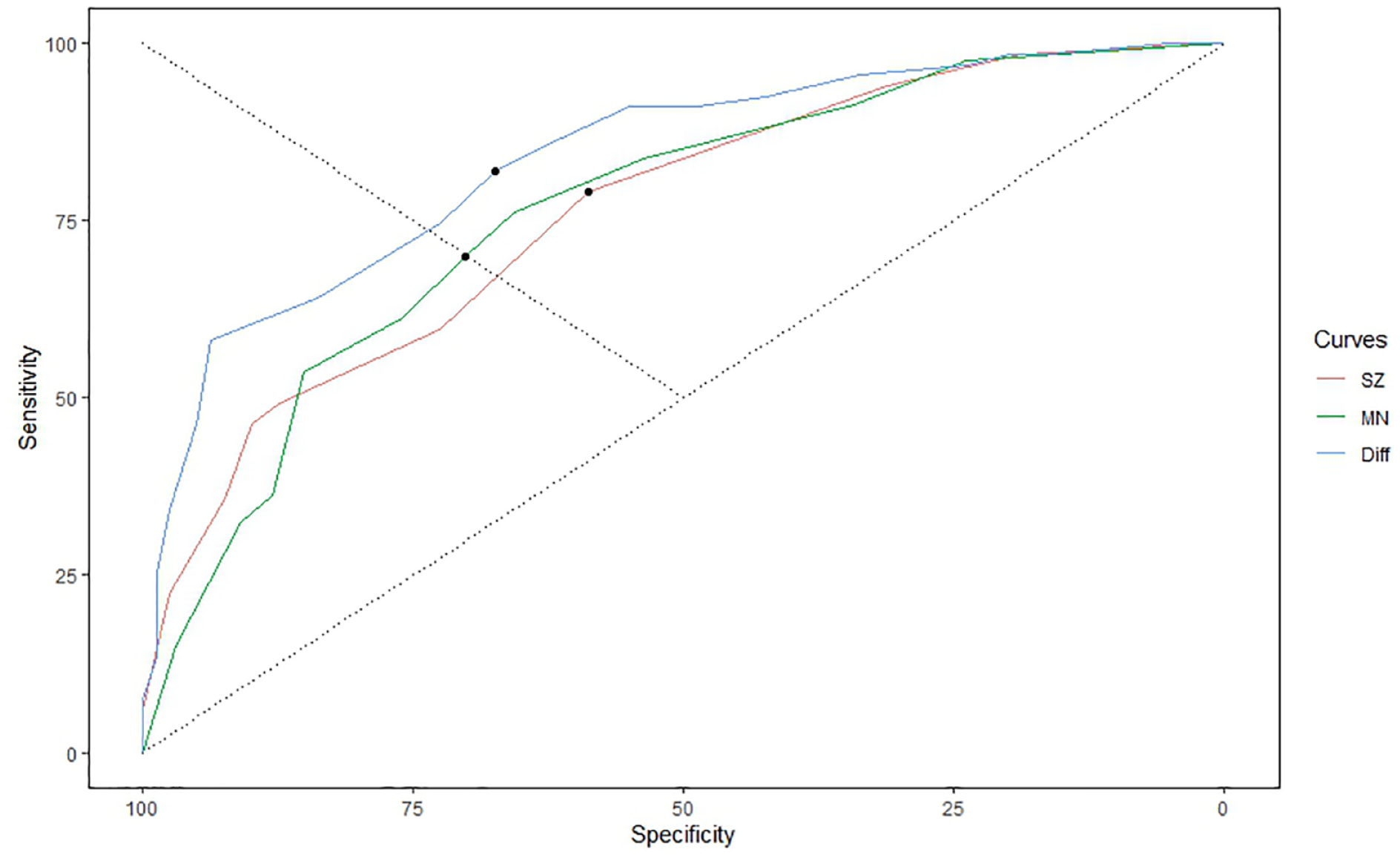
ROC curves and optimum cut-offs (marked as dots) for the SZ, MN, and difference scores.
While overall performance is informative in determining the best cut-off balancing sensitivity and specificity, we also considered the capacity of the measure to predict diagnoses correctly given that a score was equal to or exceeded (i.e. a larger score for SZ, a smaller score for MN) some threshold. Quantifying probabilities across all thresholds would provide an indication of the measure’s ability to account for more probable SZ or MN cases as the scores become more extreme (for example, we would expect the probability of having an SZ diagnosis would be much higher for an individual with a difference score of +10 compared to +2). Table 3 therefore reports the probabilities of an individual having schizophrenia or mania in relation to all possible difference score thresholds. SZ subject allocation was very high (over 80%), with difference score minimum thresholds in the +1 to +5 range, over 90% for scores of +6 to +9, and 100% for a score of +10. For the MN group, MN subject allocation exceeded 80% for a score of −3 and −4, over 90% for scores between −5 and −7, and 100% for scores between −8 and −10.
Probabilities of an individual having schizophrenia or mania given that they meet specific difference score thresholds.
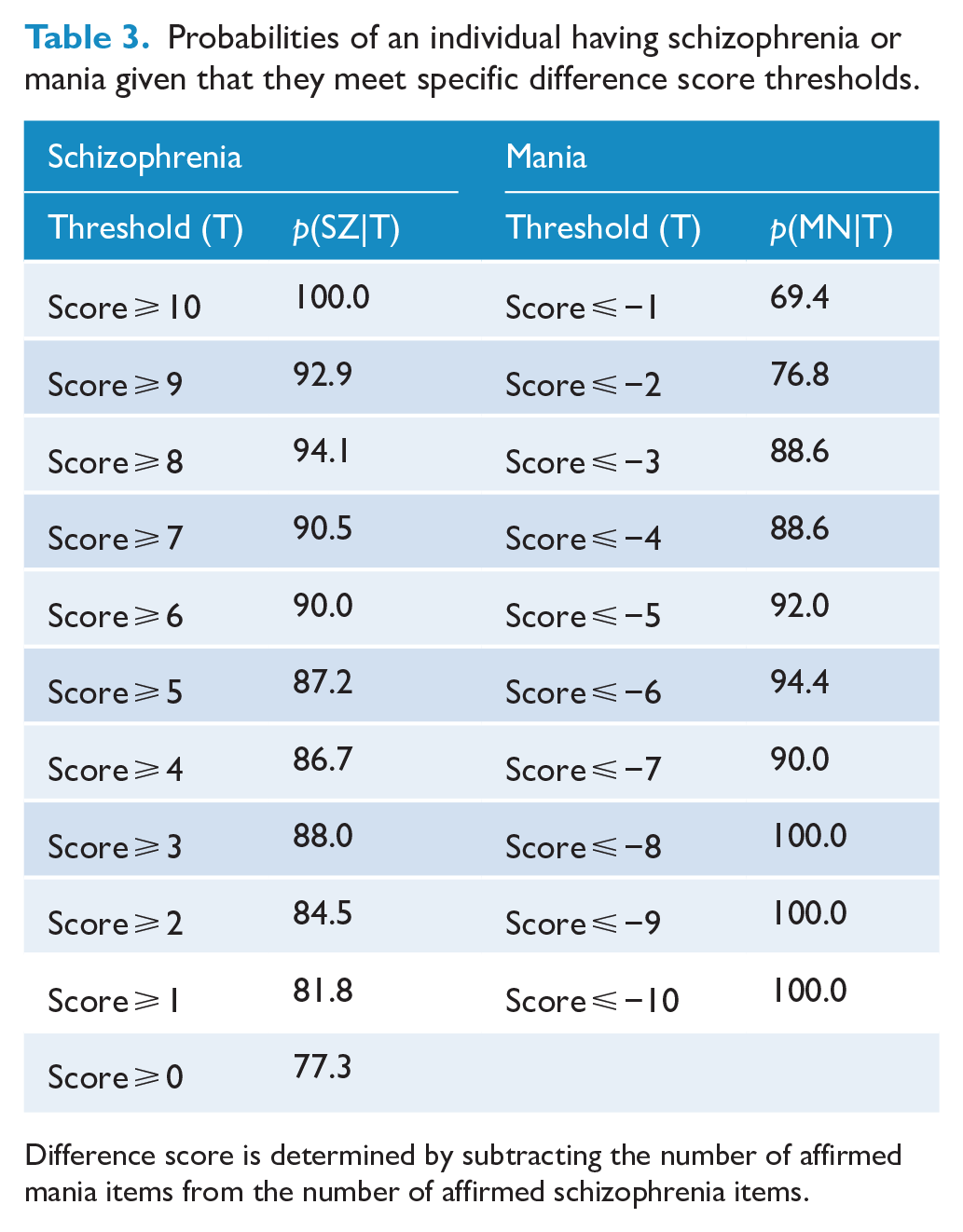
Difference score is determined by subtracting the number of affirmed mania items from the number of affirmed schizophrenia items.
A final ROC analysis was undertaken contrasting only those with ‘definite’ schizophrenia or mania. Here, a cut-off score of −1 was identified as optimal (sensitivity 0.85, specificity 0.67). However, the previous cut-off of +1 also performed quite similarly to this optimal cut-off (sensitivity 0.69, specificity 0.80).
Finally, with regards to the SA group, after applying our previously identified difference cut-off score of +1, 53.8% and 46.2% of patients were assigned a schizophrenia and mania diagnosis, respectively.
Discussion
We first note a few limitations. This study was conducted at a single hospital in patients with acute psychotic episodes, and thus the generalisability of the derived measure to other ward and patient settings is yet to be determined. We did not record the length of time the patient had been admitted for, or the period of time over which psychiatrists or nurses observed the patients. We positioned the psychiatrists’ diagnoses as the reference standard without independent verification, nor could we verify nurses’ compliance with our request to complete their forms independently as requested. Some study patients showed differing levels of disturbance over their time in the ward(s), which may have influenced severity ratings, but temporal stability was not examined. We undertook univariate, rather than multivariate analyses which would have identified underlying latent constructs that could have been used to differentiate the two conditions. Our intermediate (qua schizoaffective group) was comparatively small and had a limited role in informing the development of the scale. This means that the capacity for the measure to classify borderline cases remains unknown. Finally, some patients’ behaviours (e.g. lack of spontaneity, lack of emotional expression) might have reflected medication impact rather than reflect the psychotic state.
Our objective was to develop an observational measure that would assist in distinguishing schizophrenia and mania in psychiatric in-patients. In current practice, such distinctions are largely based only on a psychiatrist interview, which are recognised as being problematic at times. As noted in the Introduction, the risks are of invalid diagnostic assignment and less than adequate management options. It may also result in patients receiving the indeterminate diagnosis of a schizoaffective condition when a diagnosis of schizophrenia or mania may be more appropriate.
We selected a large number of candidate differentiating features respecting historically described prototypic symptoms and signs of each condition, and added several others that we judged as having potential utility. As the consultant psychiatrists’ diagnoses were expected to have a level of accuracy but not absolute validity, we therefore initially introduced ‘definite’ and ‘probable’ diagnostic options for the psychiatrists as well as an indeterminate category. Specifically, 27.7% of the sample were assigned to the ‘probable’ categories, while only 57.2% of study subjects received a diagnosis of definite schizophrenia or definite mania, supporting the common difficulty in being able to make a definitive diagnostic decision in those with functional psychoses. We assumed that the ‘indeterminate’ categorical option would capture those with a schizoaffective condition, but as it may have simply reflected the psychiatrists’ being unsure about diagnostic allocation, we cannot make any conclusion about our final algorithm assigning 54% and 46% of that group with ‘schizophrenia’ and ‘mania’ diagnoses. Future studies should focus on the capacity of the measure to assign such ‘intermediate’ patients to one or other diagnostic categories or, assuming that schizoaffective illness is a valid entity, seek to determine cut-off criteria for its identification.
By excluding items that did not differentiate the two clinical diagnostic groups significantly in univariate analyses, we were able to reduce the candidate item pool from 49 to 29 items. We then elected to limit the item set to the 10 items most distinctly differentiating the patients with schizophrenia from those with mania subjects, and a further 10 items best differentiating patients with mania from those with schizophrenia subjects, to ensure that the derived measure would not be too lengthy.
By having two nurses independently rate subjects, we were able to evaluate the reliability of ratings. Agreement in rating each of the refined set of 20 items was moderate, with a mean Cohen’s κ of 0.31. Such data could potentially suggest that accurate judgement of many of the behaviours was not always straightforward, that there was a lack of motivation in some of the nurses and/or that there were temporal changes in patients’ behaviours between their two ratings.
Analyses examining the capacity of SZ items alone to predict SZ (and similarly for MN items and MN) suggested that having at least seven of the items affirmed performed best for both sub-scales in terms of distinguishing SZ and MN patients from each other. However, as we judged that the final measure should have items reflecting both schizophrenia and mania, we emphasise results from the third ROC analysis which determined the optimal cut-off for the difference score (i.e. score on the 10 schizophrenia items minus the score on the 10 manic items). It was established that a score of +1 (i.e. one or more schizophrenia items being affirmed than the number of mania items affirmed) was the optimal cut-off for overall classification and in balancing sensitivity and specificity. Using such a single cut-off score, we achieved moderate sensitivity (i.e. 67%) and quite high specificity (i.e. 82%). However, our Table 3 data show linear trends indicating that more extreme difference scores (i.e. higher for SZ, lower for MN) were associated with a greater probability of obtaining the relevant corresponding diagnosis. Specifically, difference scores of +1 to +6 correctly assigned more than 80% of the SZ subjects, while difference scores of +6 to +10 assigned 90% to 100%. Similarly, difference scores of −3 to −10 correctly assigned 88%–100% of the MN subjects. Thus, in addition to validation studies, researchers and clinicians may wish to impose differing and higher cut-off scores if the aim is to advance sensitivity (and determine ‘true cases’).
We view these findings as supporting the utility of the final measure, which is listed in Table 4 with guidelines for rating and scoring. In application, we note that the 20-item measure preserves the original item listing order (so as to not consolidate the items assessing schizophrenia and mania items as sets and risk a rating bias). Scoring then involves generating a total schizophrenia score, a total mania score, and a difference score; with the latter providing the most definitive assessment of the probability of one diagnosis over the other. Replication studies would clearly be of benefit, while the measure may also allow those with a schizoaffective illness to be identified with greater probability by both highlighting the key defining and contributing items to its definition and by considering rates of those assigned a diagnosis of schizophrenia or mania by the measure. While the measure was designed for the assessment of acutely psychotic patients as rated by nurses, extension studies might also pursue its utility in patients with non-acute states of schizophrenia and mania or in raters occupying other mental health roles. It would also be beneficial to evaluate cut-off scores in other samples to determine the likely validity of the cut-off scores quantified in this study. The derived measure is named the SPOT (Sydney Psychosis Observation Tool) as it optimally allows the two considered conditions to be assessed as a spot test.
Final version of the SPOT.
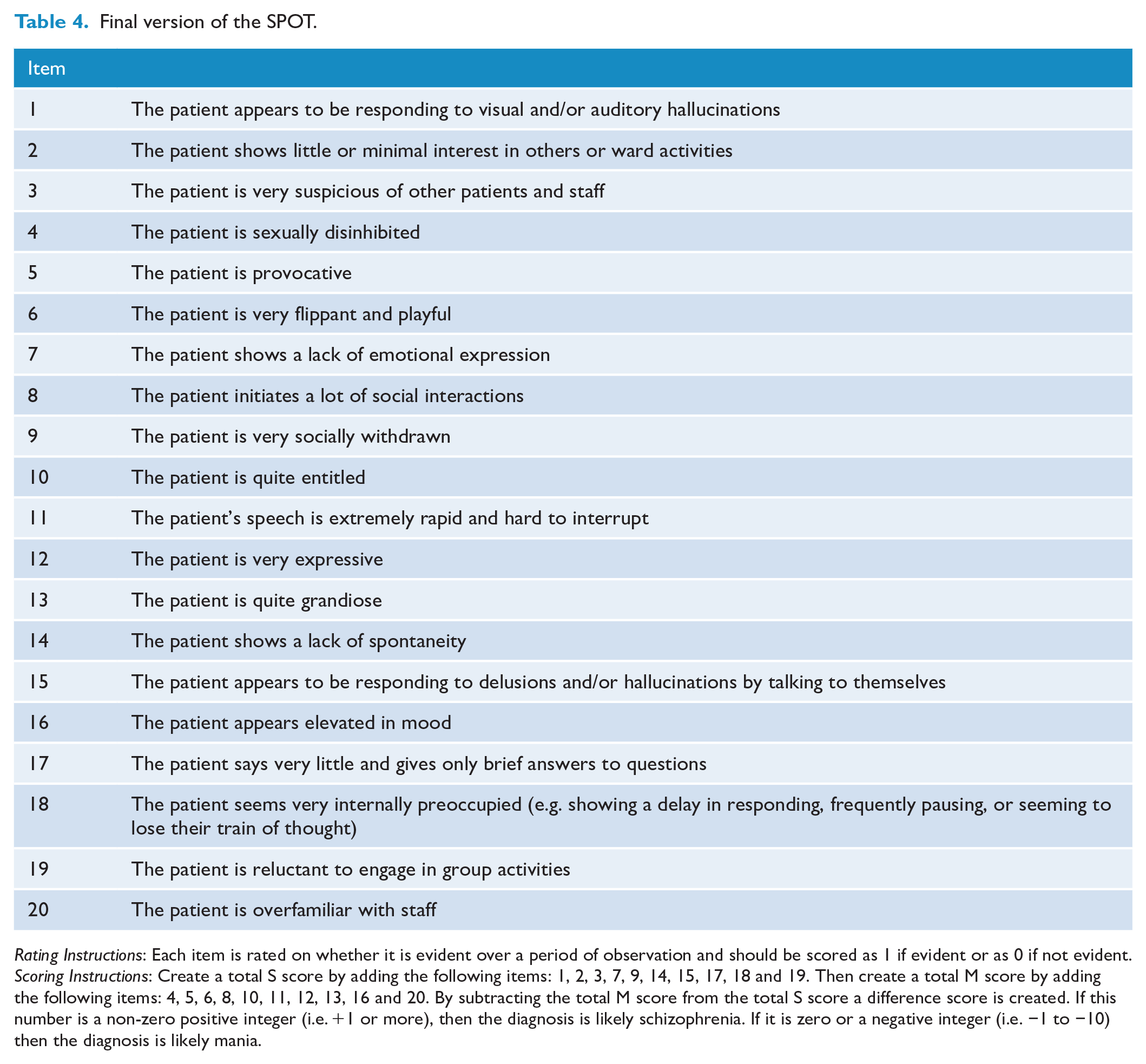
Rating Instructions: Each item is rated on whether it is evident over a period of observation and should be scored as 1 if evident or as 0 if not evident.
Scoring Instructions: Create a total S score by adding the following items: 1, 2, 3, 7, 9, 14, 15, 17, 18 and 19. Then create a total M score by adding the following items: 4, 5, 6, 8, 10, 11, 12, 13, 16 and 20. By subtracting the total M score from the total S score a difference score is created. If this number is a non-zero positive integer (i.e. +1 or more), then the diagnosis is likely schizophrenia. If it is zero or a negative integer (i.e. −1 to −10) then the diagnosis is likely mania.
Supplemental Material
sj-docx-1-anp-10.1177_00048674231201545 – Supplemental material for An observer-rated strategy for differentiating schizophrenic and manic states in inpatient settings
Supplemental material, sj-docx-1-anp-10.1177_00048674231201545 for An observer-rated strategy for differentiating schizophrenic and manic states in inpatient settings by Gordon Parker, Michael J Spoelma, Samuel J Skidmore, Amelia Reid, Samuel Morris, Greta Ferguson and Michael H Connors in Australian & New Zealand Journal of Psychiatry
Footnotes
Acknowledgements
The authors would like to thank Matthew Large, Penny Sawdy, Cecilia Muniz and Michael Paton for their assistance and all the nurses, patients and consultants who supported the study.
Author Contributions
G.P. was the primary author of the manuscript. M.J.S. co-designed the study, conducted the literature review, and performed the statistical analyses. M.H.C. coordinated recruitment and co-designed the study. S.J.S., A.R., S.M. and G.F. recruited patients for the study. All authors reviewed and edited the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Australian National Health and Medical Research Council (NHMRC; grant no. GNT1176689). The contents of the published material are solely the responsibility of the individual authors and do not reflect the views of the NHMRC.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
