Abstract
The Lundby study 1947 was set up as a prevalence study in a rural area of Sweden [1]. Re-investigations of the original cohort, including people added to the area 1947–1957, were conducted in 1957 and 1972 [2, 3]. In 1997 a further follow up was conducted [4]. At each point information was obtained about the development of mental and physical health for all individuals alive at the previous investigation, providing an opportunity to observe changes in incidence of mental disorders over 50 years.
The aim was to study how incidence of mental disorders in the Lundby cohort changed when different historical time periods were compared.
Method
Sample
The study population lived in an area situated 15 km from the medium-sized university town Lund and 25 km from the larger city Malmö in the southern part of Sweden. In 1947 the Lundby area was a farming district, including a village of 1000, and had altogether 2550 inhabitants, aged 0–92 years. In 1957 1013 newcomers were included. The present study includes the total cohort (n = 3563, male 1823, female 1740).
In the 1940s and 1950s the sample was representative of a rural population, but by 1972 gradual urbanization had changed the Lundby area from a farming district into a suburban area, where most people commuted to neighbouring cities [4], a development that by 1997 had become even more pronounced.
In 1972, many had moved out. Of the 2827 still alive on the 1972 cut-off date, 1424 remained while 1403 had moved, 191 to rural areas and 1210 to urban/suburban areas.
In 1997, 602 of the 1797 survivors lived in the Lundby area while 1195 lived outside, 126 in rural areas and 916 in urban/suburban areas (in 153 cases whereabouts were unknown). Table 1 details the sample.
The aim of the study was to obtain information about all episodes of mental disorders and somatic illnesses since inclusion into the cohort. At all investigations longitudinal data since the last investigation were collected using several sources of information: interviews, case registers [5–7], case notes and key informants.
The attrition in 1972–1997 was highest in those under 50, approximating 7% and 10% in male and female subjects, respectively. In those over 50 attrition 1972–1997 was 2% in male and 3% in female subjects.
Diagnostic assessment
In 1947, four psychiatrists led by Essen-Möller conducted the study. In 1957 Hagnell alone, and in 1972 Hagnell together with Öjesjö, did the follow ups. In 1997 a team led by Nettelbladt conducted the field work. The diagnoses 1972–1997 were set jointly by the 1997 research team (M.B., C.M., P.N.) along with Hagnell and Öjesjö, employing a best-estimate consensus approach, using all available information. The diagnostic principles in 1997 were the same as those used before [2, 3]. The diagnostic groups were thought of as a way to get “a rough picture of the relative distribution of the main types of mental illness in the population” [2].
Neurotic and psychotic disorders were meant to be disorders in which there was no evidence of an underlying organic aetiology. Neurotic, as opposed to psychotic, indicated a disorder with preserved insight.
Organic brain disorders included conditions with marked cognitive dysfunction or gross personality or behavioural disturbance, where organic causation was obvious.
Personality disorder, mental retardation and substance abuse were recorded separately.
Only one diagnosis per episode of disorder was recorded. The system is hierarchical, with organic brain disorders taking precedence over psychotic disorders, which in turn overrides neurotic disorders.
Every episode of disorder was rated along with the degree of dysfunction caused by it (minimal, mild, medium, severe or very severe) according to the criteria defined by Leighton et al. [8]. Conditions with severe impairment involve a marked reduction in functional capacity, for example depression with retardation or delusions (approximately corresponding to Global Assessment of Functioning Scale (GAF) 50-31). People with very severe impairment depend on daily help (GAF 30-1). Mild impairment means a lowered achievement or some symptoms but, for example, daily work is usually possible (GAF 70-61). Medium impairment is unsharply delimited between mild and severe impairment (GAF 60-51). Episodes with impairment levels medium, severe or very severe, which approximates GAF 60-1 [9]), were chosen to indicate caseness in the present paper. The reason for choosing this threshold was manifold. Psychiatric episodes giving minimal and mild impairment (GAF 80-61) are probably easy to forget and may not be considered as psychiatric disorders by the individual and will, therefore, often remain unreported even if remembered. Furthermore, individuals with mild psychiatric conditions may not seek professional help and hence data from these episodes will not be found in registers or in case notes. In order to minimize information bias when different historical periods were compared, episodes of minimal and mild impairment were excluded. Besides, the chosen caseness threshold corresponds with the criteria for clinical significance of many diagnostic categories in the DSM-IV, a GAF score of 60 or lower [10]. For all episodes, onset and termination were assessed as closely as possible using all sources of information.
Patterns of psychiatric diagnosis have changed over the last 50 years [11]. Notwithstanding this, we decided in 1997 to use the same diagnostic system as earlier in the Lundby investigations in order to be able to compare diagnoses over the 50 year period.
The diagnostic agreement for the assessment of the broad categories of neurotic, psychotic and organic brain disorders between the research teams in 1957/1972 and 1997 was tested [4], yielding a kappa value of 0.6, indicating moderate to substantial agreement [12].
Statistical procedures
First-incidence rates were obtained as the number of first-onsets of the disorder divided by the number of observation years free of the disorder in question [13]. The first-onsets were obtained for individuals who had been previously free from the disorder. Furthermore, individuals who had contracted outcomes of diagnostically higher hierarchical value than the studied outcome no longer contributed with observation years.
Incidence rates were calculated for neurotic, psychotic and organic brain disorders.
It was decided to calculate age- and sex-specific rates for the two 25 year periods, 1947–1972 and 1972–1997, and also for ten 5 year periods from 1947 to 1997. Ninety-five per cent confidence intervals (CI) for the incidence rates were calculated. To compute age-standardized incidence-rates, weights were obtained by dividing the number of observation years in each age interval during the study period 1947–1997 with the total number of observation years from all age intervals for the same period. For the 5 year period rates after 1987 separate weights were used, excluding the age interval 15–29, because no probands were younger than 30 after 1987.
The difference between age-standardized incidence-rates was tested by means of 95%CI for their ratio [14]. A ratio having the value 1 occurs when the rates are equal. A confidence interval excluding the value 1 was taken to indicate a statistically significant difference. SPSS for Windows, version 11.5.1 (SPSS, Chicago, IL, USA) was used for the calculations.
Data sources for living and dead individuals at follow up

†Original cohort (2550) + newcomers (1013).
Results
Neurotic disorders
When age- and sex-specific incidences of neurotic disorders in the first and second 25 year period were compared, the rates were found to have fallen consistently, except in men aged 70–99, for whom an increase was observed. The age-standardized first-incidence of neurotic disorders in both male and female subjects showed a statistically significant decrease (see Table 2).
Neurotic disorders of medium+ (GAF 60-1) impairment†
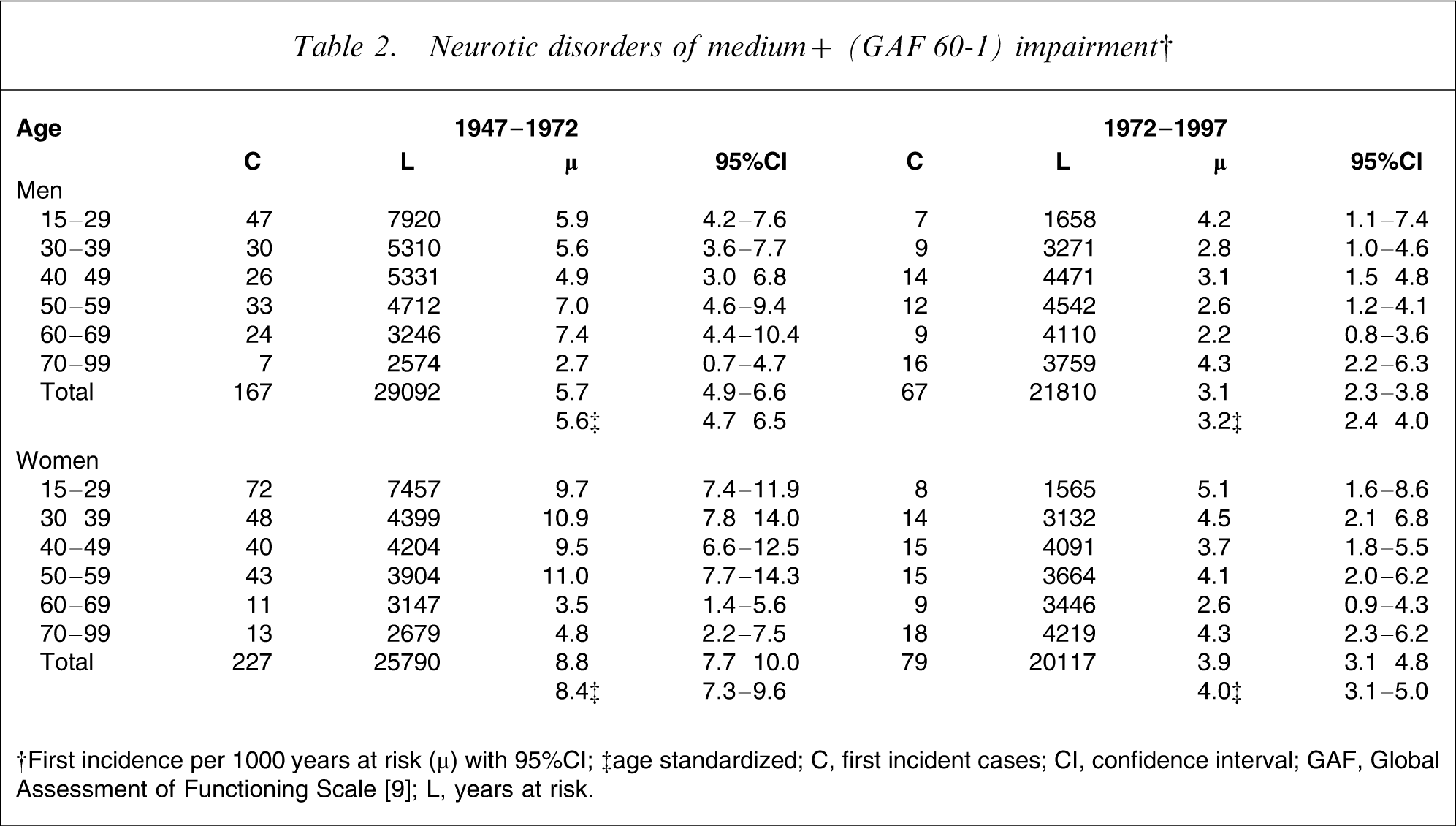
†First incidence per 1000 years at risk (µ) with 95%CI; ‡age standardized
C, first incident cases; CI, confidence interval; GAF, Global Assessment of Functioning Scale [9]; L, years at risk.
When incidences of neurotic disorders in male and female subjects were compared, female subjects had higher rates than male subjects in both 25 year periods. The difference was statistically significant in the first but not in the second period.
When, as shown in Figure 1, the development of the age-standardized incidences of neurotic disorders was observed every 5 years for 1947–1997, a decrease following 1972 was seen in both sexes.
Age-standardized first-incidence of neurotic disorders of medium+ (GAF 60-1) and severe+ (GAF 50-1) impairment by 5 year periods 1947–1997 (figure published with permission from Blackwell Publishing Ltd. Earlier published by Nettelbladt et al. (2005) Does it make sense to do repeated surveys? The Lundby Study, 1947–1997. Acta Psychiatr Scand 2005; 111:444–452.)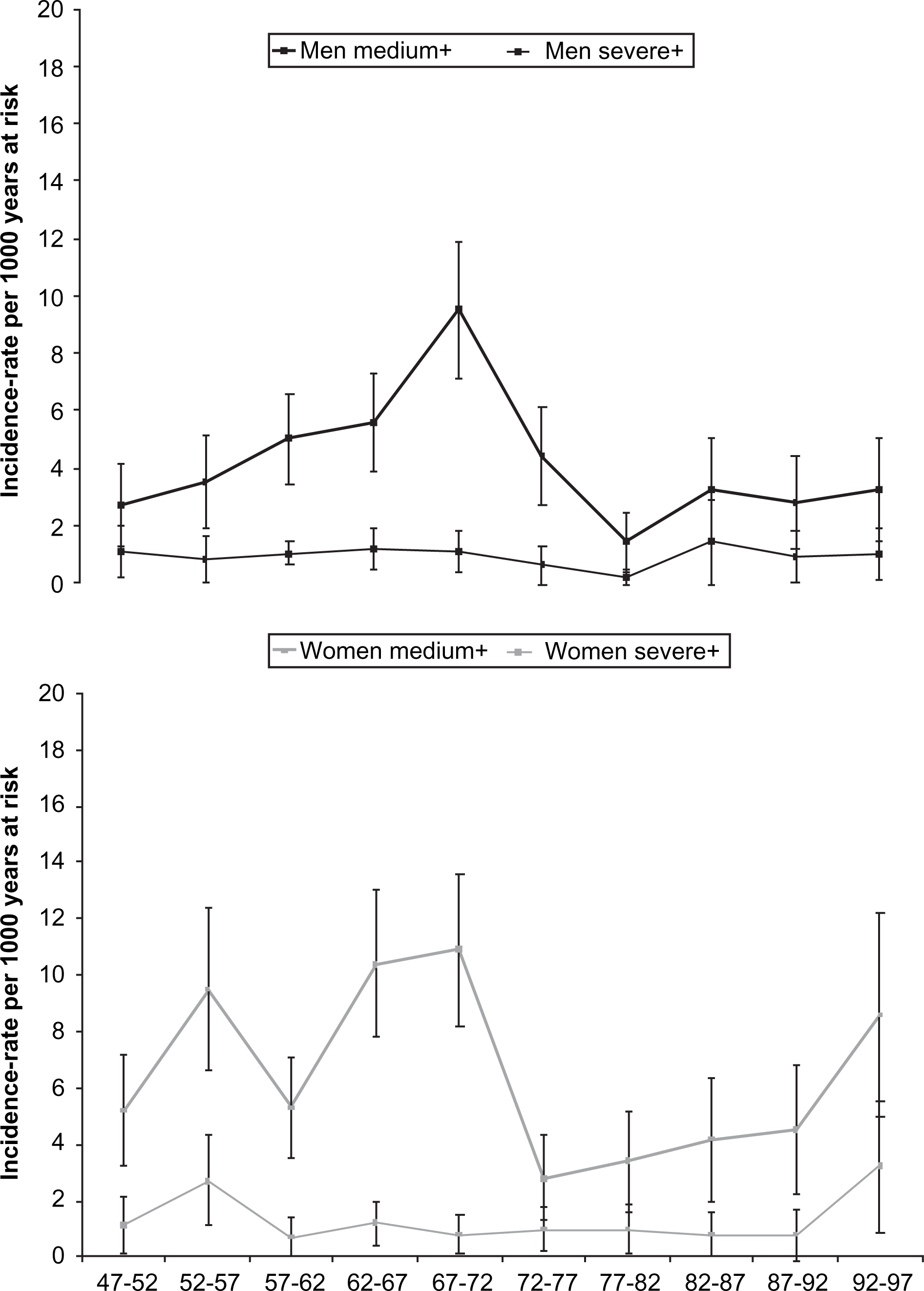
Furthermore, when the threshold for neurotic disorders was raised to severe impairment, a decrease in the age-standardized first-incidence, from the first to the second 25 year period, was still apparent in male subjects, for whom the rate fell from 1.1 to 0.6 per 1000 years at risk, whereas no change was seen among female subjects, in whom the rates remained the same, 1.3 per 1000 years at risk, in the first and second periods, respectively.
When the threshold for caseness was set at mild impairment the age-standardized first-incidence of neurotic disorders in male subjects decreased from 9.4 to 5.4 and in female subjects from 15.0 to 7.8 per 1000 years at risk.
Psychotic disorders
When age- and sex-specific incidences of psychotic disorders in the period 1947–1972 were compared to those in the period 1972–1997, the incidence rates increased consistently across all age groups in male subjects, whereas they decreased in all age intervals except for 30–39 and 70–99 in female subjects. The male age-standardized first-incidence rate of psychotic disorders increased significantly for 1972–1997, while the female age-standardized rate decreased insignificantly (see Table 3).
Psychotic disorders of medium+ (GAF 60-1) impairment†
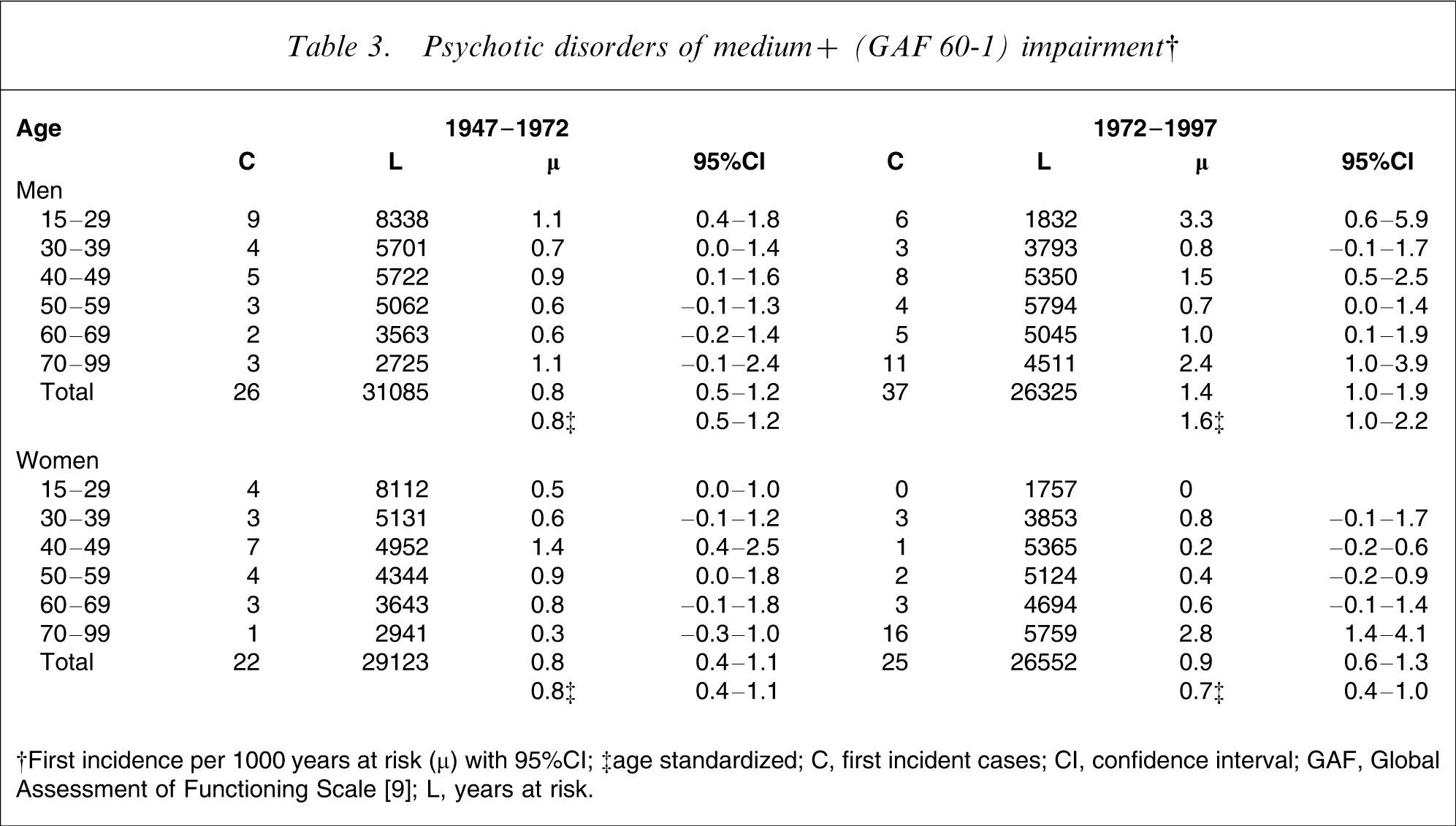
†First incidence per 1000 years at risk (µ) with 95%CI; ‡age standardized
C, first incident cases; CI, confidence interval; GAF, Global Assessment of Functioning Scale [9]; L, years at risk.
When age-standardized incidence rates of psychotic disorders in male subjects were compared to the rates in female subjects for the periods 1947–1972 and 1972–1997, no significant difference was found in 1947–1972. However, a statistically significant difference was observed in 1972–1997.
Figure 2 illustrates the fluctuation of rates of age-standardized first-incidence of psychotic disorders by 5 year intervals 1947–1997.
Age-standardized first-incidence of psychotic disorders of medium+ (GAF 60-1) impairment by 5 year periods 1947–1997.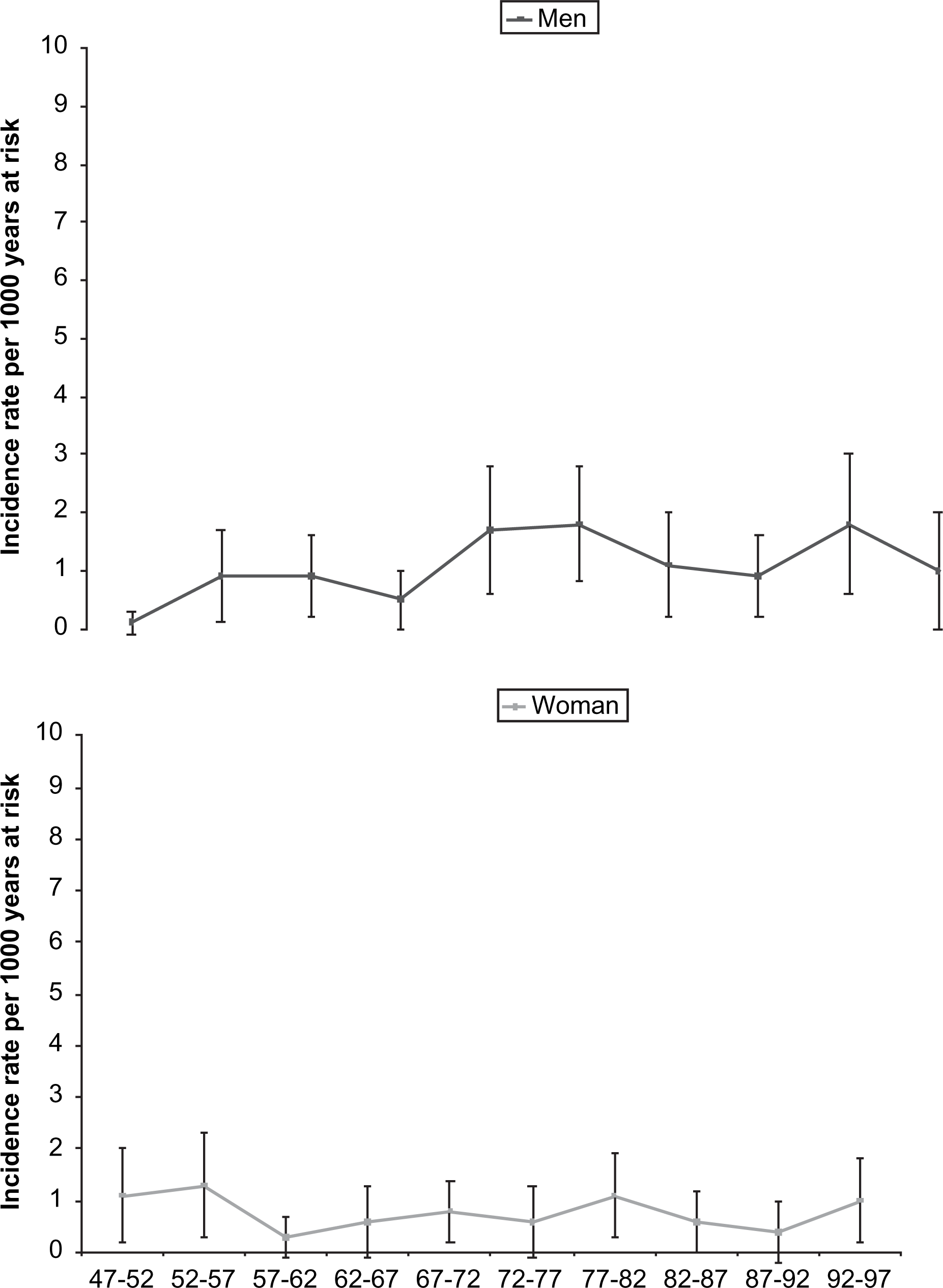
When the caseness threshold for psychotic disorders was raised to impairment level severe, the age-standardized incidences increased from 0.8 per 1000 years at risk in 1947–1972 to 1.4 in 1972–1997 in male, and from 0.6 to 0.7 in female subjects.
There were no psychosis cases with impairment level below medium.
Organic brain disorders
When age- and sex-specific incidences of organic brain disorders in 1947–1972 and 1972–1997 were compared, the rates had decreased consistently from the first to the last period in all age intervals except for 40–49. Both male and female age-standardized first-incidence rates of organic brain disorders decreased significantly from the period 1947–1972 to the period 1972–1997 (see Table 4).
Organic brain disorders of medium+ (GAF 60-1) impairment†
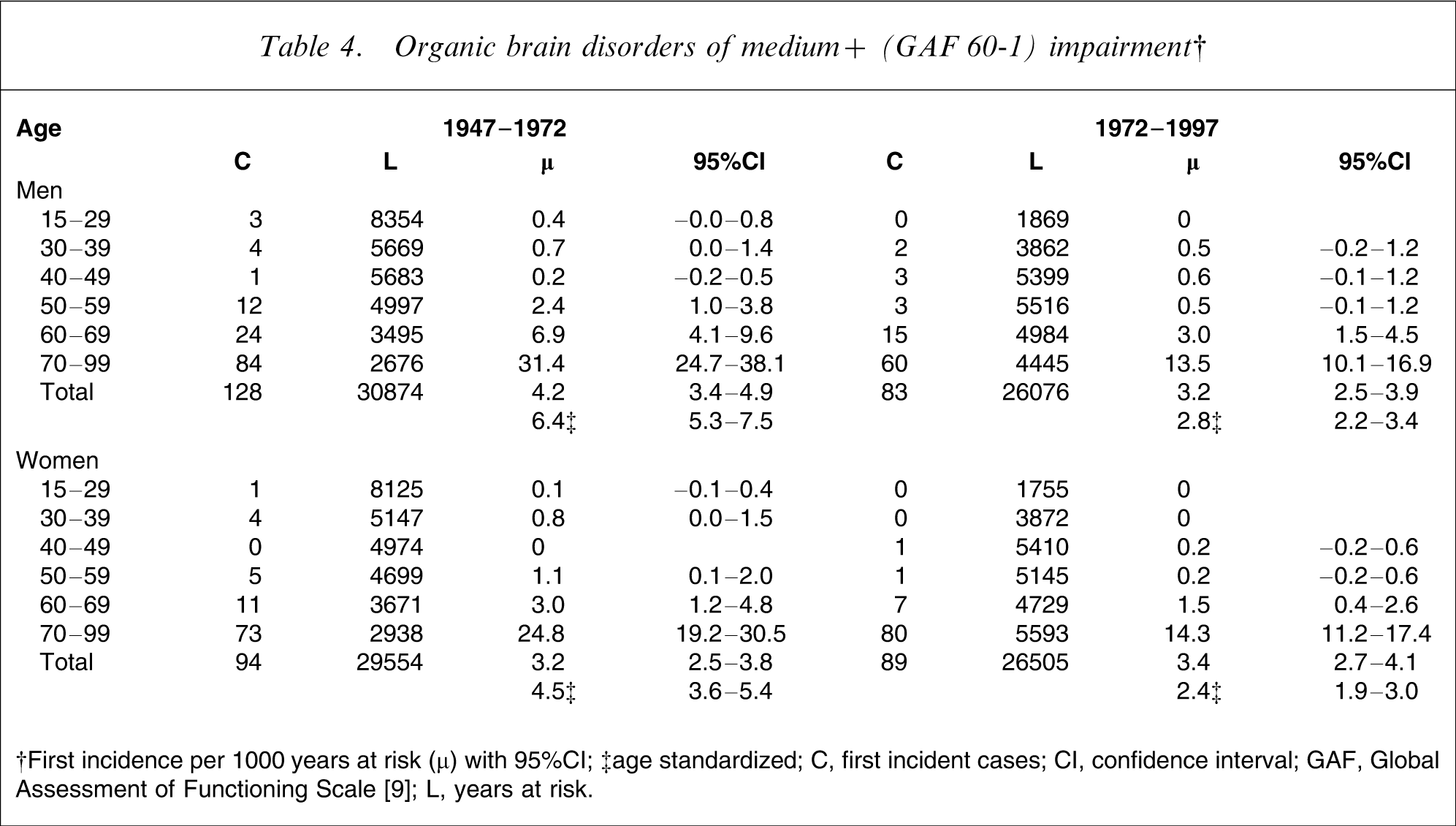
†First incidence per 1000 years at risk (µ) with 95%CI; ‡age standardized
C, first incident cases; CI, confidence interval; GAF, Global Assessment of Functioning Scale [9]; L, years at risk.
When the rate of organic brain disorders in male subjects was compared to the corresponding rate in female subjects for the periods 1947–1972 and 1972–1997 the difference of the age-standardized incidence rates was significant in 1947–1972 but not in 1972–1997.
Male and female age-standardized first-incidence rates of dementia and other organic brain disorders every fifth year from 1947 to 1997 are seen in Figure 3. In both male and female subjects a trend of decreasing incidences of dementia can be observed from 1947 to 1997, while 5 year incidences of other organic brain disorders remain fairly constant through the whole period.
Age-standardised first-incidence of dementia and other organic brain disorders of medium+ (GAF 60-1) impairment by 5 year periods 1947–1997.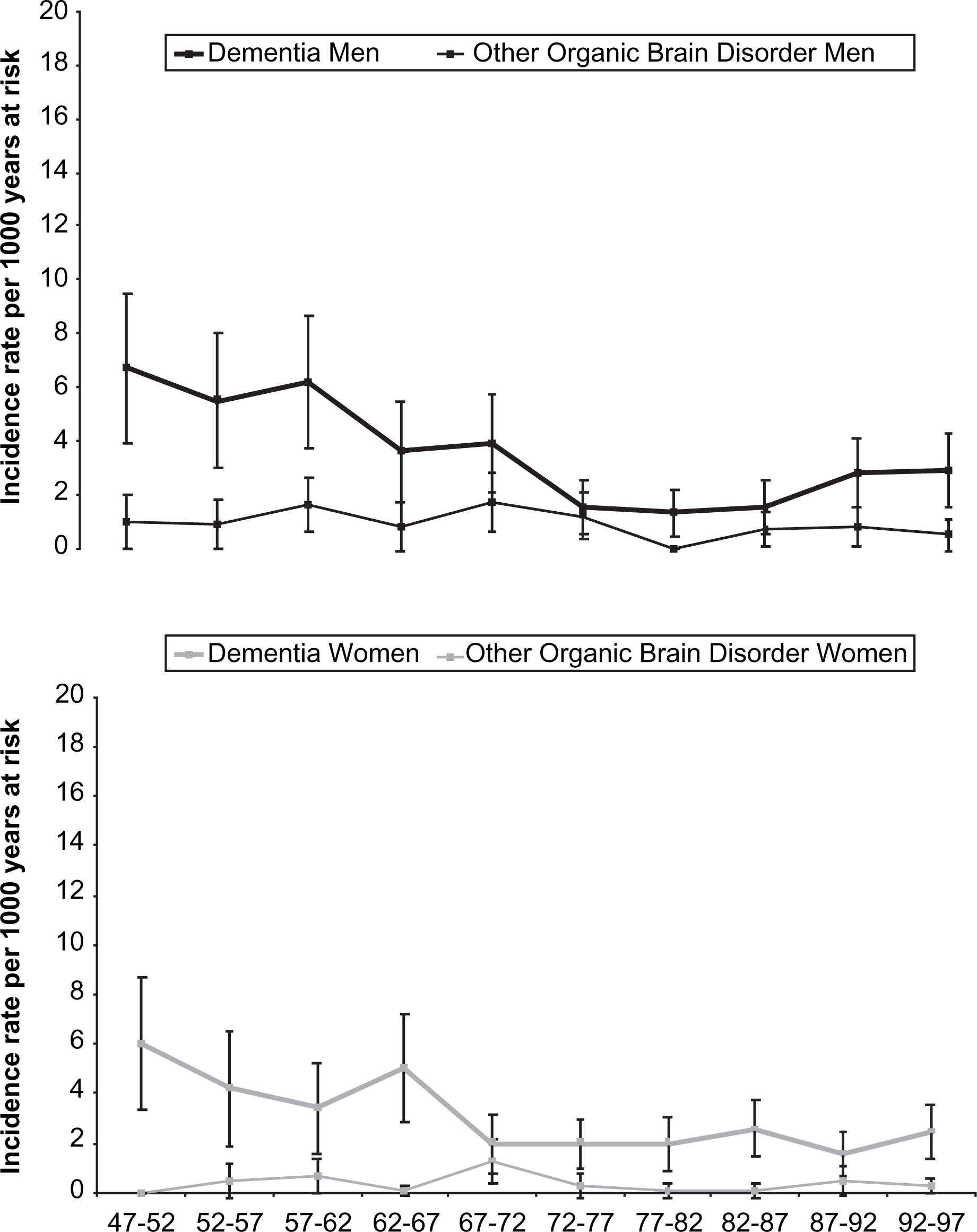
When organic brain disorder was divided into dementia and other organic brain disorders not reaching the dementia threshold one can still, for both groups, see a decrease of the age-standardized first-incidences from the first to the second 25 year investigation period. In male subjects the age-standardized first-incidence of dementia fell from 5.1 to 2.1 per 1000 years at risk and in female subjects from 4.0 to 2.1. The age-standardized first-incidence of organic brain disorders not reaching the dementia threshold fell from 1.2 to 0.6 in male subjects. Among female subjects there was only a marginal decrease from 0.5 to 0.3.
In the period 1947–1972 dementia constituted 80% and milder organic brain disorders 20% of the category organic brain disorders. In the period 1972–1997 the corresponding figures were fairly similar at 83% and 17%, respectively.
The age interval responsible for the reduction of the age-standardized first-incidence of dementia is the 70–99 interval, while the age intervals associated with the reduction of milder organic brain disorders are those under 70. In men aged 70–99 the first-incidence of dementia decreased from 28.4 to 10.9 per 1000 years at risk. In women the first-incidence decreased from 23.4 to 12.6.
When the impairment threshold to indicate caseness was changed from medium to severe, the age-standardized first-incidence per 1000 years at risk of organic brain disorders declined from 3.4 to 2.0 in male subjects and from 2.9 to 2.0 in female subjects from 1947–1972 to 1972–1997.
There were no cases of organic brain disorder with impairment level under medium.
Discussion
Because the Lundby cohort has been followed prospectively it offers an opportunity to study how the rates of mental disorders have changed during a period of great transition in society.
The first-incidence of mental disorder had decreased in both male and female subjects. This trend applied for neurotic and organic brain disorders and was consistent for most age- and sex-specific intervals. The incidence of psychotic disorders seems to have increased in male subjects and decreased in most age intervals in female subjects.
The Lundby Study spans a period when different principles of classification have emerged; this must naturally affect diagnostic patterns [15]. The dramatic fall in the first-incidence of neurotic disorders after 1972, seen in Figure 1, can thus be due to methodological artefacts. However, the fall is not likely to be explained simply by changing diagnostic patterns.
Attrition could lead to lower incidence rates if the probands who dropped out in 1997 represented first-incident cases after 1972. However, again the attrition rates are quite low.
One partial explanation for the decline of the incidence after 1972 could be that some of the neurotic and psychotic episodes registered as first ever episodes between 1947 and 1972 actually were recurrent episodes, because no complete screening was possible before 1947.
Another possible explanation for a decline in incidence of foremost neurotic disorders is that the very long study period between 1972 and 1997 may have given rise to a high degree of recall bias in the probands who were interviewed in 1997. The problem of unreported episodes could be counterbalanced by other sources of information if the proband had been admitted to hospital [5] or if a key informant could give information. However, far from all episodes resulted in hospital care and data from key informants could not be gathered for everyone.
Yet another important difference between the study periods was the number of supplementary sources of information concerning outpatient care and key informants, which in 1947–1972 was greater than in 1972–1997. Also, this would primarily contribute to the difficulties in assessing neurotic episodes after 1972. Furthermore, the impact of this was emphasized by the fact that many probands had died between 1972 and 1997, making follow up in these cases solely dependent on supplementary sources of information. However, approximately 50% of the probands who died between 1972 and 1997 lived in the Lundby area until their death and Lundby area residents were covered by a local primary health care and hospital outpatient register.
The very long timespan of the investigation has also raised difficulties in assessing psychiatric syndromes equally, especially clinical diagnoses, because the way people conceptualize, experience and express symptoms may well have changed with time [16]. New incidence of substance use disorders was not assessed in the present study and a partial explanation to the observed decrease in incidence of neurotic disorders could be that there had been a corresponding increase in substance use disorders. This gains partial support from figures of point prevalence of alcoholism, which showed an increasing trend in women, in whom the point prevalence increased from 0.2% and 0.1% in 1947 and 1957 to 0.7% and 1.8% in 1972 and 1997, respectively. In men, however, the point prevalence remained fairly stable: the corresponding numbers were 10.5%; 7.1%; 13.1%; and 10.2%. There are also problems with interrater reliability affecting the comparability of the estimated rates. However, the interrater data indicate that the field workers in 1997 had a lower threshold for caseness than the previous field workers, which would, contrary to the other biases, counteract a decrease in first-incidence in the second study period.
One further difference between the two 25 year study periods is the extent to which the probands were interviewed within each period. In the first period more probands were alive and, furthermore, the original probands of the 1947 cohort were interviewed both in 1957 and in 1972.
In 1997, the youngest members of the cohort were 40 years old, hence after 1987 there was no one under the age of 30, which may affect the incidence rate estimations in the age group 15–29, an age group in which neurotic and psychotic disorders sometimes emerge for the first time.
Thus, there are several methodological limitations capable of explaining the changes; especially the fall of the neurotic disorders.
In the case of psychotic disorders, the rates are probably not so much attributable to artefacts but the change could, because of few cases, be due to chance. But again, in the neurotic disorders it is certainly possible that a true decrease is mirrored and in the organic brain disorders the changes may be separable from bias because their decrease started before 1972 and cases were not so difficult to track. Furthermore, attrition in probands over 50 was very small.
In formulating hypotheses capable of explaining these changes one may consider several points. Notwithstanding the difficulties separating cohort, age and period effects, a change, with more or less consistently decreasing incidence, within the same sample, from one period to another, would most likely be due to a change of the period-specific environmental exposures acting proximal to illness onset [17].
Factors influencing risk may be socioenvironmental, biological and physical.
The socioenvironmental experience of the Lundby cohort encompasses increased welfare, urbanization and a change of the societal structures, including development of public health care and education, entrance of women into the labour market, birth control, changes in family structure, lessening of the cohesive power of family, church and community and changing roles of men and women. The change of biological and physical factors, subsumed in the socioenvironmental change, include changes of lifestyle (e.g. food, smoking) and increased availability of medical care (e.g. antihypertensive and antidiabetic treatment) as well as changes in the physical environment.
Socioenvironmental dynamics are often suspected to lie behind secular trends [17] and the change of social structures may be capable of explaining the decreasing incidences of neurotic disorders, for instance, through influencing the degree of freedom of choice at different points along the life trajectory. Increased freedom, in this sense, may reduce the risk of becoming depressed, worried or tired.
Modern society has been described as increasingly manifesting a process of individualization within a process of structural societal change in which, for example, former social classes and gender roles start to dissolve [18]. This transformation, also characterized by technological, medical and institutional changes, emphasizes, among other things, individual choice rather than habit and belief in authority in decisions related to health behaviours. Partly through this mechanism, changing health patterns could emerge [19].
The decrease of organic brain disorders may be the result of a healthier lifestyle and availability of treatments protecting against the development of organic brain lesions.
If the present results can be accepted as having internal validity the question arises as to their external validity. Admittedly, the Lundby cohort is an ageing, originally rural, cohort and it contains no immigrants but the findings may still be representative of the effects of the post-war societal changes on the pre-war generations in Sweden (only 228 newborn were included after 1947).
Many studies have shown higher rates of mental disorder in younger age groups than in older, which may be interpreted as a trend of increasing incidence.
In the general population National Comorbidity Survey Replication lifetime prevalence of DSM-IV Composite International Diagnostic Interview (CIDI)-rated anxiety, mood, impulse control and substance use disorders were higher in recent than in earlier cohorts [20].
Klerman et al. reported from the high-risk family Collaboration Program on the Psychobiology of Depression and the ECA community survey that successive birth cohorts showed increasing rates of depression [21, 22].
However, not all studies have shown increasing trends; in a study on longitudinal changes in prevalences of mental disorders in a normal population cohort in Finland it was found that follow ups indicated stability of prevalences, and the youngest cohort did not reach the same prevalence as the cohort 10 years older upon reaching the same age [23].
Also, in the follow up of the Stirling study [11], the prevalence rates of aggregated anxiety and depression disorders remained stable (although there was a higher rate of depression in women born after World War II, which was interpreted as a birth cohort effect).
When comparing the findings from these studies with the Lundby study several differences must be taken into account: the NIMH studies were cross-sectional and used retrospectively assessed lifetime prevalence and their perspective of study was that of birth cohorts [20–22], while in the Lundby study first-incidence rate was assessed prospectively and the perspective of the present paper is to compare periods.
Furthermore, the increasing rates in the NIMH studies were seen mainly in birth cohorts born after World War II [21, 22], whereas the majority of the Lundby sample was born earlier.
It might also be that the cross-sectional findings of higher lifetime risk in recent birth cohorts may be due to undercounting of past depressive episodes [24].
Age-related recall bias in the form of failure to recall lifetime disorders and incorrect recall of age at onset were discussed by Kessler et al. as capable of creating the false appearance of a cohort effect in the National Comorbidity Survey Replication [20].
Diminished recall behind the cohort effect has also been suggested in a simulation study by Giuffra and Risch [25].
Another difference between the cross-sectional studies cited and the Lundby study is that the Lundby data were collected through several sources and that best estimate diagnostic groups and an impairment threshold were used, while in the studies mentioned assessments were dependent on structured interviews and diagnostic algorithms, which may account for broader criteria [22].
Limitations, capable of explaining the cohort effect, mentioned by Klerman, such as selective mortality, greater rates of institutionalization in the older age groups and geographic mobility [21] would not have played such a great role in the Lundby study, and limitations due to memory effects and changes in the meaning and labelling attitudes regarding earlier experiences and behaviours are probably not quite as big a bias problem in the Lundby study due to the prospective design.
One reason for the seeming divergence of the present results from those of the studies in Finland and Stirling county may be that we measured incidence rate, while they measured current prevalence. Furthermore, changes of incidence are not incompatible with stable prevalence. Also, in this case, the diverging results could be due to differences in diagnostics.
There is a limited number of studies that have tried to ascertain secular trends of dementia incidence. In a study from Rochester, Minnesota no significant changes of the incidence of dementia were seen from 1960 to 1984 [26, 27]. One difference from the Lundby study was the length of observation period. A long period of study might be needed to be able to see a slow change over many years [26].
To sum up, after a period with increasing rates (1947–1972) a decrease of neurotic disorders was observed in both male and female subjects in all age intervals, except in men aged 70–99 years (1972–1997). A decrease of organic brain disorders was observed in all age intervals, except 40–49, in both male and female subjects, in particular in the age interval 70–99 years (1947–1997). A possible interpretation is that structural changes, healthier lifestyle and medical advances in society could explain these results.
Footnotes
Acknowledgements
This work was supported by grants from the Swedish Council for Planning and Coordination of Research, Swedish Council for Social Research, Swedish Research Council, Swedish Medical Research Council, Medical Faculty Lund University, Provincial Government of Skåne, Söderström-Königska Foundation, Bror Gadelius Foundation, Gorthon Foundation, Sjöbring Foundation, O.M. Persson Foundation and the Pfannenstill Foundation. The authors wish to acknowledge help from the Steering Committee of the Lundby Study. Povl Munk-Jørgensen has received non-conditioned educational grants from Eli Lily and Wyeth, the latter terminating in spring 2002. He has received minor lecturing honorariums from Lundbeck, GlaxoSmithKline, AstraZeneca and Wyeth. Dinesh Bhugra has received financial support from Jansen-Cilag, Eli-Lilly and AstraZeneca.
