Abstract
Objective:
While people experiencing severe mental illness have a greater prevalence of physical multimorbidity, data on incidence are largely limited to the onset of specific physical conditions. We assessed whether people with severe mental illness have increased incidence rates of physical multimorbidity compared to people with other psychiatric conditions.
Methods:
This retrospective observational cohort study reported on a longitudinal psychiatric inpatient sample (2010–2024) in a metropolitan service in Brisbane, Australia. Within a subgroup of individuals with no pre-existing physical conditions, we compared individuals with and without a history of severe mental illness (schizophrenia-spectrum or bipolar disorder). With a denominator of person-years, we calculated the incidence of different thresholds of physical multimorbidity using adjusted Fine-Gray subdistribution hazard ratios.
Results:
Among the 3310 individuals with severe mental illness, 298 developed physical multimorbidity (two chronic physical conditions) across 21893 person-years, compared to 52 among the 2850 individuals and 18,112 person-years in the comparison group. When adjusted for clinical and demographic covariates, people with severe mental illness had an increased risk of developing one (subdistribution hazard ratio = 3.36; 95% confidence interval = 2.79, 4.03), two (subdistribution hazard ratio = 4.06; 95% confidence interval = 3.02, 5.46), three (subdistribution hazard ratio = 5.36; 95% confidence interval = 3.35, 8.59), and four (subdistribution hazard ratio = 4.84; 95% confidence interval = 2.49, 9.40) chronic physical conditions. Except for malignancy and genitourinary disease, people with severe mental illness had increased incidence of chronic physical conditions in all other organ systems.
Conclusions:
People with severe mental illness experienced greater incidence rates of multimorbidity at various thresholds, with a majority of organ systems affected. This highlights the need for holistic prevention and intervention strategies to curb the accumulation of physical multimorbidity.
Keywords
Background
While the greater prevalence of chronic physical disease is well established in people experiencing severe mental illness, there is less information on the incidence of multimorbidity (Firth et al., 2019; Halstead et al., 2024a). Previous studies have demonstrated the increased incidence of specific physical comorbidities in isolation, however the incidence of multiple chronic physical conditions has not been accounted for (Momen et al., 2020; Scott et al., 2016).
Given the heterogeneity inherent to this issue, physical multimorbidity, defined as the concomitant presence of two or more chronic physical conditions (Halstead et al., 2023), is proposed as a useful construct for measuring these patterns of incidence. Given the young age of onset for both schizophrenia-spectrum disorder (peak: 20.5 years) and bipolar disorder (peak: 19.5 years) (Solmi et al., 2022), we have previously hypothesised that people with severe mental illness develop physical multimorbidity at younger ages (Halstead et al., 2023; Halstead et al., 2024b). However, there is a lack of observational studies that have specifically examined incidence rates (Halstead et al., 2024a).
Hence, we examined the incidence of physical multimorbidity among people with severe mental illness using a longitudinal inpatient psychiatric cohort, and compared these incidence rates to a comparison group comprising individuals without a history of a severe mental illness. In addition to treating physical multimorbidity as a binary outcome variable, we evaluated incidence at increasing thresholds of physical multimorbidity (two, three, or four or more chronic physical conditions). To contextualise these findings, we also measured the incidence of a general single chronic physical condition as a precursor to multimorbidity, as well as the incidence of specific disease in each organ system.
Methods
Methodological reporting
We adhered to the ‘Strengthening the Reporting of Observational Studies in Epidemiology (STROBE)’ guidelines to ensure transparent reporting (Supplemental Appendix Table S.9) (Vandenbroucke et al., 2007).
Study design and sample
This study utilised a retrospective psychiatric inpatient cohort based in Brisbane, Australia, comprising individuals who had been admitted to the adult inpatient psychiatric unit of the Princess Alexandra Hospital (PAH) from January 2010 to March 2024. From this cohort, we identified a subgroup of individuals who had no history of any chronic physical conditions at the time of their entry into the sample (i.e. first admission). Individuals older than 65 years on first admission were excluded as this cohort is managed in a separate psychogeriatric unit.
Data extract
Data for the specified cohort were drawn from the Queensland Hospital Admitted Patient Data Collection (QHAPDC) with the extraction performed by the PAH Decision Support Team (Supplemental Appendix pp. 32–38). In QHAPDC, diagnostic data are coded based on discrete hospital encounters, which in this case comprised psychiatric inpatient admissions at the PAH. As this study was only approved for this setting, the data extract available to us had no diagnostic data from admissions at other hospital sites, or admissions to other departments at the PAH. More details on QHAPDC and the coding procedure that dictates how ICD-10 codes are recorded from associated discharge summaries are available at https://www.health.qld.gov.au/hsu/collections/qhapdc.
Exposure classification
As the exposure of interest was having a severe mental illness, we classified individuals based on recorded International Classification of Diseases, 10th Revision (ICD-10) diagnosis of either schizophrenia-spectrum disorder (F20–F29) or bipolar disorder (F30–F31) (World Health Organization, 2019). Individuals with a recorded history of both disorder types (e.g. listed across primary and secondary/associated diagnoses) were classified as part of the schizophrenia-spectrum disorder group, denoting schizoaffective disorder.
We acknowledge that there is heterogeneity in the literature with respect to the psychiatric diagnoses included under the umbrella term of ‘severe mental illness’, and that other psychiatric conditions can have severe and enduring presentations. We used this pragmatic severe mental illness classification to ensure our approach was clear, reproducible, and compatible with stratification by ICD-10 codes. As a point of reference, the 2019 Lancet Psychiatry Physical Health Commission demarcates schizophrenia and bipolar disorder as comprising ‘severe mental illness’, while anxiety and depression are grouped under ‘common mental disorders’ (Firth et al., 2019). Following this classification, we also ensured compatibility with our previous systematic review (Halstead et al., 2024a) as well as other multimorbidity studies (Launders et al., 2022a, 2022b; Lawrence et al., 2025).
Individuals without a specified history of severe mental illness diagnosis comprised the ‘comparison group’. This comparison group was consequently formed by individuals with history of other psychiatric conditions, such as unipolar depressive disorders, personality disorders, eating disorders, anxiety disorders, and substance use disorders.
Demographic and clinical variables
For both groups, demographic data were obtained for age, sex, country of birth (dichotomised as ‘Australia’ or ‘Overseas’), and preferred language (dichotomised as ‘English’ or ‘Other’), and history of socioeconomic disadvantage (unemployment, low income, or homelessness, as per ICD-10 codes in Supplemental Appendix Table S.1). As sociodemographic factors have been implicated in having a mediating role in multimorbidity development (Lawrence et al., 2025; Reilly et al., 2015), these variables were utilised in subsequent multivariate analyses. All deaths that occurred during the observational period were recorded with corresponding dates of death without information on cause of death. For each admission, all relevant ICD-10 codes listed in each respective discharge summary were obtained. ICD-10 codes for relevant psychiatric and physical conditions, as well as demographic variables, were filtered using an ICD-10 legend (Supplemental Appendix Table S.1).
Classification of chronic physical conditions
We extracted relevant ICD-10 codes for 268 physical conditions that have chronic manifestations (Supplemental Appendix Table S.1), longitudinal health impacts, and/or require long-term treatment (e.g. chronic infections such as HIV). Conditions that are typically acute in nature (e.g. appendicitis) were not measured as contributing to physical multimorbidity. We also organised physical conditions based upon the relevant system into the following 11 groups: cardiovascular, endocrinological, gastrointestinal, genitourinary, haematological, chronic infections, malignancy, musculoskeletal, neurological, respiratory, and a combined ophthalmic and ear, nose, and throat (ENT) group. Congenital conditions (ICD-10 Q00–99) were not evaluated due to being a pre-existing condition. While malignancies and chronic infectious diseases are not anatomically defined to a specific organ system, they were classified into these groups to reflect their common pathophysiology and general categorisation under the ICD-10.
Measurement of incident chronic physical conditions
We measured the incidence of all chronic physical conditions from the time of each individual’s earliest entry into the cohort until the time that diagnosis was first recorded. Having excluded individuals with prevalent physical illness listed on first admission, only newly developed physical conditions were recorded. As individuals entered the cohort at different points, we measured each individual’s person-years in the sample from the time of their initial admission, up until the event (diagnosis), censoring, or the competing event of death.
Evaluation of psychiatric multimorbidity
Distinct from physical multimorbidity, we have previously discussed the analogous concept of psychiatric multimorbidity which characterises the presence of multiple psychiatric conditions (Halstead et al., 2023). Here, we evaluated for the presence of other psychiatric diagnoses also using categorised ICD-10 F codes (Table S.1). To quantitatively estimate psychiatric multimorbidity burden, we measured the number of distinct psychiatric conditions each individual had been diagnosed with.
Conversely, dementia and related syndromes (F00 to F03) and mild cognitive impairment (F06.7) were instead treated as neurological conditions. History of alcohol or other substance use disorders (F10–F16, F18, F19) and history of tobacco use (F17 and relevant Z codes; both codes frequently used to denote smoking) were classified as separate covariates.
Statistical analysis
Statistical analyses were conducted in R (version 4.5.1) and the corresponding packages are detailed in the Supplementary Methods (Supplemental Appendix p. 2). We examined the similarity in baseline demographics between the group with severe mental illness and the comparison group using Wilcoxon rank-sum and chi-square tests as appropriate.
We measured the incidence of physical multimorbidity at the following thresholds (developing one, two, three, or four chronic conditions) with a denominator of person-years, accounting for the time between first admission until event (physical multimorbidity development) or censoring. For each respective multimorbidity threshold, we calculated the incidence rate for both groups individually, facilitating the calculation of the unadjusted incidence rate ratio between groups. We examined the contributing conditions and systems for each respective ordinal physical disease developed.
We then used the Fine-Gray subdistribution hazards regression model to evaluate the incidence of physical multimorbidity with the competing risk of death, while adjusting for other demographic and clinical covariates: age (at time of study entry), sex, country of birth, preferred language, history of socioeconomic disadvantage, history of tobacco use, history of alcohol or other substance use, and psychiatric multimorbidity total. Taking into account the competing risk of death and time to censoring, we also estimated and plotted the cumulative incidence of physical multimorbidity for both groups.
We undertook a series of sensitivity analyses to explore the impact of the aforementioned covariates. We also repeated these sensitivity analyses and specifically examined the incidence of disease in each of the 11 systems. To account for individuals that had greater opportunity for incident physical conditions to have been recorded longitudinally, we also undertook further sensitivity analyses examining (1) individuals that had ⩾2 encounters in the longitudinal sample, and (2) individuals that had ⩽7 versus >7 years of follow-up. Moreover, to examine the impact of the differential readmission rate as a source of ascertainment bias, we also performed a separate series of sensitivity analyses where we adjusted for admission frequency as a time-varying covariate using Cox proportional hazards models (Sensitivity Analyses: Part XV p 343).
Ethics
Data of all individuals were de-identified from the researchers and ethical approval was granted by the Metro South Health Human Research Ethics Committee (reference: HREC/2021/QMS/80978) (Supplemental Appendix pp. 32–38).
Results
Composition of groups
This retrospective cohort study featured 3310 individuals (1364 females, 1942 males, 4 with sex listed as ‘other’) with severe mental illness, 2528 (76.37%) of whom had schizophrenia-spectrum disorder and 782 (23.63%) had bipolar disorder (Table 1). The comparison group featured 2850 individuals (1325 females, 1523 males, 2 with sex listed as ‘other’) without a history of severe mental illness; the following primary diagnoses (Supplemental Appendix Table S.2) were the most commonly listed: affective disorder (33.05%, n = 942), substance use disorder (24.88%, n = 709), anxiety or neurotic disorder (18.63%, n = 531) and personality disorder (13.58%, n = 387). Further demographic and clinical details of both groups are listed in Table 1.
Overview of demographic and clinical covariates for the group with severe mental illness and the comparison group.
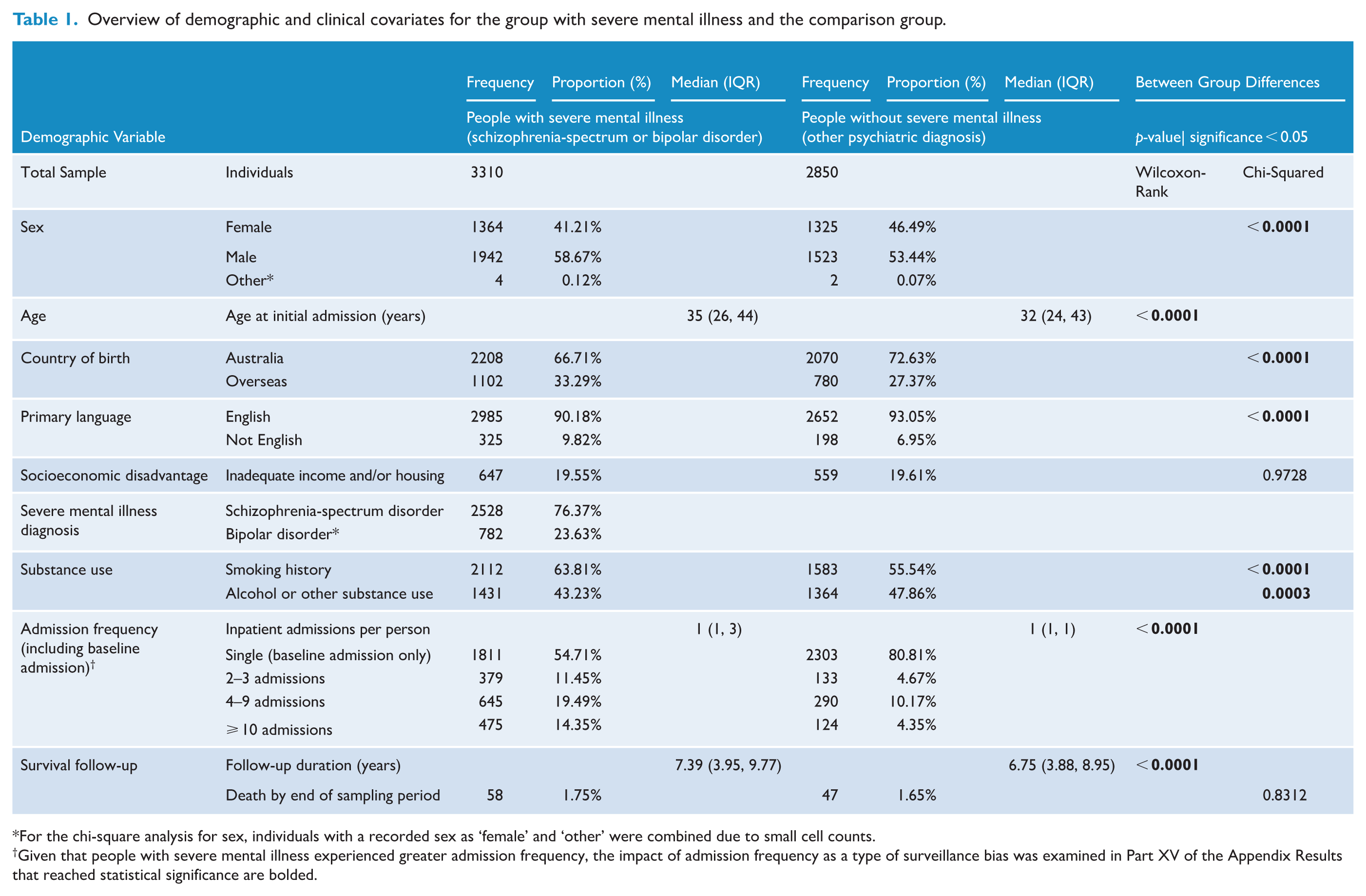
For the chi-square analysis for sex, individuals with a recorded sex as ‘female’ and ‘other’ were combined due to small cell counts.
Given that people with severe mental illness experienced greater admission frequency, the impact of admission frequency as a type of surveillance bias was examined in Part XV of the Appendix Results that reached statistical significance are bolded.
Incidence of at least one chronic physical disease
Measured here as a precursor to physical multimorbidity, people with severe mental illness had an increased incidence of a single chronic physical condition, demonstrated through both the unadjusted incidence rate ratio (IRR) of 3.88 (95% CI = 3.24, 4.65) and the Fine-Gray adjusted subdistribution hazard ratio (SHR) of 3.36 (95% CI = 2.79, 4.03) (Table 2). Several other covariates had a statistically significant impact, namely psychiatric multimorbidity, age, socioeconomic disadvantage, history of smoking, and history of alcohol or other substance use disorder (Table 3). The estimated cumulative incidence of a single physical disease for both groups is depicted in Figure 1A.
Incidence rates of physical multimorbidity at increasing thresholds for people with and without severe mental illness, measured through (i) unadjusted incidence rate ratios and (ii) adjusted subdistribution hazard ratios using Fine-Gray subdistribution hazard regression modelling.
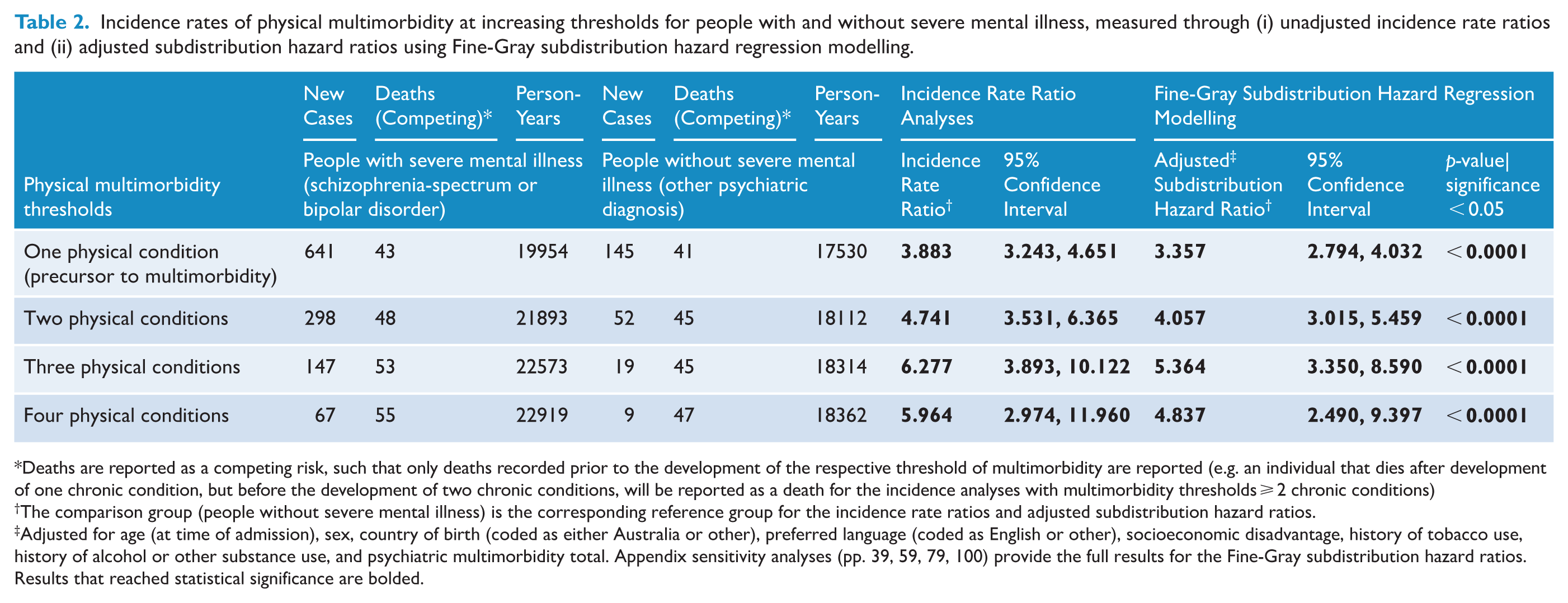
Deaths are reported as a competing risk, such that only deaths recorded prior to the development of the respective threshold of multimorbidity are reported (e.g. an individual that dies after development of one chronic condition, but before the development of two chronic conditions, will be reported as a death for the incidence analyses with multimorbidity thresholds ⩾ 2 chronic conditions)
The comparison group (people without severe mental illness) is the corresponding reference group for the incidence rate ratios and adjusted subdistribution hazard ratios.
Adjusted for age (at time of admission), sex, country of birth (coded as either Australia or other), preferred language (coded as English or other), socioeconomic disadvantage, history of tobacco use, history of alcohol or other substance use, and psychiatric multimorbidity total. Appendix sensitivity analyses (pp. 39, 59, 79, 100) provide the full results for the Fine-Gray subdistribution hazard ratios. Results that reached statistical significance are bolded.
Adjusted multivariate analyses for the subdistribution hazard ratios using Fine-Gray subdistribution hazard regression modelling.
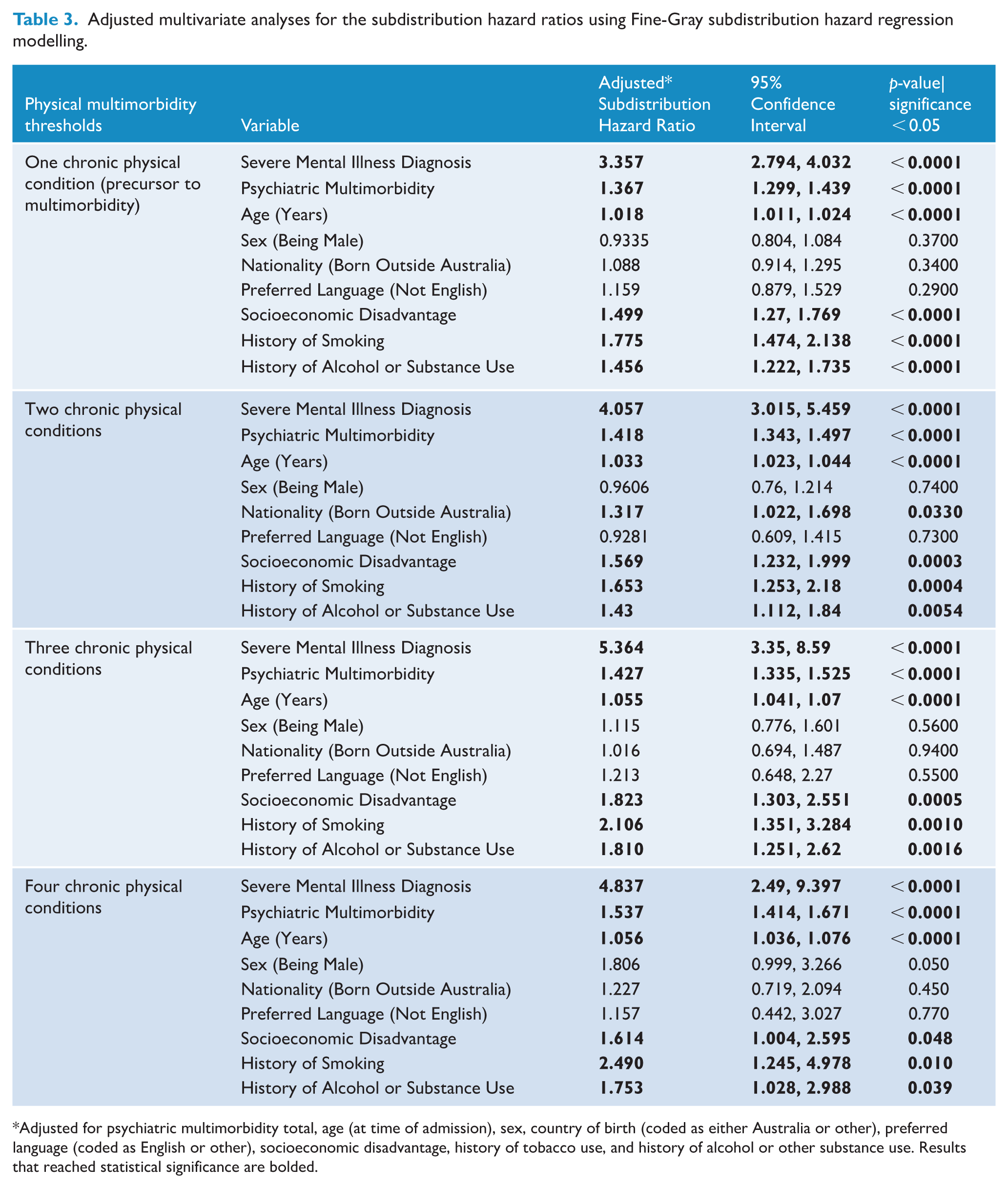
Adjusted for psychiatric multimorbidity total, age (at time of admission), sex, country of birth (coded as either Australia or other), preferred language (coded as English or other), socioeconomic disadvantage, history of tobacco use, and history of alcohol or other substance use. Results that reached statistical significance are bolded.
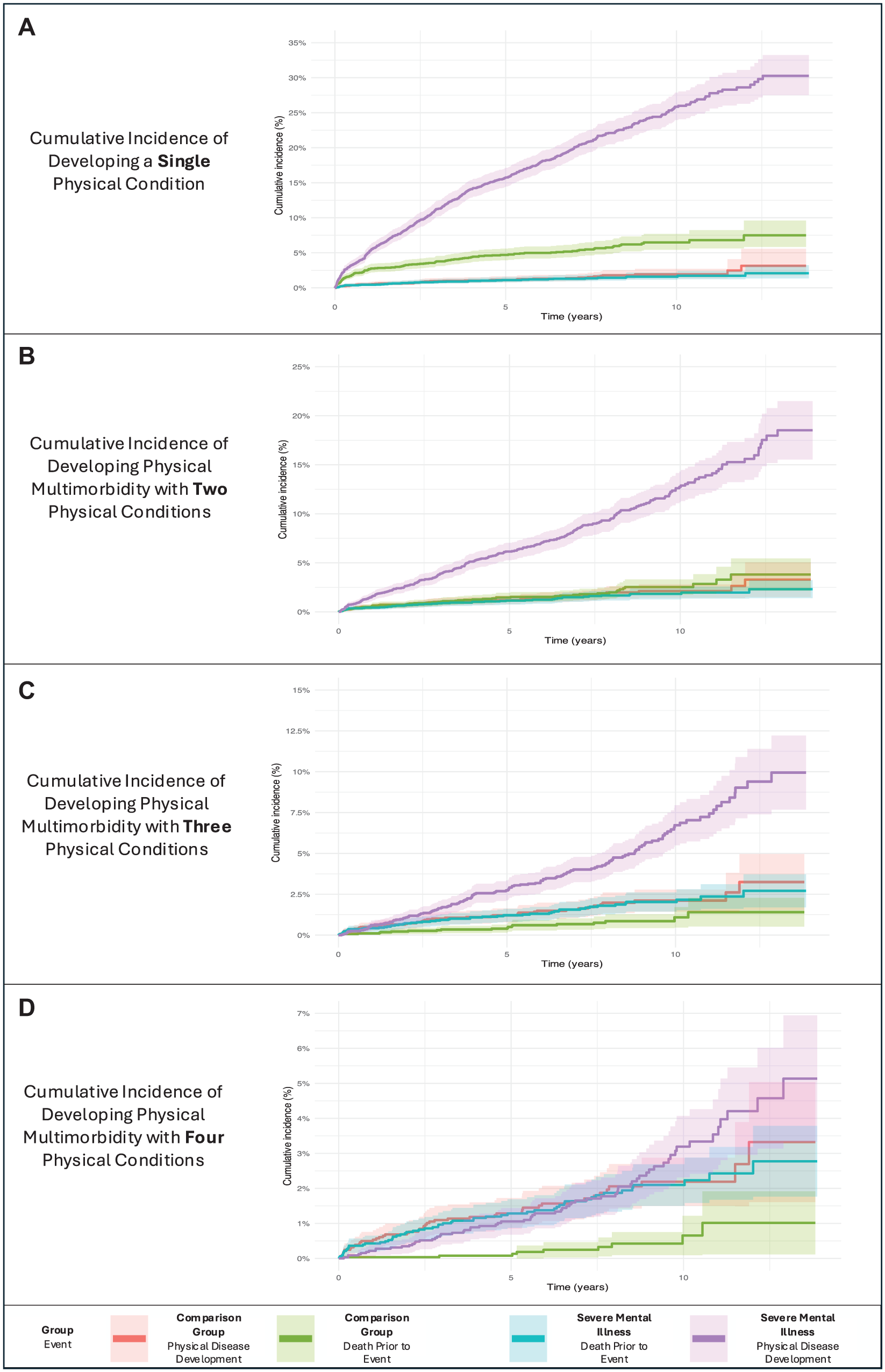
Cumulative incidence of physical multimorbidity at four thresholds (A, B, C, D) for people with and without severe mental illness, accounting for the competing risk of death prior to multimorbidity development.
Incidence of physical multimorbidity with two chronic physical conditions
298 individuals with severe mental illness developed physical multimorbidity with at least two chronic physical conditions, relative to 52 individuals in the comparison, producing an unadjusted IRR of 4.74 (95% CI = 3.53, 6.37) (Table 2). Fine-Gray subdistribution method modelling demonstrated an SHR of 4.06 (95% CI = 3.02, 5.46) after adjustment for covariates. Psychiatric multimorbidity, age, socioeconomic disadvantage, history of smoking, and history of an alcohol or substance use disorder were likewise statistically significant covariates in this adjusted analysis, with having been born outside of Australia also statistically significant (Table 3). Figure 1B provides a graphical representation of the gap in cumulative incidence.
Incidence of higher thresholds of physical multimorbidity
Among those with severe mental illness, 147 and 67 individuals went on to develop a third and fourth chronic physical condition, compared to 19 and 9 individuals among the comparison group, respectively (Table 2). Furthermore, the SHRs for the risk of developing a third and fourth chronic physical condition were 5.36 (95% CI = 3.35, 8.59) and 4.84 (95% CI = 2.49, 9.40) respectively, taking into account the competing risk of death. Age, psychiatric multimorbidity, socioeconomic disadvantage, history of smoking and of alcohol or substance use remained statistically significant covariates for both analyses (Table 3).
Types of conditions developed
Examining the incidence of chronic physical disease across individual organ systems (Table 4), statistically significant adjusted SHRs were demonstrated for developing a cardiovascular, endocrinological, gastrointestinal, haematological, chronic infectious, musculoskeletal, neurological, ophthalmic or ENT, and respiratory disorder. While the unadjusted incidence rate ratio was statistically significant for developing a genitourinary disorder (IRR: 2.73; 95% CI = 1.18, 6.34), the SHR was equivocal (SHR = 2.32; 95% CI = 0.99, 5.44) after adjustment. Malignancy rates were equivocal between groups, noting that there were few cases overall. Full details on the adjusted models are provided in the appendix (Sensitivity Analyses: Parts V to XIV).
Incidence of new chronic physical disease in each respective system for people with and without severe mental illness.
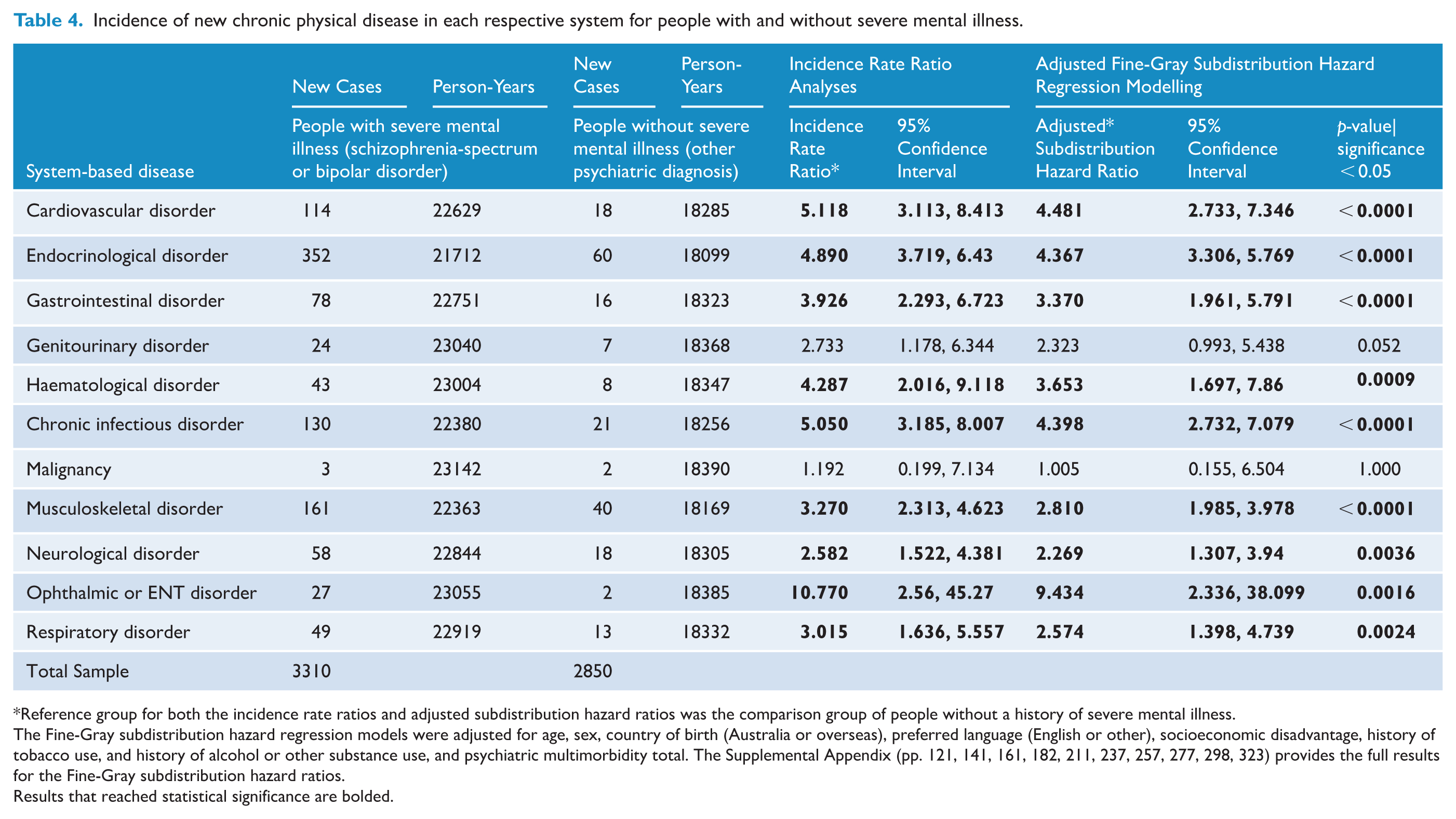
Reference group for both the incidence rate ratios and adjusted subdistribution hazard ratios was the comparison group of people without a history of severe mental illness.
The Fine-Gray subdistribution hazard regression models were adjusted for age, sex, country of birth (Australia or overseas), preferred language (English or other), socioeconomic disadvantage, history of tobacco use, and history of alcohol or other substance use, and psychiatric multimorbidity total. The Supplemental Appendix (pp. 121, 141, 161, 182, 211, 237, 257, 277, 298, 323) provides the full results for the Fine-Gray subdistribution hazard ratios. Results that reached statistical significance are bolded.
137 different types of physical conditions were observed across the two groups. The ranked frequencies of all conditions are listed in the Supplemental Appendix (Table S.4). These are further stratified by their ordinal position in the development of multimorbidity (Supplemental Appendix Tables S.3-S.8).
Sensitivity analyses
Across a majority of the sensitivity analyses, severe mental illness maintained statistical significance as a risk factor for the development of physical multimorbidity (Supplemental Appendix: Sensitivity Analyses: Part I to Part IV). Several other interesting trends could be gleaned for specific covariates. With respect to age, widened gaps in incidence were observed between groups when a younger subset (⩽ 40 years) was examined, with almost doubled IRRs among the younger subgroup compared to the older subgroup analysis (Supplemental Appendix Sensitivity Analyses Part III, pp. 81–82). A similar trend was also observed for the sensitivity analyses that stratified by presence or absence of a history of alcohol or other substance use disorder; widened gaps in incidence rates were again observed for the subgroup with an alcohol and substance use history (Supplemental Appendix pp. 54–55, 74–75, 94–95, 115–116).
After adjusting for admission frequency as its own time-varying covariate in a series of Cox proportional hazards models (Supplemental Appendix Sensitivity Analyses Part XV, pp. 343), a modest reduction in hazard ratio effect size was observed for the various multimorbidity thresholds, with severe mental illness itself retaining statistical significance between adjusted and unadjusted models.
All sensitivity analyses are reported in the Supplemental Appendix (Sensitivity Analyses: Part I to Part XV).
Discussion
There is a paucity of evidence on the incidence patterns of physical multimorbidity for people with severe mental illness. This retrospective cohort study measured the incidence rates of different thresholds of physical multimorbidity over a 14-year period for an inpatient psychiatric cohort. Relative to a mental health comparison group without a history of schizophrenia-spectrum or bipolar disorder, people with severe mental illness had statistically significant greater rates of developing physical multimorbidity at multiple thresholds. Concerningly, the gap in incidence rates between groups appeared wider among a younger subgroup of patients, highlighting that people with severe mental illness are prone to developing chronic physical conditions at younger ages compared to individuals with other psychiatric conditions. These incidence trajectories highlight the need for early and preventive interventions for multimorbidity.
The increased burden of physical multimorbidity among people with severe mental illness appears to be a consequence of increased incidence of various chronic physical conditions across multiple systems. In agreement with our results, a comprehensive Danish registry cohort study demonstrated that people with a schizophrenia or mood disorders diagnosis had a greater risk of developing a circulatory, endocrine, pulmonary, gastrointestinal, urogenital, haematologic disease, or neurologic disease compared to people without a history of mental illness (Momen et al., 2020). The relatively increased incidence rates observed among the younger subgroup in our study echoes the suggestion by Momen et al. (2020) that mental illness may lead to the onset of medical conditions being ‘brought forward’. Notably, our study builds upon previous studies of single comorbidity incidence by demonstrating comparative incidence rates at demarcated thresholds of multimorbidity (Momen et al., 2020; Scott et al., 2016). Previous comorbidity indices, such as the Charlson Comorbidity Index (Charlson et al., 1987) and the Elixhauser Comorbidity Measure (Elixhauser et al., 1998), have also been used to prognosticate mortality risk based upon weighted disease counts (of 17 and 30 disease categories respectively) for the general population. While these indices predominantly feature pertinent chronic physical conditions associated with organ failure and mortality (e.g. congestive heart failure), these indices do not appear to capture the heterogeneity of physical disease that contribute to the health challenges for people with severe mental illness, particularly as conditions such as osteoarthritis and obstructive sleep apnoea are not included. Our study adds insights into the diverse array of chronic physical conditions contributing to physical multimorbidity.
Several key factors appeared to have a synergistic deleterious impact on physical multimorbidity incidence for this cohort. While the link between physical multimorbidity and ageing is already established, both mental illness and socioeconomic deprivation have also been previously highlighted as risk factors for physical multimorbidity (Barnett et al., 2012). In supporting the notion of a synergistic effect for those with severe mental illness, higher rates of various physical comorbidities and multimorbidity have been observed in areas and regions with higher socioeconomic deprivation for people with severe mental illness (Reilly et al., 2015; Rodrigues et al., 2022).
Tobacco, alcohol, and other drugs are established risk factors for a multitude of physical health conditions and physical multimorbidity (Chan et al., 2022; Gomez et al., 2023; Kabashi et al., 2021; Osibogun et al., 2024). Here, in addition to being general risk factors for physical multimorbidity overall, risk appeared to be more pronounced in systems where tobacco and substances have a direct role in disease aetiology, such as respiratory and chronic infectious disease. Psychiatric multimorbidity was also a general risk factor applicable to both groups.
Other factors, both non-modifiable (e.g. genetic factors and developmental exposures) and modifiable (e.g. physical activity and nutrition), that could not be adjusted for in the current study are suspected to also contribute to these trends. While data on psychiatric medication prescription was lacking, it is speculated that the long-term side-effects of psychotropics, such as cardiometabolic disturbance and hyperprolactinaemia from antipsychotics (Halstead et al., 2025), contributed to the gap in multimorbidity incidence between groups, as supported by conditions like type 2 diabetes mellitus and hyperfunction of the pituitary having occurred more frequently in the group with severe mental illness in contrast to the comparison group (Supplemental Appendix Table S.4). Moreover, systemic factors also add to the complexity of how physical diseases are detected, diagnosed, and treated for people with severe mental illness. For example, screening rates of cancers and cardiovascular disease have been found to be inadequate for people with severe mental illness compared to the general population (Jakobs et al., 2020; Kisely et al., 2024; Solmi et al., 2020; Tuesley et al., 2019). Issues such as stigma and diagnostic overshadowing can result in reduced access and poorer quality of healthcare in all settings (David et al., 2013; Giddings, 2013; Jopp and Keys, 2001; Ostrow et al., 2014; Thornicroft, 2008; Thornicroft et al., 2022).
We have previously made a case for models of care that ensure integrated and multidisciplinary treatment of both physical and mental health (Halstead et al., 2023). Given the increased risk among people with severe mental illness of developing physical multimorbidity, prevention and early intervention for chronic physical disease is a must. Monitoring and treatment of psychotropic side-effects is one core area where psychiatrists have an assertive role in addressing physical multimorbidity (Halstead et al., 2025; Siskind et al., 2025). In addition to improving access to screening, there is a desperate need for addressing modifiable risk factors through tailored lifestyle-based innovative interventions targeting physical health which may overcome traditional systemic barriers, such as siloing and stigma in healthcare, and improve accessibility (Arnautovska et al., 2025a, 2025b; Johnston et al., 2025). Importantly, while psychiatrists have a core role in advocacy for people with severe mental illness, psychiatrists and the broader medical community have a shared responsibility together for tackling the significant, multifaceted health inequities faced by this population (Firth et al., 2019; Halstead et al., 2024b; Thornicroft et al., 2022).
Limitations
There are several limitations to acknowledge that may impact the generalisability of the results.
First, while we were able to adjust for socioeconomic disadvantage, we did not have access to other pertinent social and demographic details such as level of educational attainment and marital status. While there is mixed evidence in the literature with regard to the roles of these specific factors as predictors or mediators of multimorbidity among people with severe mental illness, as per a systematic review by Lawrence et al. (2025), general social and economic determinants of health are implicated in the development of multimorbidity involving physical and mental disorders (Kivimäki et al., 2020; Kirkbride et al., 2024). We also did not have data on ethnicity, including Indigenous status, and consequently were only able to examine nationality and preferred language as proxies for demographic and cultural diversity. Previous work has highlighted the differential burden of multimorbidity between ethnic groups generally (Bendayan et al., 2022; Fonseca de Freitas et al., 2022), and specifically the disproportionate multimorbidity experienced by Aboriginal and Torres Strait Islander peoples (Carman et al., 2022). For future population-based studies, it is essential that ethnicity is accounted for when analysing the epidemiology of multimorbidity.
We also did not have access to other clinical variables, such as previous and external healthcare utilisation, exposure to medications, and anthropometrics such as body mass index.
As this was a psychiatric inpatient sample, we did not have access to a separate control group without a history of any psychiatric condition which would have further contextualised these findings. Of the two groups examined here, the severe mental illness group had more frequent admissions, likely owing to a greater need for psychiatric inpatient care, facilitating a greater opportunity for physical diagnoses to have been recorded in this study. We explored this in multiple sensitivity analyses that restricted the sample to only individuals who had ⩾2 inpatient admissions; while the incidence gap between groups was smaller, people with severe mental illness were still at a distinct and statistically significant increased risk of developing physical multimorbidity. The excess incidence of physical multimorbidity in the severe mental illness group remained robust after adjustment for admission frequency as a time-varying covariate.
Because we were not able to take into account physical diagnoses recorded elsewhere (e.g. other hospital admissions and primary care), this study relied on secondary physical diagnoses recorded in discharge summaries from psychiatric inpatient units, and therefore the incidence rates in this setting may not be generalisable to population-based incidence rates. The use of secondary diagnoses also meant that we were not able to account for physical disease severity meaningfully; while it was not available here, data on medical hospitalisation rates for physical conditions would be useful to capture the incidence of discrete ‘severe’ chronic physical conditions requiring hospitalisation. Given this was a psychiatric sample, it is unclear whether some of the physical conditions were due to distinct pathological processes, as opposed to being associated with or exacerbated by psychiatric illness (e.g. spinal pain exacerbated by psychiatric illness with somatic symptoms).
Future research that can utilise larger cohorts with linked diagnostic data from multiple sources (e.g. primary and hospital care) is required to further investigate the trends explored here. Understanding incidence patterns of physical multimorbidity at a population-based level is essential. Such models will facilitate comparative incidence rates against a control population that is more representative of the general population without severe mental illness. Large populations would also facilitate modelling that can account for differing thresholds of physical disease severity (e.g. conditions requiring medical hospitalisation). Clarifying the types, combinations, and clusters of physical conditions that individuals with severe mental illness are at risk of is needed to guide the development of holistic prevention and intervention strategies, informing the development of integrated multidisciplinary healthcare models (Arnautovska et al., 2025b) and tailoring of clinical guidelines relevant to people living with severe mental illness (McCutcheon et al., 2025).
In conclusion, this study demonstrated increased incidence rates of physical multimorbidity in people with severe mental illness relative to a comparison group. Supporting the notion that people with severe mental illness tend to develop multiple chronic physical conditions at younger ages, there is a need for assertive prevention and holistic intervention programmes that are better able to curb the incidence of multimorbidity.
Supplemental Material
sj-pdf-1-anp-10.1177_00048674261426908 – Supplemental material for Comparative incidence rates of physical multimorbidity in a psychiatric inpatient population among people with and without severe mental illness: A retrospective cohort study
Supplemental material, sj-pdf-1-anp-10.1177_00048674261426908 for Comparative incidence rates of physical multimorbidity in a psychiatric inpatient population among people with and without severe mental illness: A retrospective cohort study by Sean Halstead, Urska Arnautovska, Steve Kisely, Dan Siskind and Nicola Warren in Australian & New Zealand Journal of Psychiatry
Footnotes
Acknowledgements
SH is supported by an Australian Research Training Programme scholarship and a Royal Australian and New Zealand College of Psychiatrists Foundation Partners PhD scholarship. DS is supported by an NHMRC Investigator Grant GNT 1194635.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
