Abstract
While the core symptoms of major depressive disorder (MDD) relate to disturbances in mood and affect, there is growing evidence of significant neurocognitive dysfunction [1, 2]. Comparatively few studies have examined the same patient group during an acute episode and then again following treatment response or recovery. Of particular interest is the extent to which neurocognitive deficits persist following symptomatic remission or recovery. The identification of a differential neurocognitive profile between those patients who respond or remit in the short term would be of great clinical utility.
In a recent study, we examined a group of MDD patients who had been medication-free for at least 6 weeks (including n = 26 who were entirely drug-naïve) and matched healthy controls [3]. Patients were predominantly first-episode, non-melancholic outpatients with moderate to severe depressive symptoms. In comparison to controls, patients displayed significant impairment on tests of sustained attention and executive function, visuospatial declarative memory and working memory. However, verbal declarative memory and (psycho)motor functions were unimpaired. Importantly, depression severity was correlated with almost all tests of learning and memory but not with tests of executive function. This profile may suggest that executive functions are more sensitive, being affected adversely irrespective of the severity of depression, while verbal and visuospatial learning and memory are relatively intact in milder depressive episodes but become more pronounced as severity increases. Alternatively, executive deficits could represent a relatively stable trait marker, whereas mnemonic impairment is related to clinical state [3, 4].
The present study examines a subgroup of the original cohort who returned for follow-up assessment. We hypothesized that, first, patients who recovered within the follow-up period would have exhibited less pronounced neurocognitive impairments at index assessment, and second, executive functioning would remain stable between index assessment and follow up, irrespective of symptomatic remission, while declarative learning and memory would improve significantly in patients who remitted.
Methods
Patients aged 18–65 years with a DSM–IV confirmed diagnosis of MDD (single episode or recurrent) were recruited. Patients had been entirely psychotropic medication-free for at least 6 weeks and were excluded if: currently taking other central nervous system-active medication; had a comorbid medical/psychiatric diagnosis or a recent history of alcohol or substance abuse. Symptom severity was assessed using the Montgomery–Asberg Depression Rating Scale (MADRS) [5], the 17-item Hamilton Depression Rating Scale (HDRS-17) [6] and the Beck Depression Inventory (BDI) [7]. DSM-IV criteria for melancholia were noted. The Modified Mini-Mental State Examination [8] was also administered.
The control group consisted of subjects who were psychiatrically and physically fit and who had no recent history of illicit drug use/alcohol abuse. Controls were excluded if they had: a history of psychiatric illness (personally or first-degree relative); BDI score >7; or a current alcohol intake >28 units per week (>21units week−1 for female subjects). For all participants, psychiatric history and diagnosis was confirmed/excluded by semi-structured clinical interview following the DSM–IV criteria.
Patients and controls were matched for age, sex, pre-morbid IQ (National Adult Reading Test (NART)) [9], years of formal education, and season of testing. Female subjects were matched for phase of menstrual cycle (Table 1). All subjects had English as a first language. Subjects were tested as soon after recruitment as possible to minimize delay in treatment and in all cases treatment was commenced within 1 week of the initial assessment. The study was approved by the Newcastle and North Tyneside Local Research Ethics Committee and all subjects gave written informed consent.
Demographic details for patient and control groups
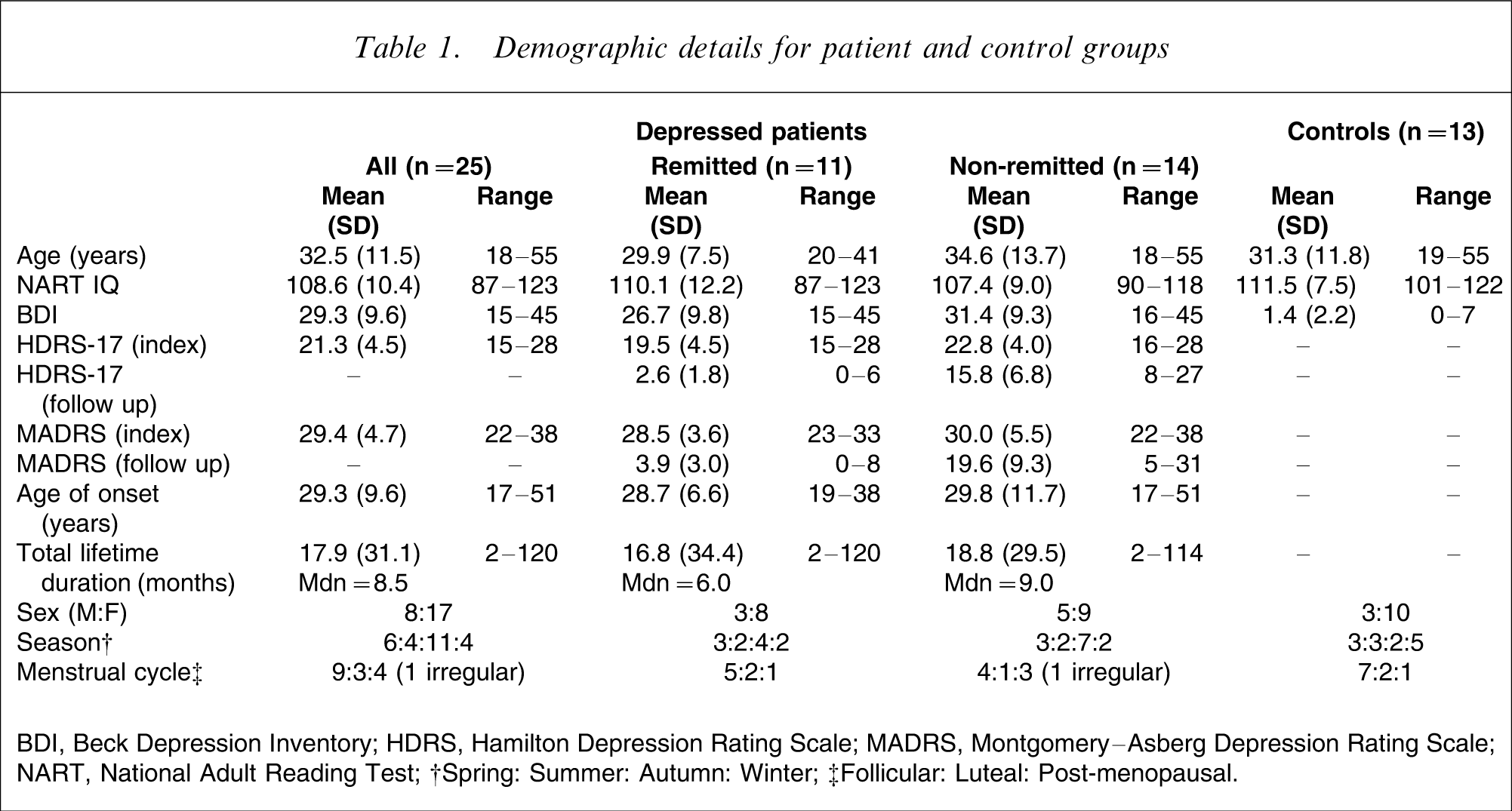
BDI, Beck Depression Inventory; HDRS, Hamilton Depression Rating Scale; MADRS, Montgomery–Asberg Depression Rating Scale; NART, National Adult Reading Test.
†Spring: Summer: Autumn: Winter.
‡Follicular: Luteal: Post-menopausal.
Neurocognitive testing
To avoid the confounding effect of diurnal variation [10], all subjects were tested at 14.00 hours. Pen-and-paper tasks and computerized tests from the Cambridge Neuropsychological Test Automated Battery (CANTAB) were utilized. These tests are summarized here; detailed descriptions are available elsewhere [3].
Psychomotor performance
Digit Symbol Substitution Task
This is a test of psychomotor speed that involves set-shifting and selective/sustained attention [11]. Total number correct in 90 s is recorded.
Learning and memory (verbal)
Rey Auditory Verbal Learning Test
This is a test of verbal learning, including delayed recall and recognition [12]. Number of words correct is recorded and percentage recall for the delayed recall trial (calculated as a percentage of the maximum score from the first five trials).
Learning and memory (visuospatial)
Paired Associates Learning (CANTAB)
Subjects learn and then replicate the matching of complex stimuli to specific spatial locations on the screen. The number of stimulus-location pairs then increases from 3 up to 8. Number of first trials correct is recorded.
Pattern recognition (CANTAB)
Subjects learn a series of 12 complex patterns before being presented with pairs of patterns and are required to identify the familiar one. Two sets are presented. Number correct and response latency are recorded.
Spatial recognition (CANTAB)
Subjects are required to learn the on-screen spatial position of five serially presented squares, with subsequent forced-choice recognition between two locations. Four trials are completed. Number correct and response latency are recorded.
Delayed Matching to Sample (CANTAB)
Subjects must recognize a previously presented stimulus item from among four very similar stimuli after a delay of either 0, 4 or 12 s. Number correct and response latency are recorded at each level.
Sustained attention and executive function
Controlled Oral Word Association Test (Benton's FAS)
This is a verbal fluency test in which subjects generate words beginning with ‘F’, ‘A’ and ‘S’, following a prescribed set of rules [13]. Total number of words correct is recorded.
‘Exclude Letter’ Fluency Test
This is a verbal fluency test in which participants generate words that do not contain the letters ‘E’, ‘A’ or ‘I’ [14]. Total number of words correct is recorded.
Spatial Working Memory (CANTAB)
This is a self-ordered search task in which subjects locate counters hidden in boxes and avoid repetitious searching of locations. Number of errors is recorded and an index of strategy is generated.
Tower of London (CANTAB)
This is a test of planning that taxes central executive function. Subjects rearrange a set of spheres to match a given target arrangement in a specified number of moves. Accuracy and latency are recorded.
Statistical analysis
Demographic data were analysed using independent samples t-test. Neurocognitive test data were converted to standardized z-scores based on the mean and SD of control data at each time point (index assessment or follow up). Composite scores were then derived for each neurocognitive domain: executive function, verbal memory, visuospatial memory, and psychomotor speed (Appendix I). These scores were derived by summing the standardized z-scores within each domain.
The patient group was divided into those who had remitted (HDRS < 8) or had not remitted (HDRS ≥ 8), based on HDRS at follow up [see the ‘asymptomatic’ definition from 15]. The composite scores of remitted and non-remitted patients were then compared: (i) at index assessment; (ii) at follow-up assessment; and (iii) for the change between visits (baseline minus follow up) using independent samples t-tests (directional hypothesis; one-tailed) [16]. Estimates of effect size (with 95% confidence interval (CI)) were calculated using the formula. Statistical analyses were carried out using SPSS version 9 (SPSS, Chicago, IL, USA).
Results
Forty-four patients with MDD were originally recruited and these results are reported elsewhere [3]. Reported here are results of the 25 MDD subjects and 13 controls who returned for follow-up assessment. Comparison of the baseline demographic and illness characteristics of those patients who returned at follow up versus those who did not indicated that there was no significant difference in the age, number of previous episodes, HDRS scores or MADRS scores between groups (p > 0.15 for all).
Three patients fulfilled DSM-IV criteria for melancholia. Eighteen patients were first-episode (72%), with the remaining seven (28%) having had one previous depressive episode. Fifteen patients (60%) were entirely drug-naïve at the time of initial testing; of the 10 who had previously received antidepressant medication, the average (median) duration drug-free was 12 months (range = 6 weeks–7 years). No patient had previously received electroconvulsive therapy.
Follow-up interval and clinical status
All subjects (patients and controls) were followed up 2–6 months after their index assessment (mean = 3.7 months, SD = 0.8). Of the 25 MDD patients who returned at follow up, 11 were defined as remitted and 14 as not remitted. There was no significant difference in the follow-up interval between patients and controls (t = 0.100, df = 36, p = 0.921) or between remitted and non-remitted patients (t = 0.582, df = 23, p = 0.576).
Following index assessment, patients had been treated according to clinical need, predominantly with standard pharmacotherapy. A full medication review was completed at follow-up assessment, documenting pharmacotherapy administered in the intervening period. In the remitted group, nine patients were taking a selective serotonin re-uptake inhibitor (SSRI; fluoxetine, n = 6; paroxetine, n = 2; citalopram, n = 1) while two had declined pharmacological intervention; in the non-remitted group, nine patients were taking an SSRI (fluoxetine, n = 7; citalopram, n = 2), two were taking venlafaxine (one with the addition of trazodone), one was taking prochlorperazine, while two had declined pharmacological intervention.
Demographic and clinical data are presented in Table 1. Sex (χ2=0.202, df = 1, p = 0.653), age (t = 1.080, df = 23, p = 0.293), NART estimated IQ (t = 0.647, df = 23, p = 0.524), severity of depression (HDRS, t = 1.956, df = 23, p = 0.063; MADRS, t = 0.790, df = 21, p = 0.439), age of onset (t = 0.287, df = 23, p = 0.777), lifetime illness duration (t = 0.156, df = 23, p = 0.878), season of testing (χ2=0.465, df = 3, p = 0.927), and for female subjects, menstrual cycle phase (χ2=1.444, df = 2, p = 0.486) did not differ between remitted and non-remitted patient groups. The three patients meeting criteria for melancholia were in the non-remitted group.
Data from 13 control subjects was available for both time points (index assessment and follow up). The control and patient groups were also matched with respect to sex (χ2=0.331, df = 1, p = 0.565), age (t = 0.306, df = 36, p = 0.761), NART estimated IQ (t = 0.915, df = 23, p = 0.366), season of testing (χ2=4.105, df = 3, p = 0.250) and for female subjects, phase of menstrual cycle (χ2=0.914, df = 2, p = 0.633).
Neurocognitive tests: comparison of remitted and non-remitted patients
Analysis of the composite scores for each neurocognitive domain revealed that at the index episode, significantly less psychomotor dysfunction was evident in patients who remitted within the follow-up interval compared to those who did not (Table 2). To examine the effect of depressive symptomatology on this difference, the correlation between HDRS-17 scores and psychomotor domain scores was examined. There was no significant relationship for the patient group as a whole (r = − 0.220, p = 0.290), or for remitted (r = 0.057, p = 0.868) and non-remitted patients (r = − 0.194, p = 0.506) when examined separately.
Composite scores by neurocognitive domain for assessment at baseline and follow up
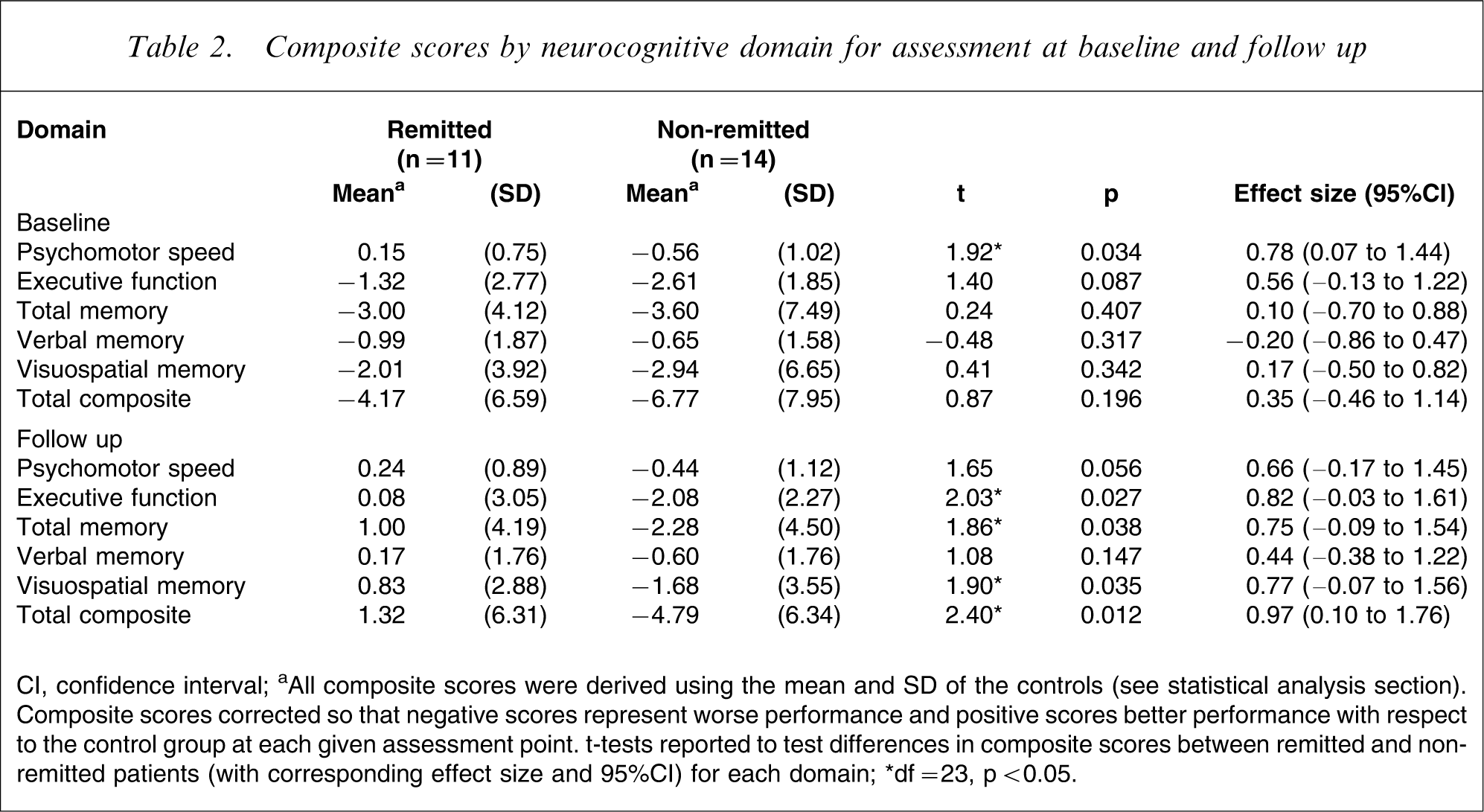
CI, confidence interval.
aAll composite scores were derived using the mean and SD of the controls (see statistical analysis section). Composite scores corrected so that negative scores represent worse performance and positive scores better performance with respect to the control group at each given assessment point. t-tests reported to test differences in composite scores between remitted and non-remitted patients (with corresponding effect size and 95%CI) for each domain.
∗df = 23, p < 0.05.
At follow up, examination of the individual neurocognitive domains showed that non-remitted patients had worse performance in executive functioning, ‘total’ memory, (specifically visuospatial memory) and a marginally significant difference in psychomotor speed (Table 2).
Analysis of the change scores between index and follow-up assessment showed a significantly greater improvement in neurocognitive function in the remitted compared to the non-remitted group (Table 3). Examination of each domain separately indicated that this effect was specifically in verbal memory. Dividing the composite verbal memory measure into its two component parts (total learning and delayed recall; Appendix I) showed that this was the result of a marked difference in the retention index of the test (remitted: mean = 0.859, SD = 0.840; non-remitted: mean = − 0.079, SD = 1.097; t = − 2.343, df = 23, p = 0.014; effect size: d = 0.94) while there was no significant difference between groups in Rey Auditory Verbal Learning Test total trials (remitted: mean = 0.293, SD = 0.632; non-remitted: mean = 0.136, SD = 0.169; t = − 0.575, df = 23, p = 0.286; effect size: d = 0.23).
Composite scores for the change from baseline to follow up within each neurocognitive domain
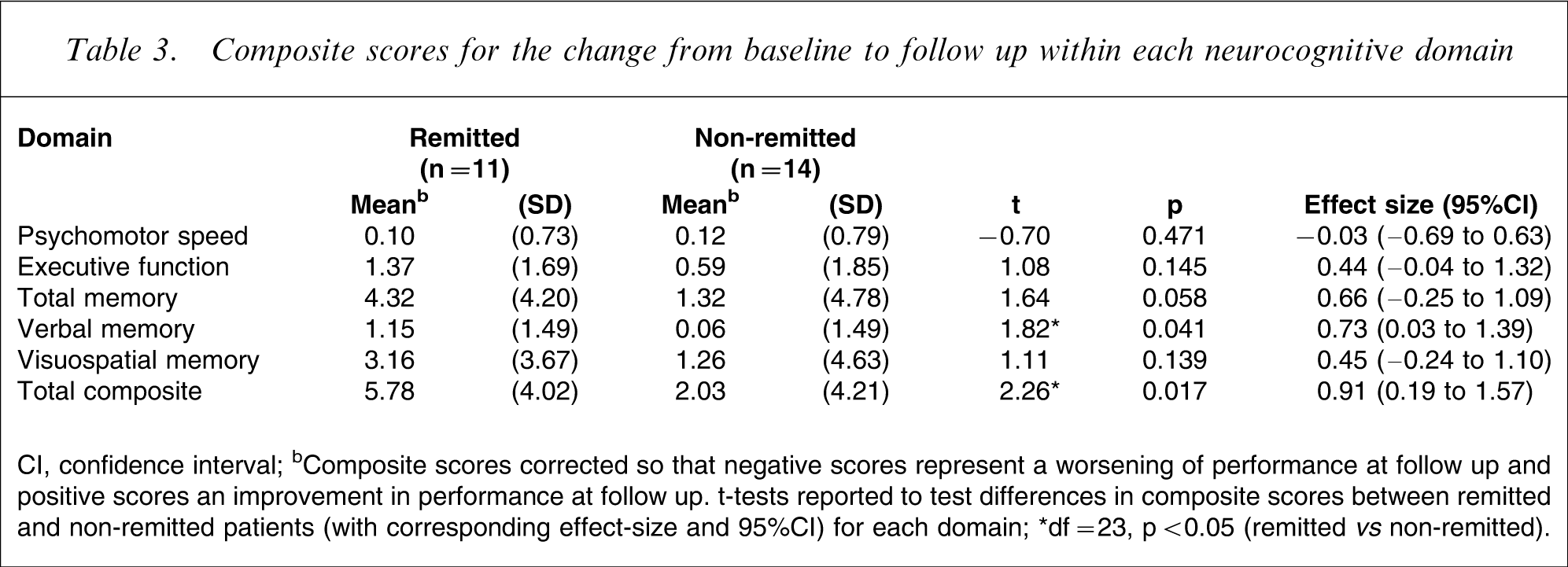
CI, confidence interval.
bComposite scores corrected so that negative scores represent a worsening of performance at follow up and positive scores an improvement in performance at follow up. t-tests reported to test differences in composite scores between remitted and non-remitted patients (with corresponding effect-size and 95%CI) for each domain.
∗df = 23, p < 0.05 (remitted vs non-remitted).
No significant differences in composite change scores for executive function, visuospatial learning and memory, and psychomotor performance were found between groups.
Discussion
The present study demonstrates subtle differences in neurocognitive functioning in adult unipolar depressed outpatients according to the short-term trajectory of their symptomatic improvement. At index episode, less psychomotor dysfunction was evident in those patients who were later found to have remitted within the follow-up period. Examination of composite scores by neurocognitive domain revealed that both groups showed some improvement in functioning at follow up (i.e. the total composite score and all individual domain scores were positive). However, the only significant difference between patient groups indicated a greater improvement in declarative verbal memory in patients who remitted compared to those who did not. This effect was specifically due to improvement in the memory retention index.
Several studies have sought to characterize the persistence of neurocognitive dysfunction in depression through assessment of patients in remission or recovery. In a follow-up study of severely depressed inpatients, Trichard et al. reported that selective executive impairments (Stroop performance) were found to persist, even after clinical remission [17]. More recently, euthymic outpatients with a history of recurrent depression have also been found to exhibit specific executive functioning deficits in the absence of declarative memory impairment [18]. The profile of neuropsychological impairment in patients with a history of recurrent melancholic depression is broader and includes both immediate and delayed visuospatial memory and executive function [19]. Similarly, non-symptomatic patients with a history of chronic depression have been found to exhibit a broad profile of impairment, including domains of immediate memory and attention, executive function and (psycho)motor speed [20]. In elderly patients, several studies have demonstrated the persistence of impairment in recovery, particularly in latency measures and aspects of (psycho)motor function [21, 22]. Interestingly, in the present study of younger outpatients, the only domain in which performance was significantly worse in non-remitting patients at index assessment was psychomotor speed. Although subtle differences between the groups may explain this finding (e.g. the three melancholic patients being in the non-remitting group), it is unlikely that this is wholly responsible. No significant psychomotor differences were observed between the remitted and non-remitted patients at follow up and the change scores similarly did not differ.
The effect of medication is an important issue to consider when evaluating findings of this nature. Recently, Constant et al. examined changes in neurocognitive function over a 7 week, subacute treatment phase with sertraline [23]. In their study, psychomotor slowing and deficits in attention and executive function were observed although these rapidly improved during the first weeks of treatment. This may be attributable to a direct modulatory effect of increased serotonin levels on cognitive function rather than the changes being associated with the improvement in depressive symptoms. The results of the present study control for this in that the medication regimens were similar for both groups and the comparison was made on the basis of symptomatic change and not response to a specific pharmacological intervention. Interest in both these areas – that is, in identifying markers of treatment response and of outcome in general – is increasing. Neuroimaging has been posited as a means of identifying ‘response-specific functional relationships’ that may not be used to identify only treatment responders/non-responders, but also those individuals at risk of relapse and those with a disease vulnerability [24]. Electrophysiological markers have also been examined as markers of response [25] and similarly, neurocognitive performance may also warrant more detailed examination as a behavioural phenotypic marker. It is important for future studies to consider neurocognitive changes at a treatment-specific level, as a potential marker of the underlying mechanisms of action of different classes of treatment.
The issue of which impairments are state markers and which are trait markers continues to be of interest. Weiland-Fiedler et al. administered a similar battery of tests to that used in the present study to drug-free, remitted MDD patients. Deficits were found in aspects of executive function and sustained attention although many of these were not significant when residual symptoms were controlled for statistically [26]. No evidence of verbal or visuospatial learning and memory impairment was found. However, more recently Paelecke-Habermann et al. have reported more widespread attentional and executive impairments in 40 remitted (defined as a period of at least 3 months) MDD patients compared to controls [27]. Taken together, these previous findings and those of the present study would seem to suggest that memory deficits are the most closely linked to depression severity and therefore clinical state. The findings for executive/attentional impairments are more equivocal; while some specific aspects may represent trait markers (although this remains to be established) it is possible that this may be a consequence of such tests being more sensitive and adversely affected even by residual, subsyndromal depression.
Several methodological issues should be highlighted with respect to the present study. First, due to the small sample size, we cannot rule out the possibility that significant effects may have been observed across additional domains with a larger cohort. Some effect sizes were of medium size (by standard criteria) but failed to reach statistical significance and the pattern of deficits/improvement may be less specific than appears here. We therefore included effect size calculations for all measures to inform future research and these findings require replication. Second, the timing of follow-up assessments in studies of this nature is critical. All subjects were tested within 6 months of their index assessment and there was no difference in the interval between testing session between the groups. Nevertheless, the temporal trajectory of neurocognitive improvement following treatment may not be well characterized in a design examining follow-up performance within a cross-sectional time frame. Tracking patients longitudinally and assessing neurocognitive function according to specific symptomatic change (e.g. at the point of 50% reduction in symptom scores and again at remission or recovery) may better determine baseline neurocognitive markers of poor response or persistence of impairment. The present study also focussed on ‘affectively neutral’ neurocognition. Future studies should include measures of affective processing that may be more sensitive [28, 29]. Finally, it is important to be cognizant of the illness history of patients: the majority of those in the present study (72%) were first-episode, drug-naïve. However, as discussed above, the profile of impairment is broader in asymptomatic patients who have a chronic or recurrent illness. Recurrent depression has also been found to be associated with greater hippocampal atrophy and – as a possible consequence – deficits in aspects of verbal declarative memory [30].
In summary, the present study found preliminary evidence for subtle differences in the neurocognitive profile (at index episode) of MDD patients who exhibited short-term remission, prior to the initiation of treatment. Following remission, these patients exhibited better neurocognitive function across a range of domains. Furthermore, the significantly greater improvement in verbal declarative memory suggests that functioning in this domain may be more closely related to clinical state. These and other previous findings indicate that aspects of neurocognitive functioning should be examined further as a means of providing a useful objective marker of response in major depression. The identification of a differential neurocognitive profile between those patients who respond or remit in the short-term would be of great clinical utility and give rise to the potential for specifically tailored treatment.
Footnotes
Acknowledgements
This study was supported by the Stanley Medical Research Institute.
Appendix
Appendix I. Neurocognitive tests included in each domain.
| Executive: | FAS; total correctELFT; total correctSWM; between search errorsTOL; proportion of perfect solutions |
| Verbal Memory: | RAVLT; total trials 1–5RAVLT; trial A7 delayed recall (% retention) |
| Visuospatial Memory: | PAL; first trials correctPRM;% correctSRM;% correct, DMTS;% correct (delayed trials) |
| Total Memory: | Sum of verbal and visuospatial memory |
| Psychomotor speed: | DSST; correct in 90s |
| Total all measures: | Sum of all measures |

DMTS, Delayed Matching to Sample; DSST, Digit Symbol Substitution Test; ELFT, Exclude letter fluency test; FAS, Benton's FAS; PAL, Paired Associates Learning; PRM, Pattern Recognition Memory; RAVLT, Rey Auditory Verbal Learning Test; SRM, Spatial Recognition Memory; SWM, Spatial Working Memory; TOL, Tower of London.
