Abstract
Objective:
Neurocognitive deficits that persist despite antidepressive treatment and affect social and vocational functioning are well documented in major depressive disorder. Cognitive training approaches have proven successful in ameliorating these deficits in other psychiatric groups, but very few studies have been conducted in unipolar depressive patients by now. In contrast to previous studies solely including outpatients, effects of a cognitive remediation intervention on neurocognitive functioning of depressed inpatients were assessed by the present study.
Method:
A randomized controlled trial was carried out with 46 depressed inpatients of a psychiatric hospital. Patients were randomly assigned to either a control group that received standard drug and non-drug (cognitive behavioural, occupational, sports, relaxation and music therapy) antidepressive treatment or a remediation group that additionally received 12 sessions of cognitive training for a total of 4 weeks (three sessions per week). An intent to treat analysis and a last observation carried forward method was used for data analyses.
Results:
Patients of the remediation group demonstrated greater improvements in neurocognitive measures of verbal and nonverbal memory, working memory and executive function (Cohen’s d effect sizes between .52 and .98).
Conclusions:
These results provide preliminary evidence that cognitive remediation interventions can be successfully applied also in psychiatric inpatients experiencing an acute depressive episode.
Introduction
Beyond the symptoms listed in diagnostic manuals, major depressive disorder (MDD) often is associated with cognitive deficits (Bora et al., 2013; Lee et al., 2012). These cognitive deficits seem to be present not only when patients are in a currently depressed, but also in milder form during euthymic remitted state (Rock et al., 2014). Although functional consequences of cognitive deficits have not been studied as extensively as in other mental diseases like schizophrenia or bipolar disorder, several hints could be found that cognitive functioning level is correlated with burden and duration of illness (Elgamal et al., 2010), social and occupational functioning (Fennig et al., 2002; Withall et al., 2009), response to cognitive behavioural therapy (CBT) (Kundermann et al., 2015) and, at least in elderly patients, even with response to antidepressive treatment (Morimoto et al., 2012). Thus, remediation of cognitive deficits could also be a promising treatment target for MDD patients.
While there is strong empirical evidence for schizophrenia patients that cognitive deficits can be ameliorated by cognitive remediation (CR) (Grynszpan et al., 2011; McGurk et al., 2007; Wykes et al., 2011), surprisingly few studies have been conducted in homogeneous samples of patients suffering from unipolar depression yet. To the knowledge of the authors, only three studies have accessed changes in neurocognitive measures following CR in nongeriatric depressive patients by now (Bowie et al., 2013; Elgamal et al., 2007; Naismith et al., 2010; see also Porter et al., 2013 for a thorough review on that topic).
All of these studies solely included outpatients and while positive effects on memory performance could be found throughout and effects on attention in two studies, the participants’ executive function level could not be significantly improved by CR. However, sample sizes were rather small (16–33 participants), and in each two of the three studies, a waitlist instead of a concurrent control group (CG) was included or the allocation to treatment or the CG was not randomized.
Hence, at the moment, nothing is known about whether CR is an effective treatment option for psychiatric inpatients. Particularly, the basic requirements are different for inpatients compared to outpatients, as the former are suffering from more severe symptoms and comparably more cognitive training sessions during a shorter period of time have to be administered.
Therefore, the aim of the present study was to investigate the effects of a CR intervention administered to patients suffering from acute depression during their inpatient stay in addition to standard drug and non-drug antidepressive therapy on memory, executive function and attention performance. In contrast to previous studies, depressed inpatients were included, participants were randomized to CG or experimental group (EG) and the intervention group was compared to a concurrent control instead of a waitlist group.
We hypothesized that CR would lead to greater improvements in memory and attention for the remediation compared to the CG. Specifically, we examined whether effects on executive function could be observed using a motivating game-like computer based remediation approach.
Methods
Participants
In all, 46 inpatients of the hospital for psychiatry and psychotherapy in Bamberg, Germany were included. All of them fulfilled the International Classification of Diseases-10 (ICD-10) as well as the Diagnostic and Statistical Manual of Mental Disorders-IV (DSM-IV) criteria for MDD and were diagnosed based on the Structured Clinical Interview for the DSM-IV (SCID, First et al., 1997) performed by physicians blind to treatment allocation. None of the participants had any previous experience with CR interventions. Exclusion criteria were medical diagnosis associated with neurocognitive impairment, prior or concurrent diagnosis of psychotic symptoms or a bipolar disorder and uncorrectable deficits in vision or hearing that would prevent performing the tests or the cognitive training tasks. After a complete description of the study, written informed consent was obtained from all subjects. The study adhered to the principles of Good Clinical Practice of the International Conference on Harmonization and the Declaration of Helsinki, and was approved by the ethics board of the University of Regensburg, Germany.
Measures
Symptom levels were obtained before and after treatment for all patients using German versions of the second edition of the Beck Depression inventory (BDI, Hautzinger et al., 2009) and the Hamilton Depression Rating Scale (HAMD, Hamilton, 1960).
Cognitive functioning was assessed using the Wisconsin Card Sorting Test (WCST, Young and Freyslinger, 1995), the digit and spatial span forward and backward, visual reproduction I and II (immediate and delayed recall of geometric line drawings), logical memory I and II (immediate and delayed recall of brief stories) subtests of the German version of the Wechsler Memory Scale (WMS-R, Wechsler, 1987), the Trail Making Test (TMT) Part A and B (Reitan, 1958) and the degraded version of the Continuous Performance Test (CPT, participants have to respond to a target blurred digit ‘3’ in a pseudorandomized sequence of blurred digits, Nuechterlein et al., 1983).
Procedure
The 46 participants were randomly assigned to either the EG or CG using a random list created before inclusion of the first participant (external randomization). A sample size of 40 participants was calculated as the minimum number to provide 80% power for reaching an effect size for the intervention of d = .8 or higher at an alpha level of .05.
All participants were voluntary inpatients and were recruited from the psychotherapeutic wards of the hospital, where they received intensive non-drug care, including continuous cognitive behavioural therapy (one to three interventions per week), two relaxation treatment sessions per week, two psychotherapeutic and one music therapy group treatment interventions per week, one partly supervised and one supervised physical training session as well as two occupational therapy sessions per week.
Baseline assessment was performed within the second week after admission for all participants. The EG received three 60-minute training sessions per week for 4 weeks (12 sessions in total) during their stay at the psychiatric ward (12 sessions in total) using the ‘game-like’ cognitive training software X-Cog®, that was explicitly designed to motivate patients as much as possible while ‘playing’ the tasks (for more details see Trapp et al., 2008, 2013). We assumed that especially for acutely depressed patients, CR tasks should be as motivating as possible to overcome avolition, to increase the subjects’ compliance to participate and to avoid unfavourable negative cognitions. Additionally, using a ‘game-like format’ might reduce participants’ fear of not being able to successfully master the tasks. Of the 19, 10 tasks that target visuomotor, memory, executive function and attention skills were selected and administered to the participants. During the processing of the tasks, subjects had to control characters that face several adventurous challenges, such as rescuing a princess which has been captured in a maze, protecting salads from hungry snails etc. Each task can be administered in five different levels of difficulty. At the beginning of the intervention, the lowest level was chosen for each participant. Every time the current level of the task was mastered successfully, this was indicated by the software, and the participants then moved up to the next level of difficulty. In the instructions for the complex attention, (working-) memory and executive functioning (EF) tasks, mainly metacognitive general strategies also applicable to cognitive demands in everyday life were proposed by the software. Additionally, participants were encouraged to apply these strategies and to develop their own when appropriate.
In general within each level, the task’s difficulty was not adjusted depending on the number of errors made by the participants. However, the first two level’s difficulty was set low enough to reduce the amount of errors rather and thus to facilitate ‘progress’ towards higher levels. In our sample, therefore, at least level three was reached by all participants. Even the more challenging tasks were constructed in a way, that correct responses clearly outweighed incorrect responses during the processing of the task. Figure 1 presents screen shots and short descriptions for some selected tasks. In small groups of three patients, the selected tasks were administered by a graduate student in clinical psychology in a quiet room.

Screen shots and descriptions of some selected tasks of the cognitive remediation software.
CG patients did not receive any additional therapeutic interventions other than the usual treatment for depression provided by the psychotherapeutic ward described in detail above. Symptom and neurocognitive measures were obtained before study entry (baseline assessment) and after 4 weeks (follow-up assessment). HAMD ratings were provided by a senior psychiatrist, while the neurocognitive tests were administered by a clinical psychologist, both were blinded to group allocation.
Data analysis
In a first step, t-tests for independent samples and chi-square tests were performed to evaluate differences in CG and EG at baseline. Then, to avoid multiple testing, multivariate analyses of variance (ANOVAs) were computed using treatment group (EG vs. CG) as group factor, ‘time’ (pretest at day 1 vs. posttest at day 28) as repeated measures factor and the following (groups of) dependent variables:
Attention (degraded CPT measures and TMT A processing time);
Executive function (WCST total errors, TMT B and TMT B minus TMT A processing time);
Working memory (WMS digit and spatial span forward and backward, visual reproduction I and logical memory I scores);
Memory (WMS visual reproduction II and logical memory II scores);
Depression (BDI and HAMD scores).
For those multivariate analyses that showed significant (p < .05) interaction effects ‘group x time’ and thus indicated differential effects of the CR intervention, univariate ANOVAs for each dependent variable were performed. Partial eta square estimators of effect size for the interaction effects were converted into Cohen’s d values according to the algorithm described in Cohen (1988). An intent to treat approach was used where missing posttest data were replaced with pretest data.
Results
Participant flow
Figure 2 shows the flow diagram of the number of participants. Follow-up data could be collected for 87% of the CG and for 91% of the EG. Participants of the EG completed 10.76 training sessions on average (minimum: 6, maximum:12, standard deviation [SD]: 1.92). One participant was excluded because an electroconvulsive therapy was initiated, two participants declined to participate in the follow-up tests and two patients were released from the psychiatric ward before follow-up assessments.
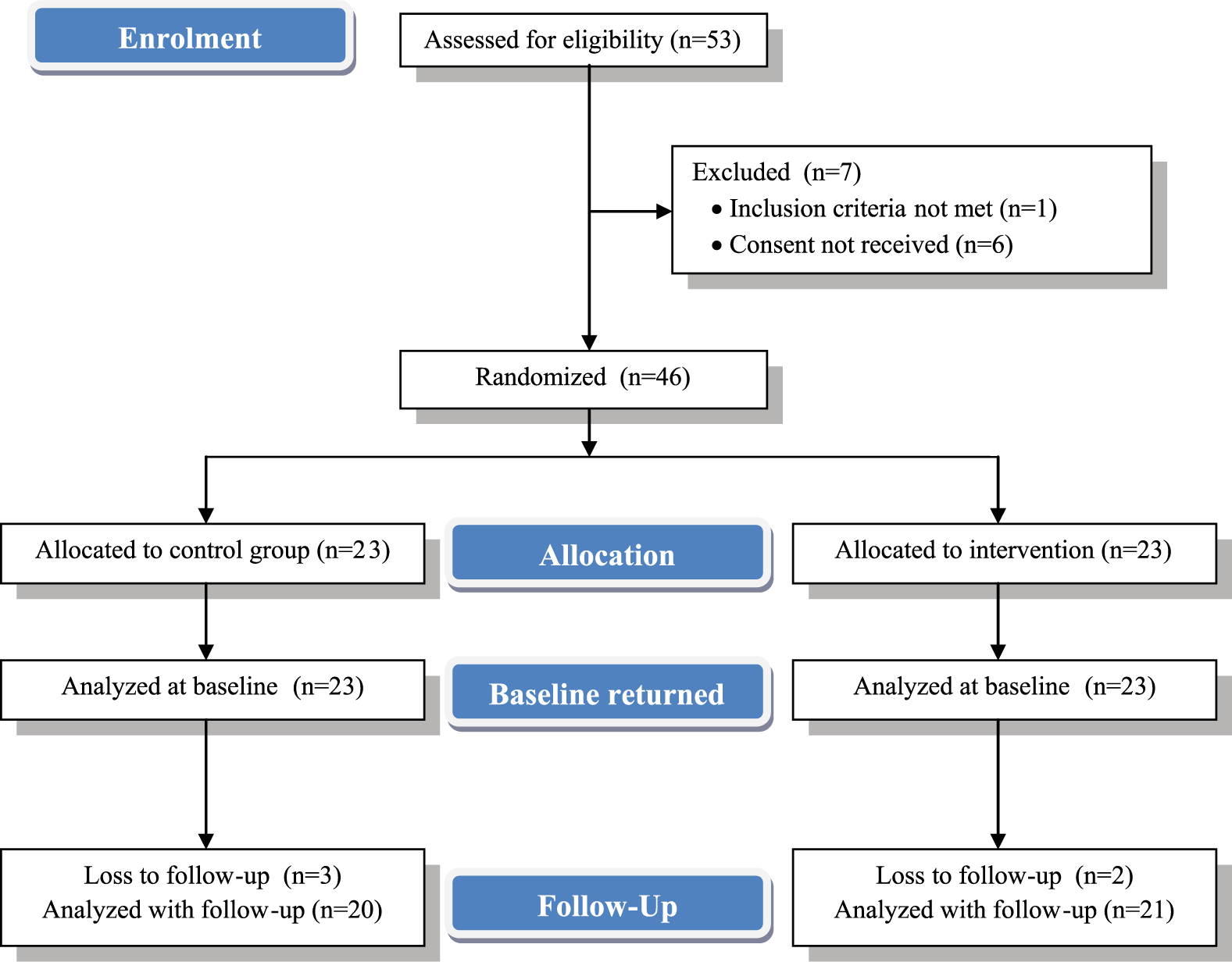
Flow diagram outlining enrolment, allocation, baseline assessment and follow-up of participants.
Baseline data
Table 1 displays clinical and demographic characteristics of all participants.
Demographic and clinical characteristics.
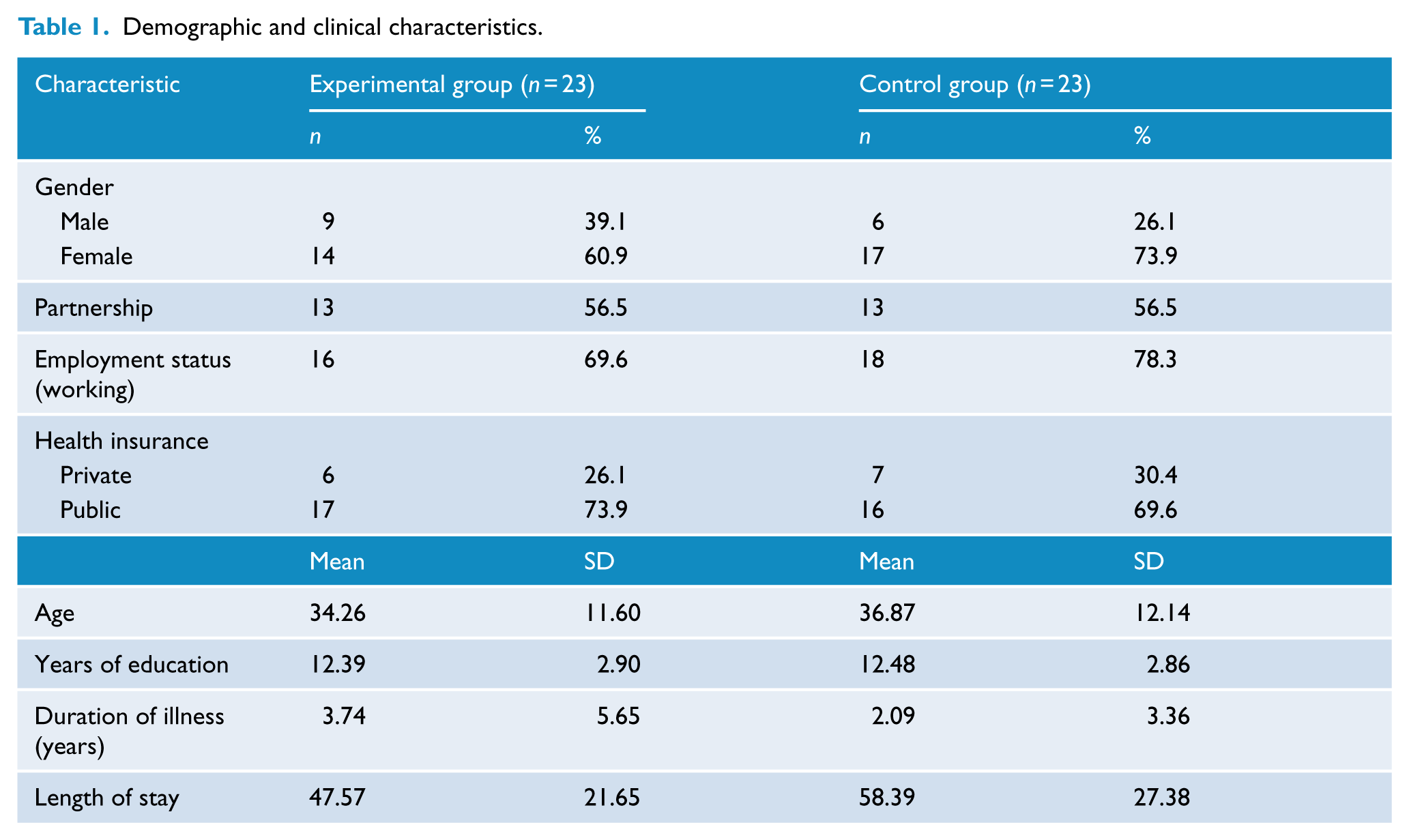
No differences between EG and CG with respect to age, years of education, length of stay and duration of illness, t(44) between .102 and 1.488, p between .919 and .144, as well as concerning male to female ratio, partnership and type of health insurance, χ2(1) between .000 and .890, p between .345 and 1.000, could be found.
Both groups presented with similar depression and neurocognitive scores, t(44) between .075 and 1.648, p between .940 and .106, except for WMS digit span forward scores, t(44) = 1.875, p = .067, and CPT omissions, t(44) = 1.990, p = .053, where EG’s scores are higher compared to the CG (see Table 2).
Mean values and results of uni- and multivariate repeated measures analyses of variance.
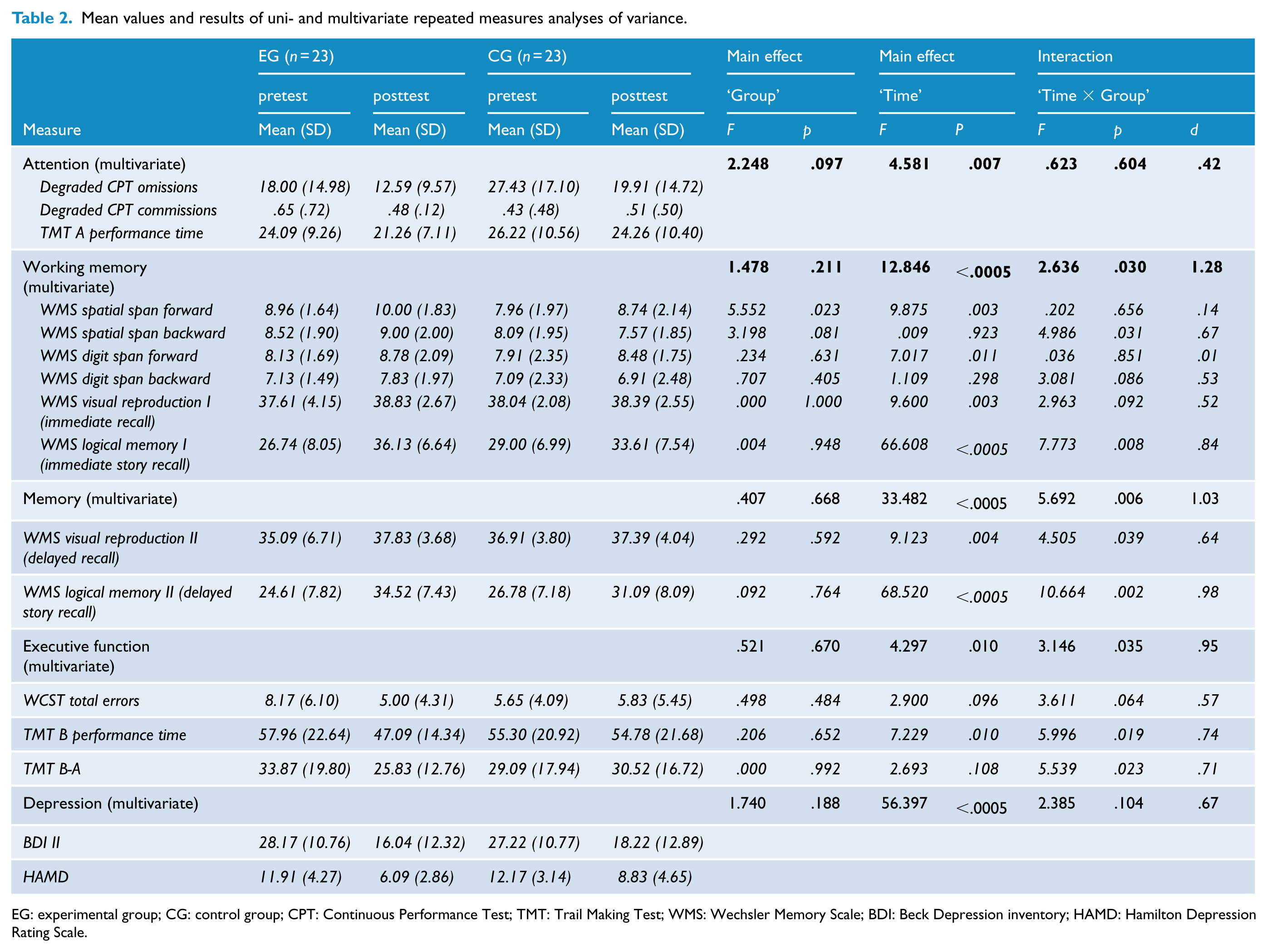
EG: experimental group; CG: control group; CPT: Continuous Performance Test; TMT: Trail Making Test; WMS: Wechsler Memory Scale; BDI: Beck Depression inventory; HAMD: Hamilton Depression Rating Scale.
Effects of the intervention
Results of multivariate and univariate ANOVAs for neurocognitive measures are shown in Table 2. Significant multivariate interaction effects could be found for working memory, memory and executive function measures while no differential changes for EG and CG from baseline to follow-up could be found for attention and depression scores.
Univariate analyses revealed significant interaction effects for WMS spatial span backward, immediate and delayed story recall and delayed recall of nonverbal contents as well as for TMT B and TMT B-A completion time on an α level of <.05 or lower. Additionally for WMS digit span backward, immediate recall of nonverbal contents and WCST errors, trend effects on a p < .10 level were obtained. Inspection of mean values shows larger improvement of performance for EG compared to CG patients throughout. To visualize the relative amount of change in all neurocognitive measures, Figure 3 displays the percentage of change from baseline to follow-up assessment for both groups.
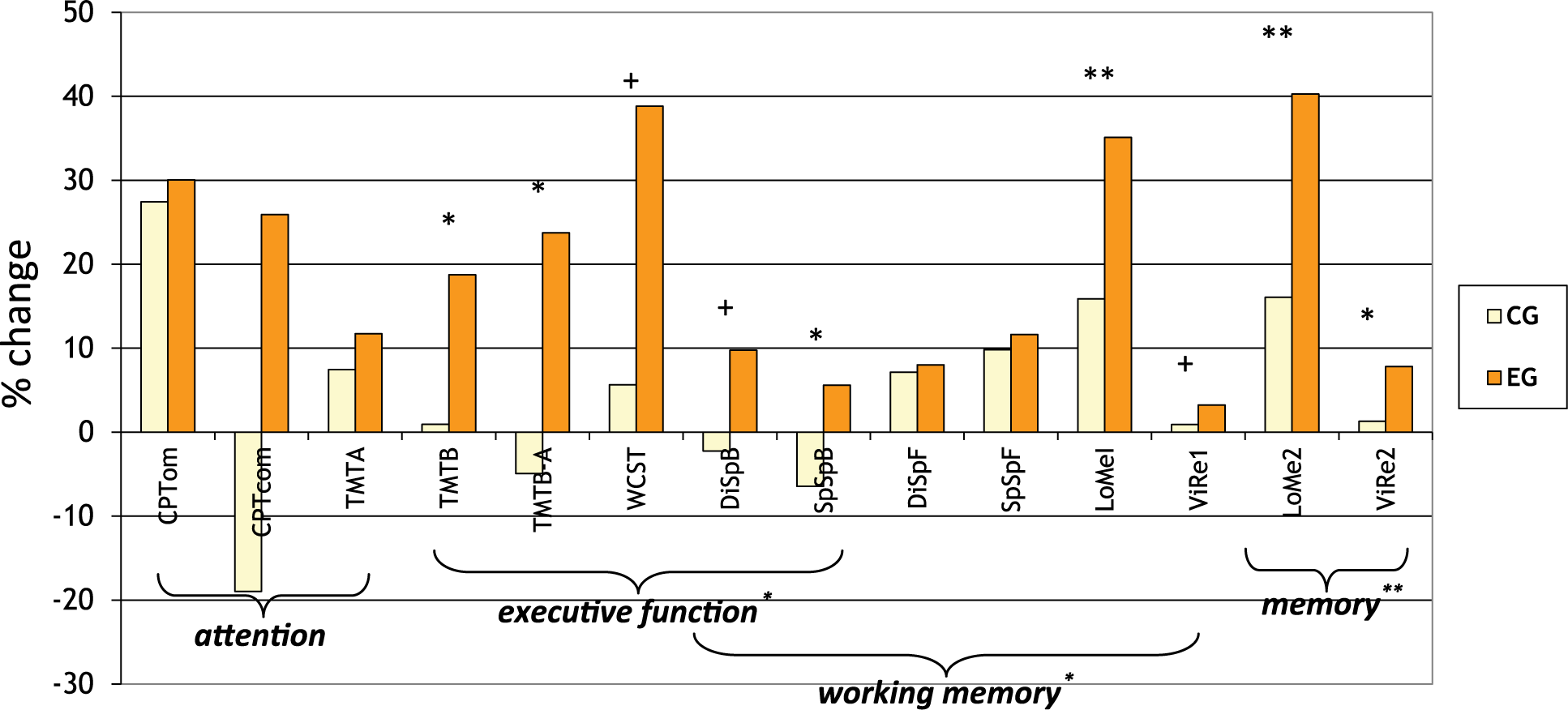
Relative Changes in neuropsychological measures from pre to posttest.
Discussion
In general, our results indicate positive effects of the CR intervention on executive function, working memory and memory of the participants, while no effects on attention could be found. As in all previous studies, also in our sample, large effect sizes indicate that verbal memory performance seems to improve as a result of CR. This is of particular importance, as, for example, structural and functional abnormalities in the hippocampus that have been shown to be related to memory impairments and also to duration of depressive illness have been repeatedly found in MDD subjects (Cole et al., 2011; Hickie et al., 2005; Sheline et al., 2003; Stratmann et al., 2014). CR might therefore counteract maladaptive brain plasticity connected with depressive illness, especially in early stages of depression, when functional abnormalities might outweigh structural changes.
Evidence concerning other memory measures than verbal memory performance is sparse and our results are partly inconsistent with other findings. In our sample, an additional effect also on nonverbal memory could be shown. However, only one of the existing publications (Naismith et al., 2010) included a nonverbal memory measure and the authors could not find any effect.
With respect to working memory measures, only results from one workgroup (Elgamal et al., 2007), reporting that digit span forward performance improved and digit span backward performance did not change following CR are available. In our sample, the finding was contrary: Although a significant multivariate interaction effect for all working memory measures could be found, subsequent univariate analyses showed that this interaction effect was mainly caused by the more complex working memory tests. A huge effect could be discovered for immediate recall of complex stories in the WMS logical memory, whereas a medium effect size for the spatial span backwards and smaller effect sizes for digit span backwards and immediate recall of geometric shapes only reaching a trend level of significance could be found. However, no improvements for the EG relative to the CG resulted for digit and spatial span forward scores, the standard measures for verbal and visuospatial memory.
As higher-level cognitive processes like EF are essential for psychosocial and occupational functioning (Evans et al., 2014), remediation of potential deficits in this domain should be a primary therapeutic target. In depressive patients, structural and functional abnormalities in prefrontal cortical areas are well documented (Levin et al., 2007; Rogers et al., 2004). These changes have been assigned a causal role in neurobiological models of depression (Mayberg, 2003) proposing a cortical limbic dysbalance in the way that that hypoactivity in neocortical regions like the dorsolateral prefrontal cortex and subgenual cingulate cortex hamper regulation of fast autonomous reactions to negative emotional input caused by hyperactivity in limbic and subcortical areas. Executive functioning also relies heavily on prefrontal areas and consequently, results from a recent meta-analysis suggest that MDD patients might be particularly impaired in EF (Snyder, 2013) and that these deficits cannot be explained by impairments in psychomotor speed or other cognitive domains. Unfortunately, however, data from existing studies implies that EF performance is more difficult to improve by CR interventions compared to other cognitive domains. Various tests for EF have been administered in existing studies with the Trail Making Test B provided in every study and no effects could be found for any measure in any of those studies. In contrast to this, the multivariate and all univariate tests of the EF measures in our data point to a large enhancing effect on EF. One putative reason for this might be that the very game-like software used in the present study bears a high potential to intrinsically motivate the participants to work on the CR tasks: As can be taken from the CONSORT diagram in Figure 1 and previous sections of the text, no drop outs because of no further interest in the CR sessions occurred and the majority of our subjects asked for additional sessions after follow-up assessments. As executive function tasks are cognitively most demanding, they might be particular vulnerable to negative self-verbalisations that hamper optimal performance and thus decrease participants’ motivation. Engaging training tasks might therefore be superior to conventional training because they may cause less disadvantageous negative self-verbalisations. Further studies could examine whether extremely game-like remediation tasks have an additional benefit.
CR as a therapeutic option for depression?
Despite ameliorating cognitive deficits CR interventions might also improve depressive symptoms: Beside importance for social and vocational functioning, cognitive deficits might also hamper progress in psychotherapy (Kundermann et al., 2015; Porter et al., 2013) as interpersonal and behavioural interventions rely on complex verbal interactions that require intact working memory, memory and executive function skills. Moreover, neuroplastic changes probably triggered by CR and leading to increased brain activity in areas like the prefrontal cortex – responsible for cognitive deficits as well as for depressive symptoms and known to be hypoactivated in depression – might improve depressive symptoms directly. Additionally, self-competency beliefs might be strengthened by perceived progress in CR tasks which may lead to an increased intrinsic motivation for other therapeutic interventions as well (Medalia and Saperstein, 2011). Very preliminary results from studies – though without adequate CG – indicate that CR might indeed improve depressive mood in currently depressed patients (Papageorgiou and Wells, 2000; Siegle et al., 2007) and that – at least in older subjects – CR might be as effective as antidepressive drug treatment (Morimoto et al., 2014).
The patients of our sample could be categorized as mildly depressed according to their HAMD scores and as moderately depressed according to their BDI scores.
The difference is startling and might partly be due to the fact that the HAMD scale contains less mood and more physical items as well as many items that are poor contributors to the measurement of depression severity (Bagby et al., 2004). Hence, the BDI scores might be better suited to show the ‘true’ severity of depression in our sample.
Unfortunately, none of the three previous studies reported scores from self-rating measures of depression, but the HAMD scores provided by two studies (Elgamal et al., 2007; Naismith et al., 2010) are similar to the scores found in the present sample. In the study of Bowie et al. (2013) including ‘treatment resistant’ patients, the Montgomery–Asberg Depression Rating Scale (MADRS) was used. According to the formula given by Zimmerman et al. (2004), the average MADRS score obtained in this sample corresponds to a HAMD score of 16. Thus, their participants could be classified as ‘mild’ to ‘moderately’ depressed and therefore presumably more severely depressed compared to our subjects.
To date, only one controlled CR study in MDD patients investigated effects of the CR intervention on depressive mood (Elgamal et al., 2007) and – like in our dataset – no advantageous effects could be found. However, former studies solely included either stable remitted, mildly depressed or treatment-resistant outpatients as participants, so maybe improvements in depressive symptom level are harder to achieve for these subpopulations. As inpatients were included in our trial, trivially, our participants experienced a larger reduction of depressive symptoms from baseline to follow-up assessment. In contrast to former studies on outpatients, possible effects of the CR intervention on depressive mood would have been incremental and additional to a broad range of antidepressive treatment options offered to participants during their inpatient stay. Although multivariate tests failed to reach statistical significance, especially the HAMD scores point to stronger improvement of the depressive mood for the EG patients in our data set. Therefore, possible additional effects on depressive mood achieved by our intervention might have been too subtle to reach statistical significance, especially as depression severity of the patients included in our sample is very mild when compared to patients of typical inpatient services in Australia, New Zealand or the United Kingdom. Nonetheless, our subjects were typical for depressive inpatients in major north-west-European psychiatric hospitals, particularly in Germany. Severity of these patients may widely vary from light to severe depression. Referral to inpatient treatment comes from psychiatrists asking for intensive multimodal treatment within time ranges of three to six weeks. Only parts of the patients are referred due to high severity or suicidality. However, for example, the average MADRS score obtained for a sample drawn from a typical New Zealand inpatient unit by Douglas and Porter (2010) and Douglas et al. (2011) who studied neuropsychological functioning and its value for the prediction of response in severe depression corresponds to a HAMD score that is about two times as high than the average HAMD scores in our sample. It is quite possible that antidepressive effects of CR interventions are more likely to occur when samples suffering from higher levels of depression are investigated. Another putative explanation for lacking effects of our intervention on depressive symptoms could be that these effects might be larger when CR is administered in later stages of the treatment process.
Limitations
Our sample size, although larger than in previous studies of CR in depressive patients and considered sufficient to detect medium to large effect sizes, is surely too small to detect small effect sizes, so future studies should use larger samples. Compared to other countries, the inpatient milieu in Germany is somewhat different as inpatient treatment of depression, especially in psychotherapeutic wards, usually is not a treatment of last resort. Additionally, the average length of inpatient stay for unipolar depressed patients is long in Germany compared with other western countries (Hölzel et al., 2011). The average length of stay of 52 days found in our sample is comparable to data reported in relevant publications regarding the treatment of depression in German hospitals for psychiatry and psychotherapy (Härter et al., 2004; Hölzel et al., 2014). Despite the fact that the intervention was administered at the beginning of the inpatient stay, these circumstances have to be considered when comparing our results with findings of future studies investigating effects of CR interventions in depressed inpatients. Even though being a randomized trial, no active placebo treatment was administered to the CG, who instead received treatment as usual. Although, as described in the methods section, both groups received an extensive drug- and non-drug antidepressive therapy, the CG received up to three hours of additional therapy per week. Thus, CG and EG are clearly non-equivalent in terms of time and amount of therapeutic input. However, our finding of a lacking effect on depressive symptoms which are most sensitive to expectancy effects (although it could have been suspected that improvements in cognition might alleviate depressive symptoms), make expectancy effects unlikely. Nevertheless, future studies should include an active CG.
A broader range of tests would have been desirable especially for the executive function domain. Future studies might use single tests for specific EF components like inhibition, shifting, updating and planning. When comparing our results with findings of previous studies, it has to be taken into account not only that all currently published CR studies in MDD patients have been conducted in outpatients, but also that these studies differ substantially from each other with respect to the features of the CR intervention (duration and frequency of training, use vs. no use of additional homework, strategy teaching vs. ‘drill and practice’, progression from basic to more complex tasks vs. complex tasks from the beginning, mild/remitted vs. treatment-resistant MDD patients).
Despite being a pilot trial, long-term effects on cognition were not assessed. No measure of social functioning was included. While a broad neurocognitive test battery was administered and social or occupational functioning was not the primary outcome target, adding at least a brief scale like the Brief Disability Questionnaire (BDQ, Von Korff et al., 1996) would have made little additional effort.
Instead of using the HAMD, it might have been preferable to use scales like the MADRS (Montgomery and Asberg, 1979), which are more sensitive to treatment changes and whose items cover current diagnostic criteria of depression provided by DSM-5 or ICD 10 to a greater extent.
Conclusion
CR may be an efficacious treatment that enhances cognitive achievement level of MDD patients and may also be offered to inpatients suffering from an acute depressive episode. Further studies should consider long-term effects of CR interventions on cognition and investigate whether these interventions might also lead to improvements in everyday functioning.
Footnotes
Acknowledgements
The authors thank Bernd Drescher, Dr. Annika Ehrlich, Dr. Andrea Eujen, Lena Otte and Christoph Ziegelmayer for their help in the recruitment of participants, conducting the symptom assessments and collecting the neurocognitive data, Susanne v. Vietinghoff for proof reading of the manuscript as well as the patients who participated in this study. WHO International Clinical Trials Registry (![]() , trial number: DRKS00006751).
, trial number: DRKS00006751).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
