Abstract
Objective:
Depressed patients display a variety of deficits in neuropsychological function, and contradictory findings in the literature may be due to disorder heterogeneity. The aim of this study was to examine the impact of severity, subtype and symptoms on cognitive control.
Methods:
Neuropsychological function across a range of cognitive control tasks was examined in melancholic (n = 65) and non-melancholic depressed patients (n = 59) relative to controls (n = 124). The relationship between subtype (melancholia vs non-melancholia) and anxiety was also examined.
Results:
Melancholia was characterised by attention and working memory deficits typically associated with the dorsolateral prefrontal cortex, while non-melancholia was characterised by verbal memory recall deficits indicative of left frontal lobe and medial temporal lobe function. The severity of anxious arousal and psychomotor disturbance contributed to cognitive impairment more than the severity of depression symptoms and anxious apprehension.
Conclusions:
Findings highlight a differential impact of depression subtype and severity, and suggest that anxious arousal and psychomotor disturbance may contribute to poorer performance on neuropsychological tasks associated with dorsolateral prefrontal cortex function.
Introduction
Major depressive disorder (MDD) corresponds with dysfunction in controlling and managing cognitive processes. Impaired performance is evident in a range of cognitive control strategies including directing attention, inhibiting behaviour, generating strategies, planning, monitoring ongoing performance and coding representations in working memory (Pizzagalli, 2010). These tasks require higher-level cognitive functions that recruit frontal regions (Stuss and Levine, 2002) and dysfunction in the frontal lobes has been proposed as a biomarker for depression (Pizzagalli, 2010). Neurobiological models have proposed that the depressed state is mediated by reduced dorsolateral prefrontal (cognition) and increased ventrolateral (affect) prefrontal cortical activity (Mayberg, 2003). Indeed, impairments are consistently observed on tasks requiring engagement of the dorsolateral prefrontal cortex (DLPFC) such as attention (e.g. switching attention) and working memory tasks (Austin et al., 1999; Murphy et al., 1999). Despite disturbances in affect, not all patients display impairment in cognitive control. This is reflected in a range of neuropsychological studies, which have failed to produce consistent results (Gorlyn et al., 2006; McClintock et al., 2010; McDermott and Ebmeier, 2009; Murphy et al., 2003; Shenal et al., 2003). These inconsistencies may, in part, relate to a lack of homogeneity in clinical samples. It is possible that cognitive impairment may be specific to more severe depression with biological disturbances such as psychomotor disturbance (Taylor and Fink, 2006) and reductions in heart rate variability (Kemp et al., 2010b; Thayer et al., 2009). Alternatively, the type of cognitive disturbances observed may differ between subtypes of depression, which would support disorder heterogeneity. The current study aims to examine the impact of severity, subtype and symptoms on neuropsychological frontal lobe function in MDD.
Specific neuropsychological deficits have been reported for melancholic patients in comparison to non-melancholic patients on tasks primarily activating the DLPFC (Stuss and Levine, 2002), such as working memory (Austin et al., 1999) and attention shifting (Austin et al., 1999; Murphy et al., 1999), but do not differ on tasks associated with the left frontal lobe (Stuss and Levine, 2002), such as verbal fluency (Austin et al., 1999). This suggests that there may be differences between melancholia and non-melancholia on tasks, which for the most part engage the DLPFC, such as working memory, and attention tasks, such that melancholia exhibits greater dysfunction. Melancholic depression is considered to be distinct from non-melancholia due to the presence of psychomotor disturbance (Parker, 2000; Schrijvers et al., 2008). There is also debate over how other symptoms such as anxiety are related to subtype (Angst et al., 2007; Coryell, 2007; Gold and Chrousos, 2002; Parker, 2000; Parker et al., 1995). Anxiety has been shown to impact upon neuropsychological task performance in depression (Basso et al., 2007). Interestingly, MDD without co-morbid anxiety displays impaired immediate recall of new information but not retrieval or retention of information, whereas MDD with co-morbid anxiety displays impaired recall and recognition memory (immediate and delayed) (Kizilbash et al., 2002) but not working memory deficits (Basso et al., 2007). Verbal memory recall has been associated with the left frontal lobe (Milner, 1974; Perret, 1974; Stuss et al., 1998) and left-sided frontal impairment has been observed in depression (Davidson, 1998; Davidson and Irwin, 1991; Heller, 1993; Heller and Nitschke, 1997; Kemp and Felmingham, 1998; Kemp et al., 2010a; Mathersul et al., 2008). However, increased right frontal activity and reduced right posterior activity has also been found in patients with depression, and this pattern of activity is more prominent in patients with melancholia and anxiety (Kemp et al., 2010a; Pizzagalli et al., 2002).
Increased depression severity is also related to greater impairment of neuropsychological function (Austin et al., 1999). As for co-morbid anxiety, depression severity may also affect cognitive function dependent on which areas of the brain are activated during the task. In a meta-analysis by McDermott and Ebmeier (2009), depression severity was related to dysfunction of episodic memory, executive function and processing speed. However, not all results are consistent; for example, Cataldo et al. (2005) reported an association between depression severity, executive functioning and processing speed, whereas Naismith et al. (2003) reported an association between depression severity, semantic processing and executive functioning, but not processing speed. Furthermore, timed and untimed subsets of cognitive tasks do not differ when it comes to the relationship between depression severity, executive function and processing speed (McDermott and Ebmeier, 2009), indicating processing speed is unaffected by pressure to complete tasks efficiently. Slowed processing speed has been observed as a key indicator of depression, and in particular melancholic depression (Taylor and Fink, 2006). While increasing disorder severity generally involves greater biological disturbance, the different cognitive profiles that have been observed in depression may relate, in part, to distinctive subtypes (i.e. melancholic and non-melancholic depression).
The primary aim for the current study was to examine the impact of depression severity, subtype and symptoms on neuropsychological function. A secondary aim of the study was to examine the relationship between subtype (melancholia vs non-melancholia) and anxiety. It was hypothesised that melancholic patients would exhibit poorer performance on working memory and attention tasks. We hypothesise that melancholic symptoms, anxious arousal and psychomotor disturbance will be primary predictors of neuropsychological performance, and impairment on working memory and attention tasks in particular. Furthermore, we expect a greater association between diagnoses of melancholia and anxiety in contrast to non-melancholia. Consistent with this we expect melancholic patients to exhibit increased anxious arousal in contrast to non-melancholic and control participants.
Method
Participants
A total of 124 participants with a primary diagnosis of MDD and 124 healthy control participants were recruited from the community in collaboration with the Brain Resource International Database (Gordon et al., 2005). All participants gave written informed consent in accordance with National Health and Medical Research Council ethical guidelines. Exclusion criteria included a history of brain injury (causing loss of consciousness for 10 minutes or more), neurological disorder, other serious medical condition, or substance abuse or dependence for greater than 1 year. All participants were medication free for at least five half-lives. All depressed participants met the criteria for MDD as determined by trained and supervised research officers using the Mini International Neuropsychological Interview (MINI) (Sheehan et al., 1998), a structured psychiatric interview based on the Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition (DSM-IV) criteria (APA, 2000). Depressed participants with co-morbid anxiety were included to maximise the generalisability of the sample. All patient participants had a primary diagnosis of MDD, while some had a variety of secondary diagnoses (generalised anxiety disorder (GAD), n = 45; panic disorder, n = 29; and post-traumatic stress disorder (PTSD), n = 14). Healthy control participants were excluded if they self-reported a history or presence of psychiatric illness.
Procedure
Participants were identified as either clinical or control participants on the basis of self-report, which was confirmed by structured interview using the MINI. Depression severity was assessed using the Hamilton Rating Scale for Depression (HRSD) (Hamilton, 1960) and psychomotor disturbance was measured by the CORE Assessment of Psychomotor Change (CORE) (Hickie, 1996). Patients were diagnosed as melancholic (MEL; n = 65) or non-melancholic (NMEL; n = 59) on the basis of the MINI. The self-report Depression, Anxiety and Stress Scales (DASS) (Lovibond and Lovibond, 1995) were completed by participants at completion of the clinical interview. The depression scale measures anhedonia, dysphoria, hopelessness, worthlessness, demotivation and lack of interest or enthusiasm, consistent with DSM-IV criteria of MDD mood disorders. The anxiety (or anxious arousal) scale measures heightened autonomic arousal and subjective experience of anxious affect (including dryness of mouth, difficulty breathing, pounding heart, sweaty palms), consistent with symptoms observed in panic disorder and PTSD. The stress (or anxious apprehension) scale measures nervous tension, difficulty relaxing and irritability, consistent with GAD. (See Mathersul et al. (2008) for supporting evidence to characterise the DASS anxiety and stress scales as anxious arousal and apprehension, respectively.) These scales, in addition to the CORE, were also used to assess diagnostic validity of participants with melancholic depression, which is considered more severe than non-melancholic depression.
All participants subsequently completed the IntegNeuro, a reliable, valid and standardized neuropsychological test battery (Clark et al., 2006; Kemp et al., 2005, 2006, 2009; Paul et al., 2005; Williams et al., 2005). The cognitive tests were administered using pre-recorded task instructions (via headphones). After a computerized visual demonstration, participants completed a practice task prior to commencing the task. Responses were given via a touch screen computer or WAV files for spoken answers. If a participant failed the test trial, the task instructions were repeated and elaborated.
Cognitive measures
Selected tasks chosen for analyses reflect those known to draw upon frontal lobe function (Picton et al., 2006; Pizzagalli, 2010; Stuss and Levine, 2002) and include directing attention, inhibiting behaviour, generating strategies, planning, monitoring ongoing performance and coding representations in working memory. These functions encompassing domains of language, attention, memory and executive functioning were chosen in order to clarify the impact of arousal and psychomotor disturbance on cognitive control. The Spot the Real Word Test was also included to provide an estimate of premorbid intelligence (Yuspeh and Vanderploeg, 2000) to screen for possible outliers, which may confound results. Measures included the following tests.
The Word Generation Test
This test is a form of letter fluency test, which is an adaptation of the Controlled Oral Word Association (COWA) test (Benton and Hamsher, 1989). The letter fluency test involves naming as many words as possible, in the space of a minute, which begin with the letters F, A and S. Participants are instructed not to use proper nouns or make variations on the same word stem. The measure taken is the average number of words produced for the three letters. The test assesses semantic knowledge and language. It engages a cognitive control strategy involving the monitoring of information and is associated with function in the left frontal lobe and the medial frontal regions (Cabeza and Nyberg, 2000).
The Verbal Recall and Recognition Task
This task assesses verbal learning and memory encoding, memory retrieval and recognition, and involves similar processes to that of the California Verbal Learning Test (Tombaugh and Hubley, 2001) and the Rey Auditory Verbal Learning and Memory task (Geffen et al., 1990; Rey, 1964). In this task, participants are presented with a list of 12 words which they are asked to memorize and recall after each presentation. Words are between four and seven characters in length and closely matched on concreteness, number of letters and frequency. After four trials a distracter list is given (trial 5) and a further two trials are completed utilizing the original list. Measures include the total number of words recalled from trials 1 to 4, the number of words recalled from the original list (trials 1–4) in trial 5 (intrusions), and the number of words recalled correctly in trials 6 (immediate recall) and 7 (delayed recall). Trials 6 and 7 were test trials relying on the learning of the original list during trials 1–4. The task engages cognitive control strategies of monitoring and has been associated with the left frontal lobe and medial temporal lobe (Milner, 1974; Perret, 1974; Stuss et al., 1998), except for trial 5 (measuring intrusions), which engages monitoring as well as inhibition and has been associated with the right DLPFC (Stuss et al., 1994).
The Time Estimation Task
This task requires participants to indicate the length of time (in seconds) a black circle takes to change colour (green). The response is indicated via the touch screen providing numbers from 1 through to 12 representing the number of seconds in a sequence across the screen. Twelve trials are completed. A bias in timing is estimated from the absolute value of the average difference between the actual duration of the stimulus and the participant’s estimate weighted by the length of the stimulus. Measures include the bias in time estimation and also the standard deviation of this bias for each participant per trial. This task measures the capacity to assess time and requires the ability to direct attention whilst engaging the DLPFC (Dias et al., 1996; Fletcher et al., 1998; Owen et al., 1993; Petrides et al., 1995; Reukert and Grafman, 1998; Rogers et al., 2000; Wilkins et al., 1987).
The Switching of Attention Task
This test is adapted from the Trail Making Tests (A and B) (Stuss et al., 2001). In Part A, the participant is required to connect 25 randomly spaced circles with numbers inside them in ascending numeric order. In Part B, participants are asked to connect ascending sequences of numbers and letters (i.e. 1-A-2-B) placed in randomly placed circles. Numbers and letters are presented one at a time over a 1-minute interval. The measures in this task are the duration taken to complete part A and part B. This task assesses cognitive flexibility and engages cognitive control strategies including directing attention, monitoring and generating strategies. The task engages the DLPFC (Dias et al., 1996; Fletcher et al., 1998; Owen et al., 1993; Petrides et al., 1995; Reukert and Grafman, 1998; Rogers et al., 2000; Wilkins et al., 2001).
The Verbal Interference Test
This is a computerized modification of the Stroop Test (Stroop, 1935). In Part 1, participants are presented with colour words printed in incongruent ink and are required to nominate the name of the word in the first trial. In Part 2, participants are presented with colour words printed in incongruent ink and are required to nominate the colour of the word in the second trial. Words are presented one at a time over a 1-minute interval. Part 2 of this test assesses the capacity to suppress automatic response tendencies (reading) (Lezak, 1995). The measures include the total score correct in Part 2. The task assesses cognitive flexibility and selective attention. It engages cognitive control strategies, such as inhibiting behaviour and generating strategies, whilst engaging the ventral prefrontal cortex (D’Esposito et al., 2000; Lezak, 1995; Stuss et al., 1983) and the medial frontal regions (Bench et al., 1993; Milner, 1974; Pardo et al., 1990; Stuss et al., 1983; Vendrell et al., 1995).
The Reverse Digit Span Task
This is a common measure of short-term working memory. In this task the participant hears a series of digits (each presented for 500 ms, separated by 1-second intervals). The participant is required to repeat the digits back in the reverse order. The number of digits in the sequence is gradually increased from three to nine, with two repetitions at each level. The measure taken is the total numbers of digits recalled in the correct order. The task engages the cognitive control strategy of monitoring and engages the DLPFC (Bench et al., 1993; Pardo et al., 1990; Perret, 1974; Stuss et al., 1989).
The Span of Visual Memory Task
The span of visual memory task is adapted from the Corsi Blocks task (Milner, 1974) and the Dot Location task (Roth and Crosson, 1985) in order to measure spatial working memory. The task assesses visual memory and new learning. In this task, nine squares on a touch screen light up in a random order. Four seconds later, participants hear a tone which indicates they have to reproduce the sequence by pressing squares on the touch screen. The measure is the total number of correct squares in the sequence without error. The task engages the cognitive control strategies of monitoring and generating strategies whilst engaging the DLPFC (Bench et al., 1993; Pardo et al., 1990; Perret, 1974; Stuss et al., 1989).
The Executive Maze Task
In this task, a dot-based maze is presented on the screen, which assesses executive functioning. The participant is required to find by trial and error a hidden path through the maze and the sequential steps taken to achieve this. The measures taken in this task include the total number of wrong moves made and the time taken to successfully complete the maze two times in a row (trials to completion). The task engages the cognitive control strategies of monitoring, inhibiting behaviour, generating strategies and planning whilst engaging the DLPFC (Bench et al., 1993; Pardo et al., 1990; Perret, 1974; Stuss et al., 1989) and the ventral prefrontal cortex (D’Esposito et al., 2000; Wilkins et al., 1987).
The Spot the Real Word Test
Premorbid intelligence was measured with the Spot the Real Word Test, a computerized adaptation of the Spot-the-Word test (Baddeley et al., 1993). This test assesses premorbid intelligence and involves differentiating between real words and nonsense words. Real word and nonsense word pairs are presented on the touch screen and the participant has to specify which one is the ‘real’ word by pressing the touch screen. The measure is the total number of correct responses.
Statistical analyses
All statistical analyses were performed using SPSS (version 17; SPSS Inc., Chicago, IL, USA). Significant effects were set at p < 0.05. Participants were excluded from the study at the point of statistical analysis if they were identified as an outlier on the premorbid intelligence test as determined by 1.5 times the interquartile range (Tukey, 1977) of all participants. A chi-squared analysis was conducted to examine differences between patients diagnosed with/without melancholia and with/without a comorbid anxiety disorder using diagnostic data from the MINI. A series of one-way ANOVAs were conducted to examine group differences for subtype across age, education, clinical and neuropsychological measures. Post hoc tests were conducted using Tukey’s (Honestly Significant Difference) HSD to adjust for multiple comparisons. A series of backward stepwise linear regressions were also conducted on clinical participants to examine whether neuropsychological function could be predicted by depression (DASS-D), anxious arousal (DASS-A), anxious apprehension (DASS-S), and psychomotor disturbance (CORE) after controlling for age, sex, IQ and years of education (i.e. DV = b0 + b1(DASS-D)1 + b2(DASS-A)2 + b3(DASS-S)3 + b4(CORE)4 + b5(age)5 + b6(sex)6 + b7(IQ)7 + b8(education)8).
Results
Participant characteristics
There were no significant differences for groups on sex (χ2 (2, n = 248) = 1.036, p = 0.596), age (F(2, 244) = 1.079, p = 0.341), years of education (F(2, 244) = 1.622, p = 0.200), or premorbid intelligence (F(2, 244) = 1.864, p = 0.167). As expected, significant differences were observed between groups (MEL; NMEL; controls, CTL) on the HRSD (F(2, 245) = 1072.345, p < 0.001), DASS-D (F(2, 245) = 517.950, p < 0.001), DASS-A (F(2, 245) = 100.533, p < 0.001), DASS-S (F(2, 245) = 256.887, p < 0.001) and the CORE total score (F(2, 245) = 96.154, p < 0.001), such that patients with melancholia displayed increased scores on all measures than those with non-melancholia and controls, respectively (see Table 1 for means and standard deviations of demographic and clinical data).
Means and standard deviations per group on demographic and clinical measures.
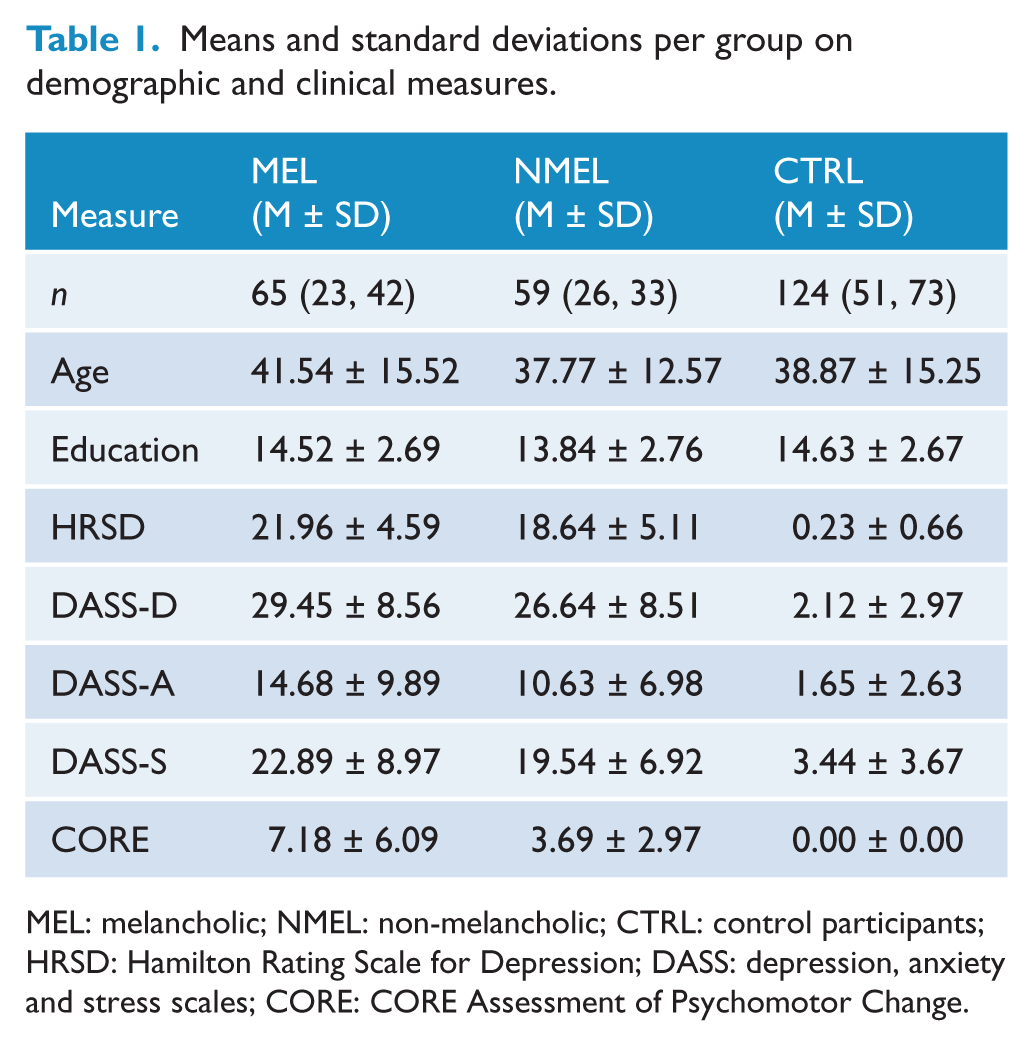
MEL: melancholic; NMEL: non-melancholic; CTRL: control participants; HRSD: Hamilton Rating Scale for Depression; DASS: depression, anxiety and stress scales; CORE: CORE Assessment of Psychomotor Change.
Melancholia was associated with the diagnosis of comorbid anxiety (χ2 (2) = 8.116, p = 0.004), such that 77% of patients with melancholic depression also had a comorbid anxiety disorder in contrast to 47% of patients with non-melancholic depression. Consistent with this, patients with melancholia displayed increased DASS anxiety scores relative to those without melancholia and controls (MEL = 14.677; NMEL = 10.627; CTRL = 1.645). Patients with melancholia were also more likely to have a diagnosis of panic disorder (χ2 (2) = 6.067, p = 0.011) but not GAD or PTSD, which were displayed equally in melancholia and non-melancholia. See Table 2 for current comorbidity data relevant to subtypes (melancholia and non-melancholia).
Current comorbidity data for melancholia vs non-melancholia.
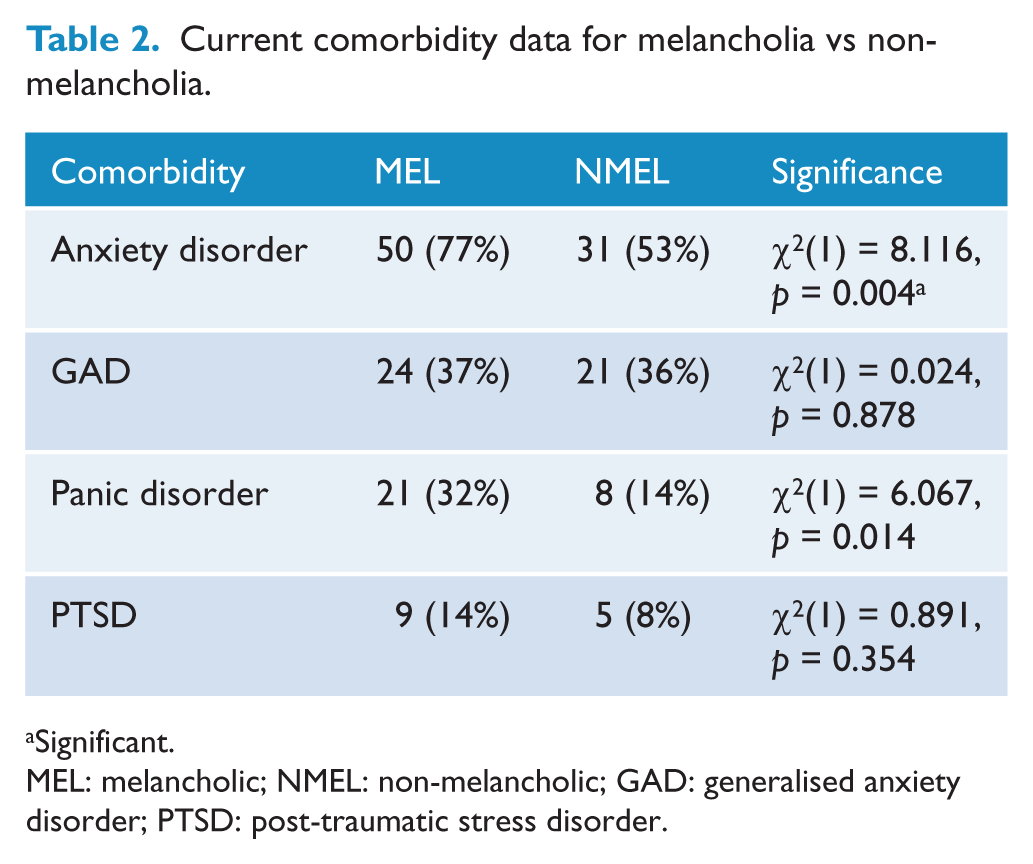
Significant.
MEL: melancholic; NMEL: non-melancholic; GAD: generalised anxiety disorder; PTSD: post-traumatic stress disorder.
ANOVA: Impact of subtype on neuropsychological function
Groups differed significantly on immediate memory recall (F(2, 244) = 3.139, p = 0.045), timing bias (F(2) = 3.120, p = 0.046), variation in timing bias (F(2, 245) = 9.773, p < 0.001), reverse digit span (F(2, 245) = 5.104, p = 0.007) and span of visual memory (F(2, 245) = 3.956, p = 0.02). Results for the ANOVA, including group means, standard deviations and significant differences on cognitive measures, are listed in Table 3.
Means and standard deviations per group on cognitive measures.
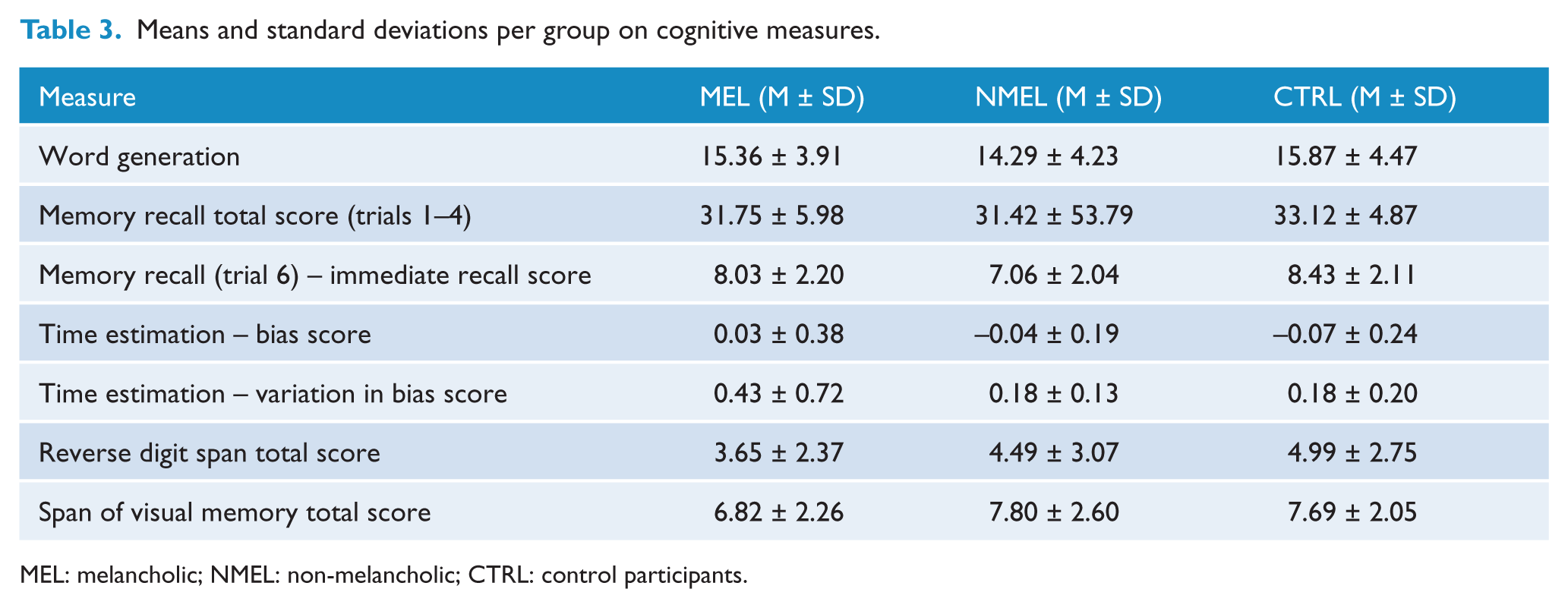
MEL: melancholic; NMEL: non-melancholic; CTRL: control participants.
Melancholia vs controls
Post-hoc tests revealed that melancholic patients differed from controls on timing bias ( MD = 0.056, p = 0.039), variation in timing bias (MD = 2.528, p < 0.001), span of visual memory (MD = −0.870, p = 0.036) and reverse digit span (MD = −1.338, p = 0.005) (see Figure 1 for means and standard errors on span of visual memory and reverse digit span tasks per group). Whilst melancholic patients exhibited poorer performance on the span of visual memory and reverse digit span tasks they demonstrated a smaller average proportional bias in an opposing direction (positive) for time estimation in comparison to controls. In effect, their timing was faster yet more accurate than controls on average. Their variability in timing across trials was, however, greater, indicating less consistency across trials in comparison to controls (see Figure 1 for means and standard errors of average and variability scores on time estimation measures per group).
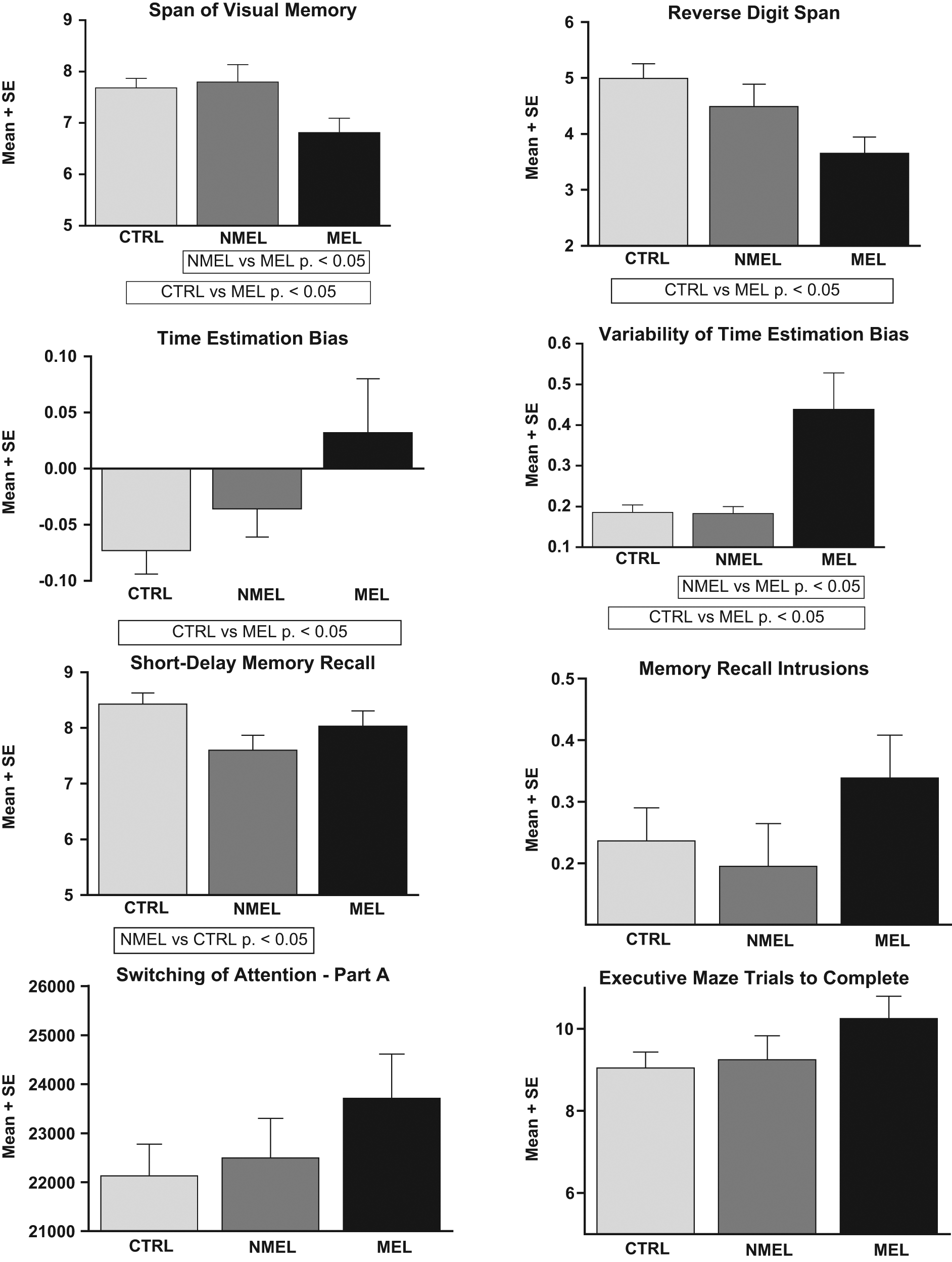
Means and standard errors for cognitive measures. The graphs demonstrate differences between group means across measures: span of visual memory; reverse digit span; time estimation bias, variability of time estimation bias; short-delay memory recall; memory recall intrusions; switching of attention – part A; and executive maze trials to completion. Significant results are indicated on relevant graphs.
Non-melancholia vs controls
Non-melancholic patients differed from controls on immediate memory recall (MD = −0.829, p = 0.043), with poorer performance in comparison to controls (see Figure 1 for means and standard errors for immediate memory recall per group).
Melancholia vs non-melancholia
Melancholic patients displayed poorer performance on variability in timing bias (MD = 2.556, p = 0.001) and span of visual memory (MD = −0.981, p = 0.047) relative to non-melancholic patients (see Figure 1 for means and standard errors for span of visual memory and variability of time estimation per group). Consistent with differences between melancholia and controls, melancholic patients exhibited greater variability in timing across trials, indicating less consistency across trials in comparison to non-melancholic patients.
Regression: Impact of symptoms on neuropsychological function
Anxious arousal
Higher levels of anxious arousal predicted poorer performance on the reverse digit span task (beta = −0.164, t(116) = −2.504, p = 0.014), executive maze (trials to completion) (beta = 0.141, t(117) = 3.142, p = 0.002) and memory intrusions (beta = 0.199, t(117) = 2.035, p = 0.044).
Psychomotor disturbance
Psychomotor disturbance (beta = 0.232, t(118) = 2.675, p = 0.009) was associated with longer duration to complete the switching of attention – part A task.
Depression and anxious apprehension
Depression and anxious apprehension did not reach levels of significance for predicting cognitive measures. Additionally, no symptoms were found to be significant predictors of word generation, memory recall, immediate delayed memory recall, bias in timing estimation and variability of bias in timing estimation, verbal interference and executive maze errors.
Discussion
The current study examined the impact of subtype, severity and symptoms on cognitive control functions. Patients with melancholia displayed poorer performance on working memory relative to patients with non-melancholia (variability in timing bias, span of visual memory) and controls (variability in timing bias, span of visual memory, reverse digit span), while patients with non-melancholia displayed immediate, short-term delayed memory recall deficits (verbal memory recall), highlighting distinctive impairments. We also observed anxious arousal and psychomotor disturbance to contribute to specific neuropsychological impairments such that higher levels of anxious arousal predicted poorer performance on measures of reverse digit span, executive maze and memory intrusions. Finally, a stronger relationship was revealed between comorbid anxiety and melancholia than for non-melancholia. Strengths of the study include a relatively large sample size, a medication-free sample, control for age, sex, education level and IQ, and the use of a validated observer-rated measure of psychomotor disturbance (Hickie, 1996).
Patients with melancholia displayed more variance in time estimation, and poorer span of visual memory, relative to patients with non-melancholia and controls, reflecting deficits in directing attention, sustaining attention and monitoring, which may relate to impairments in DLPFC (Stuss and Levine, 2002). These tasks also reflect deficits in keeping time and sustained performance, potentially relating to impairments in the superior medial regions of the frontal lobe, and right-sided frontal regions of Brodmann area 45 and subadjacent regions of the basal ganglia in particular (Kemp et al., 2005). Increased response time variability in patients with melancholia has been observed previously (relative to non-melancholia) (Winograd-Gurvich et al., 2006), and may reflect a marker of executive dysfunction, indicative of frontal or frontostriatal disturbances (Pizzagalli, 2010). Melancholic participants also demonstrated impairments on the reverse digit span in comparison to controls, which in addition to poorer performance on the span of visual memory may indicate dysfunction in coding representations and monitoring as per the working memory function, also implicating DLPFC function (Stuss and Levine, 2002). These results are consistent with the proposal that melancholic patients may perform more poorly on working memory and sustained attention tasks. By contrast, patients with non-melancholia displayed poorer memory recall (immediate delay), reflecting monitoring deficits that may relate to specific impairment in the left frontal lobe and medial temporal lobe (Stuss and Levine, 2002), highlighting distinctive neuropsychological impairment in subtypes of depression.
Contradictory findings reported in previous studies on the neuropsychological performance of depression may also be due to comorbid anxiety (Basso et al., 2007; Castaneda et al., 2008; Gualtieri and Johnson, 2008; Kaplain et al., 2006). In the current study, patients with melancholia displayed increased anxiety than those with non-melancholia (and controls). In this regard, the finding that patients with melancholia demonstrated a positive bias in time estimation may be accounted for by anxiety, with melancholic patients indicating faster average counting in comparison to controls. In addition, non-melancholic patients exhibited a negative bias in time estimation in comparison to melancholic patients, indicating slower average counting in comparison. Furthermore, it is interesting to note that immediate recall memory is more impaired in depressed patients without anxiety than those with anxiety (Kizilbash et al., 2002), a finding observed in patients with non-melancholia in the current study. Here, we revealed a relationship between melancholic depression and comorbid anxiety diagnosis, with greater contributions from panic disorder to the subtype. Convergent evidence has suggested that panic disorder is associated with symptoms of hyperarousal (Heller and Nitschke, 1997; Heller et al., 1997; Nitschke et al., 1999; Watson, 2005), such as dryness of mouth, difficulty breathing, pounding heart and sweaty palms (Davidson, 2003). These findings are consistent with models distinguishing melancholia as a disorder of pathological hyperarousal with features of rumination, fears of the future and helplessness (Gold and Chrousos, 2002).
Anxious arousal was a significant contributor to poor performance on reverse digit span, executive maze and memory intrusion measures. All these measures engage cognitive control monitoring strategies primarily activating the DLPFC (Stuss and Levine, 2002). Importantly, patients with melancholia only displayed poor performance on the reverse digit span task, but not on executive maze or memory intrusions measure, highlighting partial independence of subtype and symptoms. Whilst anxious arousal contributes to poorer performance on these tasks, subtypes do not differ on planning and capacity to inhibit information despite working memory being significantly impaired in melancholic patients. Psychomotor disturbance, another key feature of melancholia, predicted poorer performance on part A of the switching of attention task, a measure of information processing speed along with monitoring. While patients with melancholia and non-melancholia did not differ on the cognitive switching element of this task (part B) in the current study, possibly due to recruitment of (largely) outpatient rather than inpatient participants, prior studies have reported that melancholic patients perform poorly on these tasks (Austin et al., 1999). Anxious arousal and psychomotor disturbance were important contributors to impairment in our depressed sample, while depression symptoms and anxious apprehension contributed very little to impaired performance on these tasks.
In conclusion, our study reveals that melancholia is associated with more severe depression, anxiety and psychomotor disturbance, and that anxious arousal and psychomotor disturbance in particular contribute to poorer performance on tasks of working memory and sustained attention requiring activation of the DLPFC. Despite melancholia being more severe across symptom domains, patients with non-melancholia were characterised by poorer performance on short-delay memory recall, which relates to left frontal lobe and medial temporal lobe function (Milner, 1974; Perret, 1974; Stuss et al., 1998). Anxiety is a key characteristic of melancholia, which may be associated with greater deficits related to right-sided frontal regions. Thus, our findings support the proposal that depression is a heterogeneous disorder and depression does not simply appear to be a function of severity. Future studies should consider the extent to which these disturbances reflect state or trait deficits and whether neuropsychological markers are able to predict response (or non-response) to treatment (Kemp et al., 2008). Our findings have implications for the relationships between brain function and behaviour in depression for patients with melancholia and non-melancholia.
Footnotes
Funding
CRQ was supported by an Australian Postgraduate Award and AK, by a National Health and Medical Research Council Career Development Award Fellowship (571101). This research was supported by was supported by a National Health and Medical Research Council (NHMRC) Project Grant (464863) and an Australian Research Council Discovery Grant (DP0987332). AH has been awarded research funding from the NHMRC, Australian Rotary, Perpetual Trustees, Eli Lilly Australia, Janssen-Cilag Australia and the Schizophrenia Fellowship of NSW. AH has also received consultancy fees from Organon Australia, Eli Lilly and Lundbeck Australia. AH has received payments for educational sessions run for Astra Zeneca, Janssen Cilag, Eli Lilly, and Organon. AH has also run educational sessions for several medical education companies includingWellmark Australia, Reed Business Information, and CME LLC. Johnson and Johnson Pharmaceutical Research and Development, RED Europe also supported the project from which data in the current study was drawn
Declaration of interest
Sponsors played no role in analysis and interpretation of data; or in the decision to submit the paper for publication. The authors, CRQ and AK, had full access to all of the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis.
